Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Medicina y ética
versión On-line ISSN 2594-2166versión impresa ISSN 0188-5022
Med. ética vol.36 no.2 Ciudad de México abr./jun. 2025 Epub 16-Jun-2025
https://doi.org/10.36105/mye.2025v36n2.06
Articles
The origin of physicians’ positive attitudes towards drug advertising: a reflection for educators and medical students
* Professor, Faculty of Medical Sciences, Universidad de Cuenca, Ecuador. E-mail: jonathan.tipan@ucuenca.edu.ec
** Resident of the Postgraduate Course of Pathological Anatomy, Universidad Central del Ecuador, Ecuador. E-mail: andrea94freire@gmail.com
*** Professor, Faculty of Bioethics, Universidad Anahuac Mexico, Mexico. Professor, Universidad Nacional Autónoma de México. E-mail: german.novoahe@anahuac.mx
The available evidence shows that medical students are prematurely related to drug advertising. This relationship generates a phenomenon called “socialization”, where students naturally cooperate with the pharmaceutical industry. Therefore, the objective of the present review is to investigate socialization as an origin of the positive attitude towards drug advertising during professional practice. Through a review under prism methodology, the academic literature in English and Spanish from January 2019 to June 2024 was analyzed, obtaining three emerging situations: 1) negative impact of drug advertising to the health system; 2) socialization in medical students; and 3) lack of university regulatory policies. It is concluded that socialization generates a positive attitude towards drug advertising during medical education and professional practice.
Keywords: medical education; drug advertising; pharmaceutical industry; bioethics
La evidencia disponible demuestra que los estudiantes de medicina están prematuramente relacionados con la publicidad de medicamentos. Esta relación genera un fenómeno denominado: “socialización”, en donde los estudiantes cooperan naturalmente con la industria farmacéutica. Por consiguiente, el objetivo de la presente revisión es indagar sobre la socialización como origen de la actitud positiva frente a la publicidad de medicamentos durante la práctica profesional. A través de una revisión bajo metodología prisma se analizó la literatura académica en inglés y español desde enero de 2019 hasta junio de 2024, obteniendo tres situaciones emergentes: 1) impacto negativo de la publicidad de medicamentos al sistema de salud; 2) socialización en los estudiantes de medicina; y, 3) carentes políticas regulatorias universitarias. Se concluye que la socialización genera actitud positiva para la publicidad de medicamentos durante la formación médica y en el ejercicio profesional.
Palabras clave: educación médica; publicidad de medicamentos; industria farmacéutica; bioética
1. Introduction
At present, it is estimated that 95% of physicians worldwide interact with the various drug advertising strategies promoted by the pharmaceutical industry (1- 4). This relationship gives rise to biases that influence medical prescriptions up to three times compared to doctors who do not interact with drug advertising (1- 4).
Drug advertising aims to optimize the economic interests of the pharmaceutical industry, however, numerous investigations have revealed negative consequences for the healthcare system, such as persuading physicians with gifts or incentives that cloud professional judgment and conflicts of interest arise (5- 9); promotional literature that promotes the excessive use of drugs when the condition does not warrant it and over-medicalizes patients (10- 13); greater economic investment in advertising strategies in middle and low-income countries, thereby increasing the final price of drugs and health spending (14- 16); fostering misinformation about generic drugs, generating distrust of their therapeutic efficacy among healthcare professionals and the population (17- 19); disturbing the honesty and loyalty of the physician in the eyes of patients, thus affecting the physician-patient relationship (20- 23); the dissemination of scientific literature that exalts the benefits of drugs with little information on side effects (13,24); and bias in research studies sponsored by the pharmaceutical industry with repercussions on decision-making during professional practice (25- 27). All these practices encourage the irrational use of drugs and generate a public health problem (28).
Despite being well documented in the international literature on the negative effect of drug advertising on the health system (1- 4), there are studies that show that a large part of the medical community does not recognize how gifts or incentives from the pharmaceutical industry can influence their behavior when prescribing drugs (2,29,30). On the other hand, they consider it ethical to accept lowcost incentives (31), they feel that the various gifts help them to learn about new pharmaceutical products (29), they do not consider the incentives as something personal, but rather that they contribute to the improvement of patients (32), they believe that the information given to them by medical sales representatives is scientifically valid without corroborating it with other sources of medical information (29,30) and they do not believe it necessary to take precautions when dealing with the advertising strategies of pharmaceutical companies (2,29,30).
Therefore, one might ask: why do physicians, being well-trained people, continue to cooperate with the economic interests of the pharmaceutical industries even though scientific evidence demonstrates the negative effects of this phenomenon? It is therefore important to seek answers by looking into the early stages of physicians’ training.
To our knowledge, research on drug advertising has focused on health professionals (1- 4), on the other hand, few studies describe the growing interest of the pharmaceutical industry to prematurely interact with medical students and what is the impact of this premature exposure on academic training and development as a health professional (41- 47). Therefore, the purpose of this review article is to inquire about the origin of physicians’ positive attitude towards drug advertising strategies during professional practice.
2. Methodology
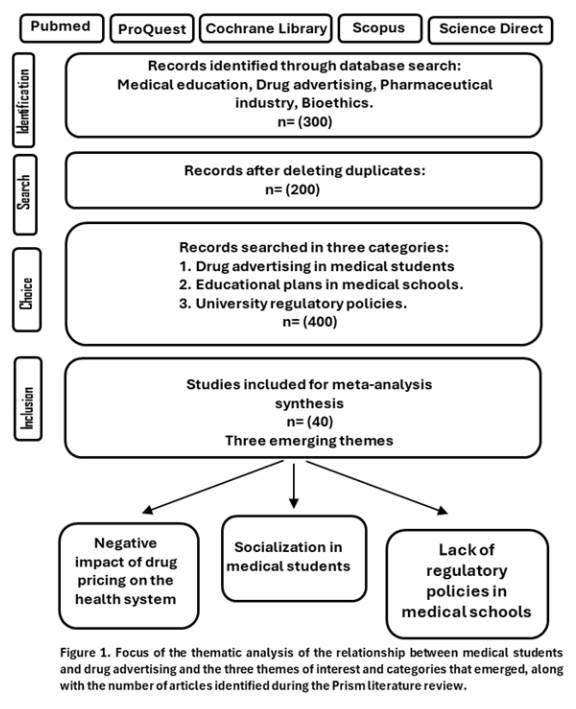
A bibliographic review was carried out under Prisma methodology. The following databases were searched: “Pubmed”, “ProQuest”, “Cochrane Library”, “Scopus”, “Science Direct”, with the keywords: “Medical Education”, “Drug Advertising”, “Pharmaceutical Industry” and “Bioethics”.
2.1. Objective
To investigate the origin of the positive attitude of physicians towards drug advertising strategies during professional practice.
2.3. Exclusion criteria
Opinion articles, letters to the editor, non-peer-reviewed literature.
Scientific articles not published in the following databases: “Pubmed”, “ProQuest”, “Cochrane Library”, “Scopus”, “Science Direct”.
Articles sponsored by the pharmaceutical industry and/or authors declaring conflicts of interest.
2.3 Data analysis
300 scientific studies were obtained from the database. After eliminating duplicate documents, 200 academic articles were examined emphasizing three categories of study: 1) drug advertising in medical students, 2) educational plans in medical schools and, 3) university regulatory policies. Of which 40 academic articles were included in the review and three relevant results emerged: 1) negative impact of drug advertising on the health system; 2) socialization in medical students; 3) lack of regulatory policies in medical schools. The data are presented below in Figure 1.
3. Development
Drug advertising, introduced as a concept by Borden in 1964 (33), combines four basic elements: product, price, place and drug promotion. This generates prescription orders by physicians, which brings pharmaceutical production to consumers and achieves sales targets for pharmaceutical companies.
The advertising of the pharmaceutical industry is a different commercial strategy; the design is aimed mainly at physicians, who are the main prescribers and therefore the target customer for pharmaceutical companies (33- 35). For this reason, the World Health Organization (WHO) encompasses the advertising strategies of pharmaceutical companies as “drug promotion”, defining it as: “all informative and persuasive activities, deployed by manufacturers and distributors, with the aim of inducing the prescription, dispensing, supply, purchase or use of medicines” (36,37).
Since 1988, the who has sought to counteract the effect of pharmaceutical advertising on health professionals and created the document: “Ethical Criteria for the Promotion of Medicines”, which aims to improve health through the rational use of medicines, using the ethical foundations of truthfulness and justice, to apply them in various advertising strategies of pharmaceutical companies.
Over the years, the ethical criteria of the who have been incorporated into the curricula to educate students in health sciences on drug promotion strategies (38). However, the criteria have not been expanded or updated in the face of new advertising activities and do not incorporate a public health ethical justification (12). In addition, governments do not prioritize resources or regulation of promotion (39,40), and in the professional environment, physicians often underestimate the influence of drug advertising on prescribing (2,29,30).
Much research has shown that drug promotion is not only focused on physicians, on the contrary, it is prematurely related to medical students (41- 47). Students participate in events organized by the pharmaceutical industry and receive a variety of gifts or incentives (41- 47). Among the most frequently offered gifts are lowcost gifts such as free drug samples, office supplies, brochures with promotional information on drugs, but also high-cost gifts such as stethoscopes, dinners and sponsorship of scientific lectures (47,48). This being so, the following is a description of the effects produced by the premature relationship between medical students and drug advertising.
3.1. Negative impact of drug advertising on the health care system
Medical students are exposed to various drug advertising strategies. Among the most frequent ones described in the literature are: free drug samples and continuing medical education, hence the majority of health science students in several countries consider these benefits to be “ethically acceptable within the health profession” (41- 48).
In relation to free drug samples, the available scientific evidence shows that they are not related to reasons for consultation or frequent treatments (1,2,49,50). On the contrary, they modify the prescribed medication and alter the initial treatment plans (50). Likewise, they generate a memory effect in health professionals, and therefore they prescribe these drugs more frequently in the future (49). And they increase the cost of the medical prescription, since the free samples of drugs do not have the complete doses of the treatments, so the patient must buy drugs of the same brand, which are generally of higher cost and with less tendency to seek pharmacological alternatives that have the same clinical efficacy (1,2).
In relation to continuing medical education, sponsored by the pharmaceutical industry, it is questioned in several situations. The industry often organizes and prioritizes the scientific contents that are given during the conferences (5), the speakers are representative health professionals in a given area, but often work for the pharmaceutical companies, who are called “key opinion leaders” (KOL) (51,52). It has been described that LCOs, having limited time to conduct in-depth research on the topics to be discussed at conferences, receive scientific information from pharmaceutical industry representatives with statistical data, relevant cases and even slides prepared for the LCOs to discuss the topic during the scientific event (52).
Key opinion leaders play the role of exhibitor providing scientifically valid, but at the same time fallacious information, since during the conferences they avoid mentioning drugs of equal or superior therapeutic efficacy, the benefits of non-pharmacological treatments and promote drugs off-label (51). Some key opinion leaders are paid to be part of the pharmaceutical industry’s research team, publishing academic articles that are mostly biased (9,53), and are crucial in defending a drug when it is under attack for side effects (11).
Therefore, key opinion leaders create for the pharmaceutical industry a strategy of independent fiction before their peers, they seek to break the skepticism of pharmaceutical product information with acceptance and trust from the medical community (51,52,54). Since for medical students and practicing physicians the activities shown as non-promotional and educational purposes are more effective when compared to those of direct commercial promotion (47,48,55- 57).
Another strategy described for medical students within drug advertising is literature handouts. This activity has been investigated and the available studies reveal that it does not meet WHO ethical criteria. The information it contains exaggerates the benefits while hiding the side effects and encouraging the irrational use of drugs as mass consumption products (13,24).
Regarding scientific studies sponsored by the pharmaceutical industry, a systematic review and meta-analysis published in Cochrane found bias (25,26). Studies sponsored by pharmaceutical companies had results with superior therapeutic efficacy and favorable conclusions compared to studies that did not receive private funding (25,26). This leads to affect decision making during medical practice if critical attitudes are not promoted during professional training (25-27,58).
Certainly, medical students in several countries consider that low-cost incentives are “ethically acceptable for health professionals” (41- 48). However, their effects on physicians prescribing behavior are superior to accepting high-cost gifts (4,31,52,59,60).
Numerous authors have shown that drug advertising has a temporary influence on physicians during prescribing, so that financial gifts can counteract this effect (51,61). Thus, it is frequently induced with low-cost incentives, such as free drug samples, dinners, office materials, promotional literature and sponsorship in continuing medical education (33,35), which is effective for the economic interests of pharmaceutical companies with a negative impact on the health system (4,28,47,62).
3.2. Socialization process in medical students
Currently, there is a growing interest in the pharmaceutical industry to relate with medical students in several countries (41- 48). This relationship starts in the first years of medical school and becomes more frequent during the last levels of the career (12,13). Thus, for each higher year completed, gifts from pharmaceutical companies increase for medical students (41,44,46).
Cialdini (54,63), in his book “the psychology of persuasion”, describes six principles that modify medical behavior during professional practice: 1) reciprocity, which consists of returning favors; 2) commitment, by being consistent with the ideas and actions of others; 3) socialization, by believing that the behavior of most people is valid; 4) sympathy, by building bonds of interest; 5) authority, because they are backed by a competent and representative personage and; 6) scarcity, by being available for a limited time. All these principles described above are employed during drug advertising.
For the author Makowska (64), the early exposure of medical students to the advertising strategies of the pharmaceutical industry is aimed at the “socialization process”. Students observe that most physicians “normally” meet in offices or other areas of health institutions with representatives of pharmaceutical companies and accept various gifts or incentives. This results in informal education, since universities do not provide them with knowledge on how to respond ethically to relations with the pharmaceutical industry and there is no regulation of the interaction of medical students with drug advertising (41- 48,64).
Correlating Makowska’s theory with what Cialdini describes the student “believes the behavior of most physicians to be normal” (socialization), and thus generates an early natural and legitimate perception of cooperating with the pharmaceutical industry and becomes accustomed to receiving gifts as privileges of the medical career (64). Consequently, this phenomenon plays a fundamental role in the origin of the positive attitude towards drug advertising during medical training and forges a strong relationship with the pharmaceutical industry in professional practice. Future physicians are prematurely socialized to cooperate with pharmaceutical industries without formal education plans to counteract socialization (54,63,64).
To make matters worse, “socialized medical students” feel invulnerable to promotional strategies after graduation, making them more likely to receive free gifts from pharmaceutical companies (42,46,47,65); oppose mandatory disclosure laws that make financial ties with industry transparent (47,64- 67); make greater use of brandname drugs over generic alternatives during clinical practice (45,68); accept incentives or free samples of drugs; and accept incentives or free samples of drugs from pharmaceutical companies (42,46,47,65); accept incentives or free samples of drugs, trying to make up for the insufficient resources of hospitals (47) and; if there are medical students who are skeptical and/or critical of drug promotion and/or advertising, the absence of education generates in them ignorance and lack of arguments before their peers to respond ethically to pharmaceutical companies (41,46,47,64,69).
3.3. Lack of regulatory policies on drug advertising in medical schools
In the health sciences faculties of several countries, formal university education does not prioritize conflicts of interest, the effects of drug promotion on the public health system, the presence of representatives of the pharmaceutical industry is not limited, and the interaction of medical students with drug advertising is not regulated (41- 48). All of the above generates risk in academic training (47), since the time spent by students in medical schools is of utmost importance for the formation of attitudes that they will carry out in their future professional practice (70).
A double-blind randomized controlled trial in the United States (71) tested the effect of medical students’ attitudes toward exposure to financial gifts offered by the pharmaceutical industry. The study showed that the positive or negative attitude of students is related to the regulatory policies of medical schools. In universities that had a restrictive policy towards pharmaceuticals, students showed a negative attitude towards drug advertising, in contrast to students with a less restrictive environment, who developed a favorable attitude towards incentives or gifts from the pharmaceutical industry.
With regard to the regulation of drug advertising, there are few countries that regulate it. Legislation in the United States, Poland and France requires health professionals to declare financial ties with pharmaceutical companies, with the aim of reducing ethically questionable economic relationships (47,51,67). However, most medical students in these countries are unaware of these legislations, but when they learn about them, they oppose regulation (47,51,67). Additionally, the relationship of physicians and/or medical students with drug advertising is far from being a priority within the formal educational plans taught by bioethics professors, generating informal education on this phenomenon, which will negatively impact professional practice and the health care system (28,47).
4. Discussion
The present research reveals how the premature relationship of medical students with the pharmaceutical industry originates a process called socialization, which consists of cooperating naturally and legitimately with pharmaceutical companies (47,54,63,65). It is suggested that this phenomenon plays a fundamental role in the origin of the positive attitude towards drug advertising during medical training and how it establishes a solid relationship with the pharmaceutical industry in professional practice (54,63,64). However, the interaction between medical students and the pharmaceutical industry has received less attention and scientific inquiry as those involving practicing physicians (4,50,72).
The interaction of medical students with pharmaceutical companies is diverse in several countries. A study in Warsaw reported that 60% of final-year medical school students received at least one gift from pharmaceutical companies and 66% had participated in educational trainings that were sponsored by industry (73). In Japan, a study of 6771 medical students revealed that students received formal conflict of interest education, yet 98% accepted incentives from the pharmaceutical industry (42). In three Baltic countries with a total of 918 medical, pharmaceutical biochemistry and nursing students, 66.2% participated in events sponsored by pharmaceutical companies (43). Although there is currently a growing interest of the industry in engaging with medical students, there are still inconsistencies in scientific studies and little research in middle and low-income countries (42,43,73).
On the other hand, there are medical students who receive classes with professors who have economic ties with pharmaceutical companies (48,74). This generates conflicts of interest, where financial associations between university professors and the pharmaceutical industry may influence the development of teaching activities, with unknowns about the veracity of the education they provide (74). The behavior of professors and the information they provide is valuable and highly influential for students, since professors are historical role models and the knowledge they impart is reflected in future health professionals (43).
While it is true, the present review describes that there is little attention to the exposure of medical students to drug advertising. However, several initiatives have been implemented in the last decade to protect students from conflicts of interest (75,76). Between the years 2007 to 2016, the American Medical Student Association published an annual ranking of U.S. health sciences schools with criteria evaluating their conflict-of-interest policies. This led to the raising of similar initiatives in France, Australia, Germany, and Canada.
The United States has made significant improvements in managing conflicts of interest at the faculty level, largely as a result of the initiatives and the considerable media attention it generated (76). In 2007, most medical schools received the worst possible grade (F), whereas in 2014 two-thirds of these scores evolved to the highest grade (A or B), following the development and strengthening of conflict of interest policies in medical schools (76).
In contrast, there are countries with no regulation at all, such is the case in Greece, medical students’ interactions with pharmaceutical companies are not subject to law or codes of ethics. In addition, there is an absence of national regulation, as well as specific institutional guidelines that address interactions between medical students and pharmaceutical companies’ advertising strategies (46). In the case of Ecuador, no published studies on medical students exposed to drug advertising were found, leaving a research gap that needs to be addressed (28).
Finally, bioethics emphasizes how time in medical schools plays a crucial role in shaping students’ attitudes that they will carry with them into their practical immersion (77- 79). Premature exposure during medical training to drug advertising leads to a natural and legitimate perception of cooperating with the industry (27- 32), which is raised in the present article as the origin of physicians’ positive attitude toward drug advertising during their academic training and in professional practice.
The lack of knowledge of health professionals on how to respond ethically to drug advertising is related to a negative impact on the health care system (28,80,81). However, physicians who received ethical training in medical schools are skeptical of promotional strategies, as well as of information from industry representatives with less influence during drug prescribing (32,58). Therefore, it is essential to create formal education policies for medical students, as well as for university educators (45,47,82). Addressing the physician and/ or medical student relationship with drug advertising strategies should be incorporated into formal bioethics education plans in the different years and with greater emphasis on the last levels of the medical career (41- 48).
The main limitation of the present narrative review was that there is little research in international literature on the relationship between medical students and the pharmaceutical industry in middle and low-income countries. However, it was approached with several studies from different countries that show their realities, so it is necessary to investigate these relationships in each city and / or nation with greater emphasis on where there is no regulation, to have an objective reality and generate public policies to regulate this phenomenon (action-research).
5. Conclusion
Drug advertising is not only focused on physicians, on the contrary, there is an increasing interest in the pharmaceutical industry to relate with medical students. This premature relationship develops a socialization process, from which the student during his academic training awakens a positive attitude towards drug promotion strategies and forges a solid relationship with the pharmaceutical industry in his practice as a professional.
Certainly, drug advertising has a negative impact on the public health system, and despite this phenomenon, there are few or no policies in medical schools that restrict incentives or gifts for students pursuing health sciences careers, and they are still far from being a priority for bioethics professors within formal educational plans.
To make matters worse, the implementation of the who Ethical Criteria is not adjusted to the new forms of drug advertising and lacks the principles and values of public health ethics. Therefore, the revision and updating of these criteria is advocated as a standard guide for educators and medical students.
From the point of view of bioethics, it is inadmissible to accept any pressure that seeks to influence drug prescription. Therefore, the present research proposes to prioritize formal education on the relationship of medical students with the pharmaceutical industry, prior to clinical immersion programs. If there is unethical drug advertising, future professionals are responsible for continuing it and affecting the public health system.
Referencias
1. Brax H, Fadlallah R, Al-Khaled L, Kahale LA, Nas H, El-Jardali F. Association between physicians’ interaction with pharmaceutical companies and their clinical practices: A systematic review and meta-analysis. PLoS ONE. 2017; 12(4):e0175493. Disponible en: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0175493 [ Links ]
2. Fickweiler F, Fickweiler W, Urbach E. Interactions between physicians and the pharmaceutical industry generally and sales representatives specifically and their association with physicians’ attitudes and prescribing habits: a systematic review. BMJ Open. 2017; 7(9):e016408. Disponible en: https://doi.org/10.1136/bmjopen-2017-016408 [ Links ]
3. Fadlallah R, Alkhaled L, Brax H, Nasser M, Rajabbik MH, Nass H. Extent of physician-pharmaceutical industry interactions in low- and middle-income countries: a systematic review. Eur J Public Health. 2018; 28(2):224-30. Disponible en: https://doi.org/10.1093/eurpub/ckx204 [ Links ]
4. Mitchell AP, Trivedi NU, Gennarelli RL, Chimonas S, Tabatabai SM, Goldberg J. Are Financial Payments from the Pharmaceutical Industry Associated with Physician Prescribing? A Systematic Review. Ann Intern Med [Internet]. 2021 [citado 15 de octubre de 2021]; 174(3):353-61. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8315858/ [ Links ]
5. Genta-Mesa G, Flórez ID. Relación médico-industria y los conflictos de interés: aspectos históricos y normativos, impactos negativos y propuestas. Iatreia [Internet]. 2019 [citado 31 de mayo de 2020]; 32(4):298-310. Disponible en: https://revistas.udea.edu.co/index.php/iatreia/article/view/333905 [ Links ]
6. Davari M, Khorasani E, Tigabu BM. Factors Influencing Prescribing Decisions of Physicians: A Review. Ethiop J Health Sci [Internet]. 2018 [citado 1 de septiembre de 2020]; 28(6):795-804. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6308758/ [ Links ]
7. Altisent R, Delgado-Marroquín MT, Astier-Peña MP. Conflictos de interés en la profesión médica. Aten Primaria [Internet]. 2019 [citado 9 de noviembre de 2021]; 51(8):506-11. Disponible en: https://www.elsevier.es/es-revista-atencion-primaria-27-articulo-conflictos-interes-profesion-medica-S0212656719303634 [ Links ]
8. Giubilini A, Savulescu J. Beyond Money: Conscientious Objection in Medicine as a Conflict of Interests. J Bioeth Inq. 2020; 17(2):229-43. https://doi.org/10.1007/s11673-020-09976-9 [ Links ]
9. Ebrahimi A, Zand S, Bagheri Amiri F, Shahi F, Jafarian A, Kaviani A. Conflict of Interest: Are Iranian Breast Cancer Specialists Prone to it? Asian Pac J Cancer Prev [Internet]. [citado 16 de mayo de 2021]; 21(6):1653-8. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7568883/ [ Links ]
10. Hollander MAG, Donohue JM, Stein BD, Krans EE, Jarlenski MP. Association between Opioid Prescribing in Medicare and Pharmaceutical Company Gifts by Physician Specialty. J Gen Intern Med. 2020; 35(8):2451-8. Disponible en: https://link.springer.com/article/10.1007/s11606-019-05470-0 [ Links ]
11. Moynihan R. Commentary: The Voice of the People, Funded Now by Your Friendly Pharmaceutical Company. Bioethical Inquiry [Internet]. 2020 [citado 31 de agosto de 2020]; 17(1):61-3. Disponible en: https://doi.org/10.1007/s11673-020-09965-y [ Links ]
12. Parker L, Williams J, Bero L. Ethical drug marketing criteria for the 21st century. BMJ [Internet]. 2018 [citado 18 de abril de 2020]; 361. Disponible en: https://www.bmj.com/content/361/bmj.k1809 [ Links ]
13. Prasad P, Bajracharya SR, Deo S, Lamichhane S, Pradhan PMS, Ghimire R. Adherence of Drug Promotional Literatures Distributed by Pharmaceutical Companies to World Health Organization Ethical Criteria for Medicinal Drug Promotion. Journal of Nepal Health Research Council [Internet]. 2019 [citado 24 de septiembre de 2020]; 17(3):345-50. Disponible en: http://jnhrc.com.np/index.php/jnhrc/article/view/1840 [ Links ]
14. Gul R, Saeed H, Saleem Z, Rasool F, Hashmi FK, Islam M. Perceptions of and barriers to ethical promotion of pharmaceuticals in Pakistan: perspectives of medical representatives and doctors. BMC Med Ethics. 2021; 22(1):2. Disponible en: https://doi.org/10.1186/s12910-020-00569-0 [ Links ]
15. Datta A, Dave D. Effects of Physician-directed Pharmaceutical Promotion on Prescription Behaviors: Longitudinal Evidence. Health Econ. 2017; 26(4):450-68. Disponible en: https://doi.org/10.1002/hec.3323 [ Links ]
16. DiStefano MJ, Levin JS. Does Incorporating Cost-Effectiveness Analysis Into Prescribing Decisions Promote Drug Access Equity? AMA journal of ethics. 2019; 21(8):E679-685. [ Links ]
17. Howard JN, Harris I, Frank G, Kiptanui Z, Qian J, Hansen R. Influencers of Generic Drug Utilization: A Systematic Review. Res Social Adm Pharm [Internet]. 2018 [citado 9 de agosto de 2020]; 14(7):619-27. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5910277/ [ Links ]
18. Das M, Choudhury S, Maity S, Hazra A, Pradhan T, Pal A. Generic versus branded medicines: An observational study among patients with chronic diseases attending a public hospital outpatient department. J Nat Sci Biol Med [Internet]. 2017 [citado 28 de septiembre de 2020]; 8(1):26-31. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5320819/ [ Links ]
19. Desai RJ, Sarpatwari A, Dejene S, Khan NF, Lii J, Rogers JR. Comparative effectiveness of generic and brand-name medication use: A database study of US health insurance claims. PLoS Med [Internet]. 2019 [citado 28 de septiembre de 2020];1 6(3). Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6415809/ [ Links ]
20. Ammous A, Bou Zein Eddine S, Dani A, Dbaibou J, El-Asmar JM, Sadder L. Awareness and attitudes of the Lebanese population with regard to physician-pharmaceutical company interaction: a survey study. BMJ open. 2017; 7(3):e013041. Disponible en: https://bmjopen.bmj.com/content/7/3/e013041 [ Links ]
21. Kanter GP, Carpenter D, Lehmann LS, Mello MM. US Nationwide Disclosure of Industry Payments and Public Trust in Physicians. JAMA Netw Open. 2019; 2(4):e191947. Disponible en: https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2730473 [ Links ]
22. Hwong AR, Sah S, Lehmann LS. The Effects of Public Disclosure of Industry Payments to Physicians on Patient Trust: A Randomized Experiment. Journal of General Internal Medicine. noviembre de 2017; 32(11):1186-92. Disponible en: https://doi.org/10.1007/s11606-017-4122-y [ Links ]
23. Fadlallah R, Nas H, Naamani D, El-Jardali F, Hammoura I, Al-Khaled L. Knowledge, Beliefs and Attitudes of Patients and the General Public towards the Interactions of Physicians with the Pharmaceutical and the Device Industry: A Systematic Review. PLoS ONE. 2016; 11(8):e0160540. Disponible en: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0160540 [ Links ]
24. Ganashree P, Bhuvana K, Sarala N. Critical review of drug promotional literature using the World Health Organization guidelines. J Res Pharm Pract [Internet]. 2016 [citado 18 de abril de 2020]; 5(3):162-5. Disponible en: https://www.ncbi.nlm. nih.gov/pmc/articles/PMC4966233/ [ Links ]
25. Lundh A, Lexchin J, Mintzes B, Schroll JB, Bero L. Industry sponsorship and research outcome: systematic review with meta-analysis. Intensive Care Med [Internet]. 2018; 44(10):1603-12. Disponible en: https://pubmed.ncbi.nlm.nih.gov/30132025/ [ Links ]
26. Lundh A, Lexchin J, Mintzes B, Schroll JB, Bero L. Industry sponsorship and research outcome. Cochrane Database Syst Rev. 16 de 2017;2:MR000033. [ Links ]
27. Devji T, Busse JW. Cochrane in CORR®: Industry Sponsorship and Research Outcome. Clin Orthop Relat Res [Internet]. 2017 [citado 29 de septiembre de 2020]; 475(9):2159-64. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5539042/ [ Links ]
28. Tipán Barros JM, Zavala-Calahorrano A, Estévez Montalvo LE. Bioética y la promoción de medicamentos. ATENEO [Internet]. 2021 [citado 3 de julio de 2021]; 23(1):89-100. Disponible en: https://colegiomedicosazuay.ec/ojs/index.php/ateneo/article/view/136 [ Links ]
29. Yimenu DK, Demeke CA, Kasahun AE, Siraj EA, Wendalem AY, Bazezew ZA. Health professional’s exposure, attitude, and acceptance of drug promotion by industry representatives: A cross-sectional study in Ethiopia. Sci Prog. 2021; 104(2):368504211029435. [ Links ]
30. Shakeel S, Nesar S, Iffat W, Fatima B, Maqbool T, Jamshed S. A quantitative insight of the interactions of prescribers with pharmaceutical organization’s representatives in clinical settings of Karachi. Integr Pharm Res Pract [Internet]. 2019 [citado 18 de abril de 2020]; 8:75-83. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6613537/ [ Links ]
31. Khazzaka M. Pharmaceutical marketing strategies’ influence on physicians’ prescribing pattern in Lebanon: ethics, gifts, and samples. BMC Health Serv Res. 2019; 19(1):80. [ Links ]
32. Altawalbeh SM, Ibrahim IA, Al-Shatnawi SF. Influence of pharmaceutical promotion on prescribers in Jordan. Int J Clin Pharm. 2020; 42(2):744-55. [ Links ]
33. Ding M, Eliashberg J, Stremersch S. Innovation and Marketing in the Pharmaceutical Industry. 1.a ed. New York: Springer; 2014. [ Links ]
34. Bhatt P. Study on Influence of Medical Representative in Conversation of Doctor’s Prescription in India. Global Journal of Management And Business Research [Internet]. 2018 [citado 30 de mayo de 2020]; Disponible en: https://pubmed.ncbi.nlm.nih.gov/33623031/ [ Links ]
35. Hailu AD, Workneh BD, Kahissay MH. Influence of pharmaceutical marketing mix strategies on physicians’ prescribing behaviors in public and private hospitals, Dessie, Ethiopia: a mixed study design. BMC Public Health. 2021; 21(1):65. [ Links ]
36. World Health Organization. Ethical criteria for medicinal drug promotion. 1988;16. Disponible en: https://apps.who.int/iris/handle/10665/38125 [ Links ]
37. Red Panamericana de Armonización de la Reglamentación Farmacéutica. OPS / OMS. 2013 [citado 24 de mayo de 2020]. Criterios éticos para la promoción, propaganda y publicidad de medicamentos. Disponible en: https://www.paho.org/hq/index.php?option=com_content&view=article&id=9145:2013-criterios-eticos-promocion-propaganda-publicidad-medicamentos-2013&Itemid=3562&lang=es [ Links ]
38. World Health Organization, Health Action International. Understanding and responding to pharmaceutical promotion: a practical guide for educators and students [Internet]. IGI Global; 2016 [citado 29 de abril de 2024]. Disponible en: http://haiweb.org/wp-content/uploads/2015/05/Pharma-Promotion-Guide-English.pdf [ Links ]
39. Lexchin J. Models for financing the regulation of pharmaceutical promotion. Global Health. 2012; 8:24. [ Links ]
40. Pashley D, Ozieranski P, Mulinari S. Disclosure of Pharmaceutical Industry Funding of Patient Organisations in Nordic Countries: Can Industry Self-Regulation Deliver on its Transparency Promise? Int J Health Serv. 2022; 52(3):347-62. [ Links ]
41. Salmane-Kulikovska I, Poplavska E, Mezinska S, Dumpe V, Dauvarte H, Lazdina L. Medical, pharmacy and nursing students in the Baltic countries: interactions with the pharmaceutical and medical device industries. BMC Med Educ [Internet]. 2020 [citado 27 de septiembre de 2020];20. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7137495/ [ Links ]
42. Saito S, Maeno T, Miyata Y, Maeno T. Medical students’ attitudes toward interactions with the pharmaceutical industry: a national survey in Japan. BMC Medical Education [Internet]. 2018 [citado 30 de mayo de 2020]; 18(1):286. Disponible en: https://doi.org/10.1186/s12909-018-1394-9 [ Links ]
43. Scheffer P, Guy-Coichard C, Outh-Gauer D, Calet-Froissart Z, Boursier M, Mintzes B. Conflict of Interest Policies at French Medical Schools: Starting from the Bottom. PLoS One [Internet]. 2017 [citado 27 de septiembre de 2020]; 12(1). Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5221756/ [ Links ]
44. Cuellar LRO, Sandoval RNC, Benavides WJP. Actitud de los estudiantes de medicina frente al marketing farmacéutico en Perú. Revista Cubana de Salud Pública [Internet]. 2020 [citado 10 de noviembre de 2021]; 46(2). Disponible en: https://pubmed.ncbi.nlm.nih.gov/33623031/ [ Links ]
45. Domeyer PJ, Katsari V, Sarafis P, Aletras V, Niakas D. Greek students’ attitudes, perception and knowledge regarding generic medicines in times of economic crisis: a cross-sectional study. BMC medical education. 2018; 18(1):262. [ Links ]
46. Filippiadou M, Kouvelas D, Garyfallos G, Tsakiridis I, Tzachanis D, Spachos D. Exposure to the drug company marketing in Greece: Interactions and attitudes in a non-regulated environment for medical students. Annals of Medicine and Surgery [Internet]. 2017 [citado 9 de octubre de 2020]; 19:23-8. Disponible en: http://www.sciencedirect.com/science/article/pii/S2049080117301577 [ Links ]
47. Makowska M, Kaczmarek E, Rodzinka M. Transparency or restricting gifts? Polish medical students’ opinions about regulating relationships with pharmaceutical sales representatives. Monash Bioeth Rev., 2021. [ Links ]
48. De Ferrari A, Gentille C, Davalos L, Huayanay L, Malaga G. Attitudes and Relationship between Physicians and the Pharmaceutical Industry in a Public General Hospital in Lima, Peru. PLoS One [Internet]. 2014 [citado 1 de septiembre de 2020]; 9(6). Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4076259/ [ Links ]
49. Rafique S, Sarwar W, Rashid A, Sheerin F. Influence of free drug samples on prescribing by physicians: A cross sectional survey. J Pak Med Assoc. 2017; 67(3):465-7. [ Links ]
50. Lussier MT, Diallo FB, Pluye P, Grad R, Lessard A, Rhéaume C. Drug samples in family medicine teaching units: a cross-sectional descriptive study. Can Fam Physician [Internet]. 2018 [citado 26 de septiembre de 2020]; 64(12):e546-52. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6371885/ [ Links ]
51. Leonardo Alves T, Lexchin J, Mintzes B. Medicines Information and the Regulation of the Promotion of Pharmaceuticals. Sci Eng Ethics [Internet]. 2019 [citado 2 de septiembre de 2022]; 25(4):1167-92. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6647516/ [ Links ]
52. Chen X. Pharmaceutical Sales Representatives in the United States and China: The Need for Professional Public Space. Health Care Anal [Internet]. 2021 [citado 2 de enero de 2022]; 1-22. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8580741/ [ Links ]
53. Piller C. Disgraced researchers can still reap drug industry payouts. Science. 2020; 370(6512):27. [ Links ]
54. Cialdini RB. The Psychology of Persuasion. [Internet]. Quill. New York; 1984 [citado 1 de septiembre de 2020]. Disponible en: https://www.goodreads.com/work/best_book/29303-influence-how-and-why-people-agree-to-things [ Links ]
55. Faisal A, Ahmad MS, Thurasamy R, Ahmed R. Doctors’ Interactions with Pharmaceutical Sales Representatives: Modelling Doctors Prescription Behaviour. Community Ment Health J. 2020; 56(3):456-63. [ Links ]
56. Jandhyala R. Influence of Pharmaceutical Company Engagement Activities on the Decision to Prescribe: A Pilot Survey of UK Rare Disease Medicine Prescribers. Pharmaceut Med. 2020; 34(2):127-34. [ Links ]
57. Leonardo Alves T, Lexchin J, Mintzes B. Medicines Information and the Regulation of the Promotion of Pharmaceuticals. Sci Eng Ethics [Internet]. 2019 [citado 11 de octubre de 2020]; 25(4):1167-92. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6647516/ [ Links ]
58. Tijdink JK, Smulders YM, Bouter LM, Vinkers CH. The effects of industry funding and positive outcomes in the interpretation of clinical trial results: a randomized trial among Dutch psychiatrists. BMC Med Ethics. 18 de 2019; 20(1):64. [ Links ]
59. Mitchell AP, Winn AN, Lund JL, Dusetzina SB. Evaluating the Strength of the Association Between Industry Payments and Prescribing Practices in Oncology. Oncologist. 2019; 24(5):632-9. [ Links ]
60. Strong C. Why academic medical centers should ban drug company gifts to individuals. Am J Bioeth. 2010; 10(1):13-5. [ Links ]
61. Hajjar R, Bassatne A, Cheaito MA, Naser El Dine R, Traboulsy S, Haddadin F. Characterizing the interaction between physicians, pharmacists and pharmaceutical representatives in a middle-income country: A qualitative study. PLoS ONE. 2017; 12(9):e0184662. [ Links ]
62. Jacob NT. Drug promotion practices: A review. Br J Clin Pharmacol. 2018; 84(8):1659-67. Disponible en: https://doi.org/10.1111/bcp.13513 [ Links ]
63. Cialdini RB. Influence: The Psychology of Persuasion. New York: Harper Collins USA; 2006. [ Links ]
64. Makowska M. How Polish medical students are socialised to cooperate with the pharmaceutical industry: a focus group study of the importance of informal, hidden and null curricula. Health Sociol Rev. 2021;1-15. [ Links ]
65. Al-Areefi MA, Ibrahim MIM, Hassali MAA, Alfadl AA. Relationships between physicians’ characteristics and practice-setting factors and their attitude towards drug promotion. Journal of Pharmaceutical Health Services Research [Internet]. 2019 [citado 9 de noviembre de 2021]; 10(3):325-32. Disponible en: https://onlinelibrary.wiley.com/doi/abs/10.1111/jphs.12284 [ Links ]
66. Williams J, Lipworth W, Mayes C, Olver I, Kerridge I. Should disclosure of conflicts of interest in medicine be made public? Medical students’ views. Med Educ. 2017; 51(12):1232-40. Disponible en: https://doi.org/10.1111/medu.13383 [ Links ]
67. Chimonas S, DeVito NJ, Rothman DJ. Bringing Transparency to Medicine: Exploring Physicians’ Views and Experiences of the Sunshine Act. Am J Bioeth. 2017; 17(6):4-18. Disponible en: https://doi.org/10.1080/15265161.2017.1313334 [ Links ]
68. Schwartz LM, Woloshin S. Medical Marketing in the United States, 1997-2016. JAMA [Internet]. 2019 [citado 31 de agosto de 2020]; 321(1):80-96. Disponible en: https://jamanetwork.com/journals/jama/fullarticle/2720029 [ Links ]
69. Stark TJ, Brownell AK, Brager NP, Berg A, Balderston R, Lockyer JM. Exploring Perceptions of Early-Career Psychiatrists About Their Relationships With the Pharmaceutical Industry. Acad Psychiatry [Internet]. 2016 [citado 12 de diciembre de 2020]; 40(2):249-54. Disponible en: https://doi.org/10.1007/s40596-015-0403-0 [ Links ]
70. Austad KE, Avorn J, Kesselheim AS. Medical students’ exposure to and attitudes about the pharmaceutical industry: a systematic review. PLoS Med. 2011; 8(5):e1001037. Disponible en: https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1001037 [ Links ]
71. Grande D, Frosch DL, Perkins AW, Kahn BE. Effect of exposure to small pharmaceutical promotional items on treatment preferences. Arch Intern Med. 2009; 169(9):887-93. Disponible en: https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/773513 [ Links ]
72. Fadare JO, Oshikoya KA, Ogunleye OO, Desalu OO, Ferrario A, Enwere OO. Drug promotional activities in Nigeria: impact on the prescribing patterns and practices of medical practitioners and the implications. Hosp Pract (1995). 2018; 46(2):77-87. Disponible en: https://doi.org/10.1080/21548331.2018.1437319 [ Links ]
73. Makowska M, Sillup G, Lee M. Pharma’s marketing influence on medical students and the need for culturally competent and stricter policy and educational curriculum in medical schools : A comparative analysis of social scientific research between Poland and the U.S. Journal of Healthcare Ethics & Administration [Internet]. 2017 [citado 1 de mayo de 2024];2(2). Disponible en: https://www.researchgate.net/publication/321873999_Pharma’s_Marketing_Influence_on_Medical_Students_and_the_Need_for_Culturally_Competent_and_Stricter_Policy_and_Educational_Curriculum_in_Medical_Schools_A_Comparative_Analysis_of_Social_ Scientific_Res [ Links ]
74. Ehringhaus SH, Weissman JS, Sears JL, Goold SD, Feibelmann S, Campbell EG. Responses of medical schools to institutional conflicts of interest. JAMA. 2008; 299(6):665-71. [ Links ]
75. Bechoux L, De Vleeschouwer O, Vanheuverzwijn C, Verhegghen F, Detiffe A, Colle F. Conflict of interest policies at Belgian medical faculties: Cross-sectional study indicates little oversight. PLoS One. 2021; 16(2):e0245736. [ Links ]
76. Carlat DJ, Fagrelius T, Ramachandran R, Ross JS, Bergh S. The updated AMSA scorecard of conflict-of-interest policies: a survey of U.S. medical schools. BMC Medical Education [Internet]. 2016 [citado 1 de mayo de 2024];16(1):202. Disponible en: https://doi.org/10.1186/s12909-016-0725-y [ Links ]
77. Casado M, López M. Manual de Bioética Laica. Edicions de la Universitat de Barcelona. Barcelona y Sevilla; 2018. [ Links ]
78. Malik F, Junaid M, Sharif I. Exploring the Role of Pharmaceutical Marketing on Physician Ethical Behaviors: A Grounded Theory Study; 1991. [ Links ]
79. Mirzaei A, Carter SR, Schneider CR. Marketing activity in the community pharmacy sector - A scoping review. Research in Social and Administrative Pharmacy [Internet]. 2018 [citado 30 de noviembre de 2020]; 14(2):127-37. Disponible en: http://www.sciencedirect.com/science/article/pii/S1551741117303315 [ Links ]
80. Workneh BD, Gebrehiwot MG, Bayo TA, Gidey MT, Belay YB, Tesfaye DM. Influence of Medical Representatives on Prescribing Practices in Mekelle, Northern Ethiopia. PLoS One [Internet]. 2016 [citado 30 de mayo de 2020]; 11(6). Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4911221/ [ Links ]
81. Kruijtbosch M, Göttgens-Jansen W, Floor-Schreudering A, van Leeuwen E, Bouvy ML. Moral dilemmas of community pharmacists: a narrative study. Int J Clin Pharm [Internet]. 2018 [citado 22 de junio de 2021]; 40(1):74-83. Disponible en: https://doi.org/10.1007/s11096-017-0561-0 [ Links ]
82. Ortiz SE, Rosenthal MB. Medical Marketing, Trust, and the Patient-Physician Relationship. JAMA [Internet]. 2019 [citado 8 de abril de 2022]; 321(1):40-1. Disponible en: https://doi.org/10.1001/jama.2018.19324 [ Links ]
Received: August 13, 2024; Accepted: December 16, 2024











 texto en
texto en