Study contribution
Capture myopathy is a disease that occurs in different animals worldwide, such as wild mammals and birds. This is a pathology probably induced by stress, which cause damage to the myofibers, that can render a bird unable to stand, walk or fly, in addition to inducing alterations in certain blood clinical biochemistry parameters like creatine kinase and aspartate aminotransferase enzymes. The aims of this study were to collect, summarize and classify taxonomic information, clinical signs, diagnostic methods, and treatments described in the literature about capture myopathy in wild birds performing a systematic review and is intended to be a reference material for students and professionals who work with these animals in clinical practice.
Introduction
Capture myopathy (CM) is a multifactorial disease associated with the capturing and handling of certain species of wild mammals and birds which can occur secondarily to other pathologies.1,2 It is a condition with worldwide distribution, with marked morbidity and mortality and their predisposing factors include specific environmental conditions (e.g., high temperature and humidity), capture techniques that induce exacerbated stress response in the animal, prolonged immobilization times, and underlying diseases that compromise the animal’s health.3,4 The disease has implications for the survival of the individuals, and this is very relevant, for example in endangered species.5
CM causes muscle tissues to break down, making it difficult for birds to stand, walk, or fly.6 Four different forms of the disease have been described: 1) hyperacute, characterized by sudden death due to heart failure after a short period (minutes), 2) acute, less severe, characterized by muscle rupture, ataxia, and death within 24-48 hours, 3) subacute, less severe, characterized by muscle and kidney damage, and death after a few days, and 4) chronic: characterized by survival for several days or months, followed by sudden death from a heart attack.7
McEntire and Sánchez8 indicated that stress and undue fear are two of the most important factors contributing to CM. The authors observed that many birds experience stress when they enter a veterinary hospital setting, which can lead to them moving excessively. This additional movement causes muscle damage and prevents its regeneration. The extreme and prolonged stress response is associated with increased catecholamines, cortisol, and aldosterone blood levels, potentially damaging cardiac and skeletal muscles. It has been suggested that ischemia and reperfusion may play a central role in CM through stress-induced muscle necrosis.9
The pathogenesis of CM is not fully understood, but it is believed to be caused by the anaerobic oxygen-free metabolic pathway, which is associated with intense muscular activity. Anaerobic metabolism ends with lactic acid accumulation, which leads to severe metabolic acidosis and secondary muscular necrosis.10,11 When muscles work at high intensity, they consume oxygen faster than the blood can deliver, resulting in oxygen deficiency. In the absence of oxygen, the muscles produce lactic acid as product instead of carbon dioxide. If lactic acid accumulates in muscle cells, it destroys their cell membranes, causing muscle damage characterized by the release of the cell content.10 When muscles are injured, the basal membrane and the sarcolemma of myofibers release cytoplasmic components such as myoglobin and creatine kinase into the bloodstream, generating an increase in their levels in the blood.12,13
This systematic review aims to 1) give taxonomic information regarding some species of wild birds that have been reported to have CM, according to the literature, 2) describe the clinical signs associated with CM in wild birds, 3) identify the methods used to diagnose CM, and 4) summarize different treatments for CM in wild birds.
Materials and methods
Study design
This corresponds to an observational, descriptive, and narrative study of scientific literature, with a systematic review design.14
Bibliographic search strategy
The information was obtained from a systematic search of scientific articles and documents in electronic documentary databases, including EBSCO Host, PubMed, Science Direct, ProQuest, and the Google Scholar search engine. In addition, book chapters in digital format were also included. The search was conducted for scientific literature published between January 1980 to March 2022.
To identify different articles in the databases and search engines, keywords were used and combined using the Boolean operators “AND and OR.” The documents were screened for the presence of the keywords in the title, abstract, or introduction. The bibliography of each document was also reviewed to identify additional sources. This information was then selected, collected, and classified. The search for scientific articles and texts was conducted in English, Spanish, and Portuguese language. Some examples of the key words used were: capture myopathy, wild birds, stress (English language), miopatía por captura, aves silvestres, estrés (Spanish language), miopatia captura, aves selvagem, estresse (Portuguese language).
Elegibility criteria of bibliography
Inclusion criteria
Scientific articles with the following designs were included: systematic reviews, narrative (bibliographic) reviews, clinical trials, and clinical case reports. The target group in the selected studies was limited to wild birds with CM. The studies included in this review were required to report data on clinical signs, treatments, or diagnostic methods regarding the pathology of CM that had been used and/or observed in wild birds.
Exclusion criteria
Theses, unpublished studies, and abstracts were not included in the review. The studies where target populations were not wild birds were also excluded, as this pathology can occur in various animal species. In addition, studies were discarded if the diagnosis of CM was not confirmatory.
Critical analysis of the information
The evaluation of the studies was carried out by conducting a critical analysis of each article, using the guideline by Bobenrieth15 regarding the introduction, objectives, materials and methods, results, conclusions, and bibliography.
Data extraction
Information regarding the taxonomy of the birds described (family, genus, and specie), clinical signs specified for CM according to the species of birds were extracted from each publication selected. Creatine kinase and aspartate animotransferase levels and the described reference values were also recorded, as well as necropsy findings (macroscopic and microscopic lesions, type of sample and species of bird where these findings were described). Finally, treatments reported in the literature for CM (fluid therapy, anti-inflammatories, vitamin treatments, mineral supplement treatments, central nervous system depressant drugs, pH control treatment, forms of rehabilitation and physiotherapy) were also compiled. The PRISMA statement (Preferred Reporting Items for Systematic Reviews and Meta-Meta-analyses)16 was followed to write this study.
Results
Bibliographic search
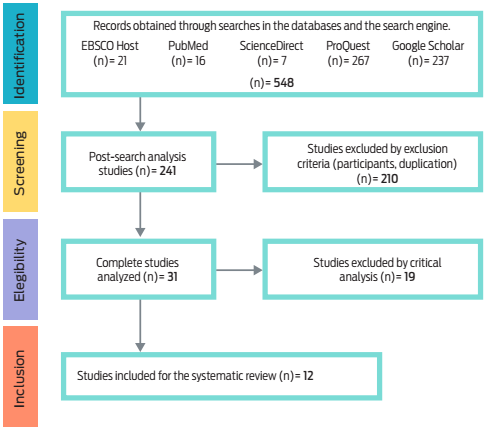
In the bibliographic search, 548 potentially eligible articles were identified in the databases and the search engine, of which 241 remained, and 210 were retrieved by the exclusion criteria. Finally, 31 studies were completely analyzed and 19 were excluded through the critical analysis, leaving a total of 12 documents chosen for the systematic review (Figure 1).
The Table 1 provides a summary of the 12 documents that were included in this systematic review, indicating the author(s), journal, year of publication, country where the study was conducted, type of publication, and database or search engine where the reference was found.
Table 1 Studies for this systematic review
| Author(s) | Year | Journal | Country | Type of publication | Database/ search engine |
|---|---|---|---|---|---|
| McEntire and Sánchez8 | 2017 | Journal of Avian Medicine and Surgery | USA | Scientific article | EBSCOhost |
| Hurtado et al.17 | 2021 | Journal of Avian Medicine and Surgery | Brazil | Scientific article | EBSCOhost |
| Businga et al.18 | 2007 | Journal of Avian Medicine and Surgery | USA | Scientific article | EBSCOhost |
| Höfle et al.19 | 2004 | Wildlife Society Bulletin | Spain | Scientific article | EBSCOhost |
| Hanley et al.20 | 2005 | Journal of Zoo and Wildlife Medicine | USA | Case report | EBSCOhost |
| Hayes et al.21 | 2003 | Journal of Wildlife Diseases | USA | Scientific article | PubMed |
| Ward et al.1 | 2011 | Journal of Wildlife Diseases | New Zealand | Scientific article | Google Scholar |
| Rogers et al.22 | 2004 | Journal of Field Ornithology | Australia | Scientific article | Google Scholar |
| Finlay y Jeske23 | 1999 | Wildfowl | USA | Scientific article | Google Scholar |
| Dunne and Miller24 | 2009 | Conference proceeding | USA | Case report | Google Scholar |
| Tully et al.25 | 1996 | Journal of Avian Medicine and Surgery | USA | Scientific article | Google Scholar |
| Carpenter et al.26 | 1991 | Journal of Zoo and Wildlife Medicine | USA | Scientific article | Google Scholar |
Taxonomic classification of wild birds with CM
The avian families Scolopacidae, Gruidae, and Anatidae showed the highest frequencies of CM. Most information on CM was recorded in countries such as the United States and Australia (Table 2).
Table 2 Classification of the birds that presented CM in the analyzed literature
| Family | Specie | Author(s) | Year | Country |
|---|---|---|---|---|
| Gruidae | Grus canadensis pulla | Carpenter et al.26 | 1991 | USA |
| Dromaiidae | Dromaius novaehollandiae | Tully et al.25 | 1996 | USA |
| Anatidae | Dendrocygna autumnalis | Finlay and Jeske23 | 1997 | USA |
| Gruidae | Antigone canadensis tabida | Hayes et al.21) Businga et al.18 | 2003 2007 | USA USA |
| Phasianidae | Alectoris rufa | Höfle et al.19 | 2004 | Spain |
| Scolopacidae | Calidris tenuirostris | Rogers et al.22 | 2004 | Australia |
| Scolopacidae | Calidris canutus | Rogers et al.22 | 2004 | Australia |
| Scolopacidae | Calidris ruficollis | Rogers et al.22 | 2004 | Australia |
| Gruidae | Grus americana | Hanley et al.20 | 2005 | USA |
| Anatidae | Branta canadensis | Dunne and Miller24 | 2009 | USA |
| Scolopacidae | Limosa lapponica Limosa lapponica baueri | Rogers et al.22 Ward et al.1 | 2004 2011 | Australia New Zealand |
| Phoenicopteridae | Phoeniconaias minor | McEntire and Sánchez8 | 2017 | USA |
| Diomedeidae | Thalassarche chlororhynchos | Hurtado et al.17 | 2021 | Brazil |
Clinical signs associated with CM in wild birds
The wild birds with CM have diverse clinical signs, which were presented and classified according to the species that developed the condition. Most of the signs were associated with the musculoskeletal system (Table 3).
Table 3 Clinical signs according to the species and authors in the literature in this review
| Clinical signs | Species | Authors |
|---|---|---|
| Paresis |
|
|
| Paralysis |
|
|
| Ataxia |
|
|
| Inability to incorporate |
|
|
| Uncoordinated flight | Alectoris rufa | Höfle et al.19 |
| Tremors | Limosa lapponica baueri | Ward et al.1 |
| Lameness |
|
|
| Lethargy |
|
|
| Dehydration | Thalassarche chlororhynchos | Hurtado et al.17 |
| Low body condition | Thalassarche chlororhynchos | Hurtado et al.17 |
| Hypotermia | Thalassarche chlororhynchos | Hurtado et al.17 |
Diagnostics methods in wild birds with CM
Clinical pathology
According to the bibliography, different biochemical analyses were carried out in the bird species with CM. The initial values of the cases of myopathy were recorded, indicating an increase in the levels of creatine kinase (CK) and serum aspartate aminotransferase (AST) enzymes in comparison with the reference values determined by different authors (Table 4).
Table 4 Biochemical analyzes as a diagnostic method for CM in wild birds of the reviewed literature
| Species | CK results (U/L) | AST results (U/L) | C reference values (U/L) | AST reference values (U/L) | Author(s) |
|---|---|---|---|---|---|
| Phoeniconaias minor | 96 019 | 4 349 | 130-1 143 | 105-415 | McEntire and Sánchez8 |
| Thalassarche chlororhynchos | 100 400 | 3 620 | 655.2 ± 21.8 | 117.6 ± 7.7 | Hurtado et al.17 |
| Alectoris rufa | 14 597 | ND* | 205-1 105 | ND* | Höfle et al.19 |
| Grus americana | 22 500 | 1 920 | 125-625 | 133-612 | Hanley et al.20 |
| Antigone canadensis tabida | 168 000 | 701 | 84-3 203 | 158-466 | Businga et al.18 |
| Dromaius novaehollandiae | ND* | 3 250 | ND* | 80-380 | Tully et al.25 |
| Dendrocygna autumnalis | 23 680 | 898 | 93-525 | 26-80 | Finlay and Jeske23 |
| Grus canadensis pulla | ND* | > 1 000 | ND* | 212-313 | Carpenter et al.(26 |
Creatine kinase (CK)
Aspartate aminotransferase (AST)
ND*: Not described in the literature.
Necropsy examination
Necropsy is another diagnostic method for CM, which is based on the post-mortem analysis of affected birds. Different types of samples were described, with the muscles of the pectoral area, gastrocnemius muscles, and muscles associated with the legs being the most affected. These muscles presented evident lesions, which are described in Table 5.
Table 5 Post-mortem findings as a diagnostic method for CM in wild birds
| Species | Samples | Macroscopic lesions | Microscopic lesions | Author(s) |
|---|---|---|---|---|
| Dendrocygna autumnalis | Gastrocnemius muscle | White muscle tissue (75 % of its structure) | Degeneration and necrosis of myofibers with mineralization and interstitial hypercellularit. Areas with dense fibrous connective tissue and tissue scarring. | Finlay and Jeske23 |
| Deep pectoral muscles | White lines near its insertion | |||
| Cardiac muscle | Very evident white lines | ND* | ||
| Grus canadensis pulla | Iliotibial, gastrocnemius, lateral and medial flexor cruris and femorotibial muscles | Pale muscle tissue with whitish lines, dull, soft and friable texture | Myofibers affected with hyaline, granular and floccular sarcoplasm with loss of striation, slightly mineralized and anucleated. Presence of necrotic myofibrils. | Carpenter et al.26 |
| Dromaius novaehollandiae Dromaius novaehollandiae | Hip abductor muscle | Presence of bleeding and muscle rupture Pale tissue with emphysema in fascia | Extensive necrosis of muscle fibers, with diffuse hyalinization. Granular degeneration, mild mineralization, macrophage infiltration, increased sarcoplasmic cell nuclei. | Tully et al.25 |
| Lateral cruris muscle | Pale tissue with emphysema in fascia | |||
| Kidney | Scattered foci of fibrosis and cortical interstitial change | Dilated tubules, with the presence of mineralization, tubular regeneration, fibrinous thrombi are observed in some veins and possibly an artery. | ||
| Alectoris rufa | Skeletal muscle& | Appearance of “cooked” muscle tissue with microhemorrhages | Degenerative changes in striated muscle fibers. | Höfle et al.19 |
| Grus americana | Pectoral muscles1 | Petechial and ecchymotic hemorrhages | Individual necrotic myofibrils, these lacked striation, were swollen with hyaline degeneration. | Hanley et al.20 |
| Leg muscles&1 | ||||
| Grus americana | Skeletal muscle& | Pale tissue with the presence of whitish lines | 70 - 90% of myofibers in skeletal muscle are necrotic and regenerative, with interspersed striations. Highly cellular areas, containing macrophages, myoblasts, and regenerative myofibers. The few remaining necrotic fibers are hyalinized or fragmented, but most were mineralized. Moderate disorganization and the presence of grouped nuclei in the center of the regenerative fibers. | Hanley et al.20 |
| Cardiac muscle | ND* | Prominent focus of mineralization. Fibroplasia in the left ventricle. | ||
| Grus americana | Leg muscles& | Diffusely pale and with pale lines | Almost 100% of myofibrils were affected in various muscles, necrotic fibers were more common and diffuse. Presence of mineralization of necrotic fibers. | Hanley et al.20 |
| Superficial and deep pectoral muscles | ||||
| Grus americana | Kidney | ND | Cytoplasmic granules are seen in the renal tubular epithelium and mild acute multifocal necrosis. | Hanley et al.20 |
ND* Not described in the literature, 1Fixation of the sample with 10 % buffered formalin, &The sample is not specifically described.
The most frequent macroscopic alterations were changes in the coloration of the muscle tissue and the presence of hemorrhagic areas due to the rupture of muscle fibers in the injured area. Microscopic lesions in skeletal muscle samples were associated with damage at the level of the muscle fibers, which may lead to necrosis and loss of the structural characteristics of the muscle tissue and organs (Table 5).
Treatments in wild birds with CM
Fluid therapy has been documented as a strategy for treating CM in birds. It involves giving the bird an electrolyte solution to improve its perfusion, helping to increase the intravascular volume. This can help to compensate for dehydration, treat hypotension and avoid metabolic acidosis, which are all physiological mechanisms that can contribute to myopathy. Lactated Ringer’s solution was the most widely used fluid in the studies analyzed in this review and the most used anti-inflammatory drug was dexamethasone (Table 6).
Table 6 Treatments used for CM in wild birds
| Drugs | Dosage | Route of administration | Interval | Author(s) | |
|---|---|---|---|---|---|
| Solutions for infusion | Lactated ringer’s solution | 10 mL/kg | EV, SC | TID | McEntire and Sánchez8 |
| ND | EV, SC | BID | Hurtado et al.(17) | ||
| ND | VO | SID | Hanley et al.20 | ||
| 1.5 L | EV | BID | Tully et al.25 | ||
| 60 mL | EV | BID | Dunne and Miller24 | ||
| Sodium chloride solution (0.9 % NaCl) | 50 mL/kg | EV | Maintenance in 48 hours | Ward et al.1 | |
| Steroidal anti-inflammatory | Dexamethasone | 2 mg/kg | IM, SC | SID | Hanley et al.(20 |
| 1-2 mg/kg | SC | BID | Businga et al.18 | ||
| 0.5 mg/kg | IM | TID | Tully et al.(25 | ||
| 0.27 mg/kg | IM | ND | Carpenter et al.26 | ||
| Non-steroidal anti- inflammatory (NSAIDs) | Ketoprofen | 1 mg/kg | IM | SID | Hanley et al.(20 |
| Meloxicam | 0.1 mg/kg | VO | SID | Ward et al.1 | |
| Piroxicam | 0.5 mg/kg | VO | SID | Hanley et al.(20 | |
| Carprofen | 2.5 mg/kg | VO | BID | Hanley et al.(20 | |
| Supplements | Vitamin supplements | 0.8 mL/kg | VO | SID | Hurtado et al.(17 |
| Vitamin E | 0.1 mg/kg | IM | SID | Hurtado et al.17 | |
| 0.1 mg/kg | IM | SID | Hanley et al.(20 | ||
| 0.06 mg/kg | IM | SID | Businga et al.(18 | ||
| Vitamin B12 | 0.25 mg/kg | IM | SID | Hurtado et al.(17 | |
| 1 mg/kg | IM | SID | Hanley et al.(20 | ||
| Selenium | 0.1 mg/kg | IM | SID | Hurtado et al.18 | |
| 0.1 mg/kg | IM | ND* | Hanley et al.(20 | ||
| 0.06 mg/kg | IM | SID | Businga et al.(18 | ||
| 200 UI | VO | SID | Dunne and Miller24 | ||
| Central nervous system depressants | Haloperidol | 0.2 mg/kg | VO | BID | McEntire and Sánchez8 |
| Diazepam | 0.2 mg/kg | VO | SID | Hurtado et al.(17 | |
| Midazolam | 1.5 mg/kg | IM | BID | Ward et al.(1 | |
| Methocarbamol | 10 mg/kg | VO | SID | McEntire and Sánchez8 | |
| 50 mg | VO | SID | Dunne and Miller24 | ||
| pH control | Sodium bicarbonate | 42 mg/kg | EV | Single dose | Hanley et al.(20 |
| 6 mEq/kg | EV | Repeat at 3 hours | Tully et al.25 |
ND*: Not described in the literature.
Vitamin E and selenium were the most used antioxidants as prophylaxis to protect against or for the treatment of CM. The authors noted that vitamins and minerals help to reduce free radicals in muscle tissues. The free radicals are produced by oxidative stress, which plays a central role in the pathophysiology of the disease. As shown in Table 6, vitamin E was one of the most frequently employed vitamins for CM, and it was administered in conjunction with selenium.
The authors highlighted that central nervous system (CNS) depressant drugs help the bird to keep calm enough to be handled and fed regularly. Methocarbamol was one of the most commonly CNS depressant agents for treating CM. This drug (methocarbamol) acts as a muscle relaxant when the signs of myopathy are not exacerbated and improves the functional regeneration of the injured muscle. Haloperidol, diazepam, and midazolam were some of the most frequent agents for reducing the physiological stress of wild birds upon capture and examination in the veterinary setting (Table 6). The intravenous administration of sodium bicarbonate was one of the most commonly used therapies for treating metabolic acidosis in wild birds with CM (Table 6). This is because sodium bicarbonate can help to raise the low pH from lactic acid accumulation.
Some authors noted that physical rehabilitation plays a central role in treating CM by restoring muscle strength, coordination, and function. The different types of treatments and physiotherapeutic methods performed in the literature are described in Table 7, along with their duration and execution intervals. One of the methods described involved keeping the birds in a small cage, preventing them from flapping or walking, thus decreasing their muscular activity. Other approaches consisted of a sling that can help to support the bird and prevent it from further injuries, facilitating the management for feeding and the application of appropriate treatments. Rogers et al.27) illustrated the making of this implement, which allows the birds to support their weight. However, legs, tails, wingtips, and cloaca must protrude through holes to prevent the bird from flapping or dirtying its sling.
Table 7 Rehabilitation treatments for capture myopathy carried out during the recovery process of wild birds
| Treatment | Therapy duration | Interval | Author(s)/year |
|---|---|---|---|
| Sling |
|
|
|
| Cage rest |
|
|
|
ND*: Not described in the literature.
Physiotherapy is a nonpharmacological therapeutic strategy to strengthen and condition muscles in birds with CM. Table 8 summarizes the different techniques used by the authors, along with the duration of each therapy.
Table 8 Physiotherapy performed during the recovery process from capture myopathy in wild birds
| Physiotherapy | Therapy duration | Interval | Authors/year |
|---|---|---|---|
| Standing and assisted walking | 10-15 minutes | BID | Hanley et al.(20 |
| Assisted walking and passive movements in the lower extremities | 15 minutes | SID | Hanley et al.(20 |
| Assisted walking, hock support and passive leg movement | 1-2 hours | SID | Businga et al.(18 |
| Muscle massage with passive flexion and extension movements | 5 minutes | BID | Dunne and Miller24 |
| Standing exercise Swimming session | 30 minutes 30 minutes | QID BID | McEntire and Sánchez8 |
Discussion
CM accounts with a considerable frequency of deaths associated with wildlife, which have considerable impacts on conservation efforts.4 The present study was aimed at analyzing, organizing, and synthesizing relevant literature information about CM in wild birds. The data collected was then used to describe the clinical signs associated with CM, diagnostic biochemical analyses, and postmortem examination. Finally, treatments applied in different studies were analyzed. The current number of studies examining CM in wild birds is limited. However, Hayes et al.21) observed that CM has been well-studied in mammals, suggesting that there is a need for further research on this disease in birds.
Taxonomic classification of birds suffering from CM (Table 2) revealed that certain families and species were overrepresented. The studies recovered in the literature were conducted in countries such as the United States and Australia, and there are not many studies on CM in Latin America, which does not mean that the disease is not present in this region. In this regard, McEntire and Sánchez.8) suggested that long-legged wading birds are especially susceptible to CM, pointing to cases in different flamingo species, including the Chilean flamingo (Phoenicopterus chilensis).
The studies included in this review found that the most common clinical signs of CM in wild birds were the inability to sit up, ataxia, paresis, and paralysis (Table 3). This is consistent with the findings of Ward et al.,1 who suggested that these clinical signs are associated with reduced muscle function as a result of the physiological cascade of CM initiated by hyperthermia, anaerobic glycolysis, metabolic acidosis, decreased tissue perfusion, and hypoxia.12 The antemortem diagnosis of CM in wild birds was performed by assessing the clinical history, clinical signs, and concentration of muscle enzymes. As shown in Table 4, CK and AST levels have been traditionally used to diagnose muscle disorders or injuries.
Hanley et al.20 indicated that although the biochemical evaluation of muscle enzymes is useful for diagnosis, it does not seem valuable for prognosis, as enzyme values can take weeks to return to normal reference ranges. The physiological cascade of CM can be initiated by increased muscle contractions and lactic acid buildup during the capture, a process that ultimately leads to cell lysis and the release of CK and AST enzymes into the blood.28 Indeed, high levels of these enzymes in the blood have been suggested to be an index of muscle damage in mammals and birds.29 Consistent with this, biochemical analyses revealed increased CK and AST levels in the included species compared to physiological levels.
West et al.30 highlighted the importance of postmortem diagnosis by necropsy and histopathology in CM. The post-mortem signs of CM may include dark red, enlarged kidneys, and an empty or reddish-brown fluid in the bladder. The flexor and extensor muscles of the hindlimbs may show soft, pale, dry areas with central white foci. This is a sign of severe muscle necrosis, a hallmark of CM.31 Myonecrosis was also described as one of the main histopathological hallmarks in CM muscles. Subacute and chronic CM manifestations were distinguished by sarcolemma proliferation, muscle regeneration, and fibrosis.12,32
The treatment for CM is complex due to the multifactorial nature of the disease. It revolves around the early administration of fluid therapy, steroidal and nonsteroidal anti-inflammatory drugs, sodium bicarbonate, vitamin E, and selenium (Table 6). The use of vitamin E combined with selenium has been reported to help reduce free radicals from the muscle. For example, Paterson et al.32 reported an association between the survival of the Virginia quail (Colinus virginianus) population and the treatment with vitamin E and selenium after capture. Central nervous system depressants, such as diazepam, have also been used as support therapy in birds recovering from CM. Diazepam is a benzodiazepine drug that can minimize stress at low doses.
Hurtado et al.17 reported that benzodiazepines are particularly useful during the last stages of rehabilitation in seabird species. Benzodiazepines can help to reduce physiological stress associated with the regurgitation process and self-inflicted injuries in the veterinary setting. Haloperidol, midazolam, and methocarbamol have been reported to have some effectiveness against myopathy in wild birds. McEntire and Sánchez8 highlighted the importance of using these tranquilizers to keep the bird calm enough to be handled and fed regularly with an esophageal tube during its recovery. The efficacy of these drugs is variable and depends on the dose administered and the bird species. Moreover, the success of the treatment will also depend on the initial care after the development of the disease and the intensive support during the recovery phase.
A treatment protocol for wild birds admitted to wildlife rehabilitation centers is needed. This study proposes to develop a protocol focused on stress control, minimizing handling, and using CNS depressants to generate muscle relaxation and reduce stress in the bird while it recovers during the hospitalization phase. The course of the bird’s recovery should be managed with a combination of treatments, including treatment of the root cause of the pathology, fluid therapy (e.g., lactated Ringer’s solution), anti-inflammatories and supplementation with vitamin E and selenium18,20,21,24. Cage rest, with a sling, may be important in myopathy therapies. This helps to immobilize the individual and prevent muscle contractions, which can help to support a successful recovery (Table 7). The use of physiotherapy (Table 8), compresses, flexion, and extension movements in the hind limbs can further help birds with CM to regain muscle mass and start the recovery process until animals are released.8,18,20,24
Conclusions
CM in wild birds is a complex disease from clinical, diagnostic, and therapeutic standpoints, with a multifactorial etiology that frequently occurs in clinical practice. Wildlife specialists must be aware of the complex nature of CM and the challenges of diagnosing and treating it. They should adopt a preventive approach, especially in birds of families Scolopacidae, Gruidae, and Anatidae, where the disease has been more frequently described. The clinical signs associated with CM are not always indicative of the disease. However, their identification is important for a correct diagnosis, which should be conducted by considering the clinical manifestation of CM and blood biochemical analyses.
The stress response of birds to capture and handling is a major factor in the development of CM. Therefore, reducing stress reactions could help to prevent the disease. Alternatively, the presence of enzymatic indicators in blood samples can be a useful diagnostic tool in clinical pathology to evidence whether an individual is suffering from the condition. In birds, postmortem diagnosis of CM can be performed by examining pectoral, gastrocnemius, and leg muscles. The most frequent macroscopic lesions are associated with changes in the coloration of the muscle tissue and the observation of hemorrhagic areas. Microscopic lesions can also be observed in skeletal muscle fibers, which can progress to necrosis and loss of the structural conformation of the muscle and organs.
The treatment for CM is complex and requires a comprehensive approach. It should include preventive measures, as well as different types of management approaches, drugs, and physiotherapy. The publicly available information on CM in wild birds is scarce, with most published data coming from the United States and Australia. There is limited research on CM in Latin America. Therefore, further national and/or local studies of this disease in Latin American countries should be undertaken. This systematic review provides a baseline of information for future research and as reference material for students, professionals, and wildlife rehabilitation centers.











 nueva página del texto (beta)
nueva página del texto (beta)