Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Veterinaria México OA
versión On-line ISSN 2448-6760
Veterinaria México OA vol.10 Ciudad de México 2023 Epub 10-Ene-2025
https://doi.org/10.22201/fmvz.24486760e.2023.1258
Original research articles
Potential of plant extracts to inhibit egg hatching and migration of Haemonchus contortus larvae
1Tecnológico Nacional de México, División de Estudio de Posgrado e Investigación/I. T. de Conkal. Yucatán, México
2Universidad Autónoma de Yucatán. Facultad de Medicina Veterinaria y Zootecnia, CCBA. Mérida, Yucatán, México
3Universidad Autónoma de Ciudad Juárez. Departamento de Ciencias Veterinarias, Ciudad Juárez, Chihuahua, México
Gastrointestinal parasitosis constitutes important health problems, worsening over the years, due to inadequate use of drugs for its control, causing anthelmintic resistance. The objective of the present study was to evaluate the potential of Azadirachta indica (AZA) and Moringa oleifera (MOR) extracts in inhibiting egg hatching and in vitro migration of Haemonchus contortus larvae. A completely randomized experimental design was carried out, where 15 treatments and a dimethyl sulfoxide negative control were evaluated: MOR75, MOR-50, MOR-25, MOR-12.5, and MOR-6.25 mg/mL; AZA-75, AZA-50, AZA-25, AZA-12.5, and AZA-6.25 mg/mL; and thiabendazole (TBZ) TBZ-200, TBZ-100, TBZ-40, TBZ-20, TBZ-10 µg/mL. The extracts did not inhibit egg hatching capacity; however, we observed a complete decrease in motility in L1 larvae. The extracts affected larval migration (P < 0.0020) compared with controls (dimethyl sulfoxide and TBZ), with migration rate inhibition above 65 %. Probit analysis showed that the median effective concentrations were 60.41 and 65.69 mg/mL for M. oleifera and A. indica, respectively. The in vitro results suggest that the water extract of both plants has an anthelmintic action against gastrointestinal nematode larvae.
Keywords: Anthelmintic; Azadirachta indica; Haemonchus contortus; Moringa oleifera; ovine
Las parasitosis gastrointestinales constituyen importantes problemas de salud, se agravan con el paso de los años debido al uso inadecuado de medicamentos para su control y provocan resistencia a los antihelmínticos. El objetivo del presente estudio fue evaluar el potencial de los extractos de Azadirachta indica (AZA) y Moringa oleifera (MOR) para inhibir la eclosión de huevos y la migración in vitro de larvas de Haemonchus contortus. Se realizó un diseño experimental completamente al azar, donde se evaluaron 15 tratamientos y un control negativo de dimetilsulfóxido: MOR-75, MOR-50, MOR-25, MOR-12.5 y MOR-6.25 mg/mL; AZA-75, AZA-50, AZA-25, AZA12,5 y AZA-6,25 mg/mL; y tiabendazol (TBZ) TBZ-200, TBZ-100, TBZ-40, TBZ-20, TBZ-10 µg/mL. Los extractos no inhibieron la capacidad de eclosión de los huevos; sin embargo, se observó una disminución de la motilidad de hasta un 100 % en las larvas L1. Los extractos afectaron la migración larval (P < 0.0020) en comparación con los controles (dimetilsulfóxido y TBZ), con una inhibición de la tasa de migración superior al 65 %. El análisis Probit mostró que las concentraciones efectivas medianas fueron 60.41 y 65.69 mg/mL para M. oleífera y A. indica, respectivamente. Los resultados in vitro sugieren que el extracto acuoso de ambas plantas tiene acción antihelmíntica contra larvas de nematodos gastrointestinales.
Palabras clave: Antihelmíntico; Azadirachta indica; Haemonchus contortus; Moringa oleifera; Ovino
Study contribution
Gastrointestinal parasitosis constitutes important health problems, worsening over time, due to inadequate use of drugs for its control, causing anthelmintic resistance. One of the main problems affecting ovine production based on the grazing system is infestation primarily caused by gastrointestinal nematodes, which affects the majority of small ruminant farms worldwide. Gastrointestinal parasitosis has a greater impact on tropical and subtropical regions, where environmental conditions encourage the proliferation of parasites. This study evaluates addressing a current complex issue regarding the potential of plant extracts in inhibiting egg hatching in gastrointestinal nematode larvae. Aiming to minimize the use of chemical products and having a sustainable livestock production and contribute by providing solutions to the problem and help minimize the use of chemical products.
Introduction
In 2016, ovine production in Mexico was approximately 118 thousand tons, of which 60 300 tons were destined for ovine meat carcasses. Mexico only covers 70 % of national demand, the remaining 30 % is imported from Australia, New Zealand, and the United States of America; therefore, this production is considered of great importance in the context of development of the livestock industry, which provides food, raw materials, employment, and, most importantly, income for rural population, being the most dedicated to this activity.1 However, a primary issue impacting ovine production in grazing systems is infestation, primarily caused by gastrointestinal nematodes (GIN), affecting the majority of small ruminants livestock farms, not only in Mexico but worldwide.
Gastrointestinal parasitosis has a more significant impact on tropical and subtropical regions, where environmental conditions promote the proliferation of parasites.2,3 It is considered to have a high economic impact leading to a decrease in animal weight and, consequently increased vulnerability to diseases with potential rise in mortality.2 Gastrointestinal parasitosis poses a global health challenge due to inappropriate use of parasiticides, contributing to the development of anthelmintic resistance (AR).4
In some countries, Haemonchus contortus has been the parasite with the most negative impact; however, in recent years, Trichonstrogylus colubriformis has also gained importance.5,6 Other genera are also present including Nematodirus, Oesophagostomum, Cooperia, Strongyloides, Teladorsagia, Chabertia, Bunostomum, Trichuris, and Dictyocaulus.7 It has been demonstrated that blood parasites from Haemonchus contortus have a profound negative impact on animals causing an approximate blood decrease of 0.05 mL per adult nematode per day.
A parasitic load of 5 000 adult nematodes in a host represents a daily blood decrease of 250 mL in sheep, leading to anemia, anorexia, and health problems. This, in turn, results in decreased productivity indicators such as daily weight gain, final weight at slaughter, and carcass weight, and an increase in feed conversion. These factors contribute to the economic inefficiency of livestock grazing systems, leading to discouragement and neglect of activity.8,9
In an effort to of minimize AR, various alternative methods have been explored to address GIN in sheep, particularly Haemonchus contortus. Among the most notable approaches is the utilization of plants with anthelmintic potential, providing a biological control strategy without inducing anthelmintic resistance. This method aims to reduce gastrointestinal larvae by leveraging, secondary metabolites, such as: saponins, tannins, and alkaloids, known for their inhibitory effects on egg hatching and larval migration.10,11
In this context, there are reports indicating that neem leaves (Azadirachta indica) yield effective compounds, particularly against endoparasites.12 It has also been observed that the use of A. indica extract inhibits more than 80 % of egg hatching and disrupts the development of larvae in more than 70 % of cases.13 Conversely, sterilized water extract from moringa leaves (Moringa oleifera) has exhibited antibacterial effects, and the alcoholic seed extract has demonstrated antifungal properties of against phytopathogenic fungi.4,14
The utilization of aqueous extracts of these plants (A. indica and M. oleifera) in ovine production systems could impede the development of parasite larvae, making it crucial to evaluate their potential positive impact. Furthermore, these alternatives are accessible to producers as they are more cost-effective than drugs can be utilized as dietary supplements for animals. The present study assessed the inhibitory activity of Azadirachta indica and Moringa oleifera extracts on egg hatching and in vitro larval migration of Haemonchus contortus.
Materials and methods
Ethical statement
All experimental procedures were approved by the Animal Experimentation Ethics Committee of the Autonomous University of Ciudad Juarez, Department of Veterinary Sciences (Resolution CEI-2023-2-980).
Area of study and collection of tree leaf
The research was conducted at the Laboratorio de Fisiología Digestiva of the Instituto Tecnológico de Conkal, situated at km 16.3 of the Antigua Carretera Mérida-Motul, Conkal, México, sited at 21°05’ N and 89°32’ W. The location features an AWo subhumid climate at an elevation of 7 m above sea level, an average annual temperature of 26 °C, and annual rainfall of 900 mm.15 Leaves from A. indica and M. oleifera were harvested in the months of April and May, during morning hours within the schedule of experimental areas of the Institute.
Collection of Parasitological material
A 4-month-old male ovine, weighing 20 kg, previously dewormed with albendazole and ivermectin, was utilized for the study. The animal underwent a 15-days confinement in a metabolic cage for diet adaptation (alfalfa pellets). Following a 10-day deworming period, fecal samples were collected every two days for a week to confirm the endoparasites including Haemonchus eggs and larvae.
Once the absence of eggs, larvae, and endoparasites was confirmed, especially Haemonchus, the animal was orally inoculated with 7 000 (L3) larvae of Haemonchus contortus (strain provided by the Facultad de Medicina Veterinaria y Zootecnia-UADY) using a 20 mL syringe (Plastipack™, México). After inoculation and pre-patent period (28 days), samples were collected for obtaining eggs for use in the assay.
Aqueous extract preparation
The leaves of each plant were subjected to a 48 hour drying process in a heating oven. Subsequently, they were ground to a particle size of 0.1 mm using a Wiley Mill (Thomas Scientific, Model 8455.6). Two stock solutions were prepared by combining 25 g of dried material (DM) with 200 mL of distilled water per plant. The resulting solution was agitated at 680 rpm using a Thermo Scientific™ agitator, Model 88880017) until complete dilution. Afterward, the solutions were refrigerated at 4 °C for 48 h, followed by filtration using gauze and a sieve with a mesh size of 38 µ. The preparation was labeled per plant as Neem stock solution (A. indica) and Moringa (M. oleifera), and various dilutions were subsequently created.16
Aqueous extract dilutions
Each original solution underwent dilution according to the following concentrations: Moringa extract at 75, 50, 25, 12.5, and 6.25 mg/mL. Neem extract at 75, 50, 25, 12.5, and 6.25 mg/mL. In addition, five dilutions of thiabendazole at concentrations of 200, 100, 40, 20 and 10 µg/mL serving as positive control and with dimethyl sulfoxide (DMSO) used as negative control (D4540-1L SIGMA©).
Assay 1
We employed the egg-hatch test (EHT), adapted from the method outlined by Marie-Magdeleine.17H. contortus eggs collected from the inoculated animal were distributed in 24-well cell plates (120 to 150 eggs/well, repeated three times) and exposed to various solutions of aqueous extracts. The plates were incubated for 48 h. Following incubation, an inverted microscope (WF10X-18 mM, VELAB®) with 10× magnification was utilized to count the number of eggs and larvae. The hatching percentage was determined using the following equation:
Assay 2
The larval migration test (LMT) was conducted by adapting the technique described by Demeler et al. (2010).17 Third-stage larvae (L3) or infective larvae in 24-well plates obtained from coproculture and exposed to various dilutions of aqueous extract. The well contents were observed using an inverted microscope (10× magnification). Subsequently, the number of migrated larvae was determined, and the percentage inhibition of larval migration (ILM) was calculated using the following equation:
Statistical analysis
We employed a completely randomized design consisting of three treatments and a negative control. The results were analyzed using PROC GLM of SAS Version 9.1.3 portable.18 Each well plate was considered as the experimental unit. Mean comparisons were conducted using Tukey’s test at a significance level of 0.05.19 Lethal concentrations (LC50, 99) were calculated using Probit through the Polo Plus program.20
Results
The secondary metabolites in both the leaves and stock solutions of Moringa oleifera and Azadiracht indica are shown in Table 1. According to variance analysis, the employed aqueous extracts did not significantly interfere (P = 0.0550) with egg hatching of Haemonchus contortus; however, they exhibited a notable effect on L1 motility, ranging from 98%-100 % (P = 0.0021).
Table 1 Chemical analysis of leaves and stock solution for secondary metabolites of Neem (Azadirachta indica) and Moringa (Moringa oleífera) (g/100 g)
| Analysis | Plant | Condensed tannins | Total tannins | Total phenols |
| Leaves | Neem | 1.00 | 0.46 | 1.84 |
| Moringa | 1.07 | 0.47 | 1.78 | |
| Stock | Neem | 0.25 | 0.115 | 0.46 |
| Solution (25 g of DM) | Moringa | 0.27 | 0.117 | 0.44 |
In the analysis, it was observed that aqueous extracts of MOR (90.17 %), AZA (89.70 %), and negative control DMSO (95.65 %) did not impact the egg hatching of H. contortus. Conversely, in TBZ group (38.81 %), there was a significant inhibition of egg hatching (P < 0.0025) when exposed to the different concentrations (Table 2), confirming the the ovicidal effect of this drug.
Table 2 Eggs and larvae of Haemonchus contortus according to treatment with aqueous extracts of plants (Mean ±SE)
| Treatment | Eggs | Larvae L1 |
| Dimethyl sulfoxide (DMSO) | 6.13 ± 4.14a | 109.58 ± 6.42a |
| Neem (Azadirachta indica) | 8.11 ± 4.47b | 105.05 ± 7.26b |
| Moringa (Moringa oleifera) | 7.80 ± 4.47b | 105.40 ± 7.50b |
| Thiabendazole | 36.21 ± 30.81c | 76.02 ± 31.35c |
a, b, c, d= different letters in the same column indicate differences (P < 0.05)
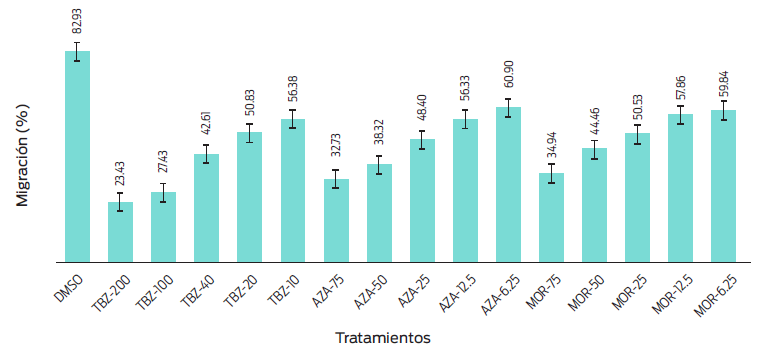
The aqueous extracts of AZA and MOR at each dilution did not exhibt a significant effect (P = 0.0550) on H. contortus egg hatching. In contrast, a dose-dependent response to TBZ was observed (P = 0.0010) (Figure 1). It was observed that aqueous extracts of MOR and AZA inhibited more than 95 % of first-stage larval motility (L1). The response of L1 to both plants was atypical of a dose-dependent reaction, as the lowest concentration achieved the lowest motility percentage (Figure 2). While the migration of first-stage larval (L1) decreased (P = 0.0031), the aqueous extract dilutions of the plants increased (Table 3 and Figure 3).
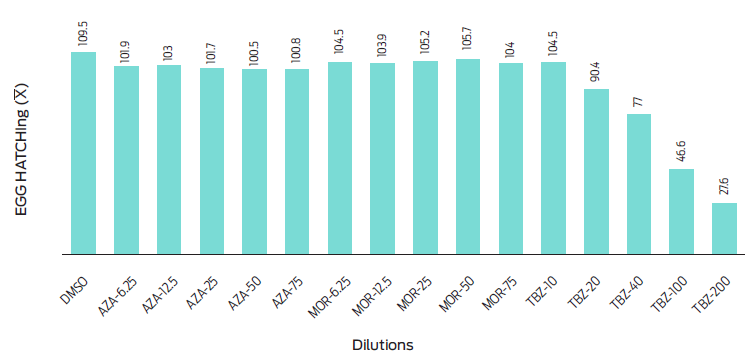
Figure 1 Egg hatching means in different extract dilutions of Azadirachta indica (AZA), Moringa oleifera (MOR), thiabendazole (TBZ), and dimethyl sulfoxide (DMSO) as negative controls.
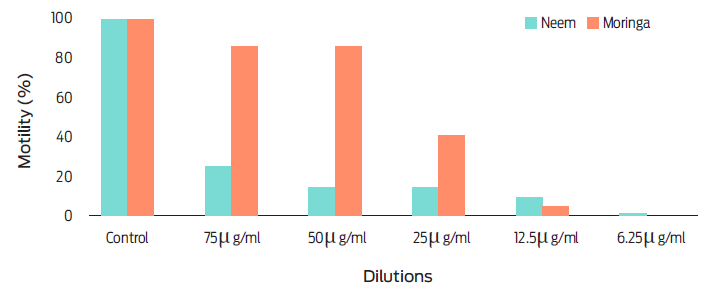
Figure 2 Motility of larvae (L1) exposed to different concentrations of aqueous extracts of Neem (Azadirachta indica) and Moringa (Moringa oleifera).
Table 3 Number of L3 larvae of Haemonchus contortus that migrated and did not migrate when exposed to different concentrations of aqueous extracts of Neem (Azadirachta indica) and Moringa (Moringa oleífera)
| Treatment | Dilution | Migration | No migration |
| Neem | 0 | 127.9 ± 9.9a | 26.5 ± 8.7e |
| Neem | 6.2 | 93.1 ± 14.1b | 59.7 ± 14.0d |
| Neem | 12.5 | 85.3 ± 17.1bc | 66.4 ± 18.4cd |
| Neem | 25 | 74.1 ± 14.5d | 79.3 ± 16.1b |
| Neem | 50 | 58.4 ± 7.7e | 94.0 ± 8.7a |
| Neem | 75 | 49.7 ± 8.6e | 102.4 ± 11.2a |
| Moringa | 0 | 126.4 ± 9.2a | 28.0 ± 9.9e |
| Moringa | 6.2 | 91.9 ± 14.8b | 61.5 ± 13.8d |
| Moringa | 12 | 88.0 ± 16.5b | 64.2 ± 17.0d |
| Moringa | 25 | 76.5 ± 14.2cd | 75.0 ± 15.1bc |
| Moringa | 50 | 67.9 ± 18.6d | 84.5 ± 18.0b |
| Moringa | 75 | 53.0 ± 21.6e | 99.2 ± 22.7a |
a, b, c, d, e= different letters in the same column indicate differences (P < 0.0550).
Discussion
In this study, we did not observe the inhibition of egg hatching when H. contortus eggs were exposed to aqueous extracts of A. indica and M. oleifera. However, a direct effect of the extracts on hatching to L1 was evident, indicating interference with the parasite’s motility. This effect is likely attributed to larvae ingesting the extracts containing metabolites such as tannins and phenols, causing damage to their intestine, which constitutes 80% of the parasite,21 thereby demonstrating inhibitory activity of the extracts.22-25
A study by Costa,26 utilized a low-dose (3.12 mg/mL) ethanolic extract of A. indica, achieving up to 97.7 % egg inhibition of H. contortus. They suggested that the ovicidal action may be associated with other secondary compounds like triterpenoids and condensed tannins. Our study aligns with these findings, as we observed a low percentage (2 %) of motile (L1) larvae when using a low-dose (6.25 mg/mL) extract of A. indica and M. oleifera.
In this context, the in vitro test involved the use of aqueous extracts to assess egg hatching and larval migration. Surprisingly, both Neem and Moringa plants exhibited an atypical response; they did not inhibit egg hatching, contrary to their larval migration. The presence of active principles interfered with the motilityof L1 larvae, incapacitating the parasite from migrating to the to the foliar part of the pastures, thus diminishing its infective capacity.
Similarly, studies conducted in Brazil12 demonstrated that the use of aqueous extracts from dried neem leaves, at a concentration of 240 000 ppm, interfered with the egg hatching capacity in feces cultures by 89 %, without differentiation between H. contortus and T. columbriformis. These effects were attributed to the presence of azadirachtin. An advantage of in vitro assays is the ability to the simultaneously explore various stages of parasite development, including validating eggs before hatching, larval development, cuticle shedding (molting), larval migration, and adult parasites, without interfering with the physiological functions of the hosts.27
In the larval migration test, the results indicated effects on the motility of L1 larvae for five different extract concentrations (Figure 3). Notably, this study found that DMSO did not interfere with (P = 0.0010) the migration capacity of L3 infective larvae, while thiabendazole exhibited migration (P = 0.0010). Thiabendazole interacts with tubulin, enhancing the formation of microtubules crucial in cell physiology and morphology: Benzimidazoles, including thiabendazole, bind to tubulin, impeding microtubules formation , disrupting parasite metabolism, hindering ATP formation, and ultimately leading to parasite death.28
It is crucial to emphasize the significant capacity (P = 0.0020) of aqueous extracts from A. indica and M. oleifera on larval migration (Table 3). This inhibitory effect on larval migration is attributed to the presence of secondary metabolites with anthelmintic properties, including condensed tannins,29 lectins,30 terpenoids,31 flavonoids.32 The affinity of phenolic compounds or tannins for glycoproteins (proline) on the parasite cuticle is noteworthy, as it binds to inhibit motility, larval development, feeding, reproduction, and ultimately leads to death. Additionally, the membranolytic action of saponins plays a role in this inhibitory process.33-35
In this study, we demonstrated a substantial inhibitory effect (> 60 %) on the migration of H. contortus larvae, attributed to the secondary compounds present in the evaluated plants. It´s noteworthy that the totality and these compounds are yet to be determined. Moreno36 assessed the in vitro impact of dry leaves on the migration of L3 infective larvae of Haemonchus contortus, Cooperia sp., Haemonchus placei, and Trichostrongylus columbriformis. Callitris endlicheri consistently hindered the migration of the studied nematodes. Specifically, the nematode species H. contortus exhibited a larval migration inhibition percentage of 88%-89.83at doses of 5 and 30 mg/mL, respectively. These in vitro studies suggest the presence of anthelmintic properties in some of the studied plant species.
In this study, water extracts of A. indica and M. oleifera exhibited an inhibitory effect on larval migration inhibition of H. contortus. Similar versatility has been reported in other studies, highlighting these plant’s interesting results in integrated pest control within the livestock sector.37,38 Specific investigations have demonstrated the nematocidal activity of plants,12,27 such as Gliricida sepium, where the inhibition of larval migration reached 95 % with the use of aqueous extracts of Gliricida sepium.39 Phytochemical screening of M. oleifera revealed the presence of alkaloids, glycosides, flavonoids, steroids, terpenoids, saponins, tannins, and anthraquinones.40 On the other hand, A. indica, is rich in terpenics (diterpens) and over fifty tetranortriterpenoids, notably azadirachtin which acts an antinutrient for insects, bacteria, and nematodes.41 Other studies focused on Gliricida sepium, a plant with diverse secondary metabolites, like saponins, tannins, and alkaloids,42 along with condensed tannin concentrations ranging from 3.6 %-4.6 %.43 These variations in ovicidal capacity and the inhibition of larval migration observed in different in vitro studies may be linked to the origin of the plant material.
The chemical composition of a plant can vary significantly even among plants of the same species duet he genetic and environmental differences, phenological state, harvesting process and drying methods, and extraction techniques, among other factors.44 Therefore, it is advisable to exercise strict control over qualitative material to be evaluated.45
In general, the types of solutions and extraction methods can significantly influence the activity of botanical compounds. This is evident with hydroalcoholic extracts, which may contain some nonpolar organic chemicals with higher polarity than aqueous extracts and have greater lipid solubility.46
Conclusions
Aqueous extracts of Azadirachta indica and Moringa oleifera did not impact egg hatching in H contortus. Nevertheless, as larvae progressed to L1, motility decreased significantly, ranging 98 % to 100 % with the lowest concentration of both extracts. In terms of larval migration, both extracts had exhibited detrimental effects on infective larvae (L3), resulting in an inhibition rate of 67 percent.
Acknowledgments
Special thanks to M.Sc. Fany Cen Chuc, Assistant of the Laboratorio de Fisiología Digestiva, for her support with laboratory analyses and advice on conducting this research.
Funding statement
Financial support was provided by the Dirección General de Educación Superior Tecnológica (DGEST) for the development of this research (5307.14-P).
Conflicts of interest
The authors declare no conflict of interest.
Author contributions
Conceptualization: E Aguilar-Urquizo.
Formal analysis: JFJ Torres-Acosta, E Aguilar-Urquizo.
Investigation: E Marcín-Marrufo.
Methodology: JFJ Torres-Acosta, E Aguilar-Urquizo.
Supervision: M Magaña-Magaña, AT Piñeiro-Vázquez, E Aguilar-Urquiso.
Validation: JFJ Torres-Acosta, E Aguilar-Urquizo.
Writing-original draft: E Marcín-Marrufo
Writing-review and editing: M Itza-Ortiz, E Aguilar-Urquizo.
REFERENCES
1. Sagarpa. SAGARPA. La ovinocultura, una actividad muy arropadora. Blog/agricultura: https://www.gob.mx/agricultura/es/articulos/la-ovinocultura-una-actividad-muy-arropadora [ Links ]
2. Torres-Acosta, JFJ, Hoste, H. Alternative or improved methods to limit gastro-intestinal parasitism in grazing sheep and goats. Small Ruminant Research. 2008;77:159-173. doi: 10.1016/j.smallrumres.2008.03.009. [ Links ]
3. Arece, J, Rodríguez, JG. Dinámica de las larvas infestantes de estrongílidos gastrointestinales en ovinos en pastoreo. Pastos y Forrajes. 2010;33(1):1-17. [ Links ]
4. Gilleard JS. Understanding anthelmintic resistance: the need for genomics and genetics. International Journal for Parasitology. 2006;36: 1227-1239. doi: 10.1016/j. ijpara.2006.06.010. [ Links ]
5. Rojas, N, Arias, M, Arece, J, Carrión, M, Pérez, K, Valerino, P. Identificación de Trichostrongylus colubriformis y Oesophagostomun columbianum en caprinos del valle del Cauto en Granma. Revista Salud Animal. 2011;33(2):116-120. [ Links ]
6. López-Ruvacalba, OA, González, GR, Osorio, AMM, Aranda, IE, Díaz, RP. Cargas y especies prevalentes de nematodos gastrointestinales en ovinos de pelo destinados al abasto. Revista Mexicana Ciencias Pecuarias. 2013;4(2):223-234. [ Links ]
7. Naem, S, Gorgani, T. Gastrointestinal parasitic infection of slaughtered sheep (Zel breed) in Fereidoonkenar city, Iran. Veterinary Research Forum, 2011;2(4):238-241. [ Links ]
8. Cordero, M, Rojo, FA. Parasitología Veterinaria. Madrid: McGraw-Hill Interamericana; 1999. [ Links ]
9. Fthenakis, GC, Papadopoulos, E. Impact of parasitism in goat productions. Small Ruminant Research. 2018;163:21-23. doi: 10.1016/j.smallrumres.2017.04.001. [ Links ]
10. Larsen M. Biological control of nematode parasites in sheep. Journal of Animal Science. 2006;84:133. doi: 10.2527/2006.8413_supple133x. [ Links ]
11. Marie-Magdeleine, C, Mahieu, M, D’Alexis, S, Philibert, L, Archimede, H. In vitro effects of Tabernaemontana citrifolia extracts on Haemonchus contortus. Research Veterinary Science. 2010;89(1):88-92. doi: 10.1016/j.rvsc.2010.01.002. [ Links ]
12. Chagas, AC, Vieira, L. Ação de Azadirachta indica (Neem) em nematódeos gastrintestinais de caprinos. Brazilian Journal of Veterinary Research and Animal Science. 2007;44(1):49-55. doi: 10.11606/issn.1678-4456.bjvras.2007.26661. [ Links ]
13. Barrabí-Puerta, M, Arece-García, J. Actividad antihelmíntica in vitro de extracto acuoso de hojas y semillas de Neem (Azadirachta indica A. Juss), I. Inhibición de la eclosión y del desarrollo larvario. Revista de Salud Animal. 2013;35(2):103-108. [ Links ]
14. Mughal, MH, Ali, G, Srivastava, PS, Iqbal, M. Improvement of drumstick (Moringa pterygosperma Gaertn) a unique source of food and medicine through tissue culture. Hamdard Medicus. 1999;42:37-42. [ Links ]
15. Flores, JS, Espejel, I. Tipos de vegetación de la Península de Yucatán. Etnoflora Yucatanense, Fascículo 3. Mérida, México: Universidad Autónoma de Yucatán; 1994. [ Links ]
16. Valderrabano, J, Uriarte, J. Efecto ovicida y larvicida de ciertos extractos vegetales, ITEA. IX Jornadas sobre Producción Animal. 2001;22(1):179-181. [ Links ]
17. Demeler, J, Kuttler, U, Samson-Himmelstjerna, G. Adaptation and evaluation of three different in vitro tests for the detection of resistance to anthelmintics in gastrointestinal nematodes of cattle. Veterinary Parasitology. 2010;170:61-70. doi: 10.1016/j.vetpar.2010.01.032. [ Links ]
18. SAS. SAS/STAT User’s guide (Version 9.1.3). Cary, NC: SAS Institute Inc; 2013. [ Links ]
19. Mendenhall WRJ. Introduction to linear model and the design and analysis of experiments. In: W Mendenhall, editor. Introduction to Probability and Statistics. Belmont, Duxbury: Brooks/Cole, Cengage Learning; 1994; 244-251. [ Links ]
20. LeOra-Software. Polo Plus Free. 2004. https://immense-ravine-20905.herokuapp.com/Leora-Software-Polo-Plus-Free-Do.pdf [ Links ]
21. Lee D. The Biology of Nematodes. Boca Raton, Florida, USA: CRC Press; 2002. pp. 1-60. [ Links ]
22. Kamaraj, C, Rahuman, AA. Efficacy of anthelmintic properties of medicinal plant extracts against Haemonchus contortus. Research in Veterinary Science. 2011;91(3):400-404. doi: 10.1016/j.rvsc.2010.09.018. [ Links ]
23. Carvalho, CO, Chagas, ACS, Cotinguiba, F, Furlan, M, Brito, LG, Chaves, FCM, Stephan, MP, Bizzo, HR, Amarante, AFT. The anthelmintic effect of plant extracts on Haemonchus contortus and Strongyloides venezuelensis. Veterinary Parasitology. 2012;183(34):260-268. doi: 10.1016/j.vetpar.2011.07.051. [ Links ]
24. Rajagopal, PL, Premaletha, K, Kiron, SS, Sreejith, KR. Phytochemical and pharmacological review on Cassia fistula Linn. “The Golden Shower”. International Journal of Pharmaceutical Chemical & Biological Sciences. 2013;3(3):672-679. [ Links ]
25. Hernández-Alvarado, J, Zaragoza-Bastida, A, López-Rodríguez, G, Peláez-Acero, A, Olmedo-Juárez, A, Rivero-Perez, N. Actividad antibacteriana y sobre nematodos gastrointestinales de metabolitos secundarios vegetales: enfoque en Medicina Veterinaria. Abanico Veterinario. 2018;8:14-27. doi: 10.21929/abavet2018.81.1. [ Links ]
26. Costa, CTC, Bevilaqua, CML, Camurça-Vasconcelos, AL, Maciel, MV, Morais, SM, Castor, CMS, Braga, RR, Oliveira, LMB. In vitro ovicidal and larvicidal activity of Azadirachta indica extracts on Haemonchus contortus. Small Ruminant Research. 2008;74:284-287. doi: 10.1016/j.smallrumres.2007.09.003. [ Links ]
27. Githiori, JB, Athanasiadou, S, Thamsborg, SM. Use of plants in novel approaches for control of gastrointestinal helminths in livestock with emphasis on small ruminants. Veterinary Parasitology. 2006;139(4):308-320. doi: 10.1016/j.vetpar.2006.04.021. [ Links ]
28. Lubega, GW, Prichard, RK. Interaction of benzimidazole anthelmintics with Haemonchus contortus tubulin: binding affinity and anthelmintic efficacy. Experimental Parasitology. 1991;73(2):203-13. doi: 10.1016/0014-4894(91)90023-p. [ Links ]
29. Nguyen, TM, Binh, DV, Kyle, DJ. Effect of foliages containing condensed tannins on gastrointestinal parasites. Animal Feed Science and Technology. 2005;121:77-87. doi: 10.1016/j.anifeedsci.2005.02.013. [ Links ]
30. Ríos de Álvarez, L, Jackson, F, Greer, A, Bartley, Y, Bartley, DJ, Grant, G, Huntley, JF. In vitro screening of plant lectins and tropical plant extracts for anthelmintic properties. Veterinary Parasitology. 2012;186(3-4):390-398. doi: 10.1016/j.vetpar.2011.11.004. [ Links ]
31. Molan, AL, Meagher, LP, Spencer, PA, Sivakumaran, S. Effect of flavan-3-ols on in vitro egg hatching, larval development and viability of infective larvae of Trichostrongylus colubriformis. International Journal for Parasitology . 2003;33(14):1691-1698. doi: 10.1016/s0020-7519(03)00207-8. [ Links ]
32. Ademola, IO, Akanbi, AI, Idowu, SO. Comparative nematocidal activity of chromatographic fractions of Leucaena leucocephala. Seed against gastrointestinal sheep nematodes. Pharmaceutical Biology. 2005;43(7):599-604. doi: 10.1080/13880200500301761. [ Links ]
33. Alonso-Díaz, MA, Torres-Acosta, JFJ, Sandoval-Castro, CA, Aguilar Caballero, AJ, Hoste, H. In vitro larval migration and kinetics of exsheathment of Haemonchus contortus exposed to four tropical tanniniferous plant extracts. Veterinary Parasitology . 2008;153:313-319. doi: 10.1016/j.vetpar.2008.01.042. [ Links ]
34. Rodríguez-Molano, CE, Cely-Reatiga, Y, Gómez-Lara, DF. Efecto in vitro del extracto de Lotus corniculatus L. sobre nemátodos gastrointestinales bovinos. Revista Cubana de Plantas Medicinales. 2016;21(2):145-156. [ Links ]
35. Zaragoza-Bastida, A, Rodríguez-Salazar, E, Valladares-Carranza, B, Rivas-Jacobo, M, Herrera-Corredor, A, Rivero-Perez, N. Cassia fistula como tratamiento alternativo contra nematodos gastrointestinales de ovino. Abanico Veterinario. 2019;9:1-10. doi: 10.21929/abavet2019.92. [ Links ]
36. Moreno, FC, Gordon, IJ, Wright, AD, Benvenuti, MA, Saumell, CA. Efecto antihelmíntico in vitro de extractos de plantas sobre larvas infectantes de nematodos gastrointestinales de rumiantes. Archivos de Medicina Veterinaria. 2010;42(3):155-163. doi: 10.4067/S0301-732X2010000300006. [ Links ]
37. Giglioti, R, Forim, MR, Oliveira, HN, Chagas, ACS, Ferrezini, J, Brito, LG, Falcoski, TORS, Albuquerque, LG, Oliveira, MCS. In vitro acaricidal activity of neem (Azadirachta indica) seed extracts with known azadirachtin concentrations against Rhipicephalus microplus. Veterinary Parasitology . 2011;181(2-4):309-315. doi: 10.1016/j.vetpar.2011.03.053. [ Links ]
38. Ahmad, N, Ansari, MS, Hasan, F. Effects of neem based insecticides on Plutella xylostella (Linn.). Crop Protection. 2012;34:18-24. doi: 10.1016/j.cropro.2011.12.010. [ Links ]
39. Puerto-Abreu, M, Arece-García, J, López-Leyva, Y, Roche, Y, Molina, M, Sanavria, A, da Fonseca, AH. Efecto in vitro de extractos acuosos de Moringa oleifera y Gliricida sepium en el desarrollo de las fases exógenas de estrongílidos gastrointestinales de ovinos. Revista de Salud Animal . 2014;36(1):28-34. [ Links ]
40. Sinha SN. Phytochemical analysis and antibacterial potential of Moringa oleifera Lam. International Journal of Science Innovations and Discoveries. 2012;2(4):401-407. [ Links ]
41. INIFAP. El árbol de nim establecimiento y aprovechamiento en la huasteca potosina. Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias. Centro de Investigación Regional del Noreste. Folleto técnico Num. 3. San Luis Potosí, SLP, México; 2004. [ Links ]
42. García, DE, Medina, MG. Composición química, metabolitos secundarios, valor nutritivo y aceptabilidad relativa de diez árboles forrajeros. Zootecnia Tropical. 2006;24(3):233-250. [ Links ]
43. Romero-Lara, CE, Palma-García, JM, López, J. Influencia del pastoreo en la concentración de fenoles totales y taninos condensados en Gliricidia sepium en el trópico seco. Livestock Research for Rural Development. 2000;12(34). [ Links ]
44. Cromm EM. Documenting and evaluating herbal remedies. Economy Botany. 1983;37(1):13-27. doi: 10.1007/BF02859302. [ Links ]
45. Hördegen, P, Hertzberg, J, Heilmann, J, Langhansd, W, Maurera, V. The anthelmintic efficacy of five plant products against gastrointestinal trichostrongylids in artificially infected lambs. Veterinary Parasitology . 2003;117(1-2):51-60. doi: 10.1016/j.vetpar.2003.07.027. [ Links ]
46. Eguale, T, Tilahun, G, Debella, A, Feleke, A, Makonnen, E. In vitro and in vivo anthelmintic activity of crude extracts of Coriandrum sativum against Haemonchus contirtus. Journal of Ethanopharmacology. 2007;110(3):428-33. doi: 10.1016/j.jep.2006.1. [ Links ]
Received: August 24, 2023; Accepted: August 30, 2023; Published: October 20, 2023











 texto en
texto en