Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Veterinaria México OA
versión On-line ISSN 2448-6760
Veterinaria México OA vol.10 Ciudad de México 2023 Epub 22-Oct-2024
https://doi.org/10.22201/fmvz.24486760e.2023.1195
Original Research Articles
Proteins from Mycobacterium bovis culture filtrate promote apoptosis independently of caspase activation in bovine macrophages
1 Universidad Nacional Autónoma de México, Facultad de Medicina Veterinaria y Zootecnia/Departamento de Microbiología e Inmunología, CDMX, México.
2 Universidad Nacional Autónoma de México, Instituto de Investigaciones Biomédicas/ Departamento de Inmunología, CDMX, México.
3 Universidad Nacional Autónoma de México, Facultad de Estudios Superiores Iztacala/ Unidad de morfología y función, Estado de México, México.
4 Universidad Nacional Autónoma de México, Instituto de Fisiología Celular/ Neuropatología molecular, CDMX, México.
Apoptosis is a normally occurring process characterized by morphological changes in the cell. The species of the Mycobacterium tuberculosis complex induce apoptosis. In bovines, Mycobacterium bovis and its protein extracts induce apoptosis independently of caspase activation. However, the identity of the proteins in the extracts is unknown. In this study, we used size-exclusion chromatography to separate protein fractions from the culture filtrate. We obtained protein fractions that induced DNA fragmentation in bovine macrophages independently of caspase-3 activation. Liquid chromatography-mass spectrometry (LC-MS) identified three candidates responsible for the biological activity: MPB70, MPB83, and 60 kDa chaperonin.
Keywords: Mycobacterium tuberculosis; Apoptosis; Protein fractions; Bovine macrophages; Caspase-3
La apoptosis es un proceso que ocurre de manera normal en los tejidos. Se caracteriza por cambios morfológicos en la célula. Las especies del complejo Mycobacterium tuberculosis inducen este fenómeno; en el modelo bovino, se demostró que tanto Mycobacterium bovis como sus extractos proteicos, inducen apoptosis independientemente de la activación de las caspasas, sin embargo, la identidad de las proteínas en los extractos proteicos se desconoce. En este estudio reportamos que, a través de cromatografía de exclusión por peso molecular, se obtuvieron fracciones proteicas con la capacidad de inducir fragmentación del DNA en macrófagos de bovinos independientemente de la activación de la caspasa 3. Asimismo, mediante cromatografía líquida-espectrometría de masas (LC-MS) se identificó la presencia de tres candidatos responsables de la actividad biológica: MPB70, MPB83 y chaperonina de 60 kDa.
Palabras clave: Mycobacterium bovis; Apoptosis; Fracciones proteicas; Macrófagos bovinos; Caspasa 3
Study contribution
Mycobacterium tuberculosis complex species and their proteins induce apoptosis in different cells. Mycobacterium bovis and its protein extracts (culture filtrate and soluble extract) can induce this phenomenon independently of caspase-3 activation. However, the identity of the proteins in the protein extracts is unknown. In this study, we identified for the first time three candidates (MPB70, MPB83, and a 60 kDa chaperonin) responsible for inducing apoptosis in bovine macrophages.
Introduction
Apoptosis is a regulated cell death process that normally occurs during tissue growth and development.1 Morphologically, cells that die by apoptosis are characterized by cell shrinkage, chromatin condensation (pyknosis), nuclear fragmentation (karyor-rhexis), exposure of phosphatidylserine to the outer side of the cell membrane, and the formation of apoptotic bodies, which are vesicles containing cellular elements.1-3Mycobacterium bovis, which is part of the Mycobacterium tuberculosis complex, is the causative agent of bovine tuberculosis. Mycobacteria have a very complex cell wall composed mostly of lipids such as mycolic acids, phenolic glycolipids, Man-LAM, and among others; however, they also contain lipoproteins,4 which are potentially virulence factors that allow the immune system to evade and the survival of these microorganisms.
Pathogenic mycobacteria use different mechanisms for intracellular survival; for example, they inhibit phagosome acidification and phagosome-lysosome fusion5 and induce apoptosis.6Mycobacterium species such as M. tuberculosis, M. bovis, M. bovis BCG, and M. avium induce cell death in macrophages. Different studies have shown that the induction of apoptosis in human or murine macrophages depends on the virulence of the strain used for the infection. Furthermore, the proteins LpqH (19 kDa lipoprotein),7,8 ESAT-6, 9 (38 kDa lipoprotein),10 PE_PGRS33,11 MPT8312 of native or recombinant M. tuberculosis, produced in M. smegmatis and E. coli, can induce apoptosis in macrophages of human origin (WBC 264-9C, HEK 293, MDM, THP-1) and murine origin (RAW262.7, BMDM). This mechanism is a host defense against the microorganism, but the microorganism can also manipulate it to evade the immune response, spread, and infect other cells.13
Previous work in a bovine model identified for the first time that M. bovis can induce chromatin condensation and DNA fragmentation in macrophages depending on the multiplicity and time of infection.6 It was also demonstrated that both bacteria and protein extracts14,15 have the ability to induce chromatin condensation and DNA fragmentation independently of caspase activation in a process that involves Apoptosis-Inducing Factor (AIF),14 and Endonuclease G (Endo G) as the main molecules driving this phenomenon.16 However, the identity of the proteins involved in cell death is still unknown. Therefore, this study identified the proteins in culture filtrate (CFE) fractions that induce apoptosis in bovine macrophages.
The results of this study demonstrated that a 15-20 kDa protein fraction (fraction of interest, FI) derived from the CFE of M. bovis induced DNA fragmentation in macrophages. Using liquid chromatography-mass spectrometry (LC-MS), we identified the presence of MPB70, MPB83, and 60-kDa chaperonin in the FI. This finding suggests that these proteins are involved in the induction of apoptosis.
Materials and methods
Ethical statement
The procedures performed on the animals were approved by the Institutional Committee for the Care and Use of Experimental Animals of the Faculty of Veterinary Medicine and Animal Science (CICUAE-FMVZ-UNAM) under number JAGP-2002.
Culture of M. bovis AN5
Mycobacterium bovis AN5 was cultured on Sauton medium for eight weeks at 37 °C under static conditions. After incubation, the culture was processed to obtain the culture filtrate extract (CFE).
Obtaining proteins from M. bovis in Sauton medium
As described previously,15 CFE was obtained by filtering the culture medium using coarse-pore filter paper to separate it from the biomass. Then, the medium was filtered through 1.2 μm, 0.45 μm, and 0.22 μm pore membranes (Millipore, Bedford, MA). Once the CFE was bacteria-free, it was precipitated with ammonium sulfate at 70 % saturation under agitation at 4 °C for 12 h. Then, the CFE was centrifuged at 10 300 × g for 20 min, and protein precipitates were collected. Proteins were dialyzed with a MWCO 3 500 (Spectra/Por) dialysis membrane against distilled water for three days (two water changes per day) and PBS 1 X overnight. The CFE was stored at -20 °C for further analysis. The total protein content was quantified using bicinchoninic acid (BCA) and 12 % SDS-PAGE gels stained with Coomassie blue; the gels were prepared to visualize the proteins by loading 15 μg of protein per lane.
Size exclusion Protein liquid chromatography
CFE was fractionated using size-exclusion chromatography. A Hi Load 16/600 Superdex 75PG column (G&E) coupled to ÄKTA Pure equipment (GE Healtcare) was used. The buffer recommended by the manufacturer (NaPO4 0.05 M, NaCl 0.15 M, pH 7.2) with a flow rate of 0.8 mL/min was used to collect 1.5 mL fractions. The protein concentration was determined for each fraction using the BCA technique, and the protein pattern was visualized on 12 % polyacrylamide gels stained with Coomassie blue.
Bovine monocyte- derived macrophages
Monocytes were obtained from the peripheral blood of healthy cattle from a herd free of bovine tuberculosis according to a previously described method.6,14,15 Briefly, 60 mL of sample was collected by jugular puncture and stored with 3.5 mL of ACD anticoagulant per 30 mL of blood. Blood was centrifuged for 30 min at 1 000 × g at room temperature (RT). The leukoplatelet layer was separated and diluted 1:1 with PBS-Citrate (PBS-C), deposited on a gradient of Histopaque 1077 (Sigma-Aldrich, St. Louis, MO), and centrifuged at 1 00 × g for 20 min at RT.
The erythrocyte-free leukoplatelet layer was collected and washed three times with PBS-C at 500 × g for 10 min at 4 °C. Finally, cells were resuspended in RPMI 1640 medium (Sigma-Aldrich, St. Louis, MO) supplemented with 2 mM L-glutamine, 1 mM sodium pyruvate, and 0.1 mM non-essential amino acids (cRPMI) (Gibco BRL, Life Technologies, Grand Island, NY) without serum. The cells were incubated for 2 h at 37 °C and 5 % CO2 to promote cell attachment. The medium was changed by adding cRPMI 1640 with 12 % autologous serum. To differentiate the cells into macrophages, the cells were cultured in ultra-low attachment plates for 12 days at 37 °C and 5 % CO2, changing the medium every three days.
Incubation of macrophages with the fraction of interest
On ultralow-attachment plates, 1 × 106 macrophages were incubated for 16 h with 100 μg/mL FI, 25 μg/mL of camptothecin (Sigma Aldrich, MO) as a positive control, and unstimulated cells as a negative control under the same culture conditions. At the end of the incubation, the cells were collected and fixed with 1 % paraformaldehyde (PFA) for 15 min at 4 °C. Then, they were stored in 70 % ethanol at -20 °C until DNA fragmentation. For the caspase-3 activity test, the cells were processed at the end of the incubation period.
DNA fragmentation
DNA fragmentation was detected by TUNEL (Terminal deoxynucleotidyl Transferase Biotin-dUTP Nick End Labeling) using the commercial assay kit APO-BrdU TUNEL (Thermo Fisher Scientific, Waltham MA), as specified by the manufacturer. Cells contained in 70 % ethanol at -20 °C were washed twice and incubated with a DNA labeling solution (containing reaction buffer, TdT enzyme, BrdUTP, and distilled water) for 1 h at 37 °C, shaking every 15 min to keep the cells in suspension. The cells were washed and centrifuged twice at 300 × g for 5 min. Fluorescein-labeled anti-BrdU antibody was added to the sample and incubated for 30 min in the dark. Cell labeling was detected by flow cytometry using an ATTUNE NxT cytometer (Thermo Fisher Scientific, Waltham, MA), and at least 10 000 events were counted.
Caspase-3 activity test
A caspase-3 activity test was performed using FI. One million (1 × 106) macrophages were incubated in ultralow-attachment plates with 100 μg/mL FI, 25 μg/mL camptothecin (Sigma Aldrich, MO) as a positive control, and unstimulated cells as a negative control for 16 h at 37 °C and 5 % CO2. After 16 h, cells were washed with 1 mL of PBS 1 X, and 115 μL of lysis buffer were added (10 mM Tris-HCl, 10 mM NaH2PO4, 130 mM NaCl, and 1 % Triton X-100), and the sample was incubated for 15 min at 4 °C. Subsequently, 100 μL of reaction buffer (40 mM HEPES, 1 M DDT, and 10 mM AcDEVD-AM) was added and incubated for 5 min at RT. Finally, fluorescence was determined in the range of 380/430-460 nm for 20 min in a Synergy HT Bio-Tek multiple detection microplate reader.
Two-dimensional electrophoresis
FI was desalted with phosphate buffer + CHAPS for isoelectrofocusing. Sodium deoxycholate (DOC) 0.02 % was added to the sample, vortexed, and incubated for 15 min at RT. Then, 10 % trichloroacetic acid (TCA) was added and incubated for 1 h at RT. The sample was centrifuged for 10 min at 18 000 × g at 4 °C. The super-natant was decanted, and acetone (-20 °C) was added. The sample was vortexed and kept on ice for 15 min, shaking every 5 min. Subsequently, the sample was centrifuged again for 10 min at 18 000 × g at 4 °C. The supernatant was decanted, the remaining acetone was absorbed with filter paper, and the pellet was allowed to dry for 5 min to facilitate resuspension. The pellet was resuspended in 150 μL of rehydration buffer with 1.5 μL of dithiothreitol (DTT) 50 X and 1 μL of ampholytes for 1.5 h at 37 °C under constant agitation.
Rehydration of the strips (ReadyStrip IPG Strips, Biorad, pH 4-7 of 7 cm) was carried out passively for 16 h with 125 μL of rehydration buffer. Isoelectrofocusing was performed at 20 °C according to the following program: 100 V-169 150 V/h, 300 V-200 V/h, by gradient 1,000 V, 300 V/h, 5,000 V 4,500 V/h, and by steps 500 V-2 000 V/h in an IPGphor III equipment (GE). After focusing, the strips were stored at -70 °C until use. For the second dimension, the strips were equilibrated with equilibration buffer (BE), adding 20 µL of DTT 10X per 2 mL of buffer. Each strip was equilibrated with 1 mL of BE for 10 min at RT under constant agitation, decanted, and equilibrated again under the same conditions (1 mL BE/10 min). Before rehydration of the strips, 10 cm × 10.5 cm gels with a thickness of 1.5 mm were prepared at a concentration of 12 %. Finally, the gels were stained with Coomassie blue.
Liquid Chromatography - Mass Spectrometry
LC-MS was performed to identify proteins in the FI. The analysis was performed using a liquid chromatograph (Eksigent nanoLC 425) coupled to a mass spectrometer (AB Sciex TripleTOF 5 600+). The data-driven search was performed using AB Sciex ProteinPilot 5.0.1 Software.
Results
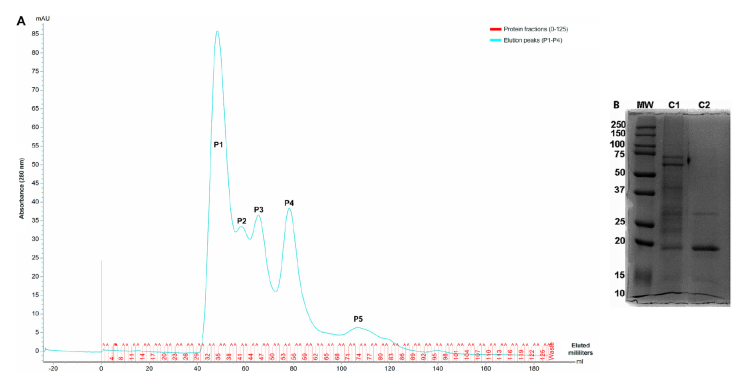
Fractionation of M. bovis AN5 proteins by liquid chromatography
CFE was fractionated using size- exclusion chromatography. Five detectable peaks were detected by absorbance at 280 nm (Figure 1A). The protein fractions obtained in peak four showed a similar electrophoretic profile; therefore, we pooled the fractions with molecular weights between 15 and 20 kDa, corresponding to fractions 51-60 FI. The CFE protein profiles were determined by 12 % SDS-PAGE and stained with Coomassie blue (Figure 1B).
DNA fragmentation and caspase-3 activity tests
DNA fragmentation in bovine macrophages was evaluated after 16 h of incubation with FI using the TUNEL technique. The percentage of DNA fragmentation of FI-incubated macrophages was 48.63 ± 9.03 %, and the percentage of fragmentation of the negative control was 7.30 ± 1.63 % (Figure 2A). Macrophage cell death occurred in the absence of caspase-3 activation (Figure 2 B).
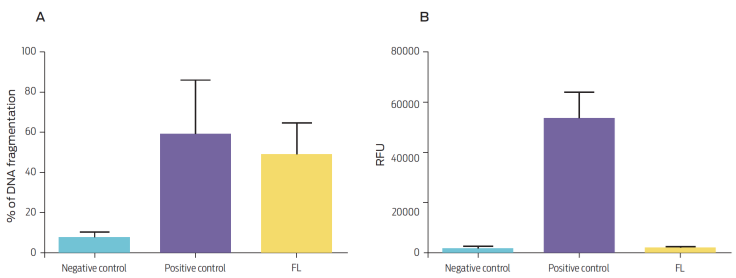
Figure 2 (A) Percentage of DNA fragmentation and (B) caspase-3 activity in bovine macrophages (106) that were either untreated (negative control), treated with camptothecin 20 μg/mL (positive control), or stimulated with 100 μg/mL FI. Results are expressed as (A) mean and standard error of three independent experiments (P = 0.1272, compared to the negative control) and (B) relative fluorescence units (RFU) of three independent experiments (P = 0.0001, compared to the negative control).
2D PAGE and LC-MS
The 2D PAGE analysis showed 25 protein spots distributed over molecular weights of 12-23 kDa; most proteins have an isoelectric point between 4.1 and 4.9 (Figure 3). Table 1 shows the proteins identified in the sequenced band.
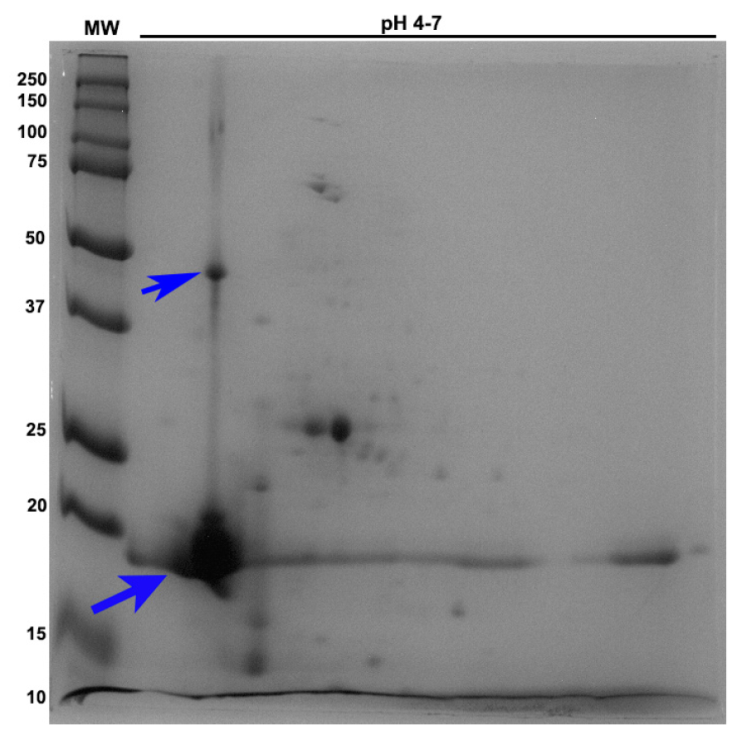
Figure 3 2D PAGE gel of FI of the CFE from M. bovis, corresponding to 15-20 kDa (12 % gel, pH 4-7). The arrow indicates the location of the proteins identified by LC-MS.
Table 1 Proteins identified from the FI of M. bovis CFE by LC-MS
| Protein | Gene | ID | Function/localization | Identity H37Rv | kDa | pI | Length (aa) |
|---|---|---|---|---|---|---|---|
| MPB70 | mpb70 | Mb2900 | Secretion protein. Cell surface lipoprotein. | 100 | 19.07 | 4.54 | 193 |
| MPB83 | mpb83 | Mb2898 | Member of the mpt70/mpt83 family. Secretion protein. Induces MMP9expression through TLR1/TLR2 stimulation in humans. | 100 | 22.07 | 4.73 | 220 |
| Chaperonin of60 kDa | groEl1 | Mb3451c | Involved in protein folding alongside cochaperonin GroES. Enables the encapsulation of non-native protein substrates and accelerates protein folding. | 100 | 56.72 | 4.74 | 539 |
ID: Identifier
pI: Isoelectric Point
Discussion
In this study, we separated the CFE of M. bovis using size exclusion FPLC. We found that a group of proteins with molecular weights between 15 and 20 kDa (FI) induced DNA fragmentation independently of caspase-3 activation. The LC-MS procedure identified the presence of MPB70, MPB83, and 60 kDa chaperonin. Characterization of the proteome and secretome of M. tuberculosis, M. bovis, M. bovis BCG, protein purified derivative (PPD), and culture filtrates under different conditions has identified the presence of highly antigenic proteins that can stimulate T lymphocytes. It has also been shown that the protein profile changes depending on the virulence and outcome of the disease in the host.17-25 2D PAGE and LC-MS have allowed the use of these proteins for developing more sensitive and specific diagnostic techniques and vaccines.18,23,26,27
Our results use the aforementioned techniques to propose possible protein candidates that induce bovine macrophage cell death in addition to their antigenic capacity. Using anion exchange chromatography, Alito et al. (2003)28 obtained 3-4 elution peaks that corresponded to proteins of different molecular weights; the effects of these proteins were measured in T lymphocytes. Similarly, our fractioning method was based solely on molecular weight, which allowed us to obtain a band of interest with a molecular weight between 15 and 20 kDa (FI).
Different investigations have shown that bacteria from the Mycobacterium tuberculosis complex can induce cell death through different pathways such as ferroptosis,29 caspase-dependent apoptosis,10 caspase-independent apoptosis,30 endoplasmic reticulum stress,31 and necroptosis32 in macrophages of human and mouse origin. Vega-Manríquez et al. (2007)14 demonstrated that incubating bovine macrophages with CFE for 16 and 24 h did not activate caspases 8, 9, and 3. This result was confirmed using the general caspase inhibitor Z-VAD-FMK. This study demonstrated that FI did not activate caspase-3, which is consistent with previous reports in bovine models.
The present information is important because some species of the Mycobacterium tuberculosis complex activate caspases when they induce apoptosis in macrophages from different origins.33 This result suggests that M. bovis attempts to interfere with apoptosis by inhibiting caspase activation. However, bovine macrophages activate an alternate apoptosis pathway independent of caspase activation. Finally, the induction of apoptosis contributes to control mycobacterial intracellular growth.16 MPB70 and MPB83 have been extensively studied in terms of their localization and the immune response they trigger in animals. Both are homologous proteins with 60 % identity at the amino acid level (MPB83 with respect to MPB70),34 but they are encoded by different genes.
These proteins present the FAS 1 or βIgH3 adhesion domain; therefore, it has been suggested that the interaction of this domain with proteins that have receptors for the same domain could explain the tropism of Mycobacterium toward certain tissues.35 Ju-Ock Nam et al., 200836 developed a polypeptide with a FAS 1 domain, which induced apoptosis in endothelial cells. Therefore, in addition to the idea that mycobacteria have a certain tropism toward some tissues due to this domain, the domain itself could participate in cell death signaling in macrophages.
Chaperonin 60 kDa is considered to be within the family of heat shock proteins. This protein can fold enzymes involved in mycolic acid synthesis.37 This protein aggregate is overexpressed under different stress conditions, including infections by microorganisms.38 The induction of apoptosis in PBMC by 60 kDa chaperonin has been reported in infections with Mycoplasma gallisepticum due to its interaction with annexin 2.39 In the case of Mycobacterium bovis, this is the first report of the induction of cell death in macrophages by this protein. However, the recombinant protein Hsp70 from M. tuberculosis induces 32.7 ± 11.63 % of DNA fragmentation in bovine macrophages.15
Heat shock proteins are associated with apoptosis induction by generating reactive oxygen species in mitochondria. There is evidence that a 60 kDa chaperonin together with a 10 kDa chaperonin are involved in caspase-3 activation signaling. These molecules have also been associated with Bax, which activates apoptosis. Our model showed that the 60 kDa chaperonin in bovine macrophages may activate apoptosis through a different mechanism.40 A limitation of the present study is that our results cannot clarify whether DNA fragmentation is due to the action of individual proteins or to the activity of the protein consortium. Incubation time should also be considered because the protein profiles of young and old cultures are different. Therefore, it is necessary to verify whether the proteins have shorter culture times in vitro and whether they participate in the pathogen-host interaction in vivo.
REFERENCES
1. Elmore, S. Apoptosis: a review of programmed cell death. Toxicologic Pathology. 2007;35(4):495-516. doi: 10.1080/01926230701320337. [ Links ]
2. Galluzzi, L., Vitale, I. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death & Differenciation. 2018;25:486-541. doi: 10.1038/s41418-017-0012-4. [ Links ]
3. Cascales Angosto, M. Bases moleculares de la apoptosis. Anales de la Real Academia Nacional de Farmacia. 2003;69(1):36-64. [ Links ]
4. Dulberger, CL., Rubin, EJ., Boutte, CC. The mycobacterial cell envelope-a moving target. Nature Reviews Microbiology. 2020;18:47-59. doi: 10.1038/ s41579-019- 0273-7. [ Links ]
5. Waters, WR., Hope, JC., Hamilton, CA., Palmer, MV., Mcnair, J., Skuce, RA., et al. Immunopathogenesis of Mycobacterium bovis infection of cattle. In: H. Mukundan, MA Chambers, WR Waters, MH Larsen, editors. Tuberculosis, Leprosy and Mycobacterial Diseases of Man and Animals: The many Hosts of Mycobacteria. Oxfordshire, UK, Boston, MA: MACABI; 2015. pp. 136-167. [ Links ]
6. Gutierrez-Pabello, JA., Mcmurray, DN., Admas, LG. Upregulation of thymosin β-10 by Mycobacterium bovis infection of bovine macrophages is associated with apoptosis. Infection and Immunity. 2002;70(4):2121-2127. doi: 10.1128/ IAI.70.4.2121-2127.2002. [ Links ]
7. Liu, L., Liu, J., Niu, G., Xu, Q., Chen, Q. Mycobacterium tuberculosis 19 kDa lipo-protein induces toll-like receptor 2-dependent peroxisome proliferator-activated receptor γ expression and promotes inflammatory responser in human macrophages. Molecular Medicine Reports. 2015;11(4):2921-2926. doi: 10.3892/ mmr.2014.3070. [ Links ]
8. Sánchez, A., Espinosa, P., García, T., Mancilla, R. The 19 kDa Mycobacterium tuberculosis lipoprotein (LpqH) induces macrophage apoptosis through extrinsic and intrinsic pathways: a role for the mithochondrial apoptosis-inducing factor. Clinical and Developmental Immunology. 2012:1-11. doi: 10.1155/2012/950503. [ Links ]
9. Derrick, SC., Morris, SL. The ESAT6 protein of Mycobacterium tuberculosis induces apoptosis of macrophages by activating caspase expression. Cellular Microbiology. 2007;9(6):1547-1555. doi: 10.1111/j.1462-5822.2007.00892.x. [ Links ]
10. Sanchez, A., Espinosa, P., Esparza, MA., Colon, M., Bernal, G., Mancilla, R. Mycobacterium tuberculosis 38-kDa lipoprotein is apoptogenic for human monocyte-derived macrophages. Scandinavian Journal of Immunology. 2009;69(1):20-28. doi: 10.1111/j.1365-3083.2008.02193.x. [ Links ]
11. Basu, S., Pathak, SK., Banerjee, A., Pathak, S., Bhattacharyya, A., Yang, Z., et al. Execution of macrophage apoptosis by PE_PGRS33 of Mycobacterium tuberculosis in mediated by toll-like receptor 2-dependent release of tumor necrosis factor-α. The Journal of Biological Chemestry. 2006;282(2):1039-1050. doi: 10.1074/jbc.M604379200. [ Links ]
12. Wang, L., Zuo, M., Chen, H., Liu, S., Wu, X., Cui, Z., et al. Mycobacterium tuberculosis lipoprotein MPT83 induces apoptosis of infected macrophages by activating the TLR2/p38/COX-2 signaling pathway. The Journal of Immunology. 2017;198(12):4772-780. doi: 10.4049/jimmunol.1700030. [ Links ]
13. Aguilo, JI., Alonso, H., Uranga, S., Marinova, D., Arbués, A., de Martino, A., et al. ESX-1-induced apoptosis in involved in cell-to-cell spread of Mycobacterium tuberculosis. Cellular Microbiology. 2013;15(12):1994-2005. doi: 10.1111/CMI.12169. [ Links ]
14. Vega-Manriquez, X., Lopez-Vidal, Y., Moran, J., Adams, LG., Gutiérrez-Pabello, JA. Apoptosis-inducing factor participation in bovine macrophage Mycobacterium bovis-induced caspase-independent cell death. Infecction and Immunity. 2007;75(3):1223-1228. doi: 10.1128/IAI.01047-06. [ Links ]
15. Rivera Maciel, A., Flores Villalva, S., Jiménez Vázquez, I., Catalan Bárcenas, O., Espitia Pinzón, C., Moran, J., et al. Mycobacterium tuberculosis and Mycobacterium bovis derived proteins induced caspase-independent apoptosis in bovine macrophages. Veterinaria México OA. 2019;6(1):1-12. doi: 10.22201/fmvz.24486760e.2019.1.560. [ Links ]
16. Benítez-Guzmán, A., Arriaga-Pizano, L., Morán, J., Gutiérrez-Pabello, JA. Endonuclease G takes part in AIF-mediated caspase-independent apoptosis in Mycobacterium bovis-infected bovine macrophages. Veterinary Research. 2018;49:69. doi: 10.1186/s13567-018-0567-1. [ Links ]
17. Nagai, S., Wiker, HG., Harboe, M., Kinomoto, M. Isolation and partial characterization of major protein antigens in the culture fluid of Mycobacterium tuberculosis. Infect Immun. 1991;59(1):372-382. doi: 0019-9567/91/010372-11$02.00. [ Links ]
18. Nagai, S., Matsumoto, J., Nagasuga, T. Specific skin-reactive protein from culture filtrate of Mycobacterium bovis BCG. Infect Immun. 1981;31(3):1152-1160. doi: 0019-9567/81/031152-09$02.00/0. [ Links ]
19. Harboe, M., Oettinger, T., Wiker, HG., Rosenkrands, I., Andersen, P. Evidence for occurrence of the ESAT-6 protein in Mycobacterium tuberculosis and virulent Mycobacterium bovis and for its absence in Mycobacterium bovis BCG. Infection and Immunity. 1996;64(1):16-22. doi: 0019-9567/96/$04.00+0. [ Links ]
20. Harboe, M., Wiker, HG., Ulvund, G., Lund-Pedersen, B., Andersen, AB., Hewinson, RG., et al. MPB70 and MPB83 as indicators of protein localization in mycobacterial cells. Infection and Immunity. 1998;66(1):289-296. doi: 0019-9567/98/$04.00+0. [ Links ]
21. Rosenkrands, I., King, A., Weldingh, K., Moniatte, M., Moertz, E., Andersen, P. Towards the proteome of Mycobacterium tuberculosis. Electrophoresis. 2000;21:3740-3756. doi: 0173-0835/00/1717-3740$17.50+.50/0. [ Links ]
22. Sabio, Y., García, J., Bigi, MM., Klepp, LI., García, EA., Blanco, FC., Bigi, F. Does Mycobacterium bovis persist in cattle in a non-replicative latent state as Mycobacterium tuberculosis in human beings? Veterinary Microbiology. 2020;247. doi: 10.1016/j.vetmic.2020.108758. [ Links ]
23. Assal, N., Rennie, B., Walrond, L., Cyr, T., Rohonczy, L., Lin, M. Proteome characterization of the culture supernatant of Mycobacterium bovis in different growth stages. Biochemestry and Biophysics Reports. 2021;28:1-4. doi: 10.1016/j.vetmic.2020.108758. [ Links ]
24. Rojas, M., Barrera, LF., Puzo, G., Garcia, LF. Differential induction of apoptosis by virulent Mycobacterium tuberculosis in resistant and susceptible murine macrophages: role of nitric oxide and mycobacterial products. The Journal of Immunology. 1997;159(3):1352-1361. doi: 159:1352-1361. [ Links ]
25. Albrethsen, J., Agner, J., Piersma, SR., Højrup, P., Pham, TV., Weldingh, K., et al. Proteomic profiling of Mycobacterium tuberculosis identifies nutrient-starvation- responsive toxin-antitoxin systems. Molecular & Cellular Proteomics. 2013;12(5):1180-1191. doi: 10.1074/MCP.M112.018846. [ Links ]
26. Forrellad, MA., Klepp, LI., Gioffré, A., Sabio y García, J., Morbidoni, HR., Santangelo, M de la P., et al. Virulence factors of the Mycobacterium tuberculosis complex. Virulence. 2013;4(1):3-66. doi: 10.4161/viru.22329. [ Links ]
27. Roperto, S., Varano, M., Russo, V., Lucà, R., Cagiola, M., Gaspari, M., et al. Proteomic analysis of protein purified derivative of Mycobacterium bovis. Journal of Translational Medicine. 2017;15:68. doi: 10.1186/s12967-017-1172-1. [ Links ]
28. Alito, A., McNair, J., Girvin, RM., Zumarraga, M., Bigi, F., Pollock, JM., et al. Identification of Mycobacterium bovis antigens by analysis of bovine T-cell responses after infection with a virulent strain. Brazilian Journal of Medical and Biological Research. 2003;36(11):1523-1531. doi: 10.1590/S0100-879X2003001100011. [ Links ]
29. Amaral, EP., Costa, DL., Namasivayam, S., Riteau, N., Kamenyeva, O., Mittereder, L., et al. A major role for ferroptosis in Mycobacterium tuberculosis-induced cell death and tissue necrosis. Journal of Experimental Medicine. 2019;216(3):556-70 doi: 10.1084/jem.20181776. [ Links ]
30. Bai, X., Kinney, WH., Su, WL., Bai, A., Ovrutsky, AR., Honda, JR., et al. Caspase-3-independent apoptotic pathways contribute to interleukin-32γ-mediated control of Mycobacterium tuberculosis infection in THP-1 cells. BioMedCentral Microbiology. 2015;15(1):1-9. doi: 10.1186/s12866-015-0366-z. [ Links ]
31. Cui, Y., Zhao, D., Sreevatsan, S., Liu, C., Yang, W., Song, Z., et al. Mycobacterium bovis Induces endoplasmic reticulum stress mediated-apoptosis by activating IRF3 in a murine macrophage cell line. Frontiers in Cellular and Infection Microbiology. 2016;6:1-12. doi: 10.3389/fcimb.2016.00182/full. [ Links ]
32. Yanti, B., Mulyadi, M., Amin, M., Harapan, H., Mertaniasih, NM., Soetjipto, S. The role of Mycobacterium tuberculosis complex species on apoptosis and necroptosis state of macrophages derived from active pulmonary tuberculosis patients. BioMed Central Research Notes. 2020;13:415. doi: 10.1186/s13104-020-05256-2. [ Links ]
33. Mohareer, K., Asalla, S., Banerjee, S. Cell death at the cross roads of host-pathogen interaction in Mycobacterium tuberculosis infection. Tuberculosis. 2018;113:99-121. doi: 10.1016/j.tube.2018.09.007. [ Links ]
34. Michell, SL., Whelan, AO., Wheeler, PR., Panico, M., Easton, RL., Etienne, AT., et al. The MPB83 antigen from Mycobacterium bovis contains O-linked mannose and (1 → 3)-mannobiose moieties. The Journal of Biological Chemistry. 2003;278(18):16423-16432. doi: 10.1074/jbc.M207959200. [ Links ]
35. Wiker, HG. MPB70 and MPB83-Major antigens of Mycobacterium bovis. Scandinavian Journal of Immunology. 2009;69(6):492-499. doi: 10.1111/J.1365-3083.2009.02256.X. [ Links ]
36. Ju-Ock, N., Jung-Eun, K., Ha-Won, J. Patent Application Publication. Pub. No.: US 2008/027.4955A1. 2008;1(19). [ Links ]
37. Colaco, CA., Macdougall, A. Mycobacterial chaperonins: the tail wags the dog. FEMS Microbiology Letters. 2014;350:20-24. doi: 10.1111/1574-6968.12276. [ Links ]
38. Qamra, R., Mande, SC. Crystal structure of the 65-kilodalton heat shock protein, chaperonin 60.2 of Mycobacterium tuberculosis. Journal of Bacteriology. 2004;186(23):8105-8113. doi: 10.1128/JB.186.23.8105-8113.2004. [ Links ]
39. Ying Yua, Lin Zhanga, Ying Chenb, Yuan Lia, Zhenzhong Wangc, Ganwu Lia, d, Gang Wanga J. X. GroEL protein (heat shock protein 60) of Mycoplasma gallisepticum induces apoptosis in host cells by interacting with annexin A2. Infection and Immunity. 2019;87(9). doi: 10.1128/IAI.00248-19. [ Links ]
40. Ikwegbue, PC., Masamba, P., Oyinloye, BE., Kappo, AP. Roles of heat shock proteins in apoptosis, oxidative stress, human inflammatory diseases, and cancer. Pharmaceuticals. 2018;11(1):1-18. doi: 10.3390/ph11010002. [ Links ]
Funding statement. This work was supported by grants PAPIIT IN220415 and CB-167488 from Universidad Nacional Autónoma de México and CONACYT (Consejo Nacional de Ciencia y Tecnología) respectively. The founding institutions played no role in the design and development of the project or paper manuscript.
Received: March 27, 2023; Accepted: July 26, 2023; Published: September 06, 2023











 texto en
texto en