Introduction
Testicular torsion is a surgical emergency that causes cessation of blood flow to the testis and surrounding structures due to the vertical rotation of the spermatic cord in newborns, children, and adolescents1. The incidence in men under the age of 25 is 1/40002. Early diagnosis and surgical intervention are required to prevent infertility and subfertility3. The diagnosis of testicular torsion should be made within the first 4-6 h. Especially if there is complete spermatic cord torsion, that is if there is a 720-degree rotation, it causes irreversible ischemic damage to the testis and loss of gonads4.
Ischemic damage during testicular torsion occurs due to oxidative stress and inflammation due to reactive oxygen species (ROS). The primary cause of testicular damage during the ischemic injury period is associated with decreased oxygen supply5. Tissue reperfusion should be provided as soon as possible to preserve the viability of ischemia-induced tissues. It is known that after detorsion of the testis, reperfusion also causes cellular damage, which is associated with an increase in ROS production and neutrophil infiltration6. The radicals formed in this process cause peroxidation of lipids in the cell membrane, denaturation of proteins, and DNA damage7.
In recent years, extensive research has sought effective strategies and drugs to minimize or prevent testicular ischemia/reperfusion (I/R) injury. However, other than trying to reduce the temperature of the scrotum, this is the only method that has been successfully applied in clinical practice8. Therefore, there is an urgent need to develop new therapeutic agents and a standard treatment protocol to reduce mortality and morbidity rates.
Exosomes are nanoscale cell-derived extracellular vesicles secreted from various cell types and found to contain bioactive components9. Exosomes, which can be obtained from saliva, urine, plasma, breast milk, and many other bodily fluids, can be used for therapeutic purposes, as they provide genetic material transfer, immune regulatory roles, and intercellular communication6,10,11. Exosomes are natural and, thanks to their very small size, can easily pass through anybody barrier. Considering these benefits, the therapeutic use of exosomes is inevitable12.
In recent studies, it has been proven that there are high concentrations of exosomes in breast milk, and they have been used in various experimental studies in the literature13,14. In this study, we aimed to investigate the protective effect of human breast milk exosomes against I/R damage in testicular torsion and their usability as a treatment modality.
Method
This study was carried out with the contributions of the Departments of Biochemistry and Pathology after being planned by the Department of Pediatric Surgery of Karadeniz Technical University (KTU) Faculty of Medicine, and the ethics committee approval with protocol number 2021/10 was obtained. The surgeries were performed in the KTU Faculty of Medicine Surgical Research Laboratory. Our work was supported by the Scientific Research Projects Coordination Unit.
Study Design
Three groups of 8 Sprague Dawley rats, which were divided randomly and each weighing 250-300 gr, were used in the study. The duration of the experiment was determined as 1 day. Until the study day, the rats were housed in cages with 8 or less sawdust under standard laboratory conditions at 22 ± 2 °C constant room temperature, 60% humidity, and 12 h light/12 h dark. Rats fasted for 24 h before surgery, and only water was given. In all surgical procedures, anesthesia was administered intraperitoneally, 10 mg/kg xylazine hydrochloride (Rompun, Bayer, Turkey) and 50 mg/kg ketamine (Ketalar, Pfizer, Turkey).
Surgical Technique: The operation area was shaved and cleaned with betadine solution. An incision made from the scrotal midline was used in the surgeries. Torsion/detorsion model; the left testis was taken out and rotated 720 degrees counterclockwise. Then, the tunica albuginea was sutured with 6/0 monofilament and fixed to the scrotum so the testis would not be detorsioned again. The scrotum was closed with 4/0 silk, and torsion was performed. After 4 h, the testis was detorted and waited. Orchiectomy was performed on the left testis 2 h after the detorsion procedure.
Group 1: after anesthesia, a vertical incision was made from the scrotal midline; the left testis was taken out by cutting the gubernaculum, then placed in the scrotum in an anatomical position without torsion. The layers were closed with 4/0 silk, and the left orchiectomy was performed 6 h later.
Group 2: a torsion/detorsion model was created for the rats in this group, and left testis orchiectomy was performed 2 h after detorsion, that is, at the 6th h of the experiment.
Group 3: a torsion/detorsion model was created for rats in this group, and 1 mg/kg ip human breast milk exosomes were given 30 min before detorsion. Left orchiectomy was performed 2 h after detorsion, at the 6th h of the experiment.
The orchiectomy material was divided into two parts. Half of the obtained testicular tissue was immediately frozen with liquid nitrogen and stored at 80 degrees until biochemical analysis. The other half was reserved for histopathological examinations.
Human breast milk collection and exosome isolation
Approximately 200-300 mL of fresh breast milk samples were collected from healthy lactating mothers who applied to the KTU University, Faculty of Medicine, Pediatric Surgery Clinic (Trabzon, Turkey). Milk samples were frozen immediately and kept at -80°C until used. Exosomes were isolated from breast milk by differential centrifugation and ultracentrifugation. All centrifugations were performed at 4 °C, and breast milk was centrifuged at 3000×g (F0650 rotor, Beckman Coulter Instruments, USA) for 10 min to separate the milk lipid layer. The supernatant was transferred to a new tube, and centrifugation was performed again at 5,000 ×g (F0650 rotor, Beckman Coulter Instruments, USA) for 30 min to eliminate large cellular debris and remaining lipids. Then, the new supernatant was transferred to a new tube, and centrifugation was performed again at 12,000 ×g (F0650 rotor, Beckman Coulter Instruments, USA) for 10 min to remove smaller cellular debris and apoptotic cells. The exosomes were obtained from the new centrifuged milk supernatant by ultracentrifugation at 110,000 ×g for 2 h at 4°C using a JS 24.15 rotor (Beckman Coulter Instruments, USA). After removing the exosome-free supernatant, the crude exosome pellet was suspended in 1000 μL phosphate-buffered saline (PBS, pH 7.4) in a 1.5 mL Eppendorf tube and stored at -80 °C until used for the experiment.
Human breast milk-derived exosome protein extraction and confirmation by western blot
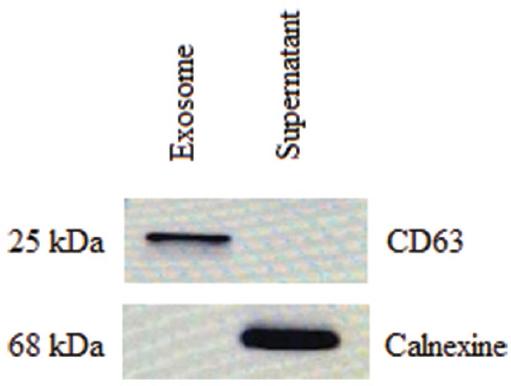
Human breast milk-derived exosomes were lysed in RIPA lysis solution (SE3924401, SERVA) containing protease and phosphatase inhibitor cocktail (PPC1010, SIGMA) was added, and exosomes were kept on ice for 15 min. Afterward, the protein was obtained by centrifugation at 15000 xg, +4°C for 15 min, and its concentrations were analyzed using the Bicinchoninic acid (BCA) kit (LOT: UD282967, Thermo Scientific, USA) and analyzed for total CD63 (positive exosome membrane marker) and calnexin (negative marker) protein expression. Lysate was separated by SDS-PAGE and transferred onto a nitrocellulose membrane. The membranes were probed with antibodies and detected using enhanced chemiluminescence detection. Primary antibodies were as follows: anti-CD63 (1:1000; Abcam, ab68418); and anti-calnexin (1:1000; Abcam, ab213243). The secondary antibody was horseradish peroxidase (HRP)-conjugated goat anti-rabbit (1:5000; Abcam, ab97200).
The protein concentration of the breast milk exosomes used in the experiment was adjusted to 1 μg/mL by the BCA kit.
Biochemical analysis
Testicular tissue was homogenized with 2 mL of cold homogenization buffer in a test tube for about 20 s until the tissues were completely disintegrated and then centrifuged at 3,000 rpm for 10 min. 750 μL of supernatant was placed in 1.5 mL Eppendorf. 600 μL of ethanol-chloroform (2:3) mixture was added to it. It was centrifuged at 10,000 xg for 30 min at +4°C. After centrifugation, protein determination, superoxide dismutase (SOD), and catalase (CAT) activity were measured in the protein phases on the upper part of the Eppendorf. SOD enzyme activity was performed by modifying the method developed by Sun and Oberley15. The Aebi method was used to determine CAT enzyme activity16.
Testicular tissue was homogenized with 2 mL of cold homogenization buffer in a test tube for about 30 s until the tissues were completely disintegrated and then centrifuged at 3,000 rpm for 10 min. The supernatant was taken, and the Malondialdehyde (MDA) level was determined. This method was carried out by modifying the method based on measuring the absorbance at 532 nm of the color of the complex formed by MDA with thiobarbituric acid in an acidic medium17.
Histopathological assay
Tissues were fixed in Bovine solution for 72 h. Fixed tissue pieces were dehydrated by passing through 70%, 90%, 96%, and 100% graded alcohol series. It was then cleared by passing through a xylene solution. After the paraffin blocks of the tissues were prepared, 5 μm thick sections were taken with a fully automatic microtome. Sections were stained with hematoxylin and eosin (H&E) after deparaffinization. The H&E stained slides were evaluated histopathologically with a light microscope (Olympus BX 51; Olympus Optical Co, Ltd, Tokyo, Japan), by an experienced pathologist unaware of the study groups. Damage to testicular tissues was graded by the Johnsen Testicular Biopsy Score (JTBS). In this scoring system, testicular tissues were evaluated semi-quantitatively in 5 high power fields (HPF) at x200 magnification by light microscopy.
Statistical analysis
SPSS 23.0 statistical package program was used in the analysis of the data. Descriptive statistics of the evaluation results were given as median and quartiles (Q1-Q3). The suitability of the data for normal distribution was checked with the Kolmogorov-Smirnov test. The Kruskal-Wallis test was used to compare the numerical values of three independent groups. A statistically significant level was taken as p < 0.05 in the Kruskal-Wallis test. Since significant results were found in the Kruskal-Wallis test, Bonferroni correction was made when making pairwise comparisons. Mann-Whitney U test was used and p < 0.017 (0.05/3) was considered significant.
Results
Confirmation of exosomes from mother milk
The presence of exosomes from human breast milk was confirmed by Western blot for the exosome membrane markers cluster of differentiation 63 (CD63) and the negative marker calnexin (Fig. 1).
Biochemical analysis
When the tissue MDA, SOD, and CAT values of the three groups were compared, a statistically significant difference was found which is shown in table 1.
Table 1 The mean values of the biochemical results of the groups
| Parameters | Group 1 Median (Q1-Q3) | Group 2 Median (Q1-Q3) | Group 3 Median (Q1-Q3) | p* | Post-hoc** |
|---|---|---|---|---|---|
| MDA (nmol/mg protein) | 28.45 (21.03-34.73) | 68.74 (44.11-91.00) | 22.38 (19.68-22.52) | < 0.001 | p1-2 = 0.015 p2-3 < 0.001 |
| SOD (U/mg protein) | 9.83 (8.67-10.65) | 10.86 (9.21-13.96) | 7.30 (6.78-9.46) | 0.012 | p2-3 = 0.004 |
| CAT (k/g protein) | 6.87 (5.54-7.38) | 3.33 (2.39-3.84) | 5.70 (3.88-6.95) | 0.007 | p1-2 = 0.002 |
*p < 0.05 was considered statistically significant. The KruskalWallis test was used.
**p < 0.017 was considered statistically significant. Bonferroni correction has been made. Mann-Whitney U test was used.MDA; malondialdehyde; SOD: superoxide dismutase; CAT: catalase.
When the pairwise MDA values of the groups were compared, a statistically significant difference was found between group 1-group 2 and group 2-group 3 (p < 0.05). There was no significant difference between group 1 and group 3.
When the pairwise SOD values of the groups were compared, a statistically significant difference was found between group 2 and group 3 (p < 0.05). There was no significant difference between group 1-group 2 and group 1-group 3.
When the pairwise CAT values of the groups were compared, a statistically significant difference was found between group 1 and group 2 (p < 0.05). There was no significant difference between group 1-group 3 and group 2-group 3. A pairwise comparison of tissue MDA, SOD, and CAT values is given in table 1.
Histopathological assay
When the left testis JTBS of different groups were compared histopathologically, the difference between them was found to be statistically significant. Values are given in table 2.
Table 2 Histopathological assay; JTBS results
| Groups | JTBS mean values Median (Q1-Q3) | p* | Post-hoc** |
|---|---|---|---|
| Group 1 | 9.5 (9.0-10.0) | < 0.001* | p1-2 < 0.001 p2-3 = 0.014 |
| Group 2 | 3.0 (3.0-4.0) | ||
| Group 3 | 8.0 (7.0-8.0) |
*p < 0.05 was considered statistically significant. Kruskal-Wallis test was used.
**p < 0.017 was considered statistically significant. Bonferroni correction has been made. Mann-Whitney U test was used. JTBS: Johnsen testicular biopsy score.
A statistically significant difference was found between group 1-group 2 and group 2-group 3 when comparing the JTBS values of the groups in pairs. There was no significant difference between Group 1 and group 3 (p < 0.05). Values are given in table 2. Histopathological images in all three groups are shown in figure 2.
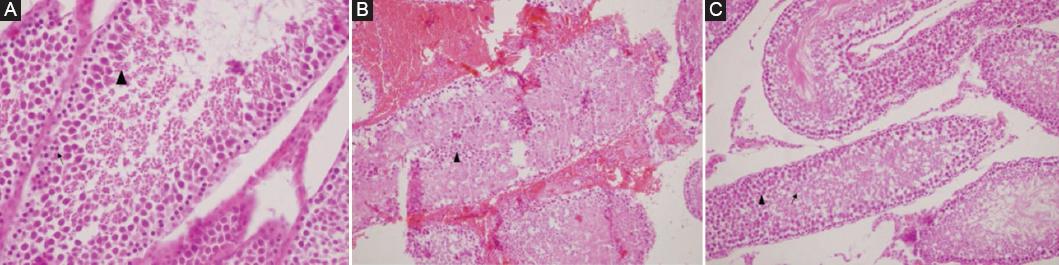
Figure 2 A: sertoli cell, spermatogonia (↑), and Spermatocyte (▲) in testicle of group 1. B: diffuse fresh bleeding in the interstitial area and exfoliated necrotic cells in seminiferous tubule lumen, few ghost germinal epithelial cells in the left testicular tubule lumen belonging to group 2 (▲) (H&E ×100). C: Spermatids (↑) and spermatozoons (▲) in the testis belonging to group 3.
Discussion
The primary pathology in testicular torsion is venous occlusion that causes testicular edema and pain. However, this venous occlusion causes hypoxia in the testis, just as in other tissues, by disrupting the arterial circulation because the tunica of the testis is not elastic. Thrombi formed at venous, arterial, and capillary levels cause testicular ischemia and subsequent necrosis18. By eliminating the factor causing ischemia in reperfusion, blood flow to the ischemic organ can be restored, irreversible damage can be prevented, and lost functions can be restored. However, "reperfusion damage" occurs due to the free oxygen radicals formed and the activated blood cells in the blood. Reperfusion after ischemia damages the tissue more than ischemia19,20.
Despite the search for alternative medical treatment methods for testicular torsion, emergency surgery is still a valid and widely applied treatment. Although the effect of testicular tissue by torsion is related to the duration and degree of torsion, the efficacy of numerous pharmacological agents and treatments have been evaluated in animal models to prevent testicular reperfusion injury since the primary preventable tissue damage occurs during the post-detorsion reperfusion period21.
Exosomes are serious cell-cell communication messengers and contain proteins, lipids, and genetic material that can be transported between cells to mediate intercellular communication and immune response22. Their existence as natural carriers, high biodistribution and stability in plasma, and relatively small size make them suitable candidates for therapeutic applications12.
A recent study suggested that exosomes produced from human breast milk may reduce oxidative stress and support immune system development23, and this encouraged us to use human breast milk exosomes in our study. Admyr et al. showed that exosomes in human breast milk can potentially affect the infant's immune system14. To investigate the effect of exosomes against I/R damage in testicular torsion due to their protective effects in reperfusion injury mentioned above and positive effects on sperm motility and viability, we used exosomes obtained from breast milk, which has not been tested in the literature before.
First, we showed that the presence of human breast milk exosome produced in the Department of Biochemistry of KTU was confirmed by the presence of CD63 and the absence of calnexin using the Western blot method. Then we applied ip low dose (1 cc) exosomes 30 min before detorsion to the torted testis. Studies show that local injection of the damaged testis has no adverse effect on the contralateral testis24,25. Our study, in which we obtained statistically significant results, shows that testicular I/R damage and necrosis occur in the testicular tissue.
Keivan et al. have shown that rats' blood serum-derived exosomes inhibit testicular damage, including seminiferous tubule degeneration and loss of interstitial tissue, due to high NO and MDA levels following testicular torsion and NO, MDA, and apoptotic gene expression levels decrease and provide healthy sperm production26. Similarly, in our study, when the MDA values of group 2 and group 3 were compared, we found a statistically significant difference between the two groups. Group 3 had significantly lower MDA values. The MDA value does not increase due to the effect of exosomes on reducing tissue damage. In addition, no statistically significant difference was found when the MDA values of group 1 and group 3 were compared.
No statistically significant difference was found when the SOD and CAT values of group 1 and group 3 were compared, and this makes us think that antioxidant enzymes are consumed less in group 3, and instead, the exosome is used as an antioxidant. As a result of these data, we think the exosome is biochemically effective in I/R damage since the MDA and SOD values are significantly different, and the CAT values are higher than Group 2, although there is no statistically significant difference.
Liu et al. showed that caspase-dependent apoptosis pathways are activated after testicular torsion injury, and spermatogenic cells are largely apoptotic, whereas treatment with exosomes significantly reduces the degree of apoptosis in spermatogenic cells24. In addition, it has been shown histopathologically that the proliferative effect of the exosomes on spermatogenic cells is not limited to primary spermatocytes, and that it can inhibit spermatogenic cell apoptosis and promote proliferation after testicular torsion-detorsion injury24. In our study, when group 2 and group 3 were compared, it was shown that the testicles of group 3 had less damage than group 2, and the difference between them was statistically significant. When group 1 and group 3 were compared, although there were histopathological differences, there was no statistically significant difference. Since exosome application significantly reduced the reperfusion damage in testicular tissue compared to the torsion group in histopathology, it was seen as an expected result that it was not statistically significant. In group 3, spermatids and spermatozoa were preserved, although not as much as normal; it has been shown that there is a significant difference compared to group 2.
Finally, the fact that exosomes were taken from human breast milk, prepared and confirmed with a series of procedures, and the application dose was taken by taking samples from previous experimental studies can be considered among the limitations of our study.
Conclusion
Exosomes are important in diagnostic and therapeutic research as they are natural liposomes. Although studies on exosomes obtained from human breast milk have succeeded and increased in recent years, there is no clinical use in humans yet. We showed that exosomes were significantly protecting the testicular tissue and germ cells compared to the torsion group and new long-term experimental studies with different doses should be needed.











 nueva página del texto (beta)
nueva página del texto (beta)