Introduction
Peri-implantitis is a common complication of oral implantation, which arises mainly due to the failure to form a stable soft tissue closure between the implant and the soft tissues, and although local antibiotics can control the infection to a certain extent, it is often difficult to remove the chronic foci of infection1. The presence of inflammation in the soft tissue of the neck of the implant affects the combination of gingival soft tissue and the smooth neck of the implant, and the inflammation gradually and continuously develops downward from the neck of the implant, resulting in the formation of a fibrous parcel on the surface of the implant, and the implant then loosens and falls off2. In the case of natural teeth, the soft-tissue closure consists of epithelium and connective tissue that penetrates the mucosal area to protect the submucosal tissues against external disturbing factors. Dental implants in contact with the epithelium and connective tissue form a closure similar to that of a natural tooth. However, compared to natural teeth, implant epithelial and connective tissues are weakly attached and susceptible to damage by external factors, leading to higher rates of implant infection3,4. Therefore, it is important to seek an effective method to strengthen the bond between soft tissues and the implant neck to resolve peri-implantitis and maintain the long-term stability of implants. 3D-printed personalized titanium implants are increasingly being studied as a new alternative to immediate implantation in the posterior region; however, due to the fact that the soft tissues around the implant are in a bacterial environment in the oral cavity, cases of poor closure of the implant with the surrounding soft tissues are common due to the effects of the rejection reaction and inflammatory response5. Relevant studies have shown that implant surface modification can effectively improve the surface properties of implants and enhance the ability of titanium surface and soft-tissue binding6, and magnesium and zinc as a commonly used preparation for oral soft tissue healing, based on this background, this paper is to build a 3D-printed titanium implant modified with magnesium and zinc ions, and implanted in the oral cavity of New Zealand great white rabbits, to explore whether it can promote the closure of the oral soft tissues, with the aim of opening up new ways to provide a number of theoretical theories for the clinical development of The purpose of this paper is to provide some theoretical references for clinical development of new ways of oral implantation.
Materials and methods
Laboratory animal
Eighteen 6-month-old healthy adult New Zealand Large White rabbits, male and female, with a body mass of 2.5-3.5 kg, were selected as experimental animals, provided by the Animal Experiment Center of Xi'an Jiaotong University, and kept in the Animal Room of the Animal Experiment Center of the Department of Medicine of Xi'an Jiaotong University.
Main reagents
S-3000N scanning electron microscope (SEM; Hitachi, Japan), E300CP/400CS hard tissue cutting and grinding system (EX-AKT Vertriebs GmH, Germany), TCS.SP8 laser scanning confocal microscope (Leica, Germany), the C43.104 electronic universal testing machine (MTS type, Shenzhen Meters Company, China). Pentobarbital sodium, calcineurin cell viability kit (Eimage Technology Co., Ltd., China).
Personalized implant design and fabrication
A computed tomography of rabbit head was taken and the obtained DICOM data were imported into the medical image processing software MIMICS 15.0 to obtain a 3D geometric surface model of the jawbone and teeth. The 3D geometric surface model of the mandible with teeth was exported as a point cloud file in STL format and imported into Geomagic 12.0 inverse software, and personalized root-shaped titanium implants were designed according to the morphology of the bilateral mandibular first premolar teeth, which were printed out using 3D printing technology (SLM, EOS-M290, Germany, average particle size of 20 μm), and a mixed-acid wash solution (HF: HNO3: H2O = 1:5:4 Vol) was diluted 4 times with ultrapure water and used to acid-wash the titanium-based material under ultrasound 3 times for 5 min each time to remove the surface oxide film. Subsequently, the samples were rinsed with ultrapure water until the surface was smooth and clean without residual traces of mixed acid wash solution, dried naturally in the air, and the samples were labeled as Ti. The above pre-treated materials were placed on the target stage of the vacuum chamber of the plasma immersion ion implantation (PIII) equipment, and the magnesium, and zinc ions were injected individually and the magnesium/zinc ions were co-injected when the vacuum degree was up to 5 × 10-2 Pa. Finally, they were washed with distilled water, autoclaved, and dried and preserved for use1. and dried and preserved for use7.
Experimental grouping and treatments
The left and right mandibular teeth of each experimental animal were randomly divided into two groups, and the bilateral first premolar teeth were extracted after general anesthesia, one implant was implanted in each of the magnesium/zinc ion surface-treated group (experimental group) and the surface-untreated group (control group), respectively, and a total of 36 implants were implanted, i.e., 18 implants in the experimental group and 18 implants in the control group. The specific methods of soft tissue implant surgery were as follows: 3% sodium pentobarbital at a dose of 1 mL/kg was used to perform auricular marginal intravenous general anesthesia, perioral skin preparation was performed after anesthesia became evident, and in accordance with aseptic surgical practices, intra and extraoral 1% povidone-iodine disinfection was performed, and routine toweling was performed. Minimally, invasive extraction of bilateral mandibular first premolar, implantation of the sterilized implant, mattress suture method to close the gingiva and implant. Postoperatively, penicillin 800,000 U was given intramuscularly once a day for 5 d to prevent infection in the operative area. Feeding and observation under routine conditions were performed to closely observe whether there was any wound infection after surgery. The sutures of the wound were removed 10d after the operation. The operation methods were all standardized to ensure that there was no significant difference between each group of experimental subjects before the test observation. The experimental animals in each group were executed by air embolization of the marginal ear vein at 2 and 4 weeks after the operation, and bilateral mandibular specimens were taken for observation and testing.
Fluorescent staining test
After the animals were executed at 2-4-weeks postoperatively, the mandibular specimens with implants were taken and fluorescently labeled with a calcein yellow chlorophyll kit, while the specimens were dehydrated, fixed, and fabricated into implant bone abrasions, which were observed under a laser scanning confocal microscope, and the fluorescence density values of the fluorescent bands were measured. The entire staining and observation process should be carried out under light protection to prevent premature quenching of fluorescence. The relevant operations were performed in strict accordance with the instructions of the reagents and instruments.
Observation indicators
(1) At 2 weeks and 4 weeks after implantation, the bonding of the implant material with the host soft tissue of the two groups was compared by visual observation and the bonding of the implant material with the host soft tissue of the two groups was evaluated by fluorescence staining. (2) Observe the fluorescence staining graphs of the two groups 2 weeks and 4 weeks after implantation and compare the fluorescence density values of the two groups.
Statistical methods
Data were analyzed using the statistical analysis software Statistical Package for the Social Sciences (SPSS) 17.0, and measurements were expressed as arithmetic mean ± standard deviation and statistically tested using one-way analysis of variance (ANOVA). Each group of variables contained at least three valid values, and the significant difference level was set at p < 0.05 to indicate that the data were statistically significantly different.
Results
Observation by naked eye
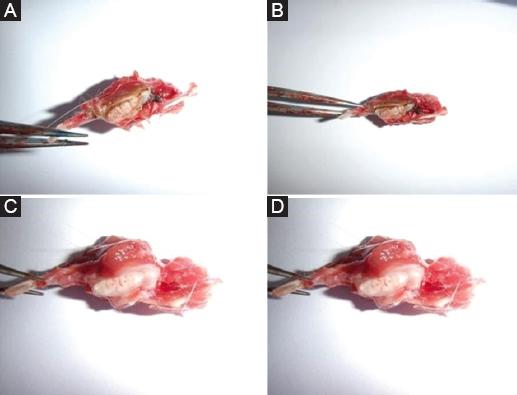
After 2 and 4 weeks of implantation, under naked eye observation, the combination of the implant material and the surrounding soft tissues in the experimental group had almost no gaps and was tighter than that of the control group, and the surrounding tissues were red and glossy, and no obvious inflammatory reaction was seen; in the control group, the combination of the implant material and the surrounding soft tissues had numerous gaps, and the surrounding tissues were poorly blooded and glossy, and there was an obvious inflammatory reaction, which was a poor result. The combination of the implant material with the surrounding soft tissue in the experimental group was significantly better than that in the control group figure 1.
Fluorescent staining test results
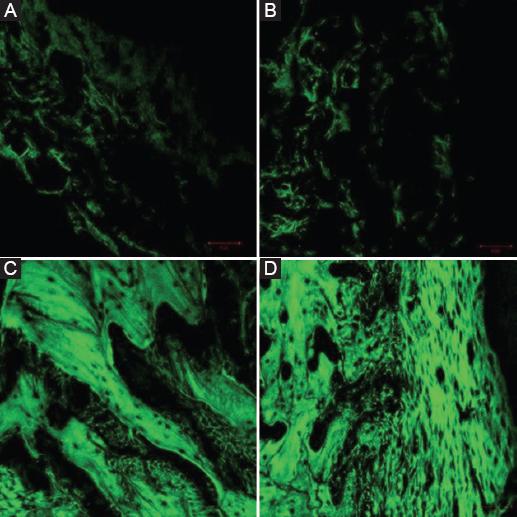
The area labeled with calcein xanthophyll showed green fluorescent bands. Since calcein xanthophyll can combine with calcified new bone and fluoresce under ultraviolet irradiation, the amount of fluorescent labeling can suggest the active degree of bone metabolism and bone proliferation in the tissues around the implant after surgery, and thus, it has been widely used in the restoration research of bone tissues. After 2 and 4 weeks of implantation, the fluorescence density values of the experimental group were (94.48 ± 33.40) and (30.78 ± 11.22), and the fluorescence density values of the control group were (68.44 ± 22.41) and (21.94 ± 7.97), respectively, and the measured data were analyzed by one-way ANOVA with SPSS 17.0 software.: At 4 weeks of planting, the fluorescence density values of both groups were significantly higher than those of this group at 2 weeks (p < 0.05); at 2 and 4 weeks of planting, the fluorescence density values of the experimental group were significantly higher than those of the control group (p < 0.05) figure 2 and table 1.
Discussion
Peri-implant soft-tissue closure consists of two parts: epithelial attachment and connective tissue attachment. Epithelial attachment is the first layer of soft-tissue closure and is the front line of defense against external risk factors8. The tooth-epithelial bonding interface is formed at the perforating gingival site immediately after the eruption of a natural tooth, and its stability depends on the binding epithelium. The binding epithelium is a lowly differentiated non-keratinized complex squamous epithelium, which is stably attached to the tooth surface by epithelial adherens and forms a closed interface against harmful biological factors9. The epithelial attachment apparatus consists of two layers, including the basal layer where the inner basement membrane binds to the outer connective tissue and the suprabasal layer where the inner basement membrane and hemibridges come into direct contact with the tooth surface. The peri-implant epithelium is also directly attached to the abutment surface through the inner basement membrane and hemi-bridging granules, but this attachment structure is distributed only in the root-side portion of the epithelial attachment interface, whereas in the natural peri-implant periodontal area, the inner basement membrane and hemi-bridging granules are widely distributed throughout the epithelial-tooth interface. Therefore, the epithelial attachment strength of implants is lower than that of the natural periodontium and is more susceptible to destruction by external forces or biological factors10-12. In addition, when inflammatory manifestations were present in the soft tissues, the tendency of the inflammatory infiltration bands on the outer side of the pocket wall cells to spread toward the root side was more pronounced in the implant periodontium compared with the natural periodontium, with a greater total extent, a higher density of inflammatory cells in the soft tissues, and the neutrophils in the natural periodontium were mainly clustered in the epithelium of the pocket wall, whereas those in the implant periodontium were also distributed in the perivascular area around the root far away from the pocket wall13,14. Animal model studies have shown that compared with periodontitis, the connective tissue of peri-implant pockets often lacks an epithelial barrier to plaque, and its inflammatory infiltration is more extensive and closer to the top of the alveolar ridge, with more neutrophils and osteoclasts, and more severe resorption of the alveolar ridge; after removing inflammatory predisposing factors, the inflammatory response in the natural periodontium is self-limiting, while peri-implant lesions can still continue to progress, triggering extensive bone resorption15,16. This shows that the inflammatory destructive capacity of the implant periodontium is higher than that of natural teeth once symptoms of infection appear. Clinical studies have also confirmed that the progression of peri-implantitis tends to increase in a non-linear fashion and progresses faster than periodontitis17. Therefore, improving the sealing properties of the implant to the oral soft tissues is crucial for preventing infections and improving the long-term success rate of implants. Most of the current implant materials are commercially pure titanium or titanium alloys, and although this untreated titanium implant material has a certain soft-tissue bonding ability, its pure metal surface inhibits cell proliferation and migration to a certain extent, thus affecting tissue healing, and its limitations presented in the field of oral implantation have become increasingly prominent18. Relevant studies have shown that the attachment of suitable concentrations of magnesium and zinc ions to the surface of titanium materials by PIII technology is important for the proliferation and migration of human gingival fibroblasts (HGFs), which is expected to influence the ability of implant binding to oral soft tissues to a considerable extent19. Therefore, the present study was conducted to investigate whether 3D-printed titanium implants based on surface modification of magnesium and zinc ions could promote oral soft-tissue closure using New Zealand Large White Rabbits, and the results were as follows.
In this study, it was found that after 2 and 4 weeks of implantation, the combination of implant material and surrounding soft tissues in the experimental group was significantly better than that in the control group under naked-eye observation; fluorescence staining analysis showed that the fluorescence density values of the experimental group at 2 and 4 weeks of implantation were (94.48 ± 33.40) and (30.78 ± 11.22), respectively, which were significantly higher than those of the control group of (68.44 ± 22.41), (21.94 ± 7.97), and the differences were all statistically significant. (21.94 ± 7.97), and the differences were statistically significant, suggesting that 3D printed titanium implants based on surface modification of magnesium and zinc ions can effectively promote the combination of the implant and its periodontal tissues, and the closure effect is remarkable. Analyzing the reasons, on the one hand, the surface modification of magnesium and zinc ions can increase the biocompatibility of titanium implants. Titanium is a commonly used implant material, but there is often a certain tissue reaction at the interface between it and the soft tissue, resulting in incomplete closure of the soft tissue to the titanium implant. By introducing magnesium and zinc ions on the titanium surface, the compatibility between the implant and the surrounding tissues can be increased and the tissue reaction to the titanium can be reduced, thus promoting the closure of the soft tissues20. In addition, HGFs are the main cells involved in soft-tissue regeneration and closure, providing the basis for the formation of the keratinocyte layer, which plays a key role in oral wound healing and soft-tissue regeneration. The number and activity of HGFs at the implant-soft tissue interface are important for the formation of a solid closure between the implant and the soft tissue21. Relevant studies have shown that surface modification means to adjust the surface morphology and chemical composition of titanium have a significant effect on the behavior of HGFs adhesion spreading, migration, and proliferation, which suggests that changes in the physicochemical properties of titanium surfaces can affect their soft-tissue closure properties22. It has been found that both magnesium and zinc ions have a certain promotion effect on the proliferation and activation of oral soft-tissue cells23. Magnesium ions can promote the migration and proliferation of soft-tissue cells and accelerate the closure process. Zinc ions have antioxidant and anti-inflammatory effects, which can promote tissue repair and healing, and further promote soft-tissue closure24. On the other hand, magnesium, a common agent in dental clinical treatment, has a significant effect on the behavior of HGFs. Ionization of magnesium into titanium surfaces by plasma immersion can effectively improve the adhesion and spreading of HGFs on the material surface and increase the potential of the material to promote soft-tissue closure. The study of the migration rate of HGFs on pure magnesium surfaces revealed that a specific concentration of magnesium ions could significantly enhance the adhesion of HGFs on the material surface although it had no significant effect on the proliferation and activity of HGFs25. Xiao et al.,26 studied and discussed the effect of magnesium ion concentration on the directional migration ability of human fibroblasts and found that this property was related to integrins in extracellular matrix proteins. Zinc is also commonly used in oral therapy, where it effectively promotes soft-tissue regeneration and facilitates wound healing. Zinc also has an important effect on the behavior of HGFs. Zogheib et al.27 cultured HGFs in a zinc-free medium and found that zinc deficiency significantly reduced HGFs migration, proliferation, and DNA synthesis, and affected cell morphology and intracellular oxidative stress levels. The addition of exogenous zinc as a supplement eliminated these conditions. Zinc has strong antimicrobial activity, implant-released zinc is effective in preventing post-operative infections, and plasma-immersed ion-incorporated zinc on titanium surfaces is effective in improving osteogenic properties and inhibiting bacterial growth. In addition, magnesium and zinc synergize to achieve complementary effects. Observations on the surface of titanium samples chelated with both magnesium and zinc ions revealed that magnesium ions can promote cell proliferation and differentiation, and facilitate the bonding of the implant with the surrounding soft tissues, while zinc ions can be highly effective in antimicrobial activity, and improve the stability and durability of the bonding of the implant with the surrounding soft tissues28. Hong29 injected magnesium and zinc simultaneously into the surface of medical titanium, and the osteogenic proliferation and differentiation of rat bone marrow MSCs could be induced by the synergistic effect of magnesium and zinc ions, and this result fits with the conclusion of the present study. In addition, it has been shown30 that magnesium and zinc ions can also affect angiogenesis in oral soft tissues. Angiogenesis is an important part of the soft-tissue repair and closure process, while magnesium and zinc ions can regulate the proliferation of vascular endothelial cells and the release of angiogenic factors, promote blood vessel growth and repair, and provide a better blood supply and nutrient support for the closure of the soft tissues, thus promoting the closure of oral soft tissues.
There are still some limitations of this study. For example, the sample size of this study is small, the study population is single, it is a single-center design, and the mechanism related to oral soft-tissue closure is relatively insufficient. All of these limitations constrain the generalization of the results of this study, and a more definitive and comprehensive conclusion of the study needs to be further confirmed by multi-sample and multi-center studies.











 nueva página del texto (beta)
nueva página del texto (beta)