Introduction
The non-physiologic retrograde flow of urine from the bladder to kidneys is defined as vesicoureteral reflux (VUR), with 1-2% prevalence and in some children has a genetic predisposition1. The triangular region between two ureterovesical junctions (UVJs) and internal ureteral meatus, namely, "the bladder trigone" has a central importance in anti-reflux mechanism and was found to derive mostly from bladder muscle with the contribution of ureteral fibers. This was approved in animal studies by the presentation of two major muscle types in the trigone; detrusor and the muscles associated with the intramural ureter2. The cellular morphology of the trigone depends on both its embryological origin and key signaling molecules including sonic hedgehog (SHH) signaling2,3. During bladder development, the smooth muscle of bladder differentiates from primitive mesenchyme under the influence of urothelium and a sufficient concentration of SHH was reported to be important in development of bladder smooth musculature4-7. Patched (Ptc 1), the membrane bound receptor for SHH is expressed in embryonic bladder mesenchyme. Ptc 1 suppresses the smoothened (Smo) in unbound state and when the SHH binds Ptc 1, this inhibitory effect disappears and initiates the cascade which activates Gli transcription factors (Gli1, Gli 2 and Gli 3) in target cell8. Gli1 and Gli 2 are the ones targeting not only the SHH but also Wnt family and bone morphogenic proteins which play role in normal embryonic development and differentiation5.
The UVJ avoids retrograde flow of urine from the variable pressure bladder to low pressure upper urinary tract (kidney and ureter) depending on the ureteral musculature at the junction particularly. Proper development of ureteral and trigonal musculature ensures this one-way flow. In case a deficiency occurs in trigonal development, some clinical disorders including VUR might occur due to improper muscle formation resulting in a relatively short intramural tunnel4. In this regard, we hypothesized that the defective SHH signaling plays a role in development of VUR due to abnormal trigonal/ureteral musculature and aimed to determine protein expression of this gene in resected UVJ segments of children with VUR. In this prospective study, we aimed to determine protein expression of SHH in resected UVJ segments of children with VUR and to our knowledge, this is the first human study trying to highlight the role of SHH protein expression in reflux etiopathogenesis.
Materials and Methods
Study design and sample preparation
This prospective study was approved by the Institutional Ethical Committee and informed consent was obtained from all the patients/parents before their inclusion in the study. (26.02.2018/161).
The study group included 19 children; 12 (63%) girls, 7 (37%) boys, who had ureteroneocystostomy (UNC) operation; 3 (15.7%) right-sided, 7 (36.8%) left-sided, 9 (47.3%) bilateral, due to primary VUR between years 2015 and 2018. The technique of UNC was Cohen in all patients and renal units except one unilateral operation done by the Politano-Leadbetter combined intra and extravesical technique. The hypoplastic and redundant distal ureter was excised and the excised hypoplastic distal segment including the intramural portion was used for the genetic analyses. Finally, a total of 28 UVJ segments were examined for Western Blot analysis to determine related protein expression level of SHH gene signaling pathway in the UVJ specimens. After the appropriate transport of the materials to the laboratory, protein isolation was carried out for each tissue and preserved at −80°C for later analysis. Protein concentrations were determined in all samples using Qubit® Protein Assay Kits (Thermo Fisher Scientific, Cat No: Q33211). Western blot band expression levels of housekeeping gene β-Actin and target gene SHH which is thought to be effective in VUR pathogenesis were transformed into numerical data using Image J program (NIH, Bethesda, MD, USA).
Housekeeping genes are known to be expressed in almost all the cells of an organism and generally considered to be the constitutive genes which are essential for the maintenance of the basic cellular functions9. In this respect, housekeeping genes are widely used as internal controls for gene expression normalization for analysis as western blotting, northern blotting, RT-PCR, etc.10. Therefore, in this study, mean abundance values of SHH protein were calculated and compared with β-actin for each related tissue thus expression levels of Western blot bands were normalized against β-actin. Clinical parameters of the patients enrolled in our study were also retrospectively reviewed.
Western blotting
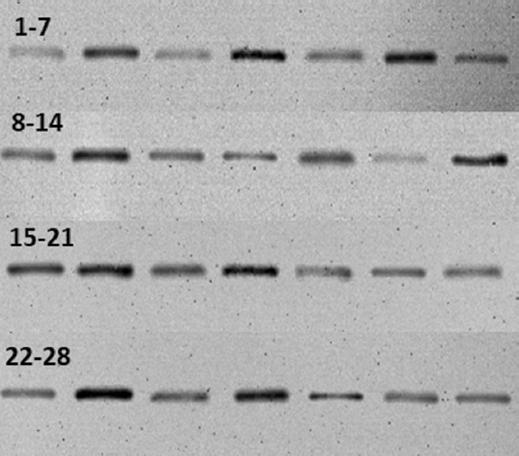
For protein denaturation, we used 100 μg from each sample and also added 4× NuPAGE LDS sample buffer (thermo fisher scientific, Cat no: NP0004) (5 μL), 10× NuPAGE sample reducing agent (thermo fisher scientific, Cat no: B0004) (2 μL) and distilled water was added to a total volume of 20 μL. This mixture was incubated at 70°C for 10 min and ice cooled for 2 min. 20 μL (100 μg) quantities of prepared protein were loaded and separated by 12% SDS-PAGE (Invitrogen, NuPAGE 4-12% Bis-Tris Gel, Cat: NP0321PK2), then transferred to a polyvinylidene difluoride membrane. Following blocking with 5% bovine serum albumin (5%BSA) in phosphate buffered saline with 0.1% tween 20 (PBS-T), the membrane was incubated overnight at 4°C with rabbit Anti-SHH Antibody (St. John’s Laboratory, Cat: STJ193168) and rabbit anti-β-actin antibody (St. John’s laboratory, Cat: STJ91464) as the loading control. Primary antibodies were diluted in 1:500 (anti-SHH) and 1:1000 (anti-β-actin). For the secondary incubation, membranes underwent hybridization with a horseradish peroxidase (HRP)-conjugated goat-anti-rabbit-IgG antibody (1:10000 dilution; advansta, Cat no: R05072-500) for 1 h at room temperature. After washing 3 times in PBS/0.1% tween 20, proteins were visualized in imaging system (ChemiDoc-It2, UVP) with using 6 ml NZY supreme enhanced chemiluminescent HRP substrate (Nzytech, Cat no: Mb19301) (Fig. 1).
Statistical analysis
Densitometry of the Western Blot protein bands was analyzed using Image J (NIH, Bethesda, MD, USA) software program. Statistical analyses were applied with the (Statistical Package for Social Sciences, Chicago, IL, USA) version 15.0 program. Descriptive analyses were presented as mean ± standard deviation and median (min-max). Normal distribution of data was analyzed using Shapiro-Wilk test (p ≤ 0.05) and non-parametric tests were used for additional statistics then. Densitometry of the target protein (SHH) band was compared to the house keeping gene, β-actin with Wilcoxon test and groups according to the grade of reflux (low-high), laterality, presence of scar and differential functions (< 40%-≥ 40%) were compared using Mann-Whitney U test. Correlation analyses were applied to test the relation between grade of VUR and protein expression level. p ≤ 0.05 was considered as statistically significant.
Results
The mean age and follow-up duration of the patients were 88.6 ± 47.7 and 26.76 ± 16.6 months, respectively. According to International Reflux Grading System 14 (50%) renal units were grouped as low-grade reflux (Grades 1-3) while 14 (50%) renal units were in high-grade reflux group (Grades 4, 5). No family history of reflux was recorded in the patients included in this study. Of the 19 patients with 28 renal units, six patients with 12 units (42.9%) had bilateral and 9 patients with 9 units (32.1%) had unilateral scar formation on DMSA scintigraphy. Renal units were also grouped according to differential functions (≥ 40% and < 40%) obtained by renal scintigraphy and then compared in terms of protein expressions. In this series, the differential renal function was < 40% in 14 (50%) renal units whereas ≥ 40% in the other 14 (50%).
The mean western blot band area of SHH gene pathway-related protein was 3880.69 (2059.55-13941.61) while the mean area of β-actin, the house-keeping gene, was 20180.25 (9530.39-26709.75) (p = 0.001). Correlation analyses between grade of reflux and protein expression of SHH gene pathway revealed no significant relation (p = 0.30). When the UV samples were grouped as low- and high-grade reflux and compared in terms of SHH protein expression levels, no statistically significant difference was found between reflux groups (p = 0.818). Analysis comparing the specimens according to presence of scar formation revealed no statistically significant difference in terms of SHH protein expression (p=0.915). In addition, there was no statistically significant difference between ≥ 40% and < 40% function groups regarding SHH band area (p = 1). As stated before 9 (47.3%) patients had bilateral reflux whereas 10 (52.7%) had unilateral VUR in this series. In case of the possible impact of bilaterality on the results, the two groups were compared and no statistically difference was found with respect to SHH protein expression between groups (p = 0.314) (Table 1).
Table 1 Sonic hedgehog protein band area calculations according to convenient groups
| Classification groups for analysis | Number of contents (renal unit) (%) | SHH protein median western blot band area (min-max) | p-value* |
|---|---|---|---|
| Low-grade reflux | 14 (50) | 4405.04 (2059.55-12515.64) | 0.818 |
| High-grade reflux | 14 (50) | 3280.69 (2169.98-13941.61) | |
| Unilateral reflux | 10 (35.7) | 3284.28 (2169.98-10296.55) | 0.314 |
| Bilateral reflux | 18 (64.3) | 4546.74 (2059.55-13941.61) | |
| Bilateral scar formation | 21 (75) | 4296.57 (2059.55-13941.61) | 0.915 |
| Unilateral scar formation | 7 (25) | 3103.74 (2746.96-13941.61) | |
| ≥ 40% differential function | 14 (50) | 3083.86 (2059.55-13941.61) | 1 |
| < 40% differential function | 14 (50) | 4405.04 (2169.98-12515.64) |
*p ≤ 0.05 is statistically significant.
Discussion
In the recent study, we found that SHH protein expression levels significantly decreased in the hypoplastic UVJ segments of the patients with VUR. On the other hand, our results did not reveal a relationship between the level of expression and severity of reflux or kidney status.
At the early gestational week 12, the condensate myoblasts of distal ureter convert into smooth muscle fibrils. These longitudinal smooth muscle fibers place at the dorsal wall of the bladder before reaching the orifice. These small diameter muscle fibrils merge the opposite fascicles and create the interureteral muscle and mucosal fold between two ureteric orifices at gestational week 14. The hypothetical function of the intertrigonal muscle is moving the two orifices medially and downward with a periodic contraction and providing a passive antireflux mechanism by the prolongation of intramural ureter11. Some studies suggested that the intramural tunnel of ureters compose of bladder muscles and develop independent from the ureters even if in the absence of a ureter, though the mechanism still remains unclear2,12. As mentioned above, the muscle development of distal ureteric segments is in mesh with bladder. The development of ureter and trigon musculature is proven to be essential to provide the one-way flow of urine from the ureters into bladder and to prevent the kidneys from reflux of urine or bacteria4. Because it is thought that trigon musculature deficiencies result in relatively short intramural tunnel.
A hedgehog family ligand, namely, SHH, controls cell fate, cell differentiation, and proliferation in embryogenesis7,13. In the literature, numerous studies have reported the SHH and its downstream signaling molecules produced by the developing bladder epithelium to have a key role in development and patterning of bladder smooth muscle and otherwise cause the bladder development to fail5-7,14,15. Histological studies indicated that either SHH or an intact urothelium is necessary to induce smooth muscle differentiation, thus the source of SHH is thought to be the urothelium8. When SHH signaling molecule binds with the transmembrane receptor Patched (Ptch), this activation causes depression of Smo and activates the Gli transcription factors in the target cell to involve in bladder development and differentiation15.
However, several studies reported that high concentrations of SHH inhibit the smooth muscle differentiation while they agreed that lower concentrations induce this process6,7,16. Cheng et al. reported that Gli2, one of the target transcription factors of SHH, upregulates the Bmp4 expression and inhibits the smooth muscle differentiation6. Another study carried out by Shiroyanagi et al. also supported the information that SHH acts as both an inducer and an inhibitor of bladder smooth muscle differentiation5.
Our results were in the same direction that SHH is required for normal development of musculature of ureter and trigone and the depressed levels of regarding protein might cause VUR. The insignificant relationship between the severity of reflux and protein expression levels might have occurred due status of other related signaling factors or downstream molecules functionary in this pathway. Furthermore, different expression levels of SHH might cause inhibition or activation and might have varied through years. All the patients were postnatally diagnosed so that there was no investigation about the Shh protein deficiency prenatally through gestational weeks. Although this is the first study in human examining the related protein expression in ureteral specimens, our study has two limitations. First, for ethical reasons, it was not possible to establish a control group to compare the expression levels of Shh protein in normal ureteral tissue. Normal ureteral tissue could be obtained from nephrectomy materials performed with another diagnosis, but this was not possible in our study group because they were pediatric patients. Therefore, further studies with an appropriate control group are needed to address this issue. The second one is relatively small sized study group to generalize our results to overall VUR patients. To the best of our knowledge, our study retains its value to be the first human study aiming to find the effect of SHH signaling pathway in VUR etiopathogenesis.
Conclusions
SHH signaling pathway which is effective in the development of bladder and ureter smooth musculature might also be effective in etiopathology of VUR. Further studies with appropriate control groups will be precious to prove our results and contribute to the diagnosis and treatment of VUR.











 nueva página del texto (beta)
nueva página del texto (beta)