Introduction
Osteoarthritis is a dynamic process that occurs due to the disruption of the balance between destruction and repair of the joint cartilage and subchondral bone. The goal of treatment is to improve the quality of life by reducing pain and mobility limitations and preserving or improving joint function. When conservative treatment options fail to reduce patient complaints, total knee arthroplasty surgery is currently considered the gold standard surgical treatment method, which is cost-effective and provides long-term survival. Modern total knee arthroplasty surgery began with the development of the tricompartmental total knee prosthesis by Insall and Ranawat in 1973, and the number of surgeries has increased significantly in the past 20 years.
It is reported that the average blood loss during total knee arthroplasty surgery is between 500 and 1500 mL, and the average transfusion rate is 20%, although there are also reports of average blood loss of up to 2000 mL and transfusion rates up to 50%1-4. Considering that the average blood volume in the body is approximately 5000 mL and these surgeries are often performed in the geriatric population, blood loss is a significant problem. There are several options to address this problem, including pre-operative iron replacement therapy and erythropoietin therapy; the use of pneumatic tourniquets, hypotensive anesthesia, and autologous blood transfusion during surgery; and following the flexion of the knee after surgery. The most commonly used method to replace this loss is allogeneic blood transfusion. Allogeneic blood transfusion carries the risk of allergic and immunological reactions2. There is also a risk of infection, especially in the case of viral diseases during the donor’s window period. In addition, according to health-care system data, it is estimated that 2 million total knee arthroplasty surgeries will be performed in the United States in 2030, and the cost of preparing and administering one unit of allogeneic erythrocyte suspension is 250-1000 $ (250 $ in Turkey, 350 $ in France, and 1000 $ in the USA)3,5-7.
Tranexamic acid (TXA) (cyclohexanecarboxylic acid) (C8H15NO2) is a synthetic derivative of lysine and is an antifibrinolytic drug that inhibits fibrinolysis by binding to the lysine region of plasminogen4. It was first described in 1968 and has been used for the control of bleeding in gynecological and hematological diseases, among others, since 1970. It continues to be used in a variety of indications in many fields. It has been included in the World Health Organization’s list of essential drugs due to its low cost and cost-effectiveness. TXA, which has been used for the control of bleeding in various fields for many years, has become increasingly popular in the recent years in reducing blood loss associated with total knee and hip arthroplasty2-4,8-11.
There have been many meta-analyses on the effectiveness of TXA, but a consensus on the optimal dosage and mode of administration has not been reached due to the wide range of dosage and modes of administration. The effectiveness of intra-articular (IA) (topical and local) and systemic (intravenous) use of TXA in orthopedics is still controversial. Different meta-analyses have reported that the use of 1 g or more of TXA IA reduces blood loss and that the use of up to 3 g is effective and safe2,4,8-11. In addition, the IA application of TXA is an effective and practical method that allows for standard dosing independent of body weight and offers the opportunity to avoid systemic thrombotic complications (deep vein thrombosis [DVT], pulmonary embolism [PE], and venous thromboembolic event [VTE]) associated with the drug9,11,12.
There are many studies on the effect of TXA on bleeding in orthopedic prosthetic surgery, and almost all of these studies compare different methods of use with each other or with a control group. The aim of our study is to contribute to the literature by evaluating the effectiveness of IA TXA administration on blood loss and transfusion rates in total knee arthroplasty surgeries, in the same study with different doses (a total of 3 drug and 1 control groups).
Materials and methods
This study includes data from 160 patients who underwent unilateral total knee arthroplasty for primary osteoarthritis at Edirne State Hospital between 2016 and 2020. The effective and safe dose range for IA TXA administration is 1-3 g, so the study groups were formed based on this data1-4,10-16. After a power analysis (80%), the study consisted of 40 patients each in 4 groups (IA 1 g, IA 2 g, IA 3 g, and control). Demographic data, estimated blood loss based on pre- and post-surgical blood parameters, drain blood loss, and transfusion data were compared and evaluated. The study included patients with Kellgren-Lawrence Stage 4 primary osteoarthritis, but excluded patients with secondary osteoarthritis (traumatic arthritis, inflammatory arthritis, etc.), a history of cardiovascular treatment, more than 6 months of anti-coagulant use, and a diagnosis or suspicion of thromboembolic events.
Patients were operated on by 2 surgeons. After the surgical drape, the extremity was elevated for 10 min and wrapped with the help of Esmark bandage from distal to proximal and then inflated to 100 mmHg above the patient’s systolic blood pressure and a pneumatic tourniquet was used. No patient’s tourniquet pressure exceeded 300 mmHg because the surgery of patients with high systolic pressure (> 200 mmHg) was postponed. All surgeries were performed under spinal anesthesia with pneumatic tourniquet17, using the gap balancing technique18 to apply a cemented posterior stabilized total knee arthroplasty (PS-TKA) (Total Knee System, Tıpsan, İzmir, Turkey; Knee Prosthesis System, Tıpmed, İzmir, Turkey). After the repair of the patellar tendon, TXA was administered to the TXA groups as a 50 mL solution with TXA (Tranexel 250 mg/5% iv/im injection Solution, Haver Farma İlaç Anonim Şirketi, Istanbul, Turkey) mixed with physiological saline (SF, 0.9% NaCl) and 50 mL SF was administered to the control group IA. After wound closure and dressing, an anti-embolism stocking was applied, and the tourniquet was released about 15 min after IA administration. The drain was clamped for 30 min after tourniquet release and the amount of blood loss from the drain was measured and recorded at 1, 3, 6, 12, 24, and 48 h, postoperatively, with the drain being removed at 48 h.16 The results of the hemograms were recorded before and after surgery on the 1st, 2nd, and 3rd days. As per American society of anesthesiologists (ASA) criteria, patients with symptomatictic hemoglobin (hgb) levels below 8 g/dL and all patients with hgb levels below 6 g/dL received allogeneic red blood cell suspension transfusions to raise their hgb levels to at least 10 g/dL. The estimated blood loss due to surgery was calculated using the Nadler and Gross formulas19,20. According to the Gross formula, 1 unit of red blood cell suspension is considered to be 300 mL for patients who received transfusions, which was added to the estimated blood loss.
All patients were mobilized as soon as possible, within 24 h after surgery, to prevent DVT. The patients were taught bedside foot pump and leg lift exercises and were treated with anticoagulants (0.4 mL enoxaparin and subcutaneous application) and antiembolic stockings for 20 days after surgery. The patients were mobilized and followed up after the surgery with the current rehabilitation protocol21, and the patients with limited mobility were referred to the physical therapy and rehabilitation unit. After pain control, the patients were discharged from the hospital between the 4th and 6th days after surgery the patients were called to the outpatient clinic on the 20th day after surgery to have their sutures removed and to have their hemogram values checked.
Statistical analysis
Statistical analyses were performed with SPSS v.23.0 software (SPSS Inc., IBM Corporation, Armonk, New York, USA). In the power analysis, it was found that, for the acceptable blood loss amount of 70 units between the treatment groups at 80% power and 5% significance level, and the standard deviation was 100, the sample size that should be included in each group was found to be 40. The groups were analyzed in terms of distribution analysis of the data and homogeneity of variance. Data with normal distribution and homogeneous variance were analyzed with parametric tests (ANOVA), and data that did not comply with normal distribution or did not have homogeneous variance were analyzed with non-parametric tests (Kruskal-Wallis, Mann-Whitney U-test). Chi-square test was used to compare categorical variables. Since there were 6 pairwise comparisons in total, Bonferroni correction was made due to group analysis, and the p-value was calculated as 0.05/6 = 0.0083.
Results
In this study, 160 patients were included, 22 of whom were male (13.75%) and 138 were female (86.25%). In 73 patients (45.63%), the right knee was operated on, and in 87 (54.37%), the left knee was operated on. The groups were compared in terms of age, height, and body weight, and no statistically significant differences were found (Table 1A).
Table 1 Whole data of the patients in 5 groups are shown
| Section | Parameter | Group 4 (control) | Group 1 (IA 1GR) | Group 2 (IA 2GR) | Group 3 (IA 3GR) | p-values (p < 0.0083) |
|---|---|---|---|---|---|---|
| A | Age (year) | 66.38 ± 8.24 | 66.22 ± 9.28 | 68.38 ± 5.19 | 67.32 ± 7.48 | 0.4332† |
| Height (cm) | 164.25 ± 7.68 | 166.18 ± 5.70 | 161.68 ± 6.01 | 161.78 ± 6.19 | 0.0122† | |
| Weight (kg) | 81.33 ± 14.17 | 79.00 ± 11.35 | 79.80 ± 10.90 | 84.38 ± 12.99 | 0.2283‡ | |
| BMI (kg/m2)* | 30.23 ± 5.44 | 28.65 ± 4.3 | 30.60 ± 4.41 | 29.41 ± 4.23 | 0.0113‡ | |
| Gender (M/F) | 9/31 | 3/37 | 6/34 | 4/36 | 0.2194§ | |
| Side (right/left) | 19/21 | 18/22 | 17/23 | 19/21 | 0.9394§ | |
| B | ASA (I + II/III + IV) | 35/5 | 38/2 | 35/5 | 33/7 | 0.2774§ |
| Pre-operative ROM** | 100 (85-130) | 105 (75-125) | 105 (70-130) | 100 (80-130) | 0.6102† | |
| Tourniquet time (min) | 100 (60-120) | 100 (60-130) | 105 (60-130) | 105 (75-120) | 0.4472† | |
| Post-operative discharge day | 3 (3-5) | 3 (3-4) | 3 (3-5) | 3 (3-8) | 0.1572† | |
| C | Hemoglobin value (g/dL)* | 13.30 ± 1.20 | 12.79 ± 1.36 | 12.59 ± 1.01 | 12.62 ± 1.00 | 0.0232† |
| Hematocrit value (%)* | 40.32 ± 3.54 | 39.26 ± 4.15 | 38.32 ± 2.86 | 38.37 ± 2.75 | 0.0283‡ | |
| Platelet count (103/mL)* | 264.5 (153-439) | 278.5 (146-519) | 269 (122-512) | 257.5 (142-496) | 0.8642† | |
| Body blood volume (l)* | 3.54 ± 0.52 | 3.42 ± 0.39 | 3.45 ± 0.40 | 3.59 ± 0.44 | 0.4714§ | |
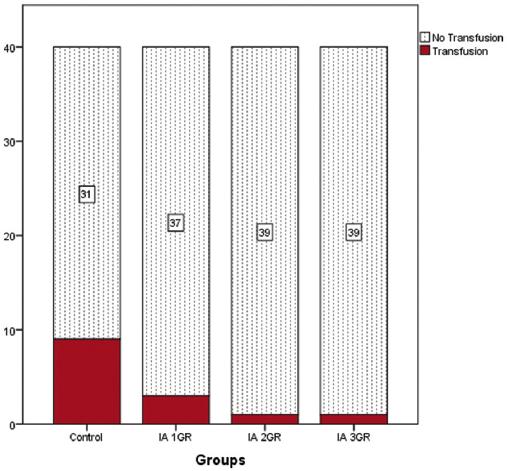
| D | Blood transfusion ( +/- ) (%) | 9/31 (22.5%) | 3/37 (7.5%) | 1/39 (2.5%) | 1/39 (2.5%) | 0.0044§ |
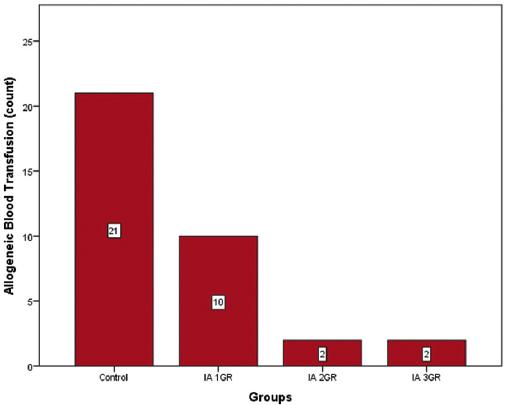
| Blood transfusion (count) | 21 | 10 | 2 | 2 | 0.0052† | |
| Blood loss from drain (mL) | 649 (250-1195) | 475 (200-1100) | 389 (120-1100) | 343 (100-1155) | 0.0012† | |
| Predictive blood loss (mL) | 1147 (457-2533) | 793 (144-2199) | 729 (198-1834) | 622 (129-2048) | 0.0012† | |
| Pbl/drain blood loss ratio | 1.61 (0.53-5.03) | 1.61 (0.29-5.03) | 1.74 (0.3-7.26) | 2.02 (0.3-5.07) | 0.1332† | |
| E | Hemoglobin value (g/dL)¶ | 11.00 ± 1.10 | 10.97 ± 0.91 | 11.02 ± 1.21 | 11.11 ± 1.05 | 0.9803‡ |
| Hematocrit value (%)¶ | 33.92 ± 3.45 | 34.27 ± 2.90 | 33.72 ± 3.64 | 33.99 ± 2.91 | 0.9843† | |
| Platelet count (103/mL)¶ | 369 (196-679) | 246 (239-325) | 345 (145-709) | 344 (205-882) | 0.3512† |
*Preoperative.
†Kruskal Wallis.
‡Anova.
§Chi-square.
¶Postoperative 3rd week.
**Range of motion.
The groups were compared in terms of ASA scores before surgery, joint range of motion measurement, surgical tourniquet time, and discharge times, and no statistically significant differences were found in any parameter (Table 1B).
The groups were compared in terms of hemoglobin (g/dL) before surgery, hematocrit (%), platelets (103/mL), and estimated body blood volume calculated according to the Nadler formula, and no statistically significant differences were found between the groups (Table 1C).
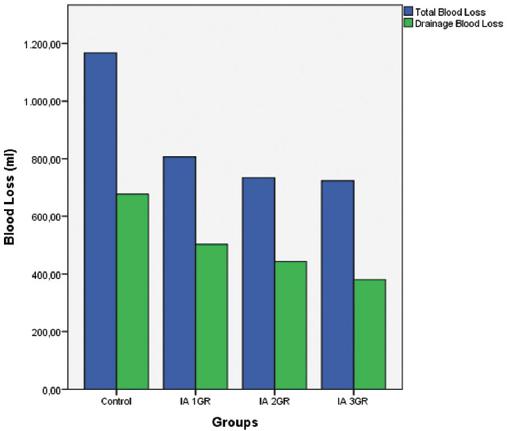
In the evaluation of post-surgical blood loss, the estimated total blood loss volume (PBL) (mL) calculated using the Gross formula, measurement of blood loss from drains (mL), and transfusion rate (%) were used (Table 1D). In statistical analyses, statistically significant differences were found in terms of estimated blood loss, transfusion rate, and blood loss from drains (p = 0.004/0.001/0.001) (Table 1D).
The relationship between total blood loss and blood loss from drains was examined, and it was found to be 1.61 (0.53-5.07) in the control group, 1.61 (0.29-5.03) in Group 1, 1.74 (0.3-7.26) in Group 2, 2.02 (0.53-4.46) in Group 3, and 1.74 (0.29-7.26) in all patients. In the analysis, no significant difference was found between the groups in terms of total blood loss/blood loss from the drain ratio. (p = 0.133) (Table 1D) (Fig. 1).
On the 20th day after surgery, no significant difference was found between the groups in terms of hemoglobin, hematocrit, and platelet counts (Table 1E). No deep surgical infections, PE, DVT, or VTEs were identified in the included patients.
When the groups were compared two-by-two, it was found that all TXA groups significantly reduced blood loss compared to the control group in terms of PBL (p = 0.001/0.001/0.001) and that there was no statistically significant difference between groups in terms of TXA dose (Table 2).
Table 2 Comparison of groups in terms of predictive blood loss and drainage blood loss
| p-values (p < 0.0083) | Group 4 (control) | Group 1 (IA 1GR) | Group 2 (IA 2GR) | Group 3 (IA 3GR) |
|---|---|---|---|---|
| Group 4 | 1.000*/1.000†/1.000‡ | 0.001§/0.001§/0.062 | 0.001§/0.001§/0.007§ | 0.001§/0.001§/0.007§ |
| Group 1 | 0.001§/0.001§/0.062 | 1.000/1.000/1.000 | 0.651/0.104/0.308 | 0.356/0.003*/0.308 |
| Group 2 | 0.001§/0.001§/0.007§ | 0.651/0.104/0.308 | 1.000/1.000/1.000 | 0.482/0.178/1.000 |
| Group 3 | 0.001§/0.001§/0.007§ | 0.356/0.003§/0.308 | 0.482/0.178/1.000 | 1.000/1.000/1.000 |
*Predictive blood loss.
†Drainage blood loss.
‡Allogeneic blood transfusion (%).
§Statistically significant level.
When the groups were evaluated in terms of transfusion rate (%), 9 patients in the control group (22.5%, 21 patients), 3 patients in the 1st group (7.5%, 10), 1 patient in the 2nd group (2.5%, 2), 1 in the 3rd group. It was determined that the patient (2.5%, 2 units) was transfused (2). Group 1 did not create a statistically significant difference in transfusion rates compared to the control group (p = 0.062), Groups 2 and 3 significantly reduced transfusion compared to the control group (p = 0.007/0.007), but there was a statistically significant difference between Groups 2 and 3 (p = 1.000) (Table 3). There was no significant difference between the control group and the first group (p = 0.133) in the double comparisons made after the difference was detected in the comparison of the number of transfusion parameters (p = 0.005). When compared to the control group, the number of transfusions was significantly lower in the 2nd and 3rd groups (p = 0.007/0.007), and no significant difference was found in the two-way comparisons between the drug groups (Table 3) (Figure 3).
Table 3 Comparison of groups in terms of allogeneic blood transfusion percentage and count
| p values (p < 0.0083) | Group 4 (control) | Group 1 (IA 1GR) | Group 2 (IA 2GR) | Group 3 (IA 3GR) |
|---|---|---|---|---|
| Group 4 | 1.000*/1.000† | 0.062/0.133 | 0.007‡/0.007‡ | 0.007‡/0.007‡ |
| Group 1 | 0.062/0.133 | 1.000/1.000 | 0.308/0.165 | 0.308/0.165 |
| Group 2 | 0.007‡/0.007‡ | 0.308/0.165 | 1.000/1.000 | 1.000/1.000 |
| Group 3 | 0.007‡/0.007‡ | 0.308/0.165 | 1.000/1.000 | 1.000/1.000 |
*Allogeneic blood transfusion (%).
†Allogeneic blood transfusion (count).
‡Statistically significant level.
When the groups were evaluated in terms of blood loss from drains, it was found that all TXA groups significantly reduced blood loss from drains compared to the control group (p = 0.001/0.001/0.001). In intra-group comparisons, it was found that the application of 3 g of TXA significantly reduced blood loss from drains compared to the application of 1 g of TXA (p = 0.003), but there was no significant difference between the application of 2 g and 3 g of TXA (p = 0.178) (Table 2).
Discussion
TXA is a cost-effective drug that has been used for bleeding control for approximately 50 years. There are many publications and meta-analyses stating that IA applications of TXA start to show its effect at doses of 1 g and above, and its use up to 3 g is effective and safe1-4,10-16.
A meta-analysis conducted by Xu et al. included 211 publications and found that TXA applications had lower transfusion rates compared to control groups and that IA (topical) use was a safe method in cases with high thrombosis risk11. In a study conducted by Kim et al., 0.5, 1, 2, and 3 g IA administration doses of TXA were compared with the control group14. In this study, TXA was found to be effective in controlling bleeding at all doses of 1 g and above, and therefore it is recommended to use TXA IA in a 1 g dose. In a study by Sahin et al., the effectiveness and safety of TXAs 2 g IA use was compared with the various combinations and doses of systemic use of TXA, and it was found that IA use of 2 g was effective and safe1. In the literature, the effectiveness of TXA in IA applications below 1 g is controversial, and therefore the lowest IA TXA group in our study was taken as 1 g.
In our study, one of the most important parameters was transfusion rates. In a study conducted by Kim et al.,14 different IA TXA doses were compared to a control group; a 19% transfusion rate was found in the control group, a 9% transfusion rate in the 1 g group, a 10% transfusion rate in the 2 g group, and an 8% transfusion rate in the 3 g group. In another study conducted by Sahin et al.1, the transfusion rate was found to be 17.64% in the control group and no transfusion was required in the 2 g IA TXA group (0%)1. In a study conducted by Georgiadis et al., the transfusion rate in the control group was 8%, whereas the rate in the TXA group was 0%; however, there was no statistically significant difference (p = 0.118)13. In a study by Lee et al., the transfusion rate in all groups treated with combined IA, IA + intravenous low and high doses of TXA was found to be 0%15. In our study, transfusion was performed at a rate of 22.5% in the control group, 7.5% in the 1 g TXA group, and 2.5% in both the 2 and 3 g TXA groups. Unlike the study by Kim et al.,14 in our study, there was no statistically significant difference in the transfusion rates between the control group and the 1 g TXA group. In addition, there was no statistically significant difference in the transfusion rates between the 2 and 3 g TXA groups. While our transfusion rates seem to be consistent with the literature, according to the meta-analysis conducted by Mi et al., transfusion is performed in 10 g/dL or less in symptomatic patients and in 8 g/dL or less in all patients in the literature, while in our study, transfusion was limited to 8 g/dL or less in symptomatic patients, and 6 g/dL or less in all patients according to the ASA criteria and the decisions of the hospital transfusion committee2.
In our study, the estimated total blood loss due to surgery was investigated. In a meta-analysis by Moskal and Capps8, the estimated total blood loss due to surgery was found to be 834.40 mL (640.48-1028.3) in the IA TXA groups and 1195.32 mL (935.63-1455.0) in the control groups. In a study conducted by Kim et al., TKA surgery was associated with a blood loss of 1503 ± 681 mL in the control group, 1190 ± 379 mL in the 1 g IA TXA group, 1147 ± 432 mL in the 2 g IA TXA group, and 1171 ± 299 mL in the 3 g IA TXA group, and no significant difference was found between the drug groups14. In a multicenter randomized controlled study conducted by Stowers et al.22, the blood loss in the IA 1.5 g TXA application group was 723 mL (620-826), and in the placebo group, it was 1090 mL (923-1257)21. In the study conducted by Şahin et al.1, 832 mL (403-1409) in the 2 g IA TXA group and 1309 mL (408-2119) in the control group were determined. In our study, blood loss was 1147 mL (457-2533) in the control group, 793 mL (144-2199) in the 1 g IA TXA group, 729 mL (198-1834) in the 2 g IA TXA group, and 622 mL (129-2048) in the 3 g IA TXA group. In our study, according with the literature, a significantly lower amount of bleeding was detected in the drug groups compared to the control group. However, no significant difference was found between the drug groups.
In our study, post-surgical drainage blood loss was an important parameter investigated. In a meta-analysis by Moskal and Capps8, post-operative blood loss from drains was found to be 353.69 mL (279.68-427.70) in the IA-TXA groups and 534.66 mL (410.28-659.05) in the control groups. In the study of Kim et al., 307.4 ± 237.7 mL of drainage blood loss was detected in the control group, 259 ± 163.4 mL in the 1 gr IA TXA group, 261.8 ± 163.9 mL in the 2 g IA TXA group, and 258.0 ± 110.8 mL in the 3 g IA TXA group14. In our study, 649 mL (250-1195) of drainage blood loss was detected in the control group, 475 mL (200-1100) in the 1 g group, 389 mL (120-1100) in the 2 g group, and 343 mL (100-1155) in the 3 g group. The relationship between drainage blood loss and total blood loss was investigated and it was determined that the ratio of the amount of drainage blood loss to total blood loss was similar at a level that did not create significant differences between the groups (1.61-2.02).
One of the most significant and serious complications of TXA that has been identified in the literature is that it creates the foundation for thromboembolic events such as DVT, PE, and VTE. A meta-analysis conducted by Moskal and Capps8 showed that the use of IA TXA does not increase the risk of thromboembolic interactions compared to other modes of use of TXA and that TXA is a cost-effective solution for total knee arthroplasty surgeries in centers with a transfusion rate of more than 25%. A meta-analysis conducted by Xu et al.11 found that any mode of use of TXA did not increase the risk of thromboembolic events when compared to the control group and that topical TXA application was effective and safer. A meta-analysis conducted by Mi et al.2, which analyzed the results of a total of 1308 patients showed that intravenous or IA administration of TXA did not increase the risk of thromboembolic events or deep surgical infection. Moskal and Capps8 found the DVT level between 2.44% and 2.55% for the IA and control groups, and no increased risk associated with the use of TXA was found. In our study, no thromboembolic events or deep surgical infections were observed in the drug groups, consistent with the literature.
Conclusion
In our study, we aimed to contribute to the literature by evaluating the effectiveness of IA TXA application on the amount of blood loss and transfusion rates in total knee arthroplasty surgeries, by comparing different doses proven to be effective in the same study (total of 3 drugs and 1 control group).
In our study, the use of 1 g TXA was not significantly effective when compared to the control group, and the use of 3 g TXA was not superior in any parameter in terms of the preventive effectiveness of blood loss compared to the use of 2 g TXA. According to our study, the use of more than 2 g of TXA IA did not increase the effectiveness of preventing blood loss in total knee replacement surgeries. Therefore, IA use of 2 g TXA is a cost-effective dose in total knee replacement surgeries, and no increase in anti-bleeding efficiency was found with dose increase. We concluded that the cost-effective dose to prevent bleeding in total knee replacement surgeries is 2 g and should be used at this dose.
When the meta-analyses in the literature are examined, the blood transfusion indications and the blood transfusion indications in our study are different for the reasons stated in the publication. Characteristics are an important shortcoming in the evaluation and statistical analysis of transfusion rates. In addition, the relationship between TXA use and returning to daily activities after surgery is important, and the inability to make this comparison in our study is an important shortcoming. Although no significant difference was found between the control and drug groups in terms of blood parameters taken on the 20th day in our study, more comprehensive studies are needed to evaluate the relationship between TXA and activity level in the early postoperative period.











 nueva página del texto (beta)
nueva página del texto (beta)