Introduction
Significant advances have been made in deep learning (DL), a subset of machine learning and artificial intelligence (AI), in the field of healthcare, particularly in orthopedic surgery. By comprehending complex algorithms, deep learning has the potential to revolutionize diagnosis, treatment planning, prediction of surgical outcomes, and even surgical procedures.
DL and machine learning (ML) methods enable computers to learn from data and make decisions or predictions without being programmed to perform specific tasks. In simple terms, ML involves teaching computers to recognize patterns or trends in data, while DL, a subset of ML, uses layers of algorithms called neural networks to analyze these data patterns more deeply.1
In the field of medical science, deep learning has become particularly valuable in analyzing medical images. By using neural networks, DL algorithms can detect intricate patterns in images such as X-Rays or MRIs, enabling them to diagnose diseases such as cancer with a high degree of accuracy. This technology acts like an expert radiologist but can work around the clock and process information at speeds and volumes far beyond human capabilities.2,3
The integration of DL and ML into medical science, especially in orthopedics, could revolutionize how we diagnose, treat, and develop interventions. These technologies significantly advance patient care, improve treatment outcomes, and drive innovation in medical research and practices. As this field continues to evolve rapidly, it holds great promise for even more remarkable breakthroughs and applications in enhancing health and quality of life.4
Advancements in deep learning in orthopedics
Preoperative planning
Deep learning algorithms have been successful in preoperative planning, particularly in predicting outcomes and complications. It also helps in selecting the most appropriate surgical approach.5
Image interpretation
Deep learning algorithms have been developed for image interpretation and have achieved expert-level accuracy in diagnosing different orthopedic conditions through MRI, X-Ray, or CT scan readings.1,2,6
Prosthesis design
AI can aid in the customization of prosthetics, leading to a better fit and increased functionality for individual patients.3,7
Robotic orthopedic surgery
Artificial intelligence-based robotics systems have gained ground in orthopedic surgery, such as hip and knee replacements, leading to greater precision and potentially better patient outcomes.8
Material and methods
This systematic review was carried out following the procedures highlighted in the ‘PRISMA-DTA’ extension for diagnostic validity studies, as well as following systematic review guidelines.9
Selection criteria
We sought studies that reveal the diagnostic accuracy and uses of DL algorithms in identifying pathologies or diseases of orthopedic interest. The primary objective was to identify the type of studies and range of diagnostic accuracy metrics. The secondary objectives included the study design and quality of reporting.
Data collection and searches
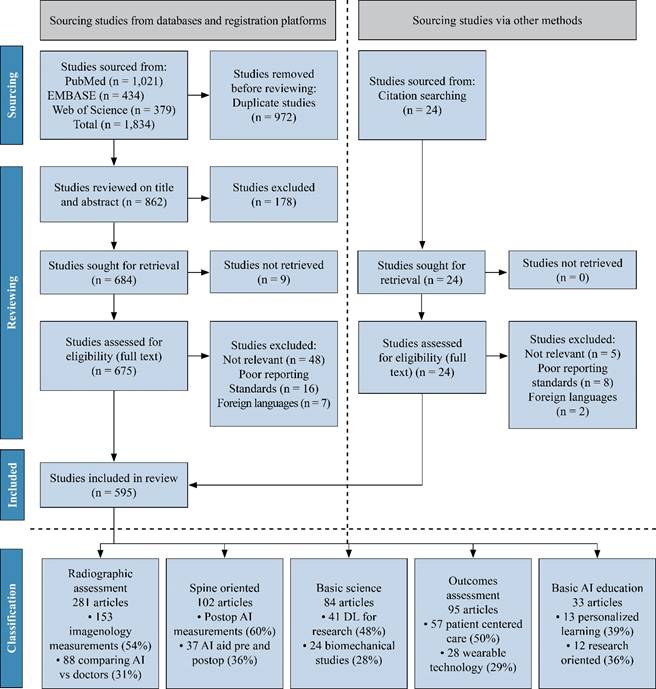
Electronic literature searches in PubMed, EMBASE and Google Scholar were also conducted from January 1st, 2020, up to October 31st, 2023. The search terms used consisted of Mesh terms and all-field search terms for «orthopedic deep learning» and «neural networks» (examples are DL, convolutional networks, etc.) and specific terms such as «imaging» (such as magnetic resonance, computed tomography, ultrasound, or X-Ray), and the results were used as «diagnostic accuracy metrics» (sensitivity and specificity). We identified 862 studies, 595 of which were included in the systematic review. A total of 281 studies about radiographic assessment, 102 about spine-oriented surgery, 95 about outcome assessment, 84 about basic AI orthopedic education and 33 about basic science application were included for review. Peer-reviewed studies that reported on the accuracy of DL algorithms for identifying pathology using medical imaging were included. Primary outcomes were diagnostic accuracy, study design and reporting standards reported in the literature. Estimates were pooled using random effects meta-analysis (Figure 1).
Inclusion criteria
Eligible studies included those evaluating the diagnostic accuracy of a DL algorithm in the orthopedic field in general and in subspecialties. Only studies that reported either diagnostic accuracy raw data or sensitivity and specificity data were incorporated in the systematic review. No restrictions were imposed on the date range, and the most recent search took place in November 2023.
Exclusion criteria
Articles not written in English were excluded. Abstracts, conference articles, preprints, reviews, and meta-analyses were not considered as the purpose was to scrutinize the methodology, reporting standards, and quality of primary research studies appearing in peer-reviewed journals.
Selection process
The study selection process entailed an initial review by two different reviewers of all titles and abstracts. This was followed by a second review by the third and fourth authors of the full-text articles. This process ensured that the remaining articles met the inclusion criteria.
A total of 153 different imaging methods for radiographic aspects were identified. A total of 185 different ML algorithms were used, with the convolutional neural network architecture being the most common (73%). Improving diagnostic accuracy and speed was the most common method used (62%)
Review
This review focuses on five principal applications of machines and deep learning methodologies in orthopedics. The key areas of focus include (1) evaluation through radiography, (2) spine-focused surgical interventions, (3) basic orthopedic science, (4) evaluation of outcomes in general orthopedic surgeries, and (5) fundamental artificial intelligence (AI) instruction for orthopedic surgeons. Given the expansive use of AI in orthopedic surgery, it is impractical to cover every facet of this review. Instead, we have opted to concentrate on a select few applications that we believe encapsulate and effectively summarize the enhancements associated with AI and deep learning; we summarize these reviews with examples in Table 1.
Table 1: Summary of deep learning techniques applied to orthopedic literature.
| Study | Aims | Deep learning method | Application |
|---|---|---|---|
| Staartjes, et al.(5) | Evaluate the feasibility of deriving robust deep learning-based predictive analytics from single-center, single-surgeon data | Deep neural network-based and logistic regression-based prediction models | Basic AI education Outcomes assessment |
| Kang, et al.(7) | Develop a machine learning–based implant recognition program and to verify its accuracy | Object detection and clustering. Model training with Keras deep learning platform | Basic AI education |
| Moon, et al.(14) | Automatically, rapidly, and accurately detect lower limb alignment by using anteroposterior standing X-Ray | YOLOv5 (Vision AI) HarDNet-MSEG image segmentation model |
Radiographic assessment |
| Tan, et al.(23) | Identify biomarkers and develop an integrated diagnostic model for predicting the onset of early intervertebral disc degeneration | LASSO, random forest, and support vector machine recursive feature elimination | Basic science |
| Anastasio, et al.(24) | Identify combinations of orthobiologic factors applied to bone healing/fusion | Artificial neural networks | Basic science Basic AI education |
| Yan, et al.(25) | Segment chondrocytes from histological images of cartilage | U-Net (convolutional neural network) | Basic science |
| Melgoza, et al.(26) | Report a new robust quantitative mouse intervertebral disc degeneration histopathological scoring system | Artificial neural networks and multilayer perceptron | Basic science |
| Zhu, et al.(28) | Develop a predictive model for postoperative osteonecrosis of the femoral head | MATLAB convolutional neural network | Basic AI education outcomes assessment |
| Maki, et al.(35) | Develop a predictive model for surgical outcomes in patients with cervical ossification of the posterior longitudinal ligament | Machine learning, LightGBM, deep learning, RadImagenet | Spine-oriented surgery Radiographic assessment |
| Patel, et al.(36) | Develop and evaluate a model for identifying orthopedic implants using radiographs | Seven convolutional neural networks U-Net segmentation network |
Basic AI education Radiographic assessment |
| Shen, et al.(37) | Developed an MRI-based system to detect early osteonecrosis of the femoral head | Convolutional neural network | Radiographic assessment |
| Guy, et al.(38) | Make the diagnosis of proximal femur fracture on radiographs | Deep learning algorithm (TensorFlow) | Radiographic assessment |
| Guo, et al.(39) | Developed a model for diagnosing supraspinatus tears shoulder MRI | Convolutional neural networks (Xception) | Radiographic assessment |
| Yi, et al.(40) | Identification, classification and differentiation for knee arthroplasty implants | Deep convolutional neural network | Radiographic assessment |
| Klempt, et al.(41) | Develop and validate a model for identification of hip and knee joint arthroplasty designs from plain radiographs | Convolutional neural network | Radiographic assessment |
| Merkely, et al.(42) | Identify cartilage defects when applied to the interpretation of knee MRI | Three convolutional neural networks | Radiographic assessment |
| Yamamoto, et al.(43) | Statistically evaluate the osteoporosis identification ability | ResNet convolutional neural network | Radiographic assessment |
| Tiwari, et al.(44) | Identify the most appropriate -based model for the detecting grade of knee osteoarthritis | Eight machine learning models (DenseNet) | Radiographic assessment Basic AI education |
| Leung, et al.(45) | Develop a prediction model for risk of knee osteoarthritis progression | Transfer learning approach (ResNet34) | Radiographic assessment |
| Borjali, et al.(46) | Develop a model for efficient and accurate hip dislocation detection following primary total hip replacement from medical narratives | Convolutional neural network natural language processing models | Basic AI education |
| Eweje, et al.(47) | Develop an algorithm that can differentiate benign and malignant bone lesions using routine MRI | EfficientNet-B0 architecture and a logistic regression model | Radiographic assessment |
| Ashkani, et al.(48) | Assess the performance of two different networks in detecting ankle fractures using radiographs | Deep convolutional neural networks Inception V3 and ResNet50 |
Radiographic assessment |
| Li, et al.(49) | Improve the diagnostic accuracy and efficiency for diagnosing meniscal tear using MRI | Mask regional convolutional neural network. ResNet50 | Radiographic assessment |
| Magneli, et al.(50) | Train and evaluate a model for AO/OTA classification of shoulder fractures | A modified CNN of the ResNet architecture | Radiographic assessment |
| Shen, et al.(51) | Exploratory investigation for the classification and prediction of mechanical states of cortical and trabecular bone tissue | Convolutional neural networks. ResNet with transfer learning | Basic science Basic AI education |
| Lau, et al.(52) | Build an image-based machine-learning model for detecting TKA loosening | Random forest classifier Xception Model, ImageNet and TensorFlow |
Radiographic assessment Basic AI education |
| Kim, et al.(53) | To automatically select and position THA components that are most suitable for the patient’s bone anatomy | Convolutional neural network | Basic AI education |
| Recht, et al.(54) | Accelerate MRI to allow a 5-minute comprehensive examination of the knee | Variational network U-Net |
Basic science Basic AI education |
| Borjali, et al.(55) | Increase accuracy, accelerate analysis time, and reduce interobserver bias by automating 3D volume assessment of syndesmosis anatomy | Three deep learning models | Radiographic assessment Basic AI education |
| Yang, et al.(56) | To assess the severity of knee osteoarthritis in portable devices | RefineDet Deep learning-based diagnostic model |
Basic AI education Radiographic assessment |
| Hernigou, et al.(57) | Provide an overview of the possibility to predict dislocation with a calculator according to the type of implant for THA | Supervised learning model Artificial neural network | Basic AI education Outcomes assessment |
| Rahman, et al.(58) | To detect loosening of the hip implant using X-Ray images | Deep Convolutional Neural Networks based novel stacking approach (HipXNet) | Basic AI education Radiographic assessment |
| Wang, et al.(59) | Develop a recovery and nursing system after total hip arthroplasty and to conduct clinical trials | Deep neural network based on Mask R-CNN | Basic AI education Outcomes assessment |
| Kinugasa, et al.(60) | Evaluate the accuracy of diagnoses made by AI on ultrasound images of developmental dysplasia of the hip | MATLAB deep learning toolbox | Basic AI education Radiographic assessment |
| Sharifi, et al.(61) | Identify spatiotemporal gait parameters, gait patterns, activity types, and changes in mobility after total knee arthroplasty | Six contemporary multivariate time series neural network architectures | Basic AI education Outcomes assessment |
| Li, et al.(62) | Evaluate the performance of DL in differentiation of benign and malignant vertebral fracture on CT | ResNet50 network | Radiographic assessment |
| Rouzrokh, et al.(63) | Identify all pelvic and hip radiographs with appropriate annotation of laterality and presence or absence of implants | Two deep-learning algorithms EfficientNetB3 classifier YOLOv5 object detector |
Basic AI education Radiographic assessment |
| Huang, et al.(64) | Automated segmentation and quantification of the vertebrae and intervertebral discs on lumbar spine MRIs | Deep learning-based program (Spine Explorer) | Radiographic assessment Spine-oriented surgery |
| Kong, et al.(65) | Develop an X-Ray-based fracture prediction model using deep learning with longitudinal data | Convolutional neural network. DeepSurv | Radiographic assessment Spine-oriented surgery |
| Zhao, et al.(66) | Create a reliable learning-based approach that provides consistent and highly accurate measurements of the Cobb angle | Convolutional neural network Deep learning SpineHRformer |
Radiographic assessment |
| Wang, et al.(67) | Analytic function for the correlation between lumbar disc herniation and angle and irregular variation of joint of lumbar facet-joint | Convolutional neural network-Based MRI image recognition algorithm | Radiographic assessment Spine-oriented surgery |
| Broida, et al.(68) | To more accurately screen surgical candidates seen in a spine clinic. | Transformer-based machine learning architecture | Spine-oriented surgery Basic AI education |
| Ito, et al.(69) | Predict postoperative complications in patients with cervical ossification of the posterior longitudinal ligament | Deep learning was used to create two predictive models | Spine-oriented surgery Outcomes assessment |
| Etzel, et al.(70) | Predict and classify whether a patient will experience a short or long hospital LOS after lumbar fusion surgery with a high degree of accuracy | Six machine learning algorithmic analyses | Spine-oriented surgery Outcomes assessment |
| Maras, et al.(71) | Differentiate normal cervical graphs and graphs of diseases that cause mechanical neck pain | Convolutional neural networks VGG-16, VGG-19, Resnet-101, and DenseNet-201 networks | Radiographic assessment |
| Campagner, et al.(72) | Define an invasiveness score for LBP procedures based on biological markers and inflammatory profiles | Supervised machine learning | Spine-oriented surgery Outcomes assessment |
| Liu, et al.(73) | Diagnosis of benign and malignant spinal tumors based on magnetic resonance imaging | Multimodel weighted fusion framework | Radiographic assessment |
| Müller, et al.(74) | Predict the multidimensional outcome of patients undergoing surgery for degenerative pathologies of the thoracic, lumbar or cervical spine | Convolutional neural network | Spine-oriented surgery Outcomes assessment |
| Mandel, et al.(75) | Forecasting the outcome of vertebral body growth modulation from skeletally immature patients | Spatial-temporal corrective networks | Spine-oriented surgery Outcomes assessment |
| Fan, et al.(76) | Simulated foraminoplasty of percutaneous endoscopic transforaminal discectomy | Deep learning-derived 3D (DL-3D) models | Spine-oriented surgery |
| Chen, et al.(77) | Analyze perioperative factors and predict the occurrence of surgical site infection following posterior lumbar spinal surgery | LASSO regression analysis, support vector machine, and random forest | Spine-oriented surgery Outcomes assessment |
| Mu, et al.(78) | Application value of magnetic resonance spectroscopy and computed tomography in the treatment of lumbar degenerative disease and osteoporosis | Deep convolutional neural network image segmentation processing technology | Spine-oriented surgery Basic AI education |
| Cho, et al.(79) | Automatically detect the tip of the instrument, localize a point, and evaluate the detection accuracy in biportal endoscopic spine surgery | RetinaNet and YOLOv2 | Spine-oriented surgery Basic AI education |
| Silva, et al.(80) | Predict spine surgery outcome | Boosted decision tree classifier (SpineCloud) | Spine-oriented surgery |
| Kuris, et al.(81) | Determine whether it could predict readmission after 3 lumbar fusion procedures | Neural network, a supervised machine learning technique | Spine-oriented surgery Outcomes assessment |
| von Atzingen, et al.(82) | Marker-less surgical navigation proof-of-concept to bending rod implants | Augmented reality with on-device machine learning | Spine-oriented surgery Basic AI education |
| Tran, et al.(83) | Semantic segmentation on X-Ray images | Multipath convolutional neural network, BiLuNet | Basic AI education Radiographic assessment |
| Chen, et al.(84) | Identify the possibility of THR in three months of hip joints by plain pelvic radiography | Sequential two-stage deep learning algorithm HipRD and SurgHipNet | Basic AI education Radiographic assessment |
| Niculescu, et al.(85) | Comparative study of the biomechanical behavior of commonly used orthopedic implants for tibial plateau fractures | Artificial Neural Network model | Basic science Basic AI education |
| Bonnheim, et al.(86) | Calculating biomarkers of cartilage endplate health using MRI images | Four independent convolutional neural networks | Basic science Radiographic assessment |
| Kasa, et al.(87) | Assess surgical performance with comparable performance to the expert human raters | Multimodal deep learning model | Basic AI education Outcomes assessment |
| Loftus, et al.(88) | Reproducibility of an automated postoperative triage classification system | Deep convolutional neural network | Basic AI education |
Application 1: radiographic assessment
DL, an AI technology, possesses the ability to analyze and interpret complex medical imaging, a capability that paves the way for more accurate diagnoses and patient-specific treatments.10 Studies have shown that DL can process X-Rays, CT scans, and MRI images effectively to identify specific pathologies, such as fractures, osteoarthritis, bone tumors, deformities, and degenerative diseases.11,12 Radiographic parameters are instrumental for accurately determining disease progression, planning surgeries, and predicting postoperative outcomes. Moreover, this approach has the potential to analyze biomechanical data and perform automated measurements, which are traditionally time-consuming.13
In addition to improving diagnostic accuracy, its application also brings forth the potential for expedited and optimized medical workflows. With its ability to swiftly analyze large volumes of radiographic data, clinicians can rapidly plan treatment trajectories and focus intensively on critical patient care.14,15 Moreover, DL models can easily facilitate the detection of subtle or complex patterns in images, which may be challenging even for experienced radiologists. This could enhance early detection and intervention for debilitating conditions, eventually improving patient outcomes. Additionally, DL can serve as an educational tool to aid trainees and junior clinicians in enhancing their understanding and interpretation of orthopedic imaging.16 Certainly, key challenges must be considered, such as ensuring the ethical use of patient data, maintaining transparency in AI decision-making processes, and eliminating interoperability issues among various healthcare systems.
However, despite its promising advantages, DL implementation in orthopedics must be approached with caution due to its dependency on the quality and size of the dataset used for training. Hence, additional research is necessary to overcome these potential limitations and standardize the use of DLs in orthopedic radiographic assessments in pursuit of precision medicine. Overcoming these obstacles will be crucial for fully realizing the potential benefits and advancements that DL offers to the field of orthopedic radiographic assessment.
Application 2: spine-oriented surgery
DL has presented significant advancements, particularly in spine-oriented surgical procedures. Researchers and practitioners are actively exploring and utilizing DLs to assist in areas ranging from diagnosis to surgical planning and prognosis prediction.17 Specific examples include the detection and classification of spinal disorders such as scoliosis, spondylolisthesis, and intervertebral disc degeneration from radiographic images. DL models have also been designed to assist in surgical planning by predicting pedicle screw placement, reducing the chances of iatrogenic injury.18,19
In addition to its use in diagnostic and treatment planning, the role of DL in enhancing intraoperative guidance and postoperative rehabilitation in spinal surgeries also demonstrates significant promise. With its advanced pattern recognition capabilities, DL can potentially provide real-time feedback during spinal surgeries, thereby increasing the precision and safety of procedures.20,21 For instance, DL algorithms can aid in identifying anatomic landmarks, enhancing visualization, and assisting tool navigation, subsequently reducing the risk of medical errors.
However, complete adoption of DL in spine-related orthopedic practices remains a challenging task. Despite the considerable progress, issues such as data privacy, algorithmic transparency, and the need for interdisciplinary collaboration between data scientists and clinicians need to be adequately addressed. As the potential of DL continues to be explored, more comprehensive guidelines governing its application in spinal surgeries have yet to be established to leverage its benefits effectively.
Application 3: Basic science (biomechanics/pathogenesis)
In the realm of basic science in orthopedics, the incorporation of DL has amplified the possibility of understanding intricate details and processes at the molecular, cellular, and tissue levels. The established DL algorithms can supplement traditional research methods by analyzing complex patterns and interrelationships among numerous biological variables. They can handle vast multidimensional datasets, aiding in biomarker discovery, modeling disease progression, and identifying potential therapeutic targets.22
DL can aid genomic research in orthopedics, where it could be used to understand patterns of gene expression pertaining to bone growth, determine the underlying genetic causes behind orthopedic diseases, or predict patient responses to treatments at the genetic level. Similarly, at the tissue level, DL-powered image analysis can provide a more in-depth analysis of histopathological samples, potentially enabling early detection of degenerative diseases and comprehensive assessment of cellular responses to different interventions.25
Moreover, DL can complement biomechanical studies by facilitating the analysis of complex motion patterns and forces, providing deeper insights into the effects of various physical activities on the musculoskeletal system.26
Application 4: outcomes assessment
DL has proven to be a critical tool in orthopedics for assessing patient outcomes. By analyzing vast sets of patient data, DL models can predict patient-specific outcomes following various interventions, thereby enabling a more personalized approach to patient care. Moreover, this approach can help identify factors contributing to optimal and suboptimal outcomes, facilitating improvements in therapeutic strategies.27,28 DL algorithms can be used to mine data to uncover complex, non intuitive correlations between patient characteristics, intervention details, and postoperative outcomes. Such correlations could be used to predict future patient recovery patterns, incidence of complications, or even the likelihood of rehospitalization, allowing clinicians to effectively plan and adjust treatments and follow-up schedules.29
Furthermore, DL can be used to analyze real-time patient data collected through wearable technology to provide a comprehensive understanding of patient function and recovery in real-world settings. By mining these rich data sources, DL can potentially uncover nontraditional metrics of orthopedic outcomes, which may prove relevant in achieving patient-centered care.29,30 DL can decipher patterns in movement data to accurately assess rehabilitation progress following joint replacement or reconstructive surgeries. Likewise, this study can provide insights into the adherence of patients to prescribed rehabilitation protocols and enable tailored interventions for improving patient compliance.31,32
While this fusion of DL and wearable technology opens a new dimension in outcome research in orthopedics, key issues, including patient acceptance, data privacy, and data validity, need careful deliberation. Nonetheless, with continuous refinement and stringent validation practices, the integration of DL can undoubtedly drive a paradigm shift in outcome assessment, taking orthopedic care a step closer to the aim of optimized, patient-centered care. As we move forward, the effective application of DLs can significantly enhance the quality and efficacy of orthopedic care by ensuring that interventions align with individuals’ expectations and desired outcomes.
Application 5: fundamental AI education for orthopedic surgeons
DL, as a subset of AI, has been progressively weaving its way into medical education, including orthopedics. With its capacity to process vast and complex datasets, DL can contribute significantly to bolstering both theoretical knowledge and practical skills among medical students and professionals in the orthopedic field. For example, DL algorithms can assist in creating immersive, personalized learning experiences by identifying individual learning patterns and offering tailored educational content.33
Beyond direct educational functions, DL can serve as an integral tool in fostering research literacy among medical students and professionals in orthopedics. With increasing focus on evidence-based practice in the current healthcare landscape, having skills to conceive, conduct, and interpret research is now considered equally important as clinical skills. DL can equip learners to handle big data analytic tasks, analyze complex research data, and more accurately interpret the findings of a study.34 Additionally, DL can be used as a pedagogical tool to teach foundational concepts of bioinformatics relevant to orthopedics, such as genomic studies in osteoarthritis or proteomic data analysis in bone healing.
The introduction of AI-driven research concepts in medical education faces several hurdles, including the need for curricular adaptations, lack of skilled educators, and the risk of overreliance on algorithms at the expense of contextual decision-making. Despite these challenges, the incorporation of DL in medical education is a necessary advancement that can mold a generation of medical professionals competent in both clinical and research domains of orthopedics.
Importantly, familiarizing future orthopedic practitioners with AI concepts and their uses also becomes fundamental to nurturing a workforce that is adept at leveraging DL in clinical problem solving.
Limitations and challenges
Certain challenges surfaced in the literature under review include the ‘black box’ nature of neural network models, which makes it difficult for users and orthopedic surgeons to comprehend and explain the basis of the outcomes produced. Moreover, there are hindrances to the applicability and implementation of these models across various healthcare institutions due to variations in standardization, procedures, and dataset parameter availability. Even when dataset parameters are available, their values may differ because of varying demographics and geographical locations. These limitations may influence the overall accuracy and predictive potency of the artificial intelligence model and could pose a risk to patients if implemented indiscriminately. Thus, further investigations and trials are indispensable for confirming the efficacy of the developed model.
Despite its impressive potential, it is crucial to note that the success of DL applications relies primarily on the quality, size, and diversity of the data used for model training. The full integration of DL into surgery, therefore, requires additional studies, robust data handling techniques, and stringent validation measures to ensure accurate and reliable utilization in clinical practice.
Future considerations
The integration of advanced technologies such as DL and AI into orthopedics marks a significant shift toward more precise, personalized, and efficient care in the field. Currently, these technologies are being applied to improve diagnostic processes, such as interpreting X-Rays and MRI scans with remarkable accuracy and modeling and simulating surgical planning. In the future, orthopedics could be profoundly impacted by further advancements in DL and AI. These technologies hold the promise of enhancing diagnostic accuracy, streamlining treatment planning, optimizing surgical interventions through robotic assistance, and tailoring patient care to individual needs with unprecedented precision. Furthermore, AI-driven predictive models could pave the way for proactive management of orthopedic conditions, potentially improving long-term outcomes.
The field of radiology research has widely embraced deep learning, a testament to the potential of this technology to revolutionize medical imaging analysis. A noticeable trend in the plethora of medical literature is the frequent use of deep learning models applied through different conception methodologies. The replication observed in radiology studies emblematic of the community’s determination to fine-tune the accuracy of these models, emphasizing the importance of deriving diverse methodologies tailored to distinct datasets and clinical scenarios, despite the commonality of the underlying algorithms. While the repetitive use of similar models facilitates a deeper understanding of their capabilities and limitations, it also poses the challenge of potentially stalling innovation by focusing on incremental improvements rather than groundbreaking discoveries.
However, this approach has undeniably propelled radiology forward, improving image analysis, helping early and accurate diagnoses, and initiating a continual dialog on best practices. In sum, although the repetitive nature prevails in radiological deep learning research, its presence is pivotal both for incremental progress and for highlighting areas where novel contributions are crucial for further advancements in the field, urging researchers to persistently seek innovative solutions.
The widespread adoption of AI in orthopedic practice must carefully address potential challenges, including data privacy concerns, the need for substantial training datasets, and ensuring equitable access to AI-driven care. Ethical considerations around patient autonomy and the role of AI in clinical decision-making also require thoughtful addressing.
Conclusion
Advancements in deep learning offer immeasurable opportunities in orthopedic surgery, from aiding in diagnosing complex conditions to performing surgical procedures with greater precision than ever before. However, integrating deep learning algorithms into orthopedics is associated with numerous challenges, including ethical concerns about data privacy and regulating the use of AI in clinical environments. More research is needed to validate and refine these tools and strategies so they can be safely and effectively incorporated into practice.
Heterogeneity was high between studies, and extensive variation in methodology, terminology and outcome measures was noted. This can lead to an overestimation of the diagnostic accuracy of DL algorithms for medical imaging. There is an immediate need for the development of artificial intelligence-specific guidelines to provide guidance around key issues in this field.
DL is a burgeoning field with immense potential across all areas of healthcare, particularly radiology. The present systematic review and meta-analysis scrutinized the quality of the literature and summarized the diagnostic accuracy of DL techniques. Despite the high diagnostic accuracy of DL currently available, interpreting these findings is vital considering the poor design, conduct, and reporting of studies, which could result in bias and overestimation of the power of these algorithms. Improving DL application requires standardized instructions related to study design and reporting, which can further clarify its clinical utility.











 nueva página del texto (beta)
nueva página del texto (beta)