Introduction
The Mayan cichlid, Mayaheros urophthalmus (Günter 1862), is a locally consumed species commonly known as the "mojarra castarrica", which is caught throughout the year (Viedma et al. 2011). This endemic species is common in southeastern Mexico, making it a widely exploited resource (Fernández-Pérez 2024). Its natural distribution in Mexico ranges from the Papaloapan River basin in southern Veracruz, Tabasco, Chiapas, Campeche, and the states of the Yucatan Peninsula (Miller 2009). Its exceptional tolerance to changes in salinity, temperature, and water level enhances its viability for range expansion, as its ability to colonize new habitats is extensive (Loftus 1987). This high tolerance also makes it susceptible to aquaculture exploitation. However, there is not enough information on its reproduction in captivity.
M. urophthalmus has been reported as a multiple spawner due to various stages of oocyte development in a mature ovary. Mature gonads are found throughout the year, with high reproductive activity during the season of elevated temperatures and the presence of rainfall (Chávez-Lomelí et al. 1989). The species is characterized by low fertility and large oocyte sizes associated with paternal brood care. The fact that it is a multiple spawner indicates that it directs energetic investment towards high offspring survival rather than the number of offspring generated, with some modifications associated with adaptation to water and environmental change throughout the year (Lang and Fitzhugh 2015). Fecundity plays a key role in reproduction and is regulated by environmental conditions, food availability, predation, and genetic factors (Kjesbu et al. 2003, Ganias 2013, Rickman et al. 2000). Due to greater ecological stability, the combination of low fecundity and large egg size is a characteristic of demersal spawners. Multiple spawnings increase the chances of survival, mainly due to increased fecundity per reproductive season and reduced competition among offspring. This suggests that the species invests heavily in larval survival at the expense of the number of offspring (De-Sousa et al. 2008). In cichlids, survival is favored by factors such as large oocyte size, large egg yolk reserve, biparental care, and multiple spawning patterns (Araújo et al. 2012). However, all these adaptive advantages vary from environment to environment, as environmental conditions can favor or limit reproductive events, offspring survival, and growth (Pankhurst and Munday 2011, Cummings et al. 2017, Abdollahpour et al. 2022). It is widely known that environmental variability, including changes in temperature, precipitation, and photoperiod, can also influence fish movements and reproductive behavior (Silva et al. 2020). In the particular case of the Mayan cichlid, it has been reported that it has a high reproductive potential (Martínez-Palacios and Ross 1994), being even greater than that of tilapia because it is a mouthbrooder.
The pressure of commercial fishing is limiting the production of this species in Southeastern Mexico (Fernández-Perez 2024); therefore, aquaculture might play a significant role in recovering fish stocks in local lagoons and rivers. However, despite the potential traits that make M. urophthalmus a species of interest (Chávez-Lomelí et al. 1989, Martínez-Palacios and Ross 1994), there have been no efforts to initiate selective breeding to improve growth in captivity or enhance fingerling production. The present research constitutes a foundational step in the formation of M. urophthalmus broodstock batches to select improved genetic lines. The reproductive management of native species intended for introduction into culture systems is imperative for evaluating their potential incorporation into aquaculture. The Mayan cichlid has been a subject of interest for the study of captive management since the 1980s (Martinez-Palacios and Ross 1992). Nonetheless, its development in this field has been gradual, primarily due to the constraints imposed by captive reproductive management, which has led to the maintenance of aquacultural practices at a modest scale (Maldonado et al. 2020, Fernandez-Perez 2024).
To assess the hypothesis that M. urophthalmus has good reproductive traits such as length of the breeding season, spawning events, fertility, and fry production from each location, a reproduction trial was conducted using pairs from each locality and all the possible crosses. These crosses will allow the foundation of a batch of broodstock that will allow us to deepen our knowledge of reproductive management in captivity. In the present study, four groups of fish from representative hydrographic basins of the Mexican Southeast were formed: Nauchinapa stream (ANB), Grijalva River, and Usumacinta River (middle and lower basins). Some differences between collection sites include lower and very low average temperatures in winter (Nauchinapa stream), minimal marine influence (Grijalva River), and marine influence (Usumacinta River). The reproductive potential per group and their crosses were analyzed to form families for each zone and create a founder effect.
Materials and methods
Origin of the organisms and management in captivity
The present study was conducted in the facilities of the Tropical Aquaculture Laboratory of the Academic Division of Biological Sciences of the Universidad Juárez Autónoma de Tabasco, located in Villahermosa, Tabasco. Organisms collected at the localities of San Juan Volador in Pajapan, Veracruz (ANB); Zapotal in Comalcalco, Tabasco (GLB); Estapilla in Tenosique, Tabasco (UMB); and Nuevo Campechito in Carmen, Campeche (ULB) were used (Figure 1). These sites were selected based on different environmental conditions, such as low temperature (ANB), brackish water from a lower basin (ULB), and freshwater from low and mid-basins (GLB and UMB). Temperature and dissolved oxygen were measured using a YSI® 55 (Springer, CA, USA) DO meter, and salinity using a refractometer (SR6 Vital Sine Premium®). At the laboratory, pH was measured using a portable pH meter (CONDUCTRONIC™).
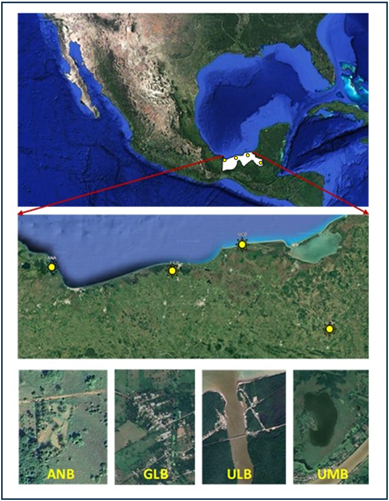
Figure 1 Geographic location of collection sites for M. urophthalmus broodstock. Geographic coordinates are: ANB (18°15'53"N 94°38'18.0"W); GLB (18°19'44"N 93°16'41"W); ULB (18°38'27.5"N 92°27'32.5"W); UMB (17°33'40"N 91°24'06"W).
The data from the collection sites are shown in Table 1. From each collection site, the fish were transported in a 1 000 L plastic tank provided with continuous aeration. Upon arrival, fish were placed in 2 000 L capacity plastic tanks. To avoid introducing disease into stock tanks, the fish were bathed for 15 minutes in water with 5 ppm of Potassium Permanganate before introducing them into the experimental area. The fish were kept separated by sex in plastic tanks with a 2 000 L capacity provided with continuous aeration, connected to a recirculation system for acclimatization and quarantine. The recirculation system consists of tanks and filtration of water (10 μm mesh size) with a biological (polyethylene mesh) and sand sieve (20 μm) at a rate of 100% every eight hours. These fish were fed three times per day (0800, 1 200, and 1 800 h) ad libitum, using 3% of their biomass with El Pedregal Silver Cup® balanced trout (45% protein and 16% lipid). Each organism was marked with a numbered spaghetti tag for identification, and before starting the reproduction trials, the fish were individually weighed and measured. The average weights and lengths of the females used in the experiment were 117.23 ± 17.45 g and 175.25 ± 8.83 mm (ANB); 124.65 ± 35.71 g and 180.77 ± 13.79 mm (GLB); 117.31 ± 22.59 g and 178.83 ± 13.88 mm (ULB); 123.30 ± 24.95 g and 179.70 ± 13.33 mm (UMB).
Table 1 Dissolved oxygen, temperature, and salinity at the collection sites.
| Details \ Basin | Arroyo Nauchinapa Basin (ANB) | Grijalva Lower Basin (GLB) | Usumacinta Lower Basin (ULB) | Usumacinta Mid-Basin (UMB) | Tropical Aquaculture Laboratory (LAT) |
|---|---|---|---|---|---|
| DO (mg/L) | 6.1 | 5.7 | 7.5 | 8.3 | 7.7 ± 0.14 |
| Temperature °C | 24.0 | 27.9 | 26.5 | 28.0 | 28.7 ± 0.52 |
| (Salinity (‰) | 0.0 | 0.0 | 24.0 | 0.0 | 0.0 |
| Collection Date | 22/10/2017 | 10/09/2017 | 08/09/2017 | 03/12/2017 | Jan-dec/2018 |
Dissolved oxygen (DO), temperature, and salinity data correspond to the day of collection. For the Laboratory (LAT), data correspond to the mean ± SD throughout the study.
Experimental design
A completely randomized one-factor design was used to generate the diallelic crosses with all possible combinations between the pairs from the different localities; for each combination between various locations, males and females from each area were used, making 16 total combinations (Table 2). After the quarantine period, thirty-two adult fish were randomly selected from the broodstock batch from each locality (16 females and 16 males). Four broodstock pairs were randomly placed under the combinations (16) in 2 000 L tanks. Each cross had one tank, and each pair was considered a replicate (four replicates per cross). The tanks were conditioned by placing rigid plastic sheets that served as protection and nests for the pairs. To maintain the water quality in the system, in addition to recirculation, 10% of the water was partially replaced daily, siphoning from the bottom to eliminate excreta and food residues. The reproduction trial ran from January to December 2018. The fish were able to select partners in each tank. No induction for spawning was used, and natural spawnings were verified daily. Feeding was maintained as previously mentioned. Culture conditions remained stable in the breeding system, maintaining average temperature values of 28.72 ± 0.52 °C (range: 27.91-29.70); Dissolved Oxygen (DO) of 7.55 ± 0.14 mg/L (range: 7.21-7.68); and pH of 7.80 ± 0.42 UI (range: 6.20-7.80).
Table 2 Diallelic crosses between the four populations of M. urophthalmus.
| ♀ ♂ | ANB | GLB | UMB | ULB |
|---|---|---|---|---|
| ANB | ANB x ANB | ANB x GLB | ANB x UMB | ANB x ULB |
| GLB | GLB x ANB | GLB x GLB | GLB x UMB | GLB x ULB |
| UMB | UMB x ANB | UMB x GLB | UMB x UMB | UMB x ULB |
| ULB | ULB x ANB | ULB x GLB | ULB x UMB | ULB x ULB |
Crosses in boxes highlight those with females and males from the same basin. ANB = Nauchinapa Stream Basin; GLB = Grijalva River Lower Basin; UMB = Usumacinta River Mid Basin; and ULB = Usumacinta River Lower Basin.
Because M. urophthalmus eggs are adhesive, they are attached to surfaces during spawning (bottom or walls); therefore, measuring diameter or counting the number of eggs produced per female during a spawning was not possible. For this reason, hatchlings were collected by siphoning on the same day of hatching, as they remain non-swimming at the nesting site. Freshly collected hatchlings were counted to determine fertility (total number of fry per spawning) and relative fertility (number of fry per gram of female), and were then randomly seeded in 100 L plastic tanks with constant aeration for growth.
Statistical analysis
The postulates of parametric statistics were verified using the standardized skewness and kurtosis coefficients for normality and Levene's homoscedasticity test. When the postulates were unmet, the corresponding non-parametric test was conducted. In the case of factorial analyses and linear regression, the postulates were verified using residual analysis. To determine statistical differences between locations for time until first spawn, time between spawns, and number of hatchlings per spawn, an analysis of covariance (ANCOVA) was used, incorporating female weight as a covariate, followed by a Tukey's HSD multiple contrast test. A linear regression Analysis was used to determine the association between the weight of the female and the number of hatchlings per spawning or female. The number of spawnings and the total number of hatchlings were compared using a Chi-square test. Fertility values were contrasted using the Kruskal-Wallis non-parametric test, followed by a Bonferroni multiple contrast test. A contingency table analysis was used to determine if the number of females spawned was associated with the crosses between basins. All statistical analyses were performed with Statgraphics Centurion® v19 software, using a significance value of α = 0.05. Graphical analyses were performed using Sigma Plot® v14.5 or Centurion® v19 software.
Results
M. urophthalmus reproduction under captive conditions occurs from March to September. However, only fish from the ANB locality reproduced from March onwards, without spawning for the other localities this month. From April onwards, spawning was obtained for fish from all localities. Towards the end of the spawning season, spawning was only obtained in fish from ANB and ULB during September. These results indicate that ANB females had the longest season, extending two months. The factorial ANCOVA, including female origin, male origin, and the covariate female weight as factors, indicates that ANB females had their first spawning in the shortest time (p < 0.01) as they required a mean (± SD) of 31.45 (± 19.52) days for first spawning; In contrast, GLB, UMB and ULB females required means of 58.60 (± 27.87), 67.70 (± 47.50) and 83.71 (± 34.01), respectively (Figure 2). The average number of days between spawning is highly significant only for female origin (ANCOVA; p < 0.001). This was lower for females from ANB, while the other three basins were not significantly different (Figure 2).
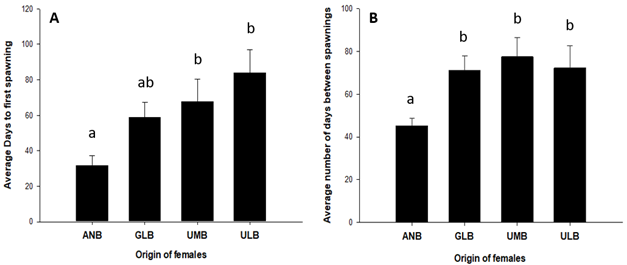
Figure 2 Mean values (± DE) of days from the beginning of the experiment to the first spawning (A) and days between spawnings (B).
The female that took the shortest time to the first spawning once the experiment was set up was ANB, with a male from the same locality, taking 12 days to initiate spawning. The crosses that took the longest to trigger a spawn coincided with the presence of ULB males (Table 3).
Table 3 Specific number of days required for the first spawning per cross to occur.
| Basin | ANB ♂ | GLB ♂ | UMB ♂ | ULB ♂ |
|---|---|---|---|---|
| ANB ♀ | 20.25 | 25.33 | 41.00 | 46.50 |
| GLB ♀ | 42.33 | 65.67 | 30.00 | 101.00 |
| UMB ♀ | 62.67 | 59.33 | 27.00 | 128.50 |
| ULB ♀ | 69.25 | 63.50 | 74.00 | 106.50 |
Numbers highlighted in green indicate the shortest times females need to spawn, while values highlighted in orange indicate the longest times.
When analyzing the number of hatchlings produced per spawning (hps), the ANCOVA indicates a highly significant effect of female weight (p < 0.001); after removing this effect, the analysis suggests a slight effect of the origin factor (p = 0.02); however, the rigor of the Tukey's HSD a posteriori contrast, indicates that these differences are not statistically significant (p > 0.05). GLB females provided the highest number of hatchlings in the experiment, followed by the ANB females. UMB and ULB females had the lowest mean production. Despite a 414 hatchling difference between the highest and the lowest mean, the large variability between spawners and the stringency of Tukey's test prevents the differences from being statistically significant. The smallest spawning had 1,696 hatchlings from a GLB female, while the largest spawning reached 4,345 from an ANB female. The greatest variability was observed in GLB, reaching a coefficient of variation of 30.60% (Table 4).
Table 4 Reproductive performance values from M. urophthalmus females, estimated from the number of hatchlings per spawning (hps) from the four localities involved in the study. Results include values obtained in all experimental crosses.
| Basin | Spawnings | Mean | Median | SD | CV | Min | Max | Range | Total |
|---|---|---|---|---|---|---|---|---|---|
| GLB | 18a | 2 733.33 | 2 578.50 | 836.39 | 30.60% | 1.696 | 4 320 | 2 624 | 49 200a |
| ANB | 39b | 2 683.69 | 2 609.00 | 575.76 | 21.45% | 1.923 | 4 345 | 2 422 | 10 664b |
| UMB | 15a | 2 493.60 | 2 541.00 | 323.49 | 12.97% | 1.863 | 2 987 | 1 124 | 37 404a |
| ULB | 11a | 2 319.00 | 2 200.00 | 407.57 | 17.58% | 1.845 | 3 051 | 1 206 | 25 509a |
SD = Standard Deviation, CV = Coefficient of de Variation, Min = Minimum hps value in a spawning, Max = maximum hps value. ANB = Nauchinapa Stream Basin; GLB = Grijalva River Lower Basin; UMB = Usumacinta River Mid Basin; and ULB = Usumacinta River Lower Basin.
Although female weight is highly significant in the ANCOVA, the association between the number of offspring obtained per spawner and female weight is not very high. The simple linear regression achieves a correlation coefficient r = 0.46, and only 21.07% of the variability in the number of hatchlings is explained by the model (No. of fry = 1425.74 + 9.49*weight). This model predicts that for each gram of female, 9.49 hatchlings are produced, a value that is far from those observed per spawner, which in general averages 21.39 hatchlings. On the other hand, there was no significant association between the number of young obtained per spawning and the number of spawns per female (p > 0.05; Pearson's Correlation Coefficient = 0.12), with females having up to 4 spawns and a high number of young; as well as females with one or two spawns and few young in their brood (Figure 3).
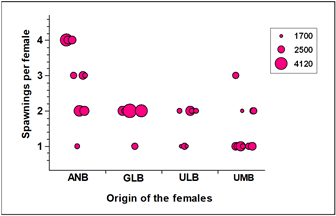
Figure 3 Bubble plot for the number of spawnings per female as a function of the locality of origin. The bubble diameter represents the average number of hatchlings produced per spawning.
The Chi-square test indicates highly significant differences in the number of spawnings and total hatchling production among localities (p < 0.001). ANB females were the most productive, accumulating 39 reproductive events among all crosses and generating more than double the amount of hatchlings that those obtained from GLB females, which had 18 reproductive events, while UMB females and ULB had 15 and 11 spawnings, respectively (Table 4). The contrast of relative fertility (expressed as hatchlings/gram) between locations indicates that there are statistically significant differences (KW; p < 0.01). The Bonferroni post hoc test indicates that ANB females were significantly more productive than UMB females (Table 5).
Table 5 Mean values of female weight (± SD) and median for hatchling per gram of female weight (± IQR).
| Basin | Mean Weight ± DE | Mean h/gr | Median h g-1 |
|---|---|---|---|
| ANB | 210.28 ± 34.46a | 23.45 ± 5.61 | 21.65 ± 6.34b |
| GLB | 232.94 ± 39.53b | 20.85 ± 5.19 | 19.01 ± 6.44ab |
| UMB | 215.47 ± 35.48ab | 18.44 ± 3.28 | 19.20 ± 4.89a |
| ULB | 220.18 ± 33.73ab | 18.99 ± 2.09 | 19.52 ± 1.01ab |
h/gr = Hatchlings per gram; ANB = Nauchinapa Stream Basin; GLB = Grijalva River Lower Basin; UMB = Usumacinta River Mid Basin; and ULB = Usumacinta River Lower Basin.
When the productivity of the different crosses generated by the combinations between locations was analyzed, the ANB-ANB (female-male) combination showed the highest spawning performance, with a total of 18 reproductive events, and the ULB-UMB combination showed the lowest performance, with only one spawning during the experiment. ANB females also presented higher reproductive efficiency for crosses with GLB and UMB males. In contrast, ULB females had more spawnings with males from the same locality (Table 6). Figure 4 shows how spawning activity was distributed based on the specific crosses, highlighting that ANB females were highly active with males from the same region. GLB and UMB females were not as selective in pairing with males from their region, as they had an equal number of spawnings with ANB males in both cases. Only ULB females had more affinity with males from their locality (Figure 4).
Table 6 Spawning data by cross. The Total F column indicates the number of spawnings for females per watershed, while the data in the Total M row indicates spawnings for males.
| Basin | ANB ♂ | GLB ♂ | UMB ♂ | ULB ♂ | Total H |
|---|---|---|---|---|---|
| ANB ♀ | 18 | 10 | 8 | 3 | 39 |
| GLB ♀ | 6 | 6 | 4 | 2 | 18 |
| UMB ♀ | 5 | 4 | 5 | 1 | 15 |
| ULB ♀ | 3 | 2 | 2 | 4 | 11 |
| Total M | 32 | 22 | 19 | 10 | 83 |
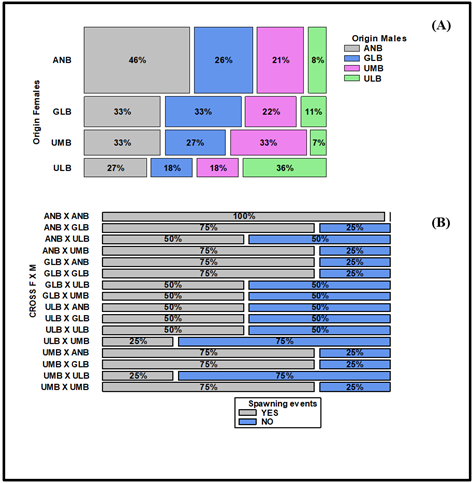
Figure 4 Mosaic plot for percentage spawning values as a function of origin for females (rows) and males (columns) (A). Mosaic plot for the percentage of spawned and non-spawned females per cross (B).
Of the total number of ANB females used in the experiment, 75% (12/16) spawned, while for GLB and UMB, 62.5% of females (10/16) spawned, while for ULB, only 43.8% (7/16) spawned. Analysis of the number of females that spawned per cross indicates that there is independence between the crosses and the number of females spawned (χ2; p > 0.05). Reproductive performance at the individual level is also high for ANB females. In the ANB female-male cross, 100% of the females spawned. The second most productive cross was of ANB females with GLB males, where one female spawned four times and two more spawned three times (75% of the total females). In seven crosses, 75% of the females spawned, and in six of the crosses, 50% spawned. The two crosses with fewer females spawning (25%) were ULB-UMB and UMB-ULB (Figure 5), with the difference that in the first one, the female spawned twice, and in the second one, only once.
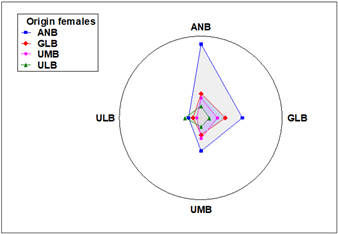
Figure 5 Radar plot for the number of spawnings as a function of the locality of origin of the females (inner polygons) and the origin of the males (reference outside the circle).
The affinity between females and males is reflected in the number of spawnings per cross. Two females of the ANB-ANB cross spawned up to five times, and two more spawned four times. Another highly productive cross was the ANB-GLB cross, as one female spawned four times, and two more spawned three times each. Two more ANB females had three spawnings, along with UMB-ULB males, and only one UMB female had three spawnings in the season. Seventeen females were recorded with two spawnings throughout the season: eight from GLB, four from ULB, three from UMB, and two from ANB. Twelve females had only one spawning throughout the study, six of them from UMB, three from ULB, two from GLB, and one from ANB (Figure 5). The size of the polygons in the figure clearly shows the reproductive capacity of females from each locality, as well as the affinity with males.
Discussion
The present research lays the foundations for introducing improvements in captive breeding. The most important result of the present study is the great reproductive potential shown by the organisms from San Juan Volador (ANB) in a small basin of the Nauchinapa stream in the state of Veracruz. The low productivity of organisms from Nuevo Campechito in the lower basin of the Usumacinta River is also outstanding. Regarding this, Ishikawa and Kitano (2020) demonstrated that stream ecotypes of the three-spined stickleback (Gasterosteus aculeatus) exhibit protracted breeding seasons, commencing earlier and concluding later than other ecotypes. Additionally, at lower latitudes, these ecotypes reproduce nearly year-round, in contrast to their higher latitude counterparts, potentially due to differences in photoperiod durations. The later authors consider that divergent natural selection on the reproductive strategy may act on the TSHβ2 locus, resulting in differential expression patterns that are potentially involved in the regulation of seasonal reproduction in anadromous-marine and stream ecotypes. Despite the absence of data regarding the dissemination of M. urophthalmus in Central America, the potential for genetic variation among populations, including epigenetic mechanisms that regulate the expression of reproductive traits, is a relevant consideration.
The results observed in the current study may be due to environmental characteristics present in the growth zone of the organisms used as broodstock, such as temperature and salinity. The organisms from ANB grew at lower average temperatures than in the area where the study was conducted. The Nauchinapa stream basin (ANB) is small, belongs to the hydrographic region of the Coatzacoalcos River, and originates west of the town of Pajapan at an elevation of 160 meters above sea level; its short journey to the Gulf of Mexico is only 9 km and is fed by small streams. According to the Horton-Strahler model, its order magnitude is three (INEGI 2016). The few existing data on the area indicate that the ambient temperature ranges between 20 and 26 °C, presenting a warm-humid climate with abundant rainfall in summer (INEGI 2010). However, there is no information on the temperature range of the Nauchinapa stream; the only data available was obtained during broodstock collection in November, which was 24.0 °C. Other information available is about the oyster lagoon, located 6.3 km in a straight line from the collection point. In this ecosystem, average high, medium, and low temperatures of 28.8 ± 1.2, 30.7 ± 0.6, and 26.4 ± 1.6, respectively, have been reported (Aguirre-León et al. 2020). In this same lagoon, Ríos-Castro (2015) reported the presence of dominant jellyfish species at temperatures between 21.5 and 29 °C. These data make us think that in the cold front season, the temperatures of the Nauchinapa stream could easily be below 20 °C, so the temperatures probably used in the Tropical Aquaculture Laboratory for reproduction-induced continuous spawning, as they were maintained between 27.91 and 29.70 °C. The temperature values observed in the present study fall between the optimal value suggested for the species since Martínez-Palacios et al. (1994) described that the M. urophthalmus in captivity presents more spawnings between June and October, highlighting that the ideal temperature for reproduction is between 28 and 31 °C and that reproductive events are inhibited below 24 °C; this last value is similar to what was observed in the Nauchinapa stream in November.
Jalabert and Zohar (1982) mentioned that reproductive periods in most teleost fishes are apparently regulated by environmental factors such as photoperiod, temperature, salinity, and rainfall. These authors emphasize that in temperate fishes, photoperiod and temperature have the greatest impact, whereas in tropical areas, variations are more limited, and temperature is rarely a limiting factor. However, Jonsson and Jonsson (2014) reviewed the impact that exposure to specific environmental conditions during early development has on life history traits such as age at maturity, egg size, maturity, and even sex determination; emphasizing the importance of temperature and the role that global change may exert on fish populations and highlighted the need to generate information on the impacts on the genetic, epigenetic, neural, and hormonal mechanisms the environment has during early development. With the resulting observations, and given the wide distribution range of M. urophthalmus, it is evident that this species tolerates low temperatures, as has been demonstrated in ecosystems of Florida, USA, where this species withstands the low temperatures left by the passage of cold fronts with low averages between 15.2 and 15.3 °C (Schofield et al. 2010). These same authors report that the species loses equilibrium at 12 °C and dies at 9.9 °C, and, remarkably, they report that in a wide range of salinities (0-35 ppt), M. urophthalmus organisms modified very little tolerance to high or low temperatures.
For Faunce and Lorenz (2000), the characteristics and length of the reproductive season among M. urophthalmus populations may be governed by differences in their life cycle. This situation is evident in our study, as the Nauchinapa Stream (ANB) population develops in a lower-temperature environment, and the Nuevo Campechito (ULB) population is exposed to higher salinities. The conditions at the Tropical Aquaculture Laboratory (28-30 °C and zero ppt) apparently stimulated ANB fish reproduction and inhibited ULB fish. It has been mentioned in several studies that the reproduction of this species in its natural range is directly related to the environmental temperature since it starts in spring and ends in autumn, from April to November, disappearing completely in winter (Martínez-Palacios and Ross 1992). This applies only to tropical areas since in Florida, Faunce and Lorenz (2000) report that the breeding season is from April to June. These same authors point out that, unlike those reported by Martínez-Palacios and Ross (1992), in Florida, the species apparently does not base its reproduction exclusively on temperature, but rather, the key for C. urophthalmus to begin spawning is apparently linked to the increase in salinity. Similarly, Loftus (1987) reported nests and a group of mature spawners in Snook Creek, recording 26 ppt of salinity in the water. This information supports what was observed in our study and supports the need for captive breeding to consider the life history characteristics of the populations involved.
The absolute fertility reported for M. urophthalmus ranges from 2 085 to 6 615 eggs per female, depending on size, while the relative fecundity varies from 20 to 30 eggs per gram of female (Martinez-Palacios and Ross 1992). The data obtained in the present study correspond to the number of hatchlings obtained per spawning, which implies the possible loss of non-spawned or unfertilized eggs, as well as unhatched embryos and dead offspring in the first hours; however, the data indicate that the number of hatchlings produced per gram of reproductive female varies between 18.44 (UMB) and 23.45 (ANB). This suggests that the species has good reproductive potential despite possible losses of oocytes or embryos. Martínez-Palacios et al. (1993) reported for the species that, under two different densities of breeders, the average number of offspring obtained per spawning was between 1 025 and 2 282. In our systems, the averages obtained were between 2 319 and 2 733 offspring per spawning, slightly above the reported data. These data confirm that the reproductive potential of the species is high, since in addition to being able to spawn multiple times in a season (up to 3 and 4 times), the number of offspring per spawning is higher than in tilapia, where between 200 and 600 offspring are obtained per spawning, depending on the size of the female (Martínez‐Palacios y Ross 1992, Martínez‐Palacios et al. 1993, El-Sayed et al. 2005, Tsadik and Bart 2007).
Concerning the founder effect, it is important to mention that, in this study, the need arises to form batches of broodstock that produce offspring that can be improved by genetic selection, leading to the production of aquaculture products that favor or present profitable characteristics such as better growth and, therefore, better yield. In this regard, although ANB specimens present a higher reproductive performance, it will be necessary to evaluate the benefit of crosses between all the populations since, among them, the best specimens could be obtained to establish a population with a founder effect, given that, in theory, the genetic variability between both populations should be higher. Wasko et al. (2004) mentioned that the main reasons for the reduction of genetic variability in captive flocks are attributed to the use of a few breeders and the crossing of related individuals. Hence, these crosses also constitute a lower possibility of inbreeding. This study is a good start to continue with aspects of genetic selection to improve reproductive performance for aquaculture purposes. It will also be indispensable to regularly introduce new specimens to the populations to maintain the founding populations in good condition, without ceasing to make use of genetic selection that will lead to exploiting the potential of the Mayan cichlid at commercial levels.
Conclusions
The ANB population showed the best reproductive potential with high fertility compared to the other populations evaluated. This shows that the reproductive performance of different populations of the same species depends on differences in their life cycle and can be enhanced or inhibited by the conditions under which production occurs. Future studies should also consider incorporating genetic criteria in breeding schemes, such as molecular diversity evaluations or genetic management plans, to avoid bottlenecks.











 nueva página del texto (beta)
nueva página del texto (beta)



