Understanding the factors that shape plant species composition and richness in extreme environments remains an important topic in ecology. In arid regions, where water availability is a fundamental limiting factor for plant life (Noy-Meir 1973), sites with persistent moisture become ecologically prominent in the surrounding arid environment. These ecosystems function as paradoxical refugia, supporting relatively high biodiversity despite severe water limitations, and contribute significantly to ecological processes at both local and regional scales (Grismer & McGuire 1993, Stevens & Meretsky 2008, McLaughlin et al. 2017, Cartwright et al. 2020). Within this context, arid palm groves stand out as a distinctive wetland type that supports specialized plant assemblages (Schwenkmeyer 1986, Cornett 2008). Yet despite their importance, the floristic diversity associated with these systems has rarely been examined from a regional or biogeographic perspective.
Palm groves in arid regions offer a compelling case study for examining how wetland systems operate under extreme climatic constraints. Typically, small and sustained by isolated water sources (Schwenkmeyer 1986, Franco-Vizcaíno et al. 2007, Garcillán et al. 2012), these groves form localized oases of productivity. Their presence generates ecological transitions, where mesic plant assemblages emerge within an arid matrix, giving rise to biodiversity hotspots embedded in desert landscapes (Franco-Vizcaíno et al. 2007, Cornett 2008). Stable moisture regimes in these sites provide critical refugia for hydrophilic flora, facilitating species persistence and contributing to regional biodiversity under severe water limitation (McLaughlin et al. 2017, Cartwright et al. 2020). Given these attributes, palm groves can be viewed as functional analogs of insular systems in island biogeography, with their floristic biodiversity shaped by wetland area (Pérez-Navarro et al. 2019) and by the geographical isolation between palm groves (Garcillán & Rebman 2025). Understanding their floristic composition is key to interpreting how these systems contribute to species persistence, microhabitat heterogeneity, and biodiversity maintenance under arid conditions.
The western region of the Sonoran Desert, spanning latitudes 23° to 34° N, encompasses nearly the entire distribution range of five palm species -Washingtonia filifera, W. robusta, Brahea armata, B. brandegeei, and B. edulis- and represents a distinct biogeographic domain of the palm family in North America (Pulido-Silva et al. 2023). This area forms a broad climatic transition zone, extending over 1,400 km. It spans from the summer-rain-influenced tropics of the southern Baja California Peninsula to the winter-rain-dominated Mediterranean climate of southern California and northwestern Baja California Peninsula. While most palm groves occur within the Sonoran Desert proper, some extend into adjacent regions: palms are found in the desert-Mediterranean ecotone and, more frequently, in the tropical dry forest and the sarcocaulescent scrub of the Cape Region. The region also marks the northwestern limit of palm species distribution in North America and includes some of the most arid habitats where palms occur.
Numerous studies have examined the flora associated with palm groves in California (Vogl & McHargue 1966, Cornett 2008) and in the Baja California Peninsula (Arriaga et al. 1997, León de la Luz et al. 1997, León de la Luz & Domínguez-Cadena 2006, Franco-Vizcaíno et al. 2007, Garcillán et al. 2012, Wehncke et al. 2012, Ruiz-Campos et al. 2014, Valov 2020, Garcillán & Rebman 2025). However, these studies have primarily focused on individual sites or subregions. To date, no comprehensive analysis has addressed how floristic composition varies across palm groves along this full latitudinal and climatic gradient, which itself defines a biogeographically coherent region. Given this pronounced, geographically structured ecological variation, a regional synthesis is essential to detect broad-scale patterns in species distribution and community structure.
Research on how wetland plant communities are distributed across large geographic gradients has produced mixed findings. Some studies report clear shifts in species composition aligned with climatic transitions, suggesting structured geographic patterns (Chappuis et al. 2012, Li et al. 2024), while others observe limited spatial differentiation, with similar species recurring across distant sites (Alahuhta et al. 2017, Zhou et al. 2022). These differing results underscore the need to assess the extent to which the regional ecological variation shapes the distribution of wetland flora, especially in arid regions of high biogeographic complexity, such as the Baja California Peninsula and southern California, where climatic, topographic, and historical factors interact in a particularly intricate combination (Garcillán et al. 2010).
In a previous study based on a selected set of palm groves, we found that floristic similarity declined with distance, and that this pattern varied according to species’ water dependence (Garcillán & Rebman 2025). In the present work, we pursue two main objectives. First, to compile the most comprehensive inventory to date of wetland-associated plant species linked to the five native palm species across the region. Second, building on this extensive floristic and geographic coverage, we analyze the geographic structure of the hydrophytic flora associated with palm groves at the regional scale. Specifically, we test the hypothesis that variation in species composition among palm groves reflects the regional patterns of climatic variation across the Baja California Peninsula and southern California.
Materials and methods
Data collection. We compiled floristic data from palm groves containing native palm species across southern California and the Baja California Peninsula using two main sources: (i) published literature and (ii) herbarium records.
Literature review.- We conducted a comprehensive review of published sources and identified 11 studies reporting plant species associated with groves of the five native palm species in the region: Brahea armata, B. brandegeei, B. edulis, Washingtonia filifera, and W. robusta (Vogl & McHargue 1966, Arriaga et al. 1997, León de la Luz et al. 1997, León de la Luz & Domínguez-Cadena 2006, Franco-Vizcaíno et al. 2007, Cornett 2008, Garcillán et al. 2012, Wehncke et al. 2012, Ruiz-Campos et al. 2014, Rascón-Ayala et al. 2018, Valov 2020). In addition to published sources, we incorporated an unpublished species list derived from floristic fieldwork conducted in 2018 at the Guadalupe Canyon palm grove, in northern Baja California Peninsula (J.P. Rebman, unpublished data).
Herbarium records review.- We queried the databases of the SD Herbarium (San Diego Natural History Museum) and the Baja California Botanical Consortium (www.bajaflora.org) using two approaches. First, we selected all specimens of the five native palm species. When geographic coordinates were missing, we estimated locations based on locality descriptions whenever possible. For each palm specimen, we recorded any co-occurring plant species listed on the label and linked them to the palm species and site. Second, we searched for other plant species specimens whose labels mentioned palms as associated taxa, applying the same georeferencing and data extraction procedures.
Data preparation. All floristic records were compiled into a unified species-by-site database. Taxonomic names were standardized following the nomenclatural framework of Rebman et al. (2016), with additional updates from Plants of the World Online (POWO 2025).
Species were categorized by wetland dependence following Lot et al. (2015), complemented by field observations: aquatic (A), species that complete their life cycle submerged and generally cannot survive outside the water; subaquatic (S), species that complete their life cycle at the water margins or in saturated soils, tolerating temporary dry conditions; tolerant (T), species that complete most of their life cycle in a dry environment but can withstand seasonal submersion. All the remaining species lacking adaptations were considered non-hydrophytes or intolerant (I). We also categorized species by lifespan (annual or perennial), growth form (grass and grass-like plants, other herbaceous species, vines, succulents, shrubs, and trees), and native status, based on Flora of North America Editorial Committee (1993), Rebman et al. (2016), and CalFlora (2022). Species that could be either annual or perennial were classified as perennial to keep the annual category as strictly ephemeral species.
From the literature, we retrieved two floristic lists for California palm groves located mostly in the western Colorado Desert near the San Andreas Fault - Vogl & McHargue (1966), covering 24 sites, and Cornett (2008), covering 122 sites- both of which lacked individual site coordinates. We therefore merged the two datasets into a single species list and assigned a representative centroid treated as a single site, to enable their inclusion in geographical analyses.
Floristic analysis. We analyzed patterns of floristic differentiation among palm groves across the study region based on the composition of wetland-associated species (aquatic, subaquatic, and tolerant). Given the heterogeneous spatial resolution of data sources - some records referred to multiple palm groves without precise locations - and variation in both sampling intensity and collection practices, we first grouped the 235 sites into 18 ecological areas. These groupings were defined based on geographic proximity and environmental coherence, using regional patterns in climate, topography, and vegetation (Figure 1). This allowed us to aggregate data meaningfully and detect broader floristic patterns.
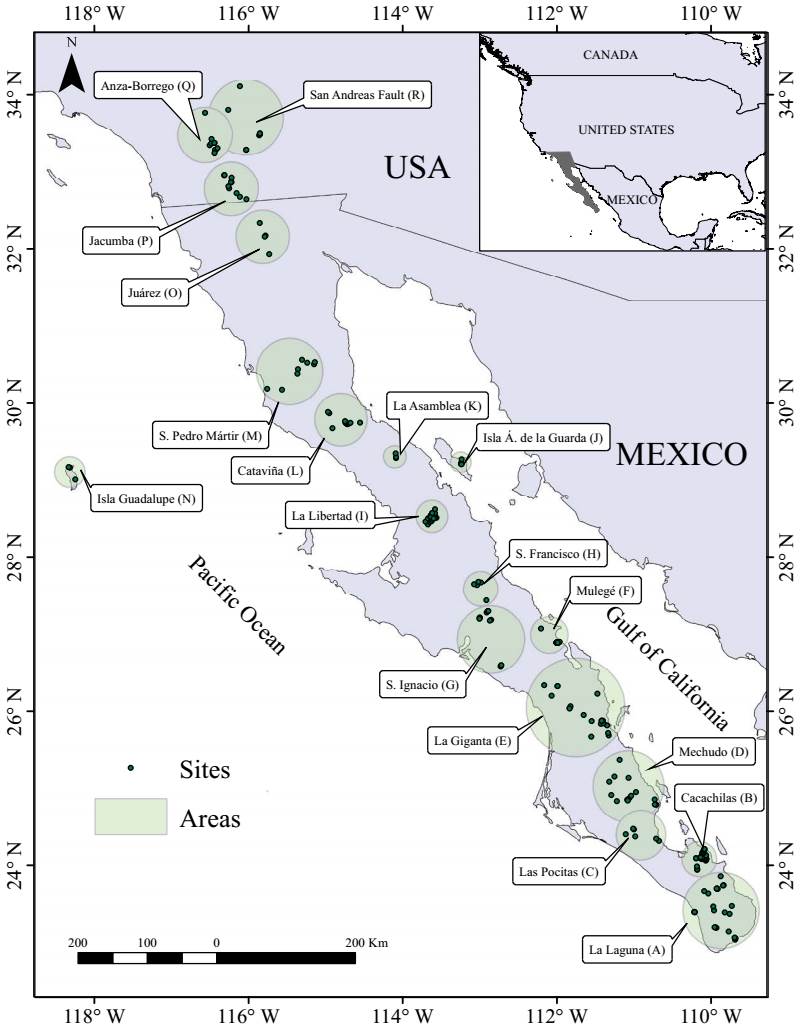
Figure 1 Distribution of 235 palm grove sites across southern California (USA) and the Baja California Peninsula (Mexico). Shaded circles indicate palm grove areas grouping sites into 18 areas listed alphabetically from South to North.
Next, to validate the environmental consistency of these groupings, we extracted 19 bioclimatic variables from the WorldClim database v. 2.1 (Fick & Hijmans 2017) at 30 arc-second resolution (~1 km²) using the coordinates of the 235 individual sites. We then performed a Principal Component Analysis (PCA) using the site-level climate data to examine whether sites grouped into the same area also clustered in climate space. To do this, we used the predefined area assignments to delineate groups and applied 95 % confidence ellipses (Student’s t-distribution) to visualize the climatic dispersion of each area. These ellipses are not the result of statistical clustering but are a graphical representation of the dispersion of each a priori group in PCA space, assuming multivariate normality and using the t-distribution to account for group sample size. The resulting PCA plot (Figure 2) showed that sites belonging to the same area tended to occupy distinct and coherent regions of environmental space, supporting the rationale for treating these ecological areas as valid units of analysis.
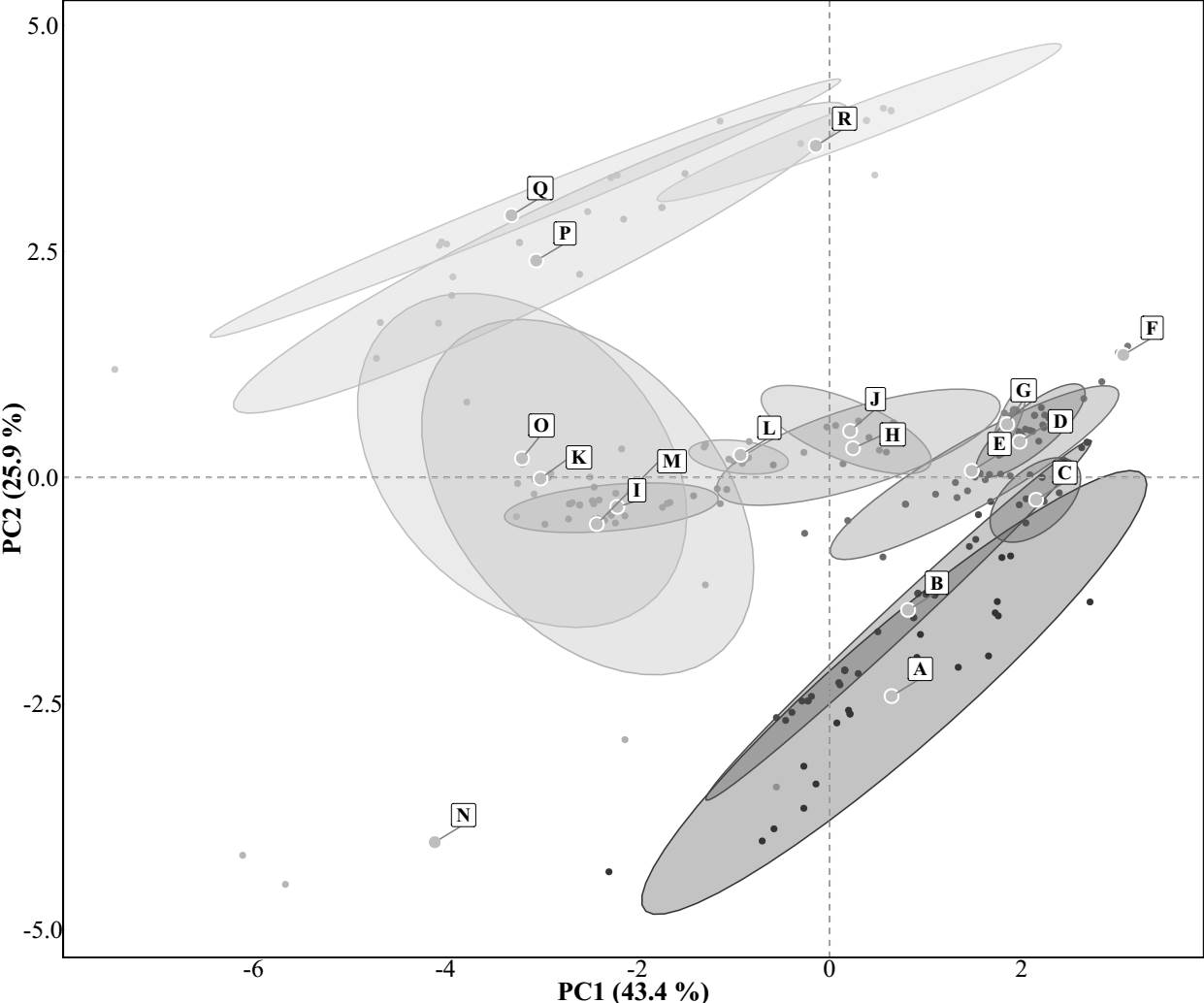
Figure 2 Principal Component Analysis of 235 palm grove sites across southern California and the Baja California Peninsula, based on 19 bioclimatic variables from the WorldClim dataset. Sites are grouped into 18 areas listed in Figure 1 (from South to North, A-R), with 95 % confidence ellipses based on Student´s t-distribution. The first two components explain 69.3 % of the climatic variation.
To characterize the structure of floristic composition across these 18 areas, we calculated pairwise Sørensen dissimilarities using the vegdist function and visualized relationships using non-metric multidimensional scaling (NMDS), both implemented in the vegan package in R (Oksanen et al. 2024, R Core Team 2024). NMDS was preferred over PCA because it does not assume linear relationships; instead, it positions sites in reduced-dimensional space by minimizing stress, which is a measure of the mismatch between ordination distances and the original dissimilarities. We further explored species-ordination relationships by fitting species vectors onto the NMDS configuration using the envfit function. Vector direction indicates increasing frequency of a species across the ordination space, while vector length reflects the strength of correlation with the axes. Significance was tested via permutation to assess whether associations were stronger than expected by chance.
To test whether floristic patterns among palm-grove areas are structured by climatic gradients, we performed a distance-based redundancy analysis (dbRDA) using the capscale function, also from the vegan package. In this framework, floristic dissimilarities (Sørensen index) were modeled as a function of climate variables (previously filtered to reduce collinearity), allowing us to quantify the proportion of variance in species composition explained by environmental factors (Legendre & Anderson 1999). This analysis directly tests the relationship between community composition and climate, providing an interpretable ordination space that links specific climatic predictors to floristic variation.
Results
We recorded 4,093 species occurrences, corresponding to 1,133 distinct plant species across 235 palm-associated sites distributed along a 1,400 km latitudinal gradient, from 23° N at the southern tip of the Baja California Peninsula, Mexico, to 34° N in California, United States (Figure 1).
Taxonomic and ecological composition. The recorded flora included 140 plant families. Asteraceae (14 % of all species), Fabaceae (9 %), and Poaceae (9 %) were the most represented, together accounting for nearly one-third (32 %) of the total species. In total, 107 species (9 %) were classified as non-native.
Nearly one-third of the recorded species (368 species, 32 %) exhibited some degree of wetland affinity: 37 aquatic (3 %), 79 subaquatic (7 %), and 252 tolerant species (22 %) (Table S1), while the remaining 765 species (68 %) were classified as intolerant to saturated soils (Figure 3A). At the family level, Poaceae was the only group consistently represented across all water-dependence categories, comprising between 6 and 16 % of species within each category (Figure 3B). Cyperaceae, although accounting for only 3 % of total species, dominated the aquatic (14 %) and subaquatic (23 %) categories, but was uncommon among tolerant (3 %) and rare among intolerant species (0.3 %). Fabaceae showed the inverse pattern -completely absent from aquatic species (0 %) and nearly absent from subaquatic species (2 %)- but it was more common among tolerant (9 %) and intolerant (10 %) groups. Asteraceae was broadly distributed, particularly among tolerant (17 %), subaquatic (14 %), and intolerant species (13 %), while rare in aquatic groups (3 %). Potamogetonaceae, a family composed exclusively of aquatic species, represented 8 % of all aquatic species in the dataset, ranking second in frequency within that category alongside Poaceae.
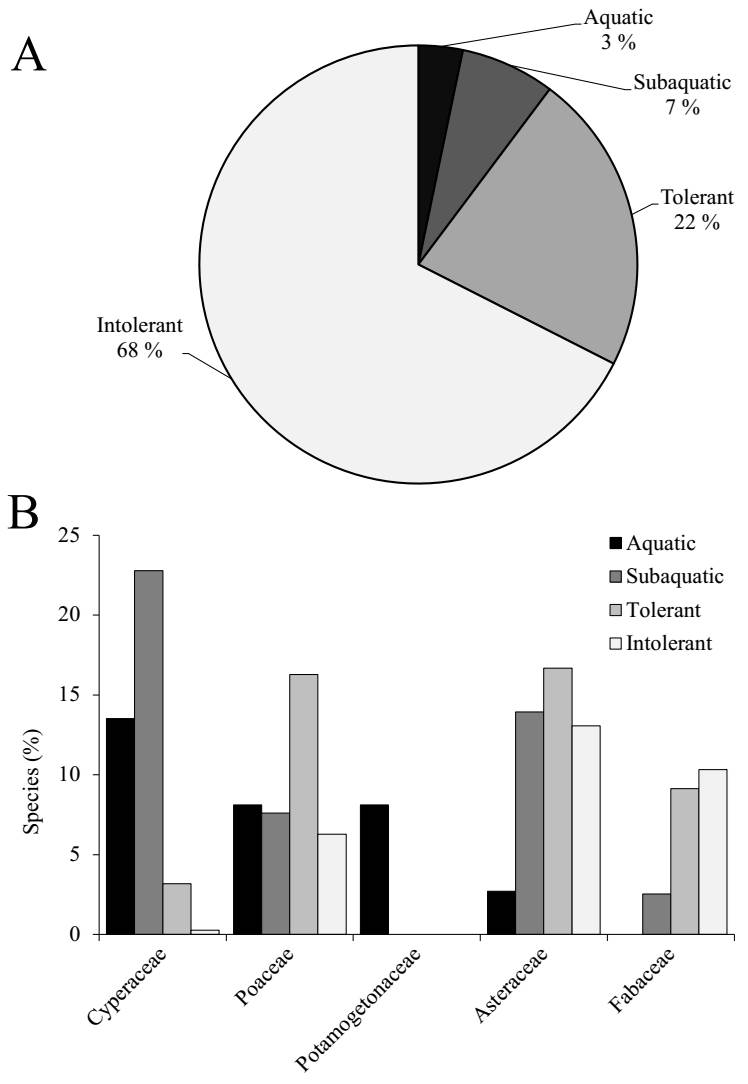
Figure 3 Species distribution by wetland category. (A) Proportion of all species across four wetland categories (aquatic, subaquatic, tolerant, and intolerant species). (B) Relative abundance (%) of species from families representing > 5 % within each wetland category: Cyperaceae, Poaceae, Potamogetonaceae, Asteraceae, and Fabaceae.
Among the 107 non-native species, the majority were classified as tolerant (56 %) or intolerant (31 %), with only a small proportion identified as subaquatic (12 %) or aquatic (1 %). In contrast, native species were predominantly intolerant (71 %), with lower proportions of tolerant (19 %), subaquatic (6 %), and aquatic species (4 %).
Perennial herbaceous species were the most common life form across the dataset (37 %), followed by annual species (29 %) and shrubs (22 %). Trees (6 %), succulents (3 %), and ferns (3 %) were comparatively less represented (Figure 4A). Life form composition varied across water-dependence categories (Figure 4B). Perennial herbaceous species dominated the aquatic group (86 %), but their frequency declined with decreasing water dependence - dropping to 51 % among subaquatic species, 35 % among tolerant species, and 33 % among intolerant species. In contrast, annual herbaceous species exhibited the opposite trend, rising from 11 % in aquatic species to 30 % in subaquatic and peaking at 43 % in tolerant species, before declining slightly to 25 % among intolerant species. Shrubs were absent from aquatic species and increased steadily across drier categories, from 5 % in subaquatic species to 14 % in tolerant and 28 % in intolerant species (Figure 4B).
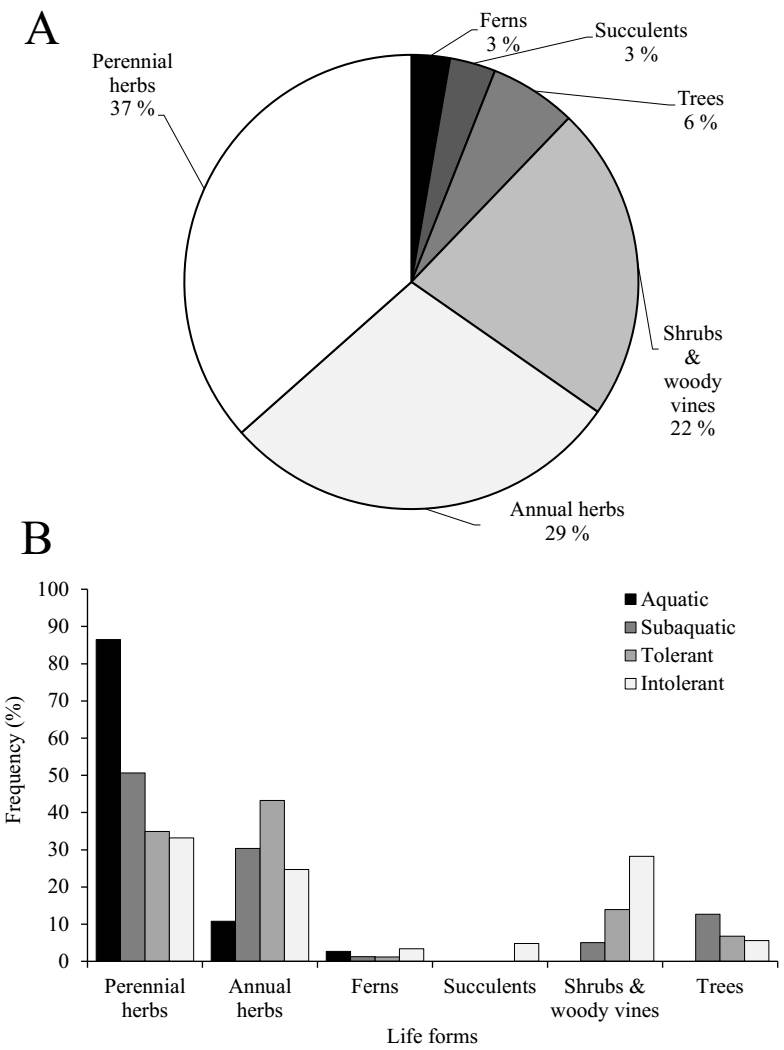
Figure 4 Distribution of plant life forms across all species. (A) Proportion of species among six main life forms. (B) Relative frequency of life forms within each wetland category.
Regional floristic patterns. Floristic differentiation among the 18 palm-grove areas revealed a well-structured regional pattern. The NMDS ordination, based on wetland-associated species (aquatic, subaquatic, and tolerant), displayed a clear spatial gradient (stress = 0.124), separating areas along both axes (Figure 5A). This value falls within the range of 0.10-0.20, which is commonly interpreted as acceptable for ecological data, indicating that the ordination provides a reliable representation of floristic dissimilarities. It is well above the range associated with potential overfitting due to extremely low stress values (i.e., values very close to zero), and below the threshold commonly linked to poor fit or underfitting (> 0.20) (Kruskal 1964, Clarke 1993). The first NMDS axis distinguished two primary floristic regions. One group (areas A-G, I), located on the negative side of NMDS1, corresponded to the southern half of the Baja California Peninsula; the second group (areas L, M, O, P, Q, R), situated on the positive side, included northern Baja California and southern California.
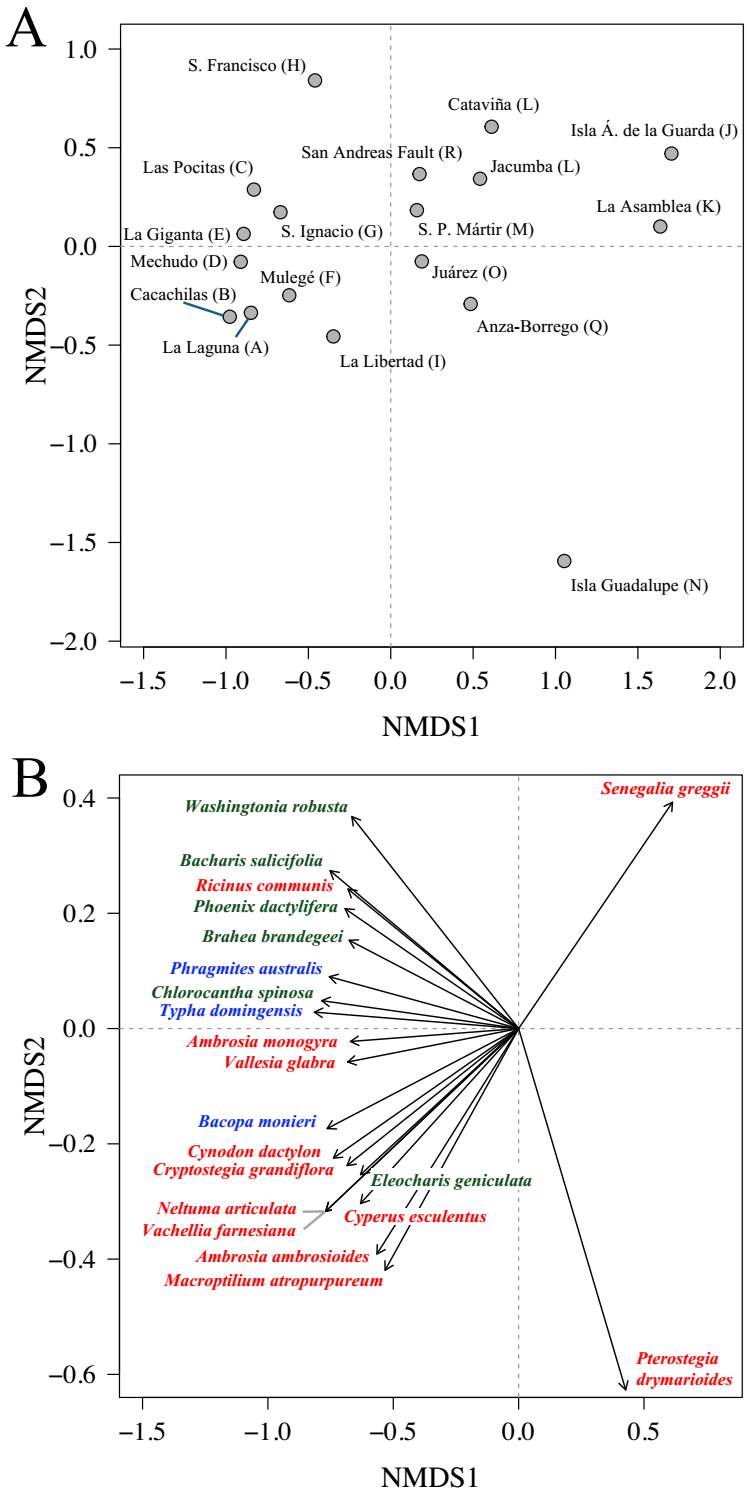
Figure 5 NMDS ordination of 18 palm grove areas listed in Figure 1 (from South to North, A-R) in southern California and the Baja California Peninsula based on species composition of hydrophytic flora (aquatic, subaquatic, and tolerant species) (stress = 0.124) . (A) Ordination of areas in two-dimensional space. (B) Species vectors were significantly associated with the ordination (P < 0.01). Axis ranges in (B) are different from (A) representing a subset of the ordination space. Species color indicates functional group: blue = aquatic, green = subaquatic, red = tolerant species.
Four areas emerged as floristic outliers: (i) Sierra de San Francisco (H), in central Baja California, positioned between the two main clusters but clearly offset along the positive end of NMDS2; (ii) Ángel de la Guarda Island (J) and (iii) Sierra de la Asamblea (K), both situated at the far right of NMDS1, indicating strong differentiation from all other regions; and, finally, (iv) Guadalupe Island (N), sharply isolated along the negative end of NMDS2, reflecting its unique biogeographic history. These spatial patterns broadly mirrored the native palm distribution across the region. Brahea armata and Washingtonia filifera occurred primarily in the northern group, while B. brandegeei and W. robusta were more common in southern areas. Brahea edulis, endemic to Guadalupe Island, underscored the floristic uniqueness of area N.
Species vectors projected onto the NMDS ordination space (Figure 5B) showed a strong directional signal, helping to interpret the spatial configuration of palm-grove areas based on floristic composition. Among the 21 species with significant associations (P < 0.01), 19 vectors were oriented toward the negative side of NMDS1, corresponding to areas in the southern Baja California Peninsula. These vectors included species classified as tolerant, subaquatic, and aquatic. A secondary gradient was captured along NMDS2. Of the 11 species vectors positioned in the negative half of NMDS2, the majority were tolerant taxa (nine tolerant, one aquatic, and one subaquatic); in contrast, the eight vectors with positive NMDS2 values were dominated by subaquatic (5) and aquatic (2) species, with only one tolerant species. Only two species -Pterostegia drymarioides and Senegalia greggii, both classified as tolerant- had vectors clearly oriented toward the positive side of NMDS1, associated with northern areas. This distribution indicates that most species with significant associations are linked to the southern portion of the gradient, with further ecological differentiation captured along the NMDS2 axis. In addition to the 21 species shown in Figure 5B, 28 other species were significantly associated with the ordination space at P < 0.05 (Table S2). These included one species (Brahea armata) located in the first quadrant (positive NMDS1 and NMDS2), a group of eight species -mostly tolerant, except B. edulis- in the second quadrant (positive NMDS1 and negative NMDS2), primarily associated with Guadalupe Island, and a larger group of 19 species in the third quadrant (negative NMDS1 and NMDS2). None of the additional 28 species showed significant associations in the fourth quadrant (negative NMDS1 and positive NMDS2).
The dbRDA model explained 55.5 % of the total floristic variation across palm-grove areas, with the first three canonical axes accounting for 75 % of the constrained variance. The overall model was statistically significant (F = 1.78, P = 0.0006), indicating that the selected climate variables explained a substantial portion of the regional compositional structure. Among them, only precipitation during the driest quarter (bio17) emerged as a statistically significant individual predictor (P = 0.0289).
Floristic assemblages across palm-grove areas were significantly associated with climatic gradients, as revealed by the distance-based redundancy analysis (dbRDA; Figure 6). The first canonical axis (CAP1) captured the primary latitudinal and climatic transition across the region. Areas on the negative side of CAP1 (A-I areas) represented the southern peninsula, characterized by warm, tropical-seasonal climates, with high annual temperature (bio1), strong precipitation seasonality (bio15), and elevated total annual precipitation (bio12). In contrast, northern areas (L-R), were positioned on the positive side of CAP1, primarily associated with higher precipitation during the driest quarter (bio17), indicative of reduced seasonality and a predominance of winter-dominated rainfall typical of Mediterranean regimes. The second canonical axis (CAP2) further separated northern areas. A compact group of Mediterranean-influenced areas (L, M, O, P, Q, R), appeared on the negative side of CAP2, linked to temperature seasonality (bio4), annual temperature range (bio7), and high maximum temperatures (bio5). In contrast, ecological outliers such as Guadalupe Island (N), the Central Desert (K), and Ángel de la Guarda Island (J) were clearly positioned on the positive side of both axes, reflecting unique climatic profiles distinct from both the Mediterranean and the southern tropical regimes. Together, the dbRDA axes underscore a strong and interpretable climate-floristics relationship, reinforcing the role of large-scale latitudinal gradients and local climatic distinctiveness in shaping palm-grove floras.
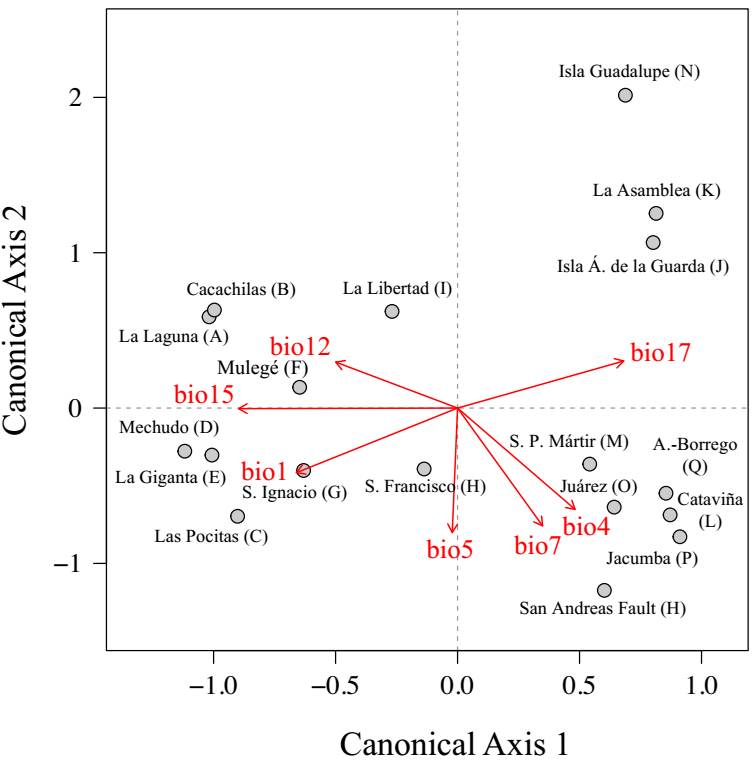
Figure 6 Distance-based redundancy analysis (dbRDA) showing the relationship between floristic composition (Sørensen dissimilarity index) and climatic gradients across 18 palm grove areas in southern California and the Baja California Peninsula. Circles represent areas and red arrows indicate the direction and strength of significant bioclimatic variables. Variable codes (from WorldClim): bio1 = annual mean temperature, bio4 = temperature seasonality, bio5 = max temperature of warmest month, bio7 = annual temperature range, bio12 = annual precipitation, bio15 = precipitation seasonality, bio17 = precipitation of driest quarter.
Discussion
This study had two primary aims: (1) to compile a comprehensive inventory of wetland-associated plant species occurring in palm groves across the Baja California Peninsula and southern California, and (2) to test whether floristic composition varies along the region’s pronounced climatic gradient. Both aims were supported by our results. We documented 368 wetland-associated species -classified as aquatic, subaquatic, or tolerant- across 235 palm groves distributed along a 1,400 km latitudinal transect. These species represent a significant component of the region’s wetland flora and provide insight into how ecological and biogeographic factors shape species distributions within these arid land oases.
Although palm groves occupy a relatively small fraction of the regional landscape, they contribute disproportionately to biodiversity, highlighting the uniqueness of wetland ecosystems. Notably, palm groves in the Baja California Peninsula harbor approximately 40 % of the strictly aquatic species previously reported for the region (Mora-Olivo et al. 2013), underscoring their role as key refugia for hydrophytic flora in water-limited environments. This likely reflects the persistence of groundwater and localized moisture, supporting plant assemblages that would otherwise be excluded from surrounding arid conditions. Similar patterns have been described for groundwater-dependent systems in the Colorado River Basin, where small springs sustain unexpectedly high levels of biodiversity despite their spatially restricted distribution (Stevens et al. 2020). In this context, arid palm groves function as ecological hotspots -concentrating distinctive floristic diversity and contributing to landscape-scale heterogeneity under strong climatic constraints.
Floristic composition exhibited a clear regional structure across the study area. The NMDS ordination revealed two major clusters of palm-grove areas arranged along a latitudinal gradient: a southern group of areas (A-G, I) associated with tropical-seasonal climates, and a northern group (L-R) aligned with Mediterranean regimes. Four areas -Guadalupe Island (N), Ángel de la Guarda Island (J), Sierra de la Asamblea (K), and Sierra de San Francisco (H)- stood out as floristic outliers, occupying peripheral positions in the ordination space and reflecting unique biogeographic and ecological conditions. Guadalupe Island, with its pronounced oceanic isolation (250 km offshore), likely shows floristic distinctiveness due to its wide spatial separation, while Ángel de la Guarda Island, although only 12 km from the mainland, being an island separated from the continent by a deep channel, may reflect limited connectivity or undersampling. Similarly, Sierra de la Asamblea and Sierra de San Francisco, two montane areas within arid matrix, may function as mountain islands, exhibiting ecological isolation comparable to that of true oceanic islands. These cases highlight how distance and ecological isolation contribute to floristic distinctiveness within the region. These patterns were reinforced by the dbRDA, which quantified the relationship between floristic composition and climatic predictors. Climatic variables explained 55.5 % of the total variation in floristic composition, with most of the explained variance captured by the first two canonical axes. CAP1 mirrored the main latitudinal contrast in the NMDS, with southern areas (A-I) linked to higher temperatures (bio1), stronger precipitation seasonality (bio15), and total annual rainfall (bio12), while northern areas (L-R) were associated with increased winter precipitation (bio17).
Species vectors projected onto the NMDS configuration provided further insight into the structuring of this compositional space. Among the 21 species significantly associated with the NMDS axes (P < 0.01), 19 pointed toward the southern portion of the ordination, indicating a concentrated signal from southern areas. This convergence of species associations and the strong climatic signal detected by CAP1 suggests that floristic distinctiveness in the southern peninsula plays a dominant role in defining the primary compositional axis across both ordination spaces. Only two species, both tolerant, had vectors pointing toward the northern areas: Senegalia greggii toward quadrant one, and Pterostegia drymarioides toward quadrant two, associated with the outlier Guadalupe Island. When expanding the analysis to include the broader set of species with P < 0.05, the overall spatial signal was reinforced rather than altered. Only one additional species -the blue palm (Brahea armata)- was associated with quadrant one. In contrast, the second quadrant incorporated eight additional species all recorded exclusively in the palm grove of Guadalupe Island, including its endemic palm B. edulis, suggesting a more complex but cohesive floristic profile for Guadalupe Island. The third quadrant, corresponding to southern desert and tropical regions, concentrated the largest number of species (19), further supporting the interpretation of this sector as a core zone of hydrophytic floristic richness. These extended patterns underscore the asymmetry in species associations across the NMDS space and suggest that the southern portion of the peninsula exhibits a more consistent and structured compositional signal, contributing disproportionately to the ordination space.
This north-south differentiation aligns with broader biogeographic patterns in the region as reviewed extensively by Garcillán et al. (2010). For instance, plant endemism increases toward the southern half of the peninsula -391 endemic taxa versus 294 in the north (Rebman et al. 2016)- and woody legume richness is also higher in the south (Garcillán et al. 2003). In this southern part, several factors converge to enhance biogeographic distinctiveness. It functions currently as an island of tropical climate that combines its geological origin from tectonic separation with its location at the end of a long, narrow peninsula. The associated peninsular effect limits dispersal from contienental through the isthmus and has sustained isolation and associated vicariant patterns over time. Alongside these historically and contextually specific features of the study region, there is an underlying north-south gradient from temperate to tropical conditions in species composition, consistent with biogeographic patterns documented at broader scales across freshwater systems. For instance, species richness and turnover in Mexican wetlands increase toward lower latitudes (Lobato-de Magalhães & Martínez 2018), and similar latitudinal patterns have been reported in European freshwater taxa (Heino 2001) and macroecological studies of freshwater diversity more generally (Heino 2011).
A secondary structuring pattern was observed along the second axis of the ecological ordinations. The secondary ordination axes (NMDS2 and CAP2) both capture additional layers of ecological variation, although their interpretations are not strictly parallel. CAP2 separates Mediterranean areas from hyperarid outliers (J, K, N), while in the south, it appears to distinguish between precipitation-influenced zones -such as the Cape region (A, B), Mulegé oasis (F), and Sierra de La Libertad (I)- and thermally stressful but likely hydrologically buffered western slope sites (C, D, E, G, H). In contrast, NMDS2 appears more directly linked to the prevalence of wetland-associated species categories. Aquatic and subaquatic species cluster toward the positive end of the axis, primarily aligning with western slope areas (e.g., C, G, E), while zones with higher overall precipitation are instead associated with tolerant species. This pattern is somewhat counterintuitive and may reflect the influence of localized moisture availability -such as spring-fed streams or shaded canyons- that allows hydrophilic taxa to persist in otherwise arid settings. Thus, while CAP2 emphasizes macroclimatic differentiation, NMDS2 appears to reflect floristic variation that may be influenced by finer-scale ecological factors such as microhabitat hydrology, partially decoupled from broad-scale precipitation regimes. Taken together, the NMDS and dbRDA reveal a dual structure in the regional organization of wetland flora: one axis reflecting broad latitudinal and climatic gradients, and a second capturing finer-scale ecological variation. This interpretation is reinforced by the close correspondence in site configuration between the two analyses and by a broad and diverse set of species associations significant at P < 0.05, which collectively provide a robust floristic basis for the compositional patterns identified.
Comparable floristic patterns have been described in diverse wetland contexts. In northeastern Brazil, aquatic plant assemblages vary across wetland types (Matias et al. 2021), and in Veracruz interdunal wetlands, vegetation shifts with the number of dry months (Peralta-Peláez & Moreno-Casasola 2009). Similar hydrology-linked structuring has been observed in riparian desert systems (Stromberg & Merritt 2016), in tropical wetlands of southern Mexico (Jimeno et al. 2025), and in Andean valleys where local topography and soil properties drive species turnover (Polk et al. 2019).
The floristic patterns also reflect broader family-level trends. Dominant wetland families such as Cyperaceae, Poaceae, and Asteraceae are widely represented across arid, tropical, and temperate wetlands in Mexico (Bonilla-Barbosa 2007, Mora-Olivo & Villaseñor 2007, Zepeda-Gómez et al. 2012) and globally (Magalhães et al. 2016, Chambers et al. 2008, Polk et al. 2019, Oliveira et al. 2019, Matias et al. 2021). Their ecological versatility -especially tolerance to fluctuating water regimes- helps explain their dominance in palm groves.
Comparisons with other regional wetland systems reveal declining similarity with increasing geographic distance and climatic contrast. Of the species recorded in this study, 80 % are also included in the National Wetland Plant List for the Arid West flora (U.S. Army Corps of Engineers 2023). This proportion drops to 30 % when compared with Mojave-Sonoran desert springs (Fraga et al. 2023), and declines further in more distant or climatically distinct regions -54 % shared with Tamaulipas (Mora-Olivo & Villaseñor 2007), 43 % with the Trans-Mexican Volcanic Belt (Bonilla-Barbosa 2007), 28 % with Guerrero (Villar-Morales 2016), and only 13 % with the Central Mexican Highlands (Lobato-de Magalhães & Martínez 2018). These trends point to biogeographic filtering and climatic affinity as major drivers of species composition.
Non-native species accounted for 9 % of palm-grove flora, a proportion comparable to regional estimates for the Baja California Peninsula (Rebman et al. 2016) and for desert springs in southern California (Fraga et al. 2023). However, they were disproportionately represented among tolerant (56 %) and subaquatic species (12 %), suggesting that invasion success may be linked to the hydrological flexibility of these non-native taxa. This trend reflects broader ecological dynamics, where species tolerant to fluctuating water levels tend to dominate in variable systems, while stable aquatic environments favor more hydrophytic taxa (Bornette & Puijalon 2011). Previous research has shown that even subtle changes in wetland hydrology -whether in average water levels or temporal variability- can shift community composition, often favoring alien over native species (Zedler & Kercher 2004, Magee & Kentula 2005). Similar patterns have been observed in prairie wetlands, where disturbances such as fire increased the proportion of tolerant species within plant assemblages (Rodríguez-Arias et al. 2018), further supporting the idea that disturbance and hydrological variability can facilitate the establishment of generalist or invasive taxa. These findings align with ecological theory identifying environmental variability as a driver of invasion, particularly in systems with recurrent fluctuation, where species adapted to both wet and dry phases may outcompete more specialized taxa (Davis et al. 2000, Richardson & Pyšek 2006).
Together, these patterns underscore how palm groves act as convergence points for ecological, climatic, and hydrological dynamics. Their unique floristic composition reflects the influence of persistent water sources, and the filtering effects of regional climate and species dispersal. In this sense, they represent distinctive functional and biogeographic nodes within arid landscapes. Our findings reinforce the importance of conserving these systems -not only as refugia for specialized wetland taxa, but also as indicators of broader ecological and climatic transitions across the Baja California Peninsula and southern California.
Supplementary material
Supplemental data for this article can be accessed here: https://doi.org/10.17129/botsci.3739











 nueva página del texto (beta)
nueva página del texto (beta)



