The family of legumes (Leguminosae) economically represents the second most important of the cultivated plants after the Poaceae (Llamas & Acedo 2016, Jimenez-Lopez et al. 2020) and is made up of approximately 770 genera and about 19,500 species (Azani et al. 2017). Legumes are important in animal nutrition, both for the nutritional value of fruits and seeds, and for the rest of the plant that is used as forage or grazing (Fontes et al. 2008a, Mazorra et al. 2016). In this sense, the biomass of trees, shrubs and especially forage legumes have a leading role due to their considerable protein content and acceptable nutritional value (Mazorra et al. 2016). In addition, legumes have a general characteristic that is to harbor atmospheric nitrogen-fixing bacteria in their roots to provide it to the plant when necessary (Llamas & Acedo 2016).
In Cuba, the most important genera are Centrosema, Desmodium, Neonotonia, Stylosanthes and Teramnus (Ruiz et al. 2015). Within the genus Teramnus, one of the most important species is the perennial herbaceous Teramnus labialis (L.f.) Spreng (Menéndez 1982, Olivera 2011, Toral et al. 2015). The greatest use of this species is as a protein source in the diet of bovine cattle (Toral et al. 2015), which is due to its acceptable biomass production and nutritional quality (Machado & Olivera 2008). However, other authors also refer to this legume as a cover crop on fruit trees such as bananas (Gutiérrez et al. 2002), the citrus (Fontes et al. 2008b) and the guava (Fontes et al. 2018).
Despite the potential of legumes, currently it has not been possible to extend their use in different agricultural systems due to the low availability of existing seeds. Additionally, the low percentage of germination limits the establishment in the field of these species (González 2011, Fontes et al. 2018) and in particular of T. labialis (Acosta et al. 2020c, 2021). In the case of this species, the low germination percentage is associated with the presence of an impermeable seed coat (Acosta et al. 2020b, 2020c, 2023b).
Of the five classes of seed dormancy recognized (Baskin & Baskin 2014, 2021), the impermeability of the seed coat is a characteristic feature of physical or combined dormancy (physical + physiological) (Wang et al. 2019, Rodrigues-Junior et al. 2020). The first step to overcome physical or combined dormancy is to eliminate the impermeability of the seed through a scarification treatment (Baskin & Baskin 2014). These treatments are grouped into three different methods: mechanical, chemical and physical (de Morais et al. 2014, Schnadelbach et al. 2016, Apodaca-Martínez et al. 2019).
The treatment of T. labialis seeds with sulfuric acid (chemical method) was described by Muñoz et al. (2009) and Acosta et al. (2023b); although the use of hot water (physical method) is the most studied procedure in this species (González & Mendoza 1991, Fontes et al. 2008a, Mazorra et al. 2016). Among the physical methods, the literature refers to the use of liquid nitrogen (LN) as a scarifying agent in seeds of many legume species (Acharya et al. 1999, Voronkova & Kholina 2010, Wu et al. 2017) and particularly in T. labialis (Acosta et al. 2019, 2020b). However, for T. labialis there are no references to the influence of these scarification treatments during germination and establishment under field conditions, so this study is aimed to compare the effect of four different scarification treatments during germination and establishment of T. labialis.
Material and methods
Seeds material. Seeds were collected in March 2019 from fifty adult plants of Teramnus labialis (L.f.) Spreng, cultivated in Ciego de Ávila, Cuba. (21˚ 89` 14.07” N; 78˚ 69` 67.53” W). The seeds had a moisture content of 7.56 % and were stored for three months as described by Acosta et al. (2020a).
Scarification treatments. Two thousand seeds were taken for each scarification treatment and the same amount for the control treatment (without scarification). All scarification treatments were performed as described by Acosta et al. (2023b) for this species:
Abrasion.- The seed coat was degraded by abrasion between two sheets of fine-grain sandpaper (number 180 for 5 s), sanding was done manually taking care not to press the seeds excessively so as not to damage the embryo.
Sulphuric acid (95 %).- Seeds were dipped in concentrated sulphuric acid for 5 min. At the end of this time, seeds were washed severally in distilled water and dried.
Hot water (80 °C).- Seeds were dipped in hot water for 5 min. At the end of this time, seeds were dried.
Liquid nitrogen (-196 °C).- Seeds were put in polypropylene cryo-vials (two thousand seeds per crio-vial) and immerse directly in LN for 30 min. After extracting the seeds, they were placed on a plastic tray for 2 h until they reached room temperature (± 25 °C).
After scarification, seeds from all treatments, including the control treatment, were used to develop each of the experiments described below.
Experiment under laboratory conditions. Seeds coat structure.- Twenty seeds, for each scarification treatment and control, were randomly selected for histological studies were performed according to Johansen (1940). Samples were fixed in FAA, dehydrated in an ethanol series, and included in Paraffin. Cross sections (5 µm thick) were cut with a hand rotary microtome (KD-202A) and collected on microscopic slides covered with gelatin solution. Sections were stained with solutions of 1 % safranin for 24 hours to dye cell walls red or deep pink. Observations were carried out using a microscope (Carl Zeiss Microlmaging GmbH 37081) at 200X, and photography by Cannon digital camera (EOS 600D).
Seeds imbibition.- To determine the imbibition capacity, three replicates of 50 seeds were used for each treatment. The seeds of each replicate were weighed in their entirety (50 seeds) and separately (each individual seed) on an analytical balance (Sartorius) and placed in Petri dishes (90 mm diameter) on filter paper moistened with 5 ml of distilled water (the seeds of each replicate in a Petri dish). After 24 h, the seeds were removed, dried and weighed again (in the same way as before placing them to be imbibed). With the mass of each individual seed (50 seeds of each replicate) the percentage of imbibed seeds (PES) was calculated according to Baskin et al. (2007), and with the total mass of the seeds of each replicate (50 seeds), the percentage of seed absorption (PSA) was calculated according to Baskin et al. (2004).
Seeds electrolyte leakage.- Three replicates of 9 g of seeds (approximately 1,320 seeds) were used in each scarification treatment, to measure electrolyte leakage (Moreno Casasola 1996). Seeds were surface sterilized with 1 % sodium hypochlorite for 2 min. They were then rinsed with distilled water and placed in a beaker with 10 mL of distilled water for 24 h at a temperature of 25 °C. Leachates were collected after 24 h of imbibition and conductivity was measured with a conductivity meter (Sartorius, pp-20).
Seeds germination trials.- Four replicates of 25 seeds, for each treatment, were tested for germination at constant temperature 30 ± 2 °C under dark at 80 % relative humidity, for 28 days in a programmable growth chamber (TOP Cloud-agri, RTOP-1000 B/D). Seeds from each replicate (25 seeds) were placed on filter paper in a 90 mm diameter Petri-dish (unsealed). The filter paper was moistened with 5 mL distilled water every 7 days. Daily germination counts, based on the length radicle (≥ 2 mm). Using the germination test data, the following numerical variables associated with seed vigor were calculated to estimate the effect of the different scarification treatments on seed viability and vigor during germination: (G) germination percentage, (T50) time necessary for 50 % of the seeds to germinate, (GI) germination index, (MGT) mean germination time and (UG) uniformity of germination (Ranal & Santana 2006).
Experiment under field conditions. Seedling growth trials.- Seeds were sown in a site representative of Typical Fersialitic soil farmland in Ciego de Avila, Cuba (21.99° 06’ 85” N; 78.76° 73’ 00” W). This type of soil, as described by Hernández et al. (2019) in the classification of Cuban soils, is moderately deep, little eroded, stony, with an effective depth of 30 cm and almost flat. For each treatment (plus the control) a plot of 4 ˟ 3 m (12 m2) was used. Four rows were made in each plot and the sowing was done at a distance of 0.30 ˟ 0.70 m, three seeds were placed in each sowing niche for a total of 170 seeds per plot. Plots were irrigated via sprinklers every 7 d until fruit formation. Border plants were not evaluated to exclude potential edge effects. The sowing was carried out in the month of June and the procedures were based on Mazorra et al. (2020).
Vegetative stage.- The following traits were measured according to Machado & Roche (2004) between 42 and 120 days after sowing (DAS): seedling emergence (%, 42 DAS), plant height (cm, 42 DAS), leaf per plant (42 DAS), leaf coverage (m2; 120 DAS).
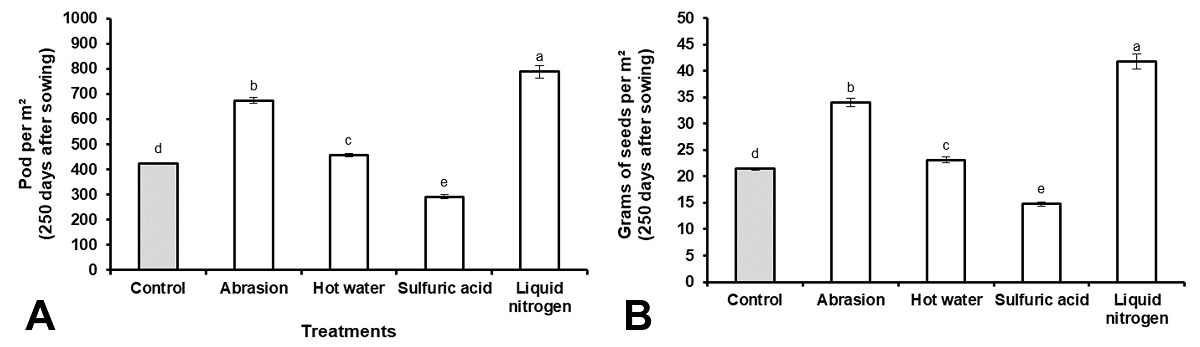
Reproductive stage.- The following traits were measured according to González (1995) at moments of harvest (250 DAS): Total pod per m2, grams of seeds per m2 (g m 2 -1).
Statistical analysis. All data were statistically analyzed using SPSS (Version 8.0 for Windows, SPSS Inc., New York, NY). Data were tested for normality using a Shapiro-Wilk test and means were compared using parametric tests (One-way ANOVA, P ≤ 0.05). Germination percentage data was transformed for analysis according to y' = 2arcosen ((y / 100)0.5).
Results
Seeds from the control treatment showed a completely intact seed coat (Figure 1A), where the cuticle, the macrosclereid cell layer and the osteosclereid cell layer could be seen. In contrast, exposure of the seeds to LN (Figure 1E) produced multiple cracks in the cuticle (Cu) and the macrosclereid cell layer, while exposure to hot water only caused small cracks (Figure 1D). Exposure of the seeds to sanding caused wear of the cuticle and part of the macrosclereid cell layer (Figure 1B), however, sulfuric acid degraded both cell layers and created corrosion and loss of the seed coat (Figure 1C).
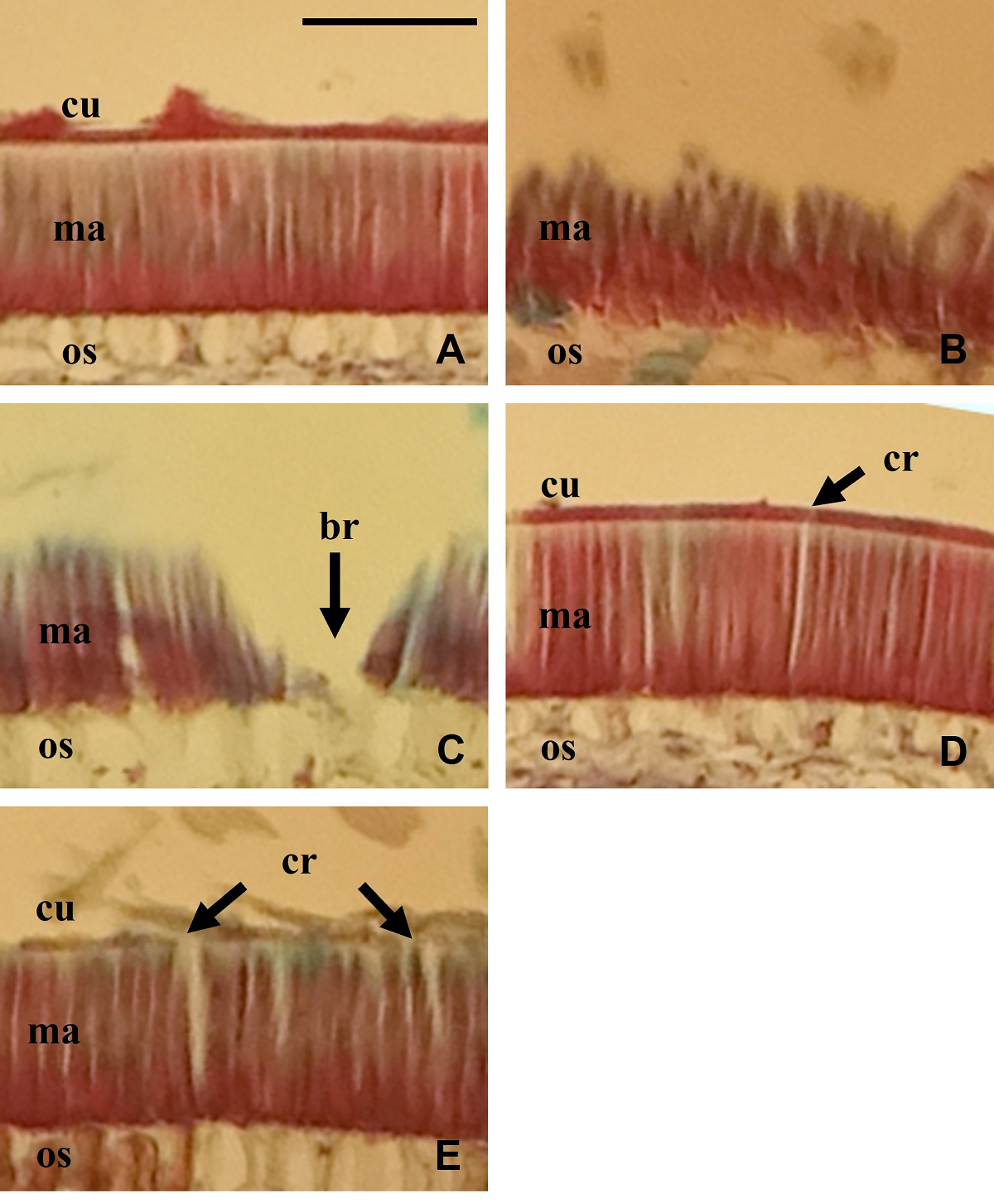
Figure 1 Teramnus labialis seeds coat morphology. (A) seeds without scarifying (control), (B) seeds scarified with sandpaper, (C) seeds scarified with sulfuric acid, (D) seeds scarified with hot water and (E) seeds scarified with LN. br = breaks, cr = cracks, cu = cuticle, ma = macrosclereid cell layer, os = osteosclereid cell layer. Bar represent 100 µm in all photos.
The percentage of seed absorption (PSA) increased with all the scarification treatments used (Figure 2A), however, the percentage of embedded seeds (PSE) was higher with the use of sulfuric acid (100 %) and LN (100 %) compared to the use of sanding (76 %) and hot water (51 %) (Figure 2B). Additionally, the loss of electrolytes during imbibition (Figure 2C) was significantly higher with the use of sulfuric acid compared to the rest of the scarification treatments, while a decrease in the loss of solutes to the medium was observed in the control treatment.
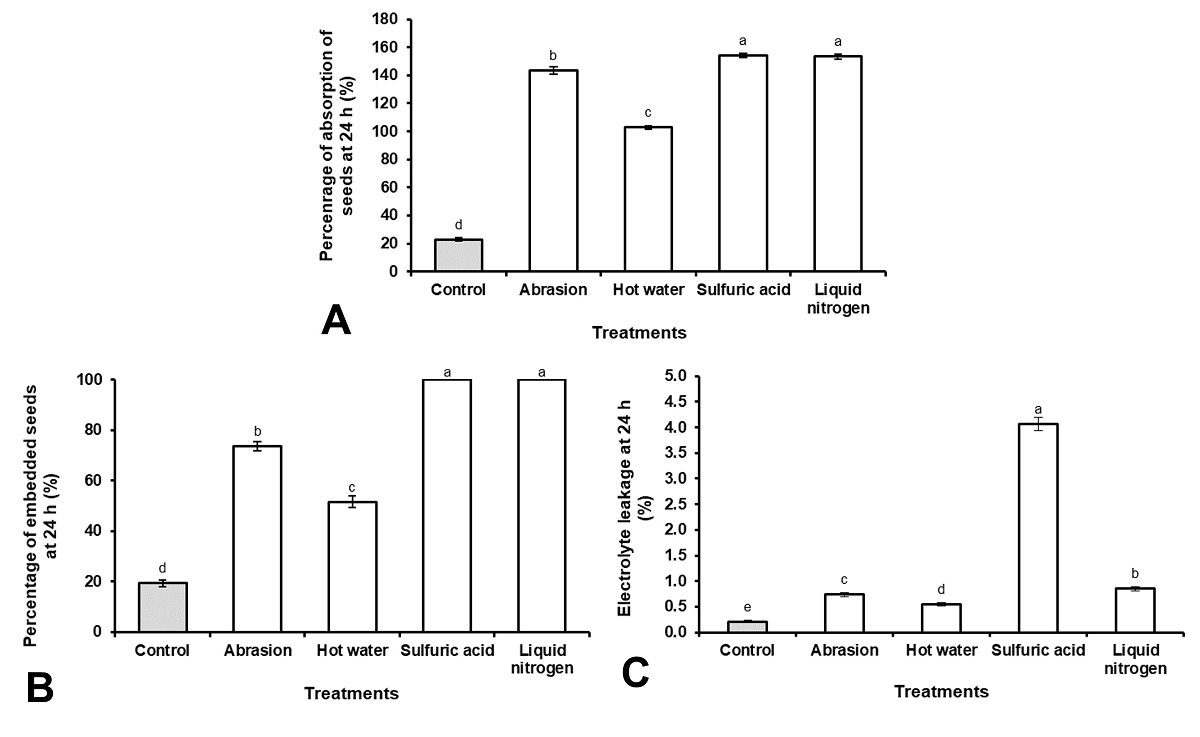
Figure 2 Teramnus labialis seeds physiological changes after scarification. (A) Imbibition capacity, (B) total seeds embedded and (C) electrolyte leakage. Results with the different letter, for each figure, are statistically different (One-way ANOVA, P ≤ 0.05, n = 3. Vertical bars represent mean ± SE.
The use of LN as a scarifying agent exhibited the highest percentage of germination (91 %), followed by sanding 79 %, sulfuric acid 68 % and hot water 62 % (Figure 3A). The use of hot water was the scarification treatment that showed the highest percentage of hard seeds (21 %) (Figure 3B), while the scarification with sulfuric acid showed the highest percentage of dead seeds (32 %) (Figure 3C).
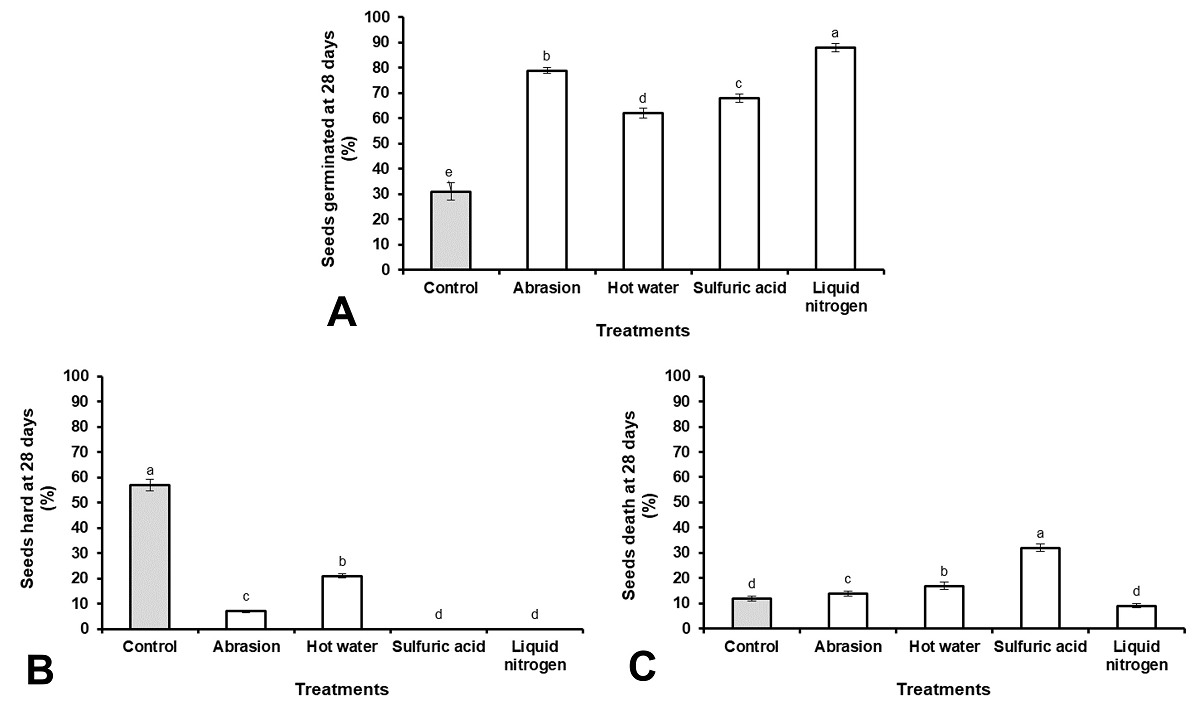
Figure 3 Teramnus labialis seeds physiological changes after scarification. (A) Seeds germinated, (B) seeds hard and (C) seeds death. Results with the different letter, for each figure, are statistically different (One-way ANOVA, P ≤ 0.05, n = 4. Vertical bars represent mean ± SE.
As expected, the use of scarification not only improved germination, but also the numerical variables associated with seed vigor (T50, GI, MGT and UG) (Table 1).
Table 1 Numerical variables associated with the vigor of unscarified (control) and scarified Teramnus labialis seeds using different treatments
| Scarification treatment |
Time required for germination of 50 % of the seeds (days) |
Mean germinated time (days) |
Germination index (seeds days-1) |
Germination uniformity (days) |
|---|---|---|---|---|
| Control | 7.94 ± 0.69 c | 1.06 ± 0.06 c | 9.12 ± 0.85 c | 11.78 ± 0.24 c |
| Abrasion | 1.99 ± 0.04 a | 7.26 ± 0.36 a | 2.81 ± 0.11 a | 2.69 ± 0.25 a |
| Hot water | 4.83 ± 0.25 b | 3.38 ± 0.17 b | 5.86 ± 0.31 b | 7.13 ± 0.44 b |
| Liquid nitrogen | 2.14 ± 0.15 a | 6.95 ± 0.28 a | 2.98 ± 0.21 a | 2.86 ± 0.23 a |
| Sulfuric acid | 1.93 ± 0.07 a | 6.93 ± 0.31 a | 2.61 ± 0.17 a | 2.47 ± 0.2 a |
Results with different letters, for each indicator, are statistically different (one-way ANOVA, P ≤ 0.05, n = 4). Only for statistical processing the data were transformed according to y´= y0.5. Intervals indicate mean ± standard error of the mean.
The percentage of plants emerged during the first 42 days after sowing was higher when LN was used as a scarification treatment (Figure 4A), as well as the height of the plants and the number of leaves per plant (Figure 4B, C). With the use of LN as a scarification treatment, 92 % of the covered area was reached 120 days after sowing (Figure 4C).
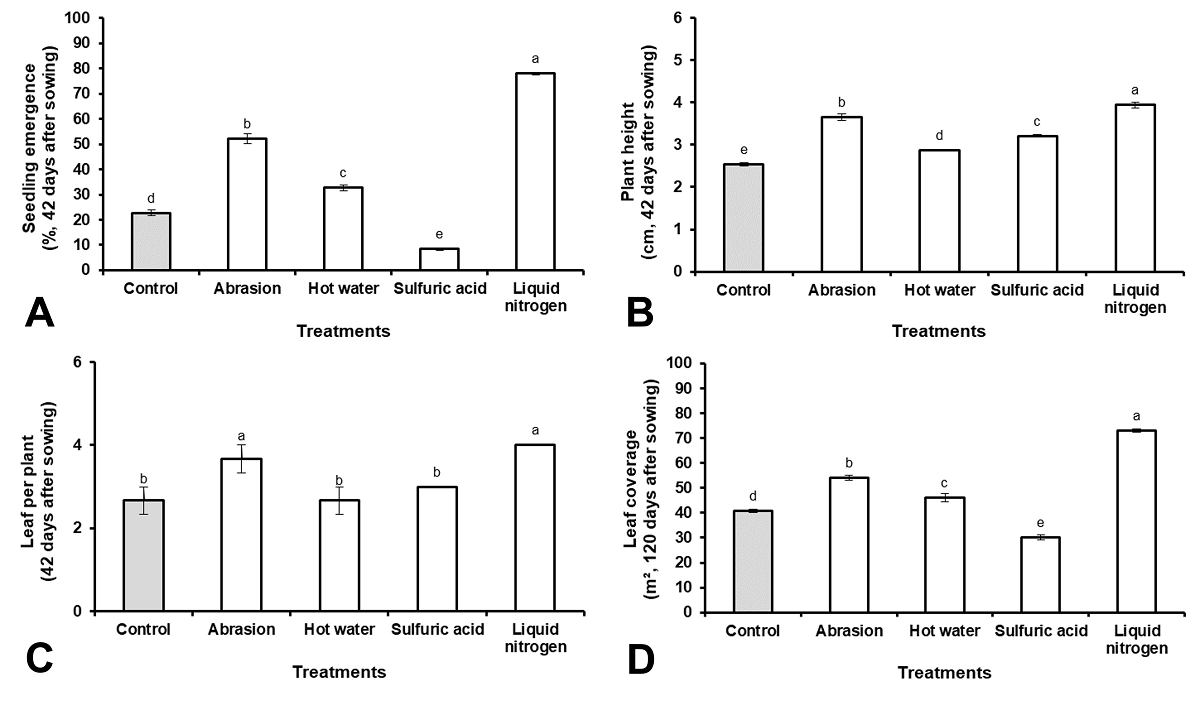
Figure 4 Teramnuslabialis plants growth during vegetative stage. (A) Seedling emergence, (B) plant height, (C) leaf per plant and (D) leaf coverage. Results with the different letter, for each figure, are statistically different (One-way ANOVA, P ≤ 0.05, n = 3). Vertical bars represent mean ± SE.
During the reproductive phase, the number of legumes per m2 (Figure 5A) and the total number of seeds per m2 (Figure 5B) were higher in the treatment of seeds scarified with LN. Additionally, the use of sanding was another treatment that showed superior results to the control, while the treatment with sulfuric acid produced the lowest number of legumes and seeds (Figure 5A, B).
Discussion
The anatomical characteristics observed in the seed coat of T. labialis seeds correspond to those described for species of the subfamily Papilionoideae by Rolston (1978), Okonwu & Ariaga (2017), da Silva et al. (2018), Shehata & Aqlan (2020) and Zemouri et al. (2020); and with the descriptions made for this species previously by Acosta et al. (2020a,b). The anatomical changes observed in the seed coat by the use of the LN are consistent with those observed in this species by Acosta et al. (2020b) and for other legume species by Luan et al. (2017), Jaganathan et al. (2018), Sun et al. (2018), Magalhães & Oliveira (2020) and Yousif et al. (2020).
The use of sulfuric acid and LN as scarification treatments allows to eliminate the impermeability of the seed coat and the imbibition of 100 % of the seeds. The depth of the corrosion and the loss of the seed coat with the use of sulfuric acid in the seeds of T. labialis agrees with what was observed in Erythrina speciosa (Andrew.) by Molizane et al. (2018). Our results coincide with the research work carried out by Müller et al. (2017), Zapata et al. (2017) and Kildisheva et al. (2018) to eliminate the impermeability of the seed coat of other leguminous species.
During imbibition, many reserve substances (sugars, fatty acids, ions, amino acids, enzymes, proteins, and inorganic ions) escape to the external medium through the membrane (Bewley et al. 2013, Marcos-Filho 2015). The increase in the leaching of solutes after scarifying the seeds corroborates the loss of integrity of the seed coat, which agrees with the studies carried out by Müller et al. (2017) and Sethi et al. (2020). However, an excessive leaching of solutes, as happened in those scarified with sulfuric acid, can lead to a decrease in germination and vigor of the seeds (Thant et al. 2017, Roach et al. 2018).
The results of imbibition, loss of electrolytes and germination that were achieved with the use of hot water as a scarification treatment were the most discreet. This is due to the fact that the treatment did not eliminate the impermeability of the seed coat in all the seeds, as reported by the studies carried out by Sánchez-Gómez et al. (2018) in Leucaena leucocephala (L.), Yousif et al. (2019) in Acacia nilotica (L.) and Singh et al. (2020) in Vigna membranacea (A.Rich.).
Previously, Muñoz et al. (2009) used scarification with sulfuric acid (95 % for 5 min.) on aged seeds of T. labialis and the results showed a high percentage of damaged seeds and, therefore, a low percentage of germination. Recently, Acosta et al. (2023b) used this treatment (95 % for 5 min.) on freshly harvested seeds of T. labialis and observed an increase in germination under controlled laboratory conditions up to 68 %. In the present investigation, scarification with sulfuric acid (95 % for 5 min.) increased the percentage of germination in seeds stored for 3 months (Table 1). The increase in germination was preceded by an increase in imbibition and the electrolyte loss, which shows that the treatment was effective in eliminating the impermeability of the seed coat. However, during field experiments, the percentage of seedling emergence decreased considerably demonstrating that the use of this treatment causes damage to the seeds as suggested by Muñoz et al. (2009) and Acosta et al. (2023b). Previous studies in other species highlight that the use of this treatment can increase the permeability of the seed coat, but also cause damage, mainly to the embryo (Galíndez et al. 2015, Zapata et al. 2017, Kildisheva et al. 2018), what affects seed viability (Okonwu & Ariaga 2017, Kheloufi et al. 2019, Yousif et al. 2019).
In relation to sanding and LN, the low percentages of dead seeds show that these treatments did not cause damage that could affect germination. The increases in water absorption, the high percentages of germination and the values obtained in all the numerical variables associated with the vigor of the seeds, demonstrate the effectiveness of these treatments for use in the seeds of T. labialis.
The speed and uniformity of the emergence after sowing is an advantage for the establishment of any species (Navarro et al. 2016, Sánchez-Gómez et al. 2018) and is related to the germination power and vigor of the seeds (Rajjou et al. 2012, Santorum et al. 2013). Ruiz & Febles (2005) reported that the establishment of a forage legume is not a simple event, but is a system integrated by planting, emergence and growth of the plant. In this sense, the emergence percentage, the size of the plants and the area covered at 120 days with the use of LN demonstrate the rapid establishment of the species. According to Fontes et al. (2008a) when the foliar cover of T. labialis reaches, at least, 70 % of the covered area, it can be classified as established.
The higher number of legumes per m2 and seeds per hectare observed in the plants that come from seeds scarified with LN, is due to the higher number of plants and foliar coverage that was obtained in this treatment. A greater leaf area in direct contact with solar radiation is a very important factor to guarantee adequate flowering and fruiting in tropical legumes (Hopkinson & Reid 1979, Matías & Matías 1995).
The study carried out during the reproductive phase also confirms that the use of LN as a scarifying agent does not cause variability at the morphological level, at least in the characters evaluated. These results coincide with those obtained in plants of Phaseolus vulgaris (L.) (Cejas et al. 2013), Solanum lycopersicum (Mill.) (Zevallos et al. 2014), Chrysanthemum morifolium (Ramat.) (Bi et al. 2016), Sorghum biclor (L.) (Villalobos et al. 2019), Cicer arietinum (L.) (Villalobos et al. 2021) and Neonotoni wightii (Wight & Arn.) (Acosta et al. 2023a).
The results of this investigation showed variability in the response to the different scarification treatments used, which is consistent with several studies carried out in other legume species (Jones et al. 2016, Lozano et al. 2016, Statwick 2016, Zapata et al. 2017, Kildisheva et al. 2018, Odirile et al. 2019, Singh et al. 2020). This is mainly related to the size and morpho-anatomical characteristics of the seeds, as well as the thickness and chemical composition of the seed coat (Jones et al. 2016, Lozano et al. 2016, Mira et al. 2017, Kheloufi et al. 2019, Odirile et al. 2019, Salazar & Ramírez 2019, Carruggio et al. 2020).
Considering the results obtained in this research, all scarification treatments increased the percentage of seed absorption, the percentage of embedded seeds, the loss of electrolytes and the percentage of germination. However, the use of sulfuric acid caused a disordered loss of solutes during imbibition, and, although the germination percentage was high, the percentage of dead seeds also increased. The damage caused to the seed with the use of this scarification treatment also caused a decrease in the percentage of seedlings emerged under field conditions and the percentage of covered area by the species at 120 d after sowing. With the use of liquid nitrogen as a scarification treatment, the best results were obtained in controlled laboratory conditions and in field conditions, reaching the establishment of the species in only 120 days, with a significant increase in the production of pods and seeds.











 nueva página del texto (beta)
nueva página del texto (beta)