Introduction
Cantharellus shows high diversity of species and ectomycorrhizal associations with several plant families, including Myrtaceae, Pinaceae, and Fagaceae (Corrales et al., 2018). Edible fungi are a non-timber forest product with high gastronomic and socioeconomic value, making them a functional food source with significant potential for sustainable use (Pérez-Moreno et al., 2021).
Despite many species records, comprehensive analyses are needed to understand the complexity associated with Cantharellus (Wang et al., 2023). One of the main challenges in defining species lies in the similarity of morphological traits, making molecular markers essential for identification (Buyck et al., 2014). Morphological identification alone can be deficient for fungi with similar external features, complicating the establishment of accurate phylogenetic relationships and the differentiation of closely related taxa. Additionally, few microscopic characteristics have been considered within Cantharellus, with the hyphae of the cuticle, and the size and shape of basidiospores being the most taxonomically relevant traits for subgenera and species (Buyck et al., 2014). In this regard, Lücking et al. (2020) emphasize that «the use of molecular data has transformed our understanding of fungal diversity, revealing cryptic species that cannot be distinguished morphologically». In recent years, taxonomic research on Cantharellus and the order Cantharellales, known as the "World Phylogenetics of Cantharellales", has allowed for clearer clade integration and the description of numerous new species in South Korea (Park et al., 2022). In Europe and Asia, phylogenetic analyses with multiple molecular markers have been conducted to elucidate and delimit species and new records (Olariaga et al., 2017; Zhang et al., 2022). Currently, around 300 species of the genus Cantharellus are known worldwide (Cao et al., 2021).
In Mexico, during explorations over the past four years in the pine forests of Pico de Orizaba National Park, Veracruz, specimens of Cantharellus were found that had been reported by Guzmán (1984) as C. odoratus; however, morphological evidence indicates that these are two new species records. Additionally, in this Mountain Region, C. violaceovinosus, C. veraecrusis, and C. tabarnensis have been found (Herrera et al., 2018; Montoya et al., 2021). These fungi differ in color according to their developmental stage and morphotype, with yellowish and brown caps and a hymenophore that is paler than those observed in the ‘new records’. Although new species of Cantharellus have been described in the region in recent years, it is believed that there may still be more yet to be discovered (Herrera et al., 2018; Montoya et al., 2021).
Edible fungi are valued and sold in several markets in the Mountain Region of central Veracruz; therefore, identifying wild fungi that grow in this region is important for revaluing the potential of this food and establishing strategies for its sustainable use and conservation. Consequently, the aim of this study was to identify the Cantharellus specimens collected in Pico de Orizaba National Park, specifically in the municipality of Calcahualco, Veracruz, both morphologically and molecularly, to understand the distribution of species within the genus.
Materials and Methods
Sampling and morphological study
The Cantharellus basidiomata were collected between July and November for four consecutive years (2019-2022) in the forests of Pico de Orizaba National Park, specifically in the municipality of Calcahualco (3 050-3 750 m) in the Mountain Region of Veracruz, Mexico. In these forests, Pinus hartwegii Lindl. dominates, although P. montezumae Lamb. and P. pseudostrobus Lindl. are also present. The species P. hartwegii is primarily found at altitudes between 3 000 and 4 000 m and is known for its tolerance to low temperatures (ranging from -10 °C to 18 °C) in mountainous areas with annual precipitation between 600 and 800 mm (Pérez-Suárez et al., 2022). The presence of P. montezumae is generally recorded at lower altitudes than P. hartwegii, while P. pseudostrobus is found in a similar altitudinal range, thus influencing the diversity of fungi involved (Gallardo-Salazar et al., 2023).
The morphoanatomical characteristics of fresh samples were described based on Largent (1973). The color notations indicated in the descriptions were defined according to Kornerup and Wanscher (1967) and the Munsell color chart (Munsell Color Company Inc., 1994).
Collected materials were dried in a drying oven at 35 °C for incorporation into a mycological collection. For the morphological review, temporary sections were prepared with 3 % KOH and stained with Congo red. A total of 35 basidiospores were measured for each sample (five specimens); their shape was interpreted using the Q statistic according to Bas (1969).
DNA extraction, PCR and sequencing
DNA was isolated from fresh material following the conditions of the protocol recommended by the manufacturer of the “Plant/Fungi DNA Isolation Kit” (Norgen Biotek Corp., Canada). The transcription elongation factor 1-alpha (tef1-α) region was amplified using the primers tef1F and tef1R (Olariaga et al., 2017) according to the protocols of O'Donnell et al. (2011).
The 5.8S-ITS2-LSU regions were amplified together using the primers ITS3C-ITS4 (Olariaga et al., 2015). The PCR amplification conditions followed those described by Olariaga et al. (2015) for the 5.8S-ITS2 regions, with the number of cycles increased to 40 for problematic samples.
PCR amplification products were purified and sequenced by Macrogen® in South Korea. Sequences were assembled and edited using AliView (Larsson, 2014) and deposited in the GenBank database.
Phylogenetic analysis
In this study, four tef1-α and ITS sequences were determined, and 125 sequences from Cantharellus species from all the main clades of the genus (after Buyck et al., 2014) were used, along with the highest similarity scores from the BLAST results downloaded from GenBank (http://www.ncbi.nlm.nih.gov/), to construct two datasets (Table 1). One dataset consisted of tef1-α sequences, and the second dataset was composed of combined tef1-α + ITS sequences. Craterellus tubaeformis was included as the outgroup.
Table 1 ITS and tef1-α sequences of 125 Cantharellus species (from GenBank) with the highest similarity to the sequences of the collected samples (indicated with an asterisk) from Pico de Orizaba National Park. ET: epitype, HT: holotype, IT: isotype, NT: neotype, PT: paratype.
| Taxon | Record number | Country | GenBank Access number | |
|---|---|---|---|---|
| ITS2 | tef1 | |||
| C. afrocibarius HT | 496/BB 96.235 | Zambia | - | JX192993 |
| C. albidolutescens HT | 457/BB 08.070 | Madagascar | - | JX192982 |
| C. alborufescens HT | MPU027371 | Morrocco | KX828764 | - |
| C. alborufescens (1) | JLS880 (AH) | Spain | KR677493 | KX828816 |
| C. alborufescens (2) | 1108/BB 12.075 | Italy | KX907209 | KX907243 |
| C. alborufescens (3) | 1109/BB 12.076 | Italy | - | KX907244 |
| C. alborufescens (4) | 1006/EC 09.91 | Italy | - | KX907242 |
| C. alborufescens (5) | AH44783 | France | KR677492 | KX828817 |
| C. alborufescens (6) | BIO-Fungi 11687 | Spain | KX828765 | KX828818 |
| C. alborufescens (7) | BB.12.088 | Italy | - | KX907245 |
| C. alborufescens (8) | BIO-Fungi 12025 | Spain | KR67749 | - |
| C. alborufescens (9) | BIO-Fungi 11691 | Spain | KX828766 | - |
| C. altipes (1) | 337/BB 07.115 | USA | JN944018 | - |
| C. altipes (2) PT | 344/BB 07162 | USA | - | GQ914945 |
| C. amethysteus NT | AH44796 | Spain | KR677512 | KX828819 |
| C. amethysteus (1) | 994/Estades 10.454 | France | KX907205 | KX907237 |
| C. amethysteus (2) | 349/BB 07.284 | Slovakia | JN944020 | GQ914953 |
| C. amethysteus (3) | 352/BB 07.309 | Slovakia | - | GQ914954 |
| C. amethysteus (4) | 993/Estades 10.453 | France | - | KX907236 |
| C. amethysteus (5) | 1003/EC 09.29 | Italy | - | KX907241 |
| C. californicus HT | OSC 122878 | USA | KX828768 | KX828820 |
| C. cibarius ET | BIO-Fungi 10986 | Sweden | KR677501 | KX828823 |
| C. cibarius (1) | AH44780 | Spain | KR677508 | KX828821 |
| C. cibarius (2) | BIO-Fungi 10780 | France | KR677503 | KX828822 |
| C. cibarius (3) | AH44778 | Spain | KX828769 | KX828824 |
| C. cibarius (4) | BIO-Fungi 12684 | Switzerland | KR677505 | KX828825 |
| C. cibarius (5) | 479/GE 07.025 | France | KX907204 | GQ914949 |
| C. cibarius (6) | 351/BB 07.300 | Slovakia | - | GQ914950 |
| C. cibarius (7) | BIO-Fungi 10477 | Spain | KX828770 | - |
| C. cibarius (8) | BIO-Fungi 12701 | Spain | KR677504 | - |
| C. cibarius var. atlanticus HT | PC0142164 | France | KX828771 | - |
| C. cibarius var. bicolor NT | BIO-Fungi 11230 | Spain | KX853517 | - |
| C. cibarius var. flavipes IT | 9312B2 (Mornand herbarium) | France | KX828772 | - |
| C. cinnabarinus NT | 312/BB 07.001 | USA | - | GQ914985 |
| C. decolorans ET | 469/BB 08.278 (PC) | Madagascar | KX907203 | GQ914968 |
| C. ferruginascens HT | E00204187 | United Kingdom | KX828773 | - |
| C. ferruginascens (1) | K(M)180009 | United Kingdom | KX828774 | - |
| C. ferruginascens (2) | AH44782 | France | KR677488 | KX828826 |
| C. ferruginascens (3) | AH44794 | Spain | KR67785 | KX828827 |
| C. ferruginascens (4) | AH44795 | Spain | KX828775 | KX828828 |
| C. ferruginascens (5) | BIO-Fungi 11700 | Spain | KR677486 | KX828829 |
| C. ferruginascens (6) | 347/BB 07.221 | Slovakia | - | KX907235 |
| C. cibarius var. flavipes IT | 9312B2 (Mornand herbarium) | France | KX828772 | - |
| C. cinnabarinus NT | 312/BB 07.001 | USA | - | GQ914985 |
| C. decolorans ET | 469/BB 08.278 (PC) | Madagascar | KX907203 | GQ914968 |
| C. ferruginascens HT | E00204187 | United Kingdom | KX828773 | - |
| C. ferruginascens (1) | K(M)180009 | United Kingdom | KX828774 | - |
| C. ferruginascens (2) | AH44782 | France | KR677488 | KX828826 |
| C. ferruginascens (3) | AH44794 | Spain | KR67785 | KX828827 |
| C. ferruginascens (4) | AH44795 | Spain | KX828775 | KX828828 |
| C. ferruginascens (5) | BIO-Fungi 11700 | Spain | KR677486 | KX828829 |
| C. ferruginascens (6) | 347/BB 07.221 | Slovakia | - | KX907235 |
| C. cibarius var. flavipes IT | 9312B2 (Mornand herbarium) | France | KX828772 | - |
| C. cinnabarinus NT | 312/BB 07.001 | USA | - | GQ914985 |
| C. decolorans ET | 469/BB 08.278(PC) | Madagascar | KX907203 | GQ914968 |
| C. ferruginascens HT | E00204187 | United Kingdom | KX828773 | - |
| C. ferruginascens (1) | K(M)180009 | United Kingdom | KX828774 | - |
| C. ferruginascens (2) | AH44782 | France | KR677488 | KX828826 |
| C. ferruginascens (3) | AH44794 | Spain | KR67785 | KX828827 |
| C. ferruginascens (4) | AH44795 | Spain | KX828775 | KX828828 |
| C. ferruginascens (5) | BIO-Fungi 11700 | Spain | KR677486 | KX828829 |
| C. ferruginascens (6) | 347/BB 07.221 | Slovakia | - | KX907235 |
| C. ferruginascens (7) | 348/BB 07.283 | Slovakia | - | GQ914952 |
| C. ferruginascens (8) | BIO-Fungi 12651 | Spain | KR677487 | - |
| C. ferruginascens (9) | AH44226 | Spain | KX828776 | - |
| C. flavus | C067_CH5 | USA | JX030467 | JX030416 |
| *C. flavus | EJHM20220923 | Mexico | OQ875963 | OQ876857 |
| C. flavus (1) | 1077/JJ | USA | - | KX857028 |
| C. flavus (2) | C067 | USA | - | JX030416 |
| C. flavus (3) | RAS464 | USA | - | OM751857 |
| C. flavus (4) | RAS473 | USA | - | OM751858 |
| C. formosus | SAR220712 (DAOM) | Canada | KR677515 | KX828830 |
| C. friesii (1) | 1004/EC 09.43 | Italy | - | KX857016 |
| C. friesii (2) | AH44798 | Spain | KR677484 | KX828831 |
| C. friesii (3) | 1001/EC 09.16 | Italy | KX907208 | - |
| C. friesii (4) | ARAN-Fungi A3020106B | Spain | KR677483 | - |
| C. gallaecicus PT | LOU-Fungi 18012 | Spain | KR677482 | - |
| C. henrici HT | PC0142165 | France | KX828777 | - |
| C. ilicis HT | BIO-Fungi 11689 | Spain | KX828778 | KX828832 |
| C. lateritius ET | 320/BB 07.025 | USA | - | GQ914959 |
| C. lateritius (1) | 332/BB 07.062 | USA | KX896767 | - |
| C. lewisii HT | 314/BB 07.003 | USA | JN944021 | GQ914962 |
| C. lewisii | 301/BB 02.197 | USA | - | GQ914961 |
| C. lilacinopruinatus HT | PC0734066 | France | KX828779 | - |
| C. lilacinopruinatus PT | PC0734067 | France | KX828767 | - |
| C. lourizanianus HT | LOU-Fungi 19494 | Spain | KX828780 | - |
| C. minor (1) | 313/BB 07.002 | USA | - | JX192978 |
| C. minor (2) | 329/BB 07.057 | USA | - | JX192979 |
| C. nigrescens | 66/BB 06.176 | Madagascar | - | JX192967 |
| C. pallens HT | PRM655551 | Spain | KX853516 | - |
| C. pallens (1) | AH44799 | Spain | KR677499 | KX828833 |
| C. pallens (2) | AH39124 | Morrocco | KX828781 | KX828834 |
| C. pallens (3) | 996/BB 09.409 | Italy | KX929162 | KX857014 |
| C. pallens (4) | 1110/BB 12.077 | Italy | KX907210 | KX857035 |
| C. pallens (5) | 997/BB 09.418 | Italy | KX907206 | KX907238 |
| C. pallens (6) | 998/BB 09.430 | Italy | KX907207 | KX907239 |
| C. pallens (7) | 999/BB 09.441 | Italy | - | KX907240 |
| C. pallens (8) | AH44784 | Spain | KR677498 | KX828835 |
| C. pallens (9) | 1115/BB 12.082 | Italy | KX907211 | KX857036 |
| C. pallens (10) | BIO-Fungi 10988 | Sweden | KR677495 | - |
| C. parviluteus IT | PC0084799 | Spain | KX828782 | - |
| C. phasmatis | C057 | USA | JX030464 | JX030417 |
| C. platyphyllus ET | 262/BB 98.126 | Tanzania | - | JX192975 |
| C. quercophilus HT | 455/BB 07.097 | USA | - | JX192981 |
| C. romagnesianus HT | PC0085043 | France | KX828783 | - |
| C. romagnesianus (1) | AH44218 | Spain | KX828784 | KX828836 |
| C. romagnesianus (2) | PC0142170 | France | KX828785 | - |
| C. romagnesianus (5) | BIO-Fungi 9933 | Spain | KR677480 | - |
| C. romagnesianus (6) | AH44788 | Spain | KR677481 | - |
| C. romagnesianus var. parvisporus HT | LOU-Fungi 19504 | Spain | KX828786 | - |
| C. roseofagetorum HT | AH44789 | Georgia | KX828789 | KX828839 |
| C. roseocanus HT | DAOM220723 | Canada | KX828787 | KX828837 |
| C. roseocanus (1) | DAOM220724 | Canada | KX828788 | KX828838 |
| C. roseocanus (2) | CC29 | USA | MK680154 | JX030415 |
| C. roseocanus (3) | JR7 | USA | OR039432 | KP639228 |
| *C. roseocanus | EJHM20220923 | Mexico | OQ875962 | OQ876856 |
| C. roseofagetorum | AH44786 | Georgia | KX828790 | KX828840 |
| C. rufipes ET | BIO-Fungi 12921 | Spain | KR677513 | |
| C. subalbidus | OSC81782 | USA | KX828791 | KX828841 |
| C. tabernensis (1) | 333/BB 07064 | USA | JN944012 | GQ914975 |
| C. tabernensis (2) | 323/BB 07040 | USA | JN944013 | GQ914977 |
| C. tenuithrix HT | 343/BB 07.125 | USA | JN944017 | - |
| C. tenuithrix (1) | 322/BB 07.035 | USA | - | GQ914946 |
| C. texensis HT | 317/BB 07.018 | USA | - | GQ914988 |
| C. tomentosus HT | 500/BB 98.060 | Tanzania | - | JX192995 |
| C. umbrinus HT | PC0142168 | France | KX828792 | - |
| Craterellus tubaeformis | 350/BB 07.293 | Slovakia | - | GQ914989 |
Sequences were aligned using Muscle (Edgar, 2004) with inconsistencies corrected manually. The phylogeny of each dataset was constructed under maximum likelihood methods. The congruence of the data sets was assessed by comparing compatible nodes in the phylogenies of each gene (Mason-Gamer & Kellogg, 1996).
Sequences were subjected to a maximum likelihood bootstrap analysis using the IQ-TREE on XSEDE tool (Trifinopoulos et al., 2016). Phylogenies from the maximum likelihood analyses were generated using IQ-Tree and visualized with FigTree v1.3.1. (Rambaut, 2009).
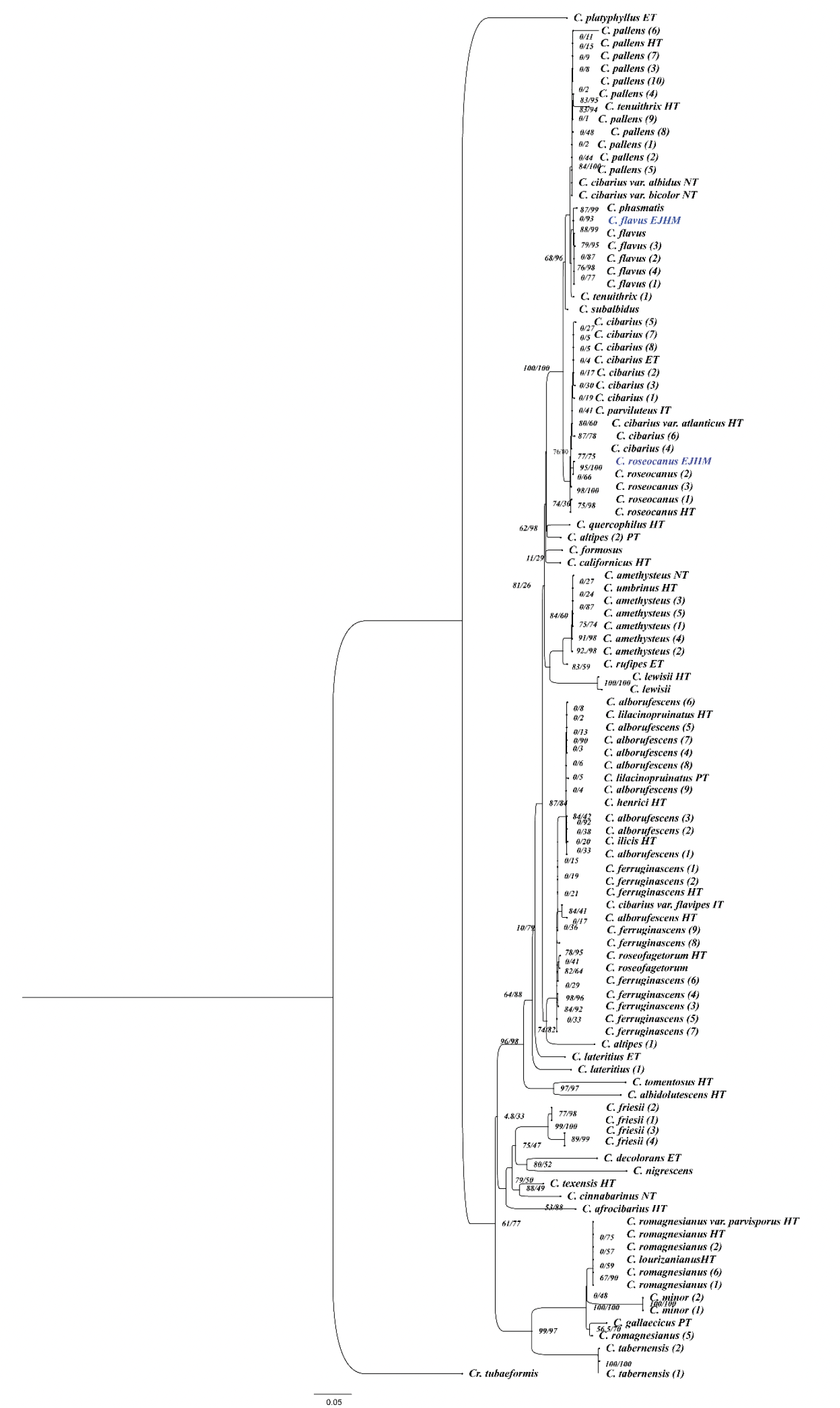
In the molecular phylogenies inferred (from ITS and tef1-α sequence datasets), support values considered were ≥70 bootstrap (BS) and ≥0.90 posterior probability (BPP), and these values were indicated as BS/BPP on the branches of the tree (Figure 2).
Results
Based on the distinctive morphological features and color variation observed in the Cantharellus specimens analyzed, together with their position in the phylogeny generated (Figure 2), it is confirmed that these represent two new records of Cantharellus in temperate forests of Pico de Orizaba National Park, Veracruz, Mexico.
New record of Cantharellus roseocanus in Mexico
Cantharellus roseocanus (Redhead, Norvell & Danell) Redhead, Norvell & Moncalvo; Index Fungorum 5: 1 (2012)
The cap measures 2.5 to 10 cm in diameter, flat-convex when young, often with a margin curled to flat or slightly depressed with an undulating and irregular edge; sticky when wet; pale yellow to egg yolk yellow or orange, often fading to nearly whitish when exposed to sunlight (Figure 3). When young, the cap color ranges from pale to dark pink, especially in areas near the margin. This color does not change significantly with damage, showing only a faint and gradual brownish hue. The flesh is whitish, with a solid and unchanging consistency after cutting.

Figure 3 Cantharellus roseocanus basidiomata collected in the Pico de Orizaba National Park, Veracruz.
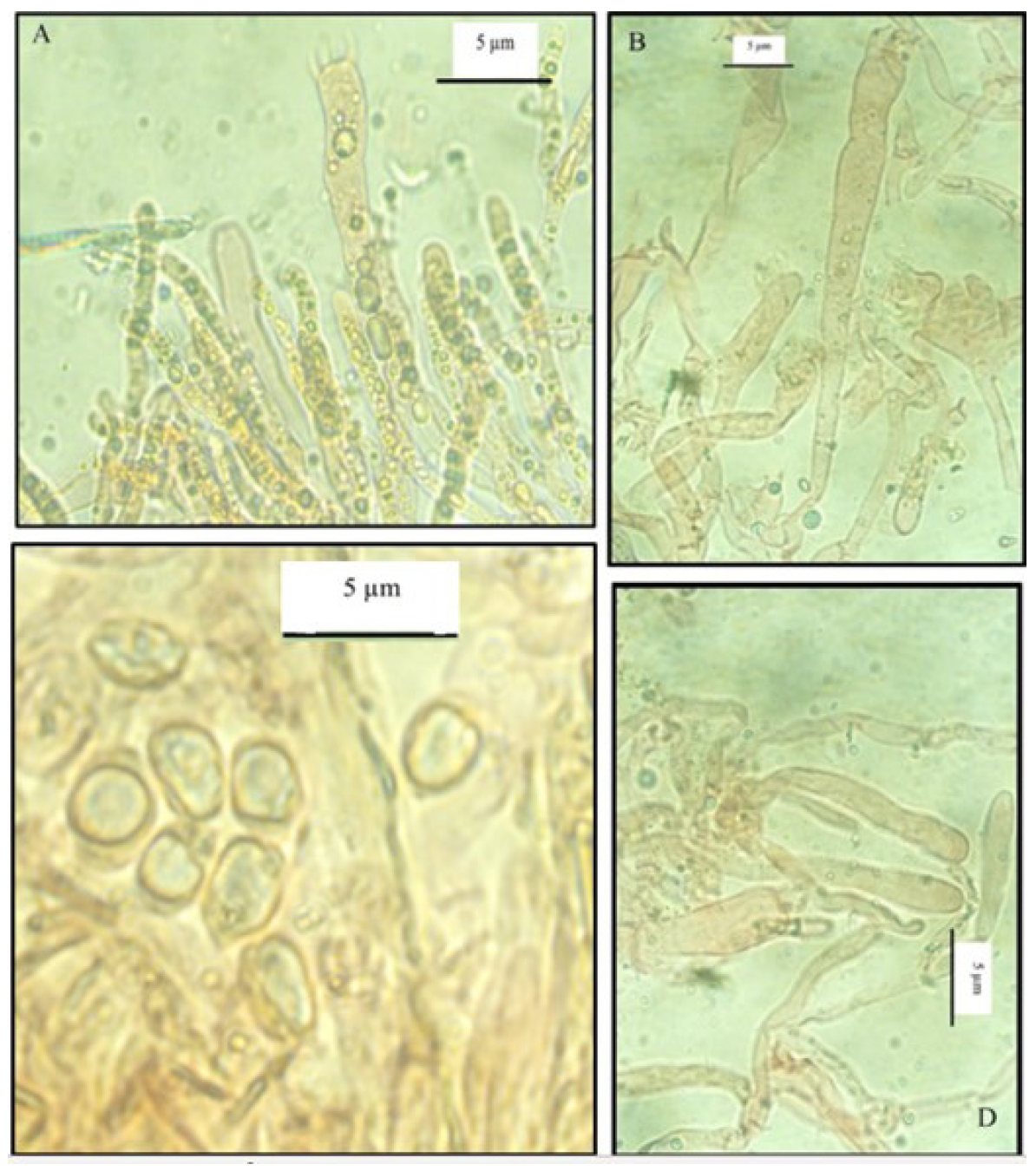
The cuticle has elements 5 to 10 µm in diameter, hyaline, cylindrical, septate and thin walled; terminal cells are cylindrical with rounded or occasionally subclavate apices (Figure 4).
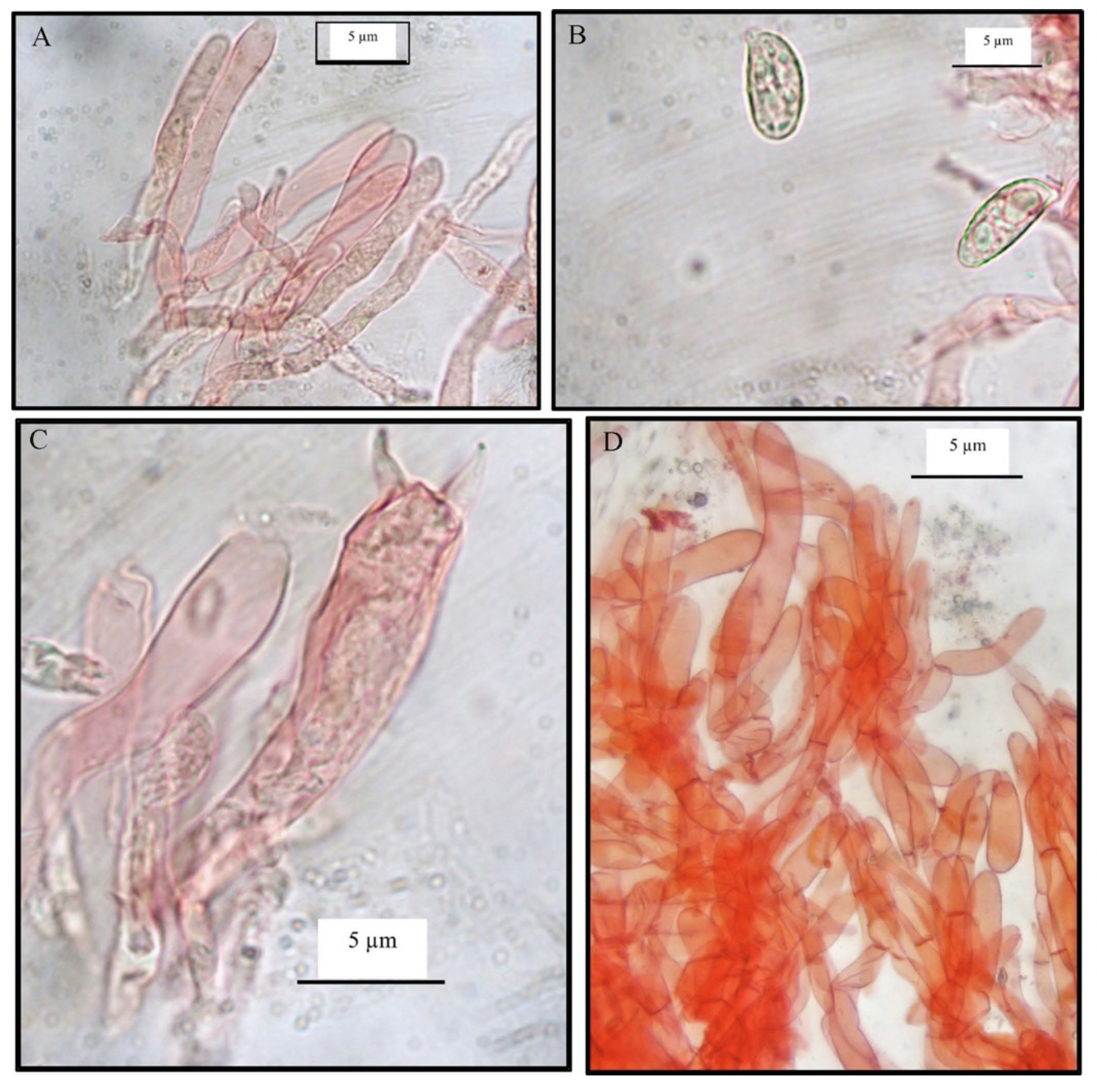
Figure 4 Cantharellus roseocanus. A) Terminal elements of the cuticle. B) Basidiospores. C) Basidium. D) Terminal elements of the cuticle.
The hymenophore shows developed veins that often show a reticulate pattern, extending through the stipe. These veins are bright orange and, in general, contrast markedly with the surface of the cap. No evident changes when damaged, but bruising can cause fungi to gradually take a brownish tone.
Basidiospores measure 7-10 x 4-5 µm, are ellipsoid, smooth, inamyloid, and weakly ochraceous in KOH; contents are granular. Basidia measure 60 to 95 µm in length and 7 to 10 µm in width, with three to four sterigmata (Figure 4). Cystidia are absent.
The stem measures 2-5 cm in length and 1-2 cm in thickness (Figure 3); it is variable in shape, though commonly fleshy, smooth, dry, with coloration similar to that of the cap or hymenium; it shows no significant changes from damage or bruising, gradually taking a brownish tone.
The habitat of C. roseocanus is gregarious, sparse in the study area, in temperate forest soil of Pinus hartwegii. The species fruits from August to September. Fungi have a mildly distinctive or slightly spicy taste, with a fragrant and sweet aroma, similar to that of peaches.
The specimens were collected along the Nueva Vaquería-El Llano road at an elevation of 3 677 m in the municipality of Calcahualco, Veracruz, Mexico. The dates and unique identification codes assigned to each specimen are as follows: September 10, 2019 (Hernández-Marañón 100919), September 12, 2020 (Hernández-Marañón 120920), September 25, 2021 (Hernández-Marañón 250921), and September 29, 2022 (Hernández-Marañón 290922). The numerical codes represent unique collection identifiers for each sample and do not correspond to a simple numerical sequence.
New record of Cantharellus flavus in Mexico
Cantharellus flavus M. J.Foltz & T. J. Volk; Mycology 105 (2): 456 (2013)
The cap reaches a diameter of 6 to 9 cm, and has an egg yolk yellow color that intensifies, especially with exposure to light or overtime (Figure 5). In its immature stage, the cap is flat-convex, evolving as it matures from flat to slightly undulated and from depressed to broad infundibuliform. The margin of the cup is typically incurved, with shapes ranging from regular to irregular, generally not turning outward at maturity, though it may occasionally exhibit lobes or sinuosities on one side. The cap texture is vibrant yellow with a spongy, watery consistency.
The hymenophore gills are decurrent, egg yolk yellow in color, often bifurcated and anastomosed. They do not display visible discoloration upon bruising; in dried specimens, bruising turns a yellowish-brown ochre color.
The basidiospores, with a yellow spore deposit, measure (7.5)8-10(11) x 3(4)4.5-6 mm, are subglobose to obovate when young, and become oblong at maturity (n = 30). The basidia measure 75-80 µm x 7-10 µm, with three to four often undulated sterigmata. Cystidia are absent.
The cuticle contains elements 5 to 8 mm in size, sometimes with thickened walls; clamp connections are present in all tissues. The fungus has a peach-like aroma and a slightly spicy taste. KOH intensifies the color of all tissues.
The stem is yellow and solid when young, sometimes showing white and mottled patches at maturity, with dimensions of 3-8 cm in length and 0.5-2 cm thick (Figure 5).
Gregarious fructification of these fungi is uncommon and is observed on the ground of temperate forests where species such as P. montezumae and P. pseudostrobus grow. Gregarious fruiting typically occurs in August and September.
Specimens were collected on the Nueva Vaquería-El Llano road (3 677 m) municipality of Calcahualco, Veracruz, Mexico, on the following dates: 10 September 2019 (Hernández-Marañón 100919), 12 September 2020 (Hernández-Marañón 120920), 25 September 2021 (Hernández-Marañón 250921) and 29 September 2022 (Hernández-Marañón 290922). Numerical codes indicate unique collection keys for each sample and do not correspond to a simple numerical sequence.
Discussion
The genus Cantharellus has been studied using molecular markers such as ITS, tef1, nLSU, rbp2, and mlsSSU (Wang et al., 2023) to advance taxonomy and facilitate the description of new species. In Mexico, there are still records and species that require more in-depth analysis, as this genus exhibits morphological characteristics that, although similar, do not correspond to homologous species.
The diversity of Cantharellus species is considerably lower in Europe compared to North America, where 22 species are recognized, excluding those that need to be transferred to Craterellus or newly described species in the process of classification (Buyck et al., 2014). These observations are supported by the study of Olariaga et al. (2017), which details the species present in Europe. A similar process would be necessary in Mexico or the North American region to determine the richness of Cantharellus and to address the knowledge gap in our country.
The species reported in this study as new records were first identified in the United States and Canada and validated through widely used genetic markers such as ITS and tef1, which provide adequate support for validating new records in Mexico. However, in Mexico, many species are still reported as C. cibarius due to morphological homogenization within the genus, despite this species being primarily restricted to the European continent (Foltz et al., 2013; Thorn et al., 2017).
The samples collected and identified as Cantharellus roseocanus and Cantharellus flavus come from areas with low levels of forest management and affected by deforestation. These ecosystems host significant ectomycorrhizal fungal diversity associated with various hosts, highlighting their vulnerability to deforestation and the resulting loss of forest and mycological diversity. These records contribute to the taxonomic knowledge of the genus Cantharellus, which still lacks complete elucidation. This finding marks only the beginning of a continuous process, given the genus's widespread distribution in our country, suggesting an extensive mycological richness yet to be discovered. In this regard, further taxonomic analysis of more specimens will be crucial to positioning Mexico among the leaders in edible species (China, the global leader, with 46 species; Zhang et al., 2022) and for revaluing the mycological potential of the Veracruz Mountain Region, based on the number of Cantharellus species. Additionally, it is important to note that the species recorded in this study have high local consumption value and are sold in local markets during the rainy season from August to November, with prices ranging from 400 to 600 MXN∙kg-1 in the Coscomatepec market in Veracruz, depending on the quality of basidiomata.
Conclusions
Based on morphological features together with their position in the generated phylogeny, the analyzed specimens were identified as Cantharellus roseocanus and Cantharellus flavus and represent two new records of the genus in the temperate forests of Pico de Orizaba National Park, Veracruz, Mexico. These records contribute to the taxonomic knowledge of Cantharellus, which still lacks complete elucidation. The taxonomic analysis of more specimens will be crucial for positioning Mexico as a leader in edible species and for the revaluation of the mycological potential of the Veracruz Mountains Region, where they are considered a non-timber forest resource of high sociocultural and economic value for the communities











 texto en
texto en