Introduction
In México, white-tailed deer (Odocoileus virginianus) is one of the foremost managed species, especially in the north of the country, where it has been demonstrated that it can be a source of economically profitable exploitation, and with ample opportunities in tropical zones from the center and southeast México (Mandujano et al. 2019). The importance of this species in México has caused it to be one of the most studied species of ungulates, with extensive information about their reproduction, habitat, foraging patterns, and behavior (Gallina-Tessaro et al. 2019).
Free water (i. e., water available for drinking) is a fundamental habitat feature for wildlife (Leopold 1987), essential for life processes (Ricklefs 2008) that may influence population dynamics and species distribution (Rosenstock et al. 1999; Simpson et al. 2011), which has been considered a limiting factor for many species in arid (Rautenstrauch and Krausman 1989; Cain et al. 2006) and seasonal environments (Mandujano and Hernández 2019). Free-standing water availability limits large herbivore populations if forage consumption does not meet water requirements, and because it helps with thermoregulation (Mogart et al. 2005; Simpson et al. 2011). For white-tailed deer, water is an essential nutrient necessary for meeting all their physiological needs, including the water lost from a deer’s body through the excretion of wastes and evaporation from the respiratory tract and skin. Also, water is lost when a doe gives birth or is lactating in such a rate that a deer’s health may be jeopardized if water is not replaced in less than 48 hours (Hewit 2011). Water bodies also are important for social interactions where evidence shows that white-tailed deer intraspecific contact is more likely in habitats where deer resources such as food and water are concentrated (Palmer et al. 2004; Kjær et al. 2010).
Water scarcity is a major challenge for wildlife habitats in arid and seasonal environments in North America and México. One way to address this issue is to construct Artificial Water Ponds (AWP), which are an integral component for maintaining and enhancing these habitats (Rosenstock et al. 1999). AWP have several purposes. Firstly, they aim to improve the performance of wildlife populations by increasing productivity, reducing mortality, and facilitating management. Secondly, they affect animal movements and distribution, which can have ecological and conservation implications (Bello et al. 2001; Destefano et al. 2000; O’Brien et al. 2006; Simpson et al. 2011). Furthermore, AWP can mitigate the loss and degradation of naturally occurring sources of free water, which are increasingly scarce and vulnerable due to climate change and human activity (Rosenstock et al. 1999; Simpson et al. 2011; Larsen et al. 2012).
The importance of water developments for white-tailed deer populations in México has been mostly studied in the north of the country (see Gallina-Tessaro et al. 2019 for a review). However, in the south, there is little information about this subject. Most of the water developments in this region are employed in protected areas (Borjes-Zapata et al. 2020) and Wildlife Management and Conservation Units (UMA per its Spanish initials; Treviño-Ruiz 2010; Gastelum-Mendoza et al. 2014). One of these UMAs is Nicte Ha, located in western Campeche. This UMA covers 2300 ha and has the objective of preserving and practicing legal hunting of white-tailed deer. Nicte Ha is the home of an extensive wetland system that undergoes severe seasonal changes. It has a marked dry season with temperatures above 40 °C (INEGI 2013), which contrasts with an extended flooding season that can last for more than eight months (Rivera-Arriaga and Villalobos-Zapata 2005). These climatic conditions strongly affect the resource availability for the wildlife inhabiting the area (Hidalgo- Mihart et al. 2017). To reduce the effect of the dry conditions in the Nicte Ha UMA, AWP were constructed along the property to preserve free water for wildlife. This is the UMA’s most important habitat management activity to cope with the extremely harsh conditions during the dry season.
Many studies agree that the conservation of natural, free water sources and the construction of artificial water reservoirs are valuable strategies for wildlife conservation and management, not only in arid climate areas but also in areas with contrasting high seasonality (Martínez-Kú et al. 2008; Delgado-Martínez et al. 2018; Mandujano and Hernández 2019; Borges-Zapata et al. 2020). Previous studies in Nicte Ha reported that the white-tailed deer responds to these contrasting environmental conditions by increasing the daily distances traveled and home range during the harshest portion of the dry season compared to the rainy season (Contreras-Moreno et al. 2019a; Contreras-Moreno et al. 2021). Additionally, it has been found that fawning synchronizes with the dry months (February- June) of the year, and the area's seasonality is one of its possible causes (Contreras-Moreno et al. 2019b).
Previous work in the Nicte Ha area showed that white-tailed deer responded to the AWP, concentrating their home ranges along these free water sources (Contreras-Moreno et al. 2021). However, to understand how the white-tailed deer is affected by the presence of AWP in Nicte Ha, it is necessary to determine other aspects, such as the visitation rate and activity patterns in these artificial water sources and in areas where the structures are not present.
White-tailed deer use the available habitats to satisfy basic requirements for food, rumination, movement, social interactions, and rest. The resulting time budget and habitat use pattern may vary with age, sex, time of day, season, weather conditions (Beier and McCullough 1990), predators, and human activities (Kilgo et al. 1998). For these reasons, the aim of this research was to describe the activity patterns and visitation rate of white-tailed deer within AWP and dirt Roads using camera traps during the dry season in the seasonal wetland area Nicte Ha, Campeche, México, and how these aspects vary across three studied years (2019, 2020, and 2021). This study will contribute to understanding the behavior of white-tailed deer in the presence of artificial water sources in tropical areas with high seasonality.
White-tailed deer use the available habitats to satisfy basic requirements for food, rumination, movement, social interactions, and rest. However, these requirements may vary depending on several factors, such as age, sex, time of day, season, weather conditions (Beier and McCullough 1990), predators, and human activities (Kilgo et al. 1998). For these reasons, the aim of this research was to describe the activity patterns and visitation rate of white-tailed deer within AWP and pathways/trails using camera traps during the dry season in the seasonal wetland area Nicte Ha, Campeche, México and how these aspects vary across three studied years (2019, 2020, and 2021). Our study will contribute to understanding how white-tailed deer adapt to the presence of artificial water sources in tropical areas with high seasonality. This knowledge could help to improve the management and conservation of this species and its habitat.
Materials and methods
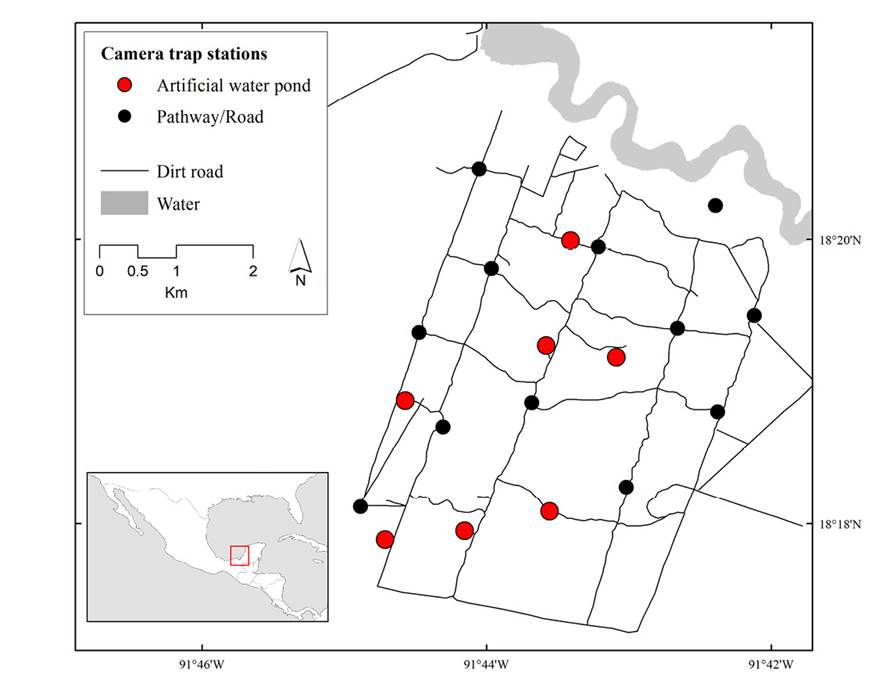
Our research was conducted at the Nicte-Ha UMA, located in southwestern Campeche, México (91° 43’ 56” W, 18° 19’ 56” N) adjacent to the Laguna de Términos Flora and Fauna Protected Area (Figure 1). The UMA has 2,300 ha of tropical lower coastal plain habitat situated between -1 and 3 m asl. The climate in the region is warm-humid, with a mean temperature of 27 °C and up to 2,000 mm of precipitation per year (INEGI 2013). The vegetation type of the area varies from hydrophilic vegetation, flooded savannas, mangroves, sub-evergreen flooded rain forests, tropical deciduous flooded forests, secondary growth forests, agricultural areas, and induced grasslands for cattle grazing (Ocaña and Lot 1996). In 2010, the owners of Nicte-Ha removed cattle from the ranch, and since then, the area has been exclusively used for sustainable white-tailed deer hunting. Human activity in the UMA is very reduced and consists of sporadic surveillance surveys, yearly road maintenance, and, during hunting season, intermittent hunting parties of no more than two or three persons per month.
Water availability is a key factor for wild ungulates in the Yucatán Peninsula, especially during the dry season (Reyna-Hurtado et al. 2010). In Nicte-Ha, a seasonal wetland area, precipitation is concentrated in a single season that usually spans from June to November. This is followed by a dry season that lasts around six months (December-May). The intensity and seasonality of the rains, combined with flat terrain, produce seasonal floods that can reach 0.5 m. The flood lasts for the entire rainy season, but once it is over, water for ungulates remains available but concentrated in numerous puddles within the area that usually dry out by mid-February. After this date, the only sources of water in Nicte Ha are the Rio del Este River and the eight AWPs constructed by the owners. It is important to note that although the Rio del Este is a permanent water body it is hardly accessible for the deer that live far from the river edges. Contreras-Moreno et al. (2019a, 2021) observed that in extreme cases when the AWP dry up, the individuals make long excursions of one or two days in which they travel several kilometers to obtain water from the river, returning immediately to their established home ranges inland.
AWPs were excavated with a bulldozer in 2010 and 2011 and initially consisted of a large hole approximately 15 m long, 8 m wide, and 2 m deep in the central part of the pond. The water ponds naturally fill with water during the rainy season and usually maintain water until the end of the dry season. However, in some extreme cases, some water ponds may dry up during the dry season. After the construction, the water ponds have not been excavated again, and because of sedimentation, the ponds have lost depth over the years. Currently, the AWP are surrounded by natural vegetation.
To determine the activity patterns and use of the AWP and Dirt Road (DR) treatments by white-tailed deer during the dry season of 2019, 2020, and 2021, we set camera trap stations at 7 AWP and at 12 sites along DR in Nicte Ha (Figure 1). The camera trap stations consisted of 1 or 2 cameras operated for at least 80 days. However, we could not maintain operating the cameras for this long due to equipment malfunction and theft on several occasions. We intended the separation between camera trap stations to be at least 1 km. However, this was not possible in some cases due to the distance in which some AWPs are distributed on the property. The camera traps were tied to trees at 50 cm above ground and programmed to function 24 hours a day. We obtained the number of white-tailed deer independent photographs at each camera trap station. We considered a record to be independent if the time interval between the photographs was more than 60 minutes (Tobler et al. 2008). We obtained the sampling effort per camera station by adding the number of camera days (i. e., a period of 24 h during which the camera station was operating), from the date when the camera was first active to the date of the last photograph, for every year and all the years together. The total sampling effort AWP and DR was obtained by adding the number of camera days each camera operated on each site each studied year. We identified the hour from each white-tailed deer independent record obtained from each one of the per year. We only use independent records for the analysis (Monroy-Vilchis et al. 2011).
We estimated the Relative Abundance Index (RAI) for each treatment per year as a measure of visitation rate (O’Brien 2011). RAI was obtained by dividing the total number of independent white-tailed deer records per camera trap per year by the camera trap effort in each station for each site per year. We used the Wilcoxon rank sums test to compare the observed RAI per camera trap station at AWS and DR for each studied year (Siegel and Castellan 1995). We compared the RAI for each camera trap station between years for each treatment using a Kruskal-Wallis test to determine if there were differences in the visit rates derived from the environmental conditions of each year. Dunns pairwise multiple comparisons test was used in case of significant differences were found in the Kruskal-Wallis analysis (Siegel and Castellan 1995). All our analyses were considered significant if P < 0.05.
We used a Kernel density estimator (Ridout and Linkie 2009) to generate the activity patterns of the white-tailed deer for each treatment and year. We also pooled all the records from the three studied years to generate a general activity pattern of the species in each treatment. To compare the white-tailed deer activity patterns between the 2 treatments and among each one of the studied years, we compared the treatment pairs per year and with the pooled data (AWP of year 2019 vs. DR of year 2019; AWP of year 2020 vs. DR of year 2020; AWP of year 2021 vs. DR of year 2021; AWP of the three summed years vs. DR of the three summed years) using the R-package overlap (Meredith and Ridout 2014). We calculated the overlap coefficient (Δ) with estimated 95% confidence intervals (CI) using a bootstrap of 1,000 samples. Following Meredith and Ridout (2014), if the smallest sample has less than 50 photographic records, we use the Δ1 estimator. Otherwise, we used the value of Δ4 as the overlap output. The estimated overlap coefficient can range from 0 (no overlap) to 1 (complete overlap). Activity was classified as (1) diurnal, 1 h after sunrise to 1 h before sunset; (2) nocturnal, 1 h after sunset to 1 h before sunrise; and (3) crepuscular, 1 h before and after sunrise and sunset (Foster et al. 2013).
Results
During the 3 years, we obtained 3,640 independent white-tailed deer records (2,584 in AWP and 1,056 in DR; Table 1) with a total camera trap effort of 3,978 camera days (1,306 camera days in AWP and 2672 in DR). The number of operating cameras in each treatment varied between years due to equipment malfunction or theft (Table 1). We obtained 1,580 records for the year 2019, 1,290 records for 2020, and 770 records for the year 2021 (Table 1).
Table 1 Number of white-tailed deer independent records, number of operating camera trap stations and camera trap effort in artificial water ponds and dirt roads located in the seasonal wetland obtained in 2019, 2020, and 2021 in Nicte Ha, Campeche, Mexico.
| Year | Treatment | Number of records | Number of operating camera trap stations | Camera trap effort |
|---|---|---|---|---|
| 2019 | ||||
| Artificial water pond | 1,079 | 6 | 435 | |
| Dirt Road | 501 | 12 | 884 | |
| 2020 | ||||
| Artificial water pond | 944 | 4 | 308 | |
| Dirt Road | 346 | 11 | 953 | |
| 2021 | ||||
| Artificial water pond | 561 | 7 | 563 | |
| Dirt Road | 209 | 11 | 835 | |
| All years combined | ||||
| Artificial water pond | 2,584 | 7 | 1,306 | |
| Dirt Road | 1,056 | 12 | 2,672 |
The RAI was bigger in the AWP than DR among the 3 studied years (Figure 2; 2019: W = 6, N = 18, P < 0.01; 2020: W = 0, N = 15; P < 0.01; 2021: W = 10, N = 17, P = 0.013). We found differences when we compared the observed RAI in the AWP among the studied years (Kruskal-Wallis H = 7.38, d. f. = 2, P =0.02; Figure 3). Dunn's test results showed differences between the RAI of AWP in 2021 with the RAI in 2019 and 2020 (2019-2021: P = 0.042; 2020-2021: P = 0.013). However, we did not find differences between 2019 and 2020 RAI (P = 0.56). In the case of the RAI, among years in the DR, we found that the RAI was similar among the studied years (Kruskal-Wallis H = 4.17, d.f. = 2, P = 0.12; Figure 3).
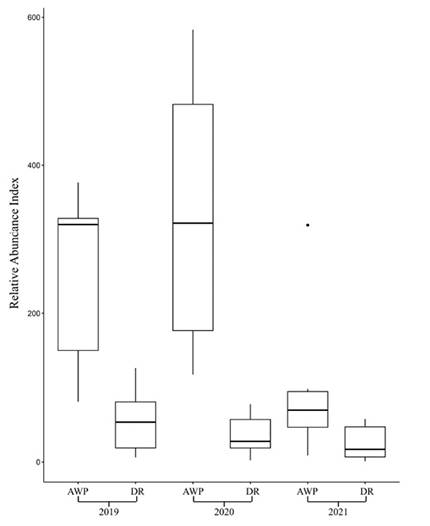
Figure 2 Comparison of the Relative abundance index (RAI) of white-tailed deer obtained in Artificial Water Ponds (AWP) and Dirt Roads (DR) during the dry season 2019, 2020, and 2019 in the UMA Nicte Ha in Campeche, México. The median of the RAI is shown as a line dividing the square, and mustaches represent confidence intervals of 95 %.
When we pooled the 3 studied years, we found that the mean activity overlaps between both treatments was Δ = 0.885 (Δ CI = 0.853-0.916; Figure 3). In the case of the individual years, we found that the mean activity overlap was Δ = 0.806 in 2019 (Δ CI = 0.762-0.854; Figure 3), Δ = 0.802 in 2020 (Δ CI = 0.745-0.851; Figure 3) and Δ =0.791 in 2021 (Δ CI = 0.726-0.851; Figure 3). Despite the significant overlap between both treatments, we observed in 3 that in the years 2019 and 2020, as well as in the pooled data, in the case of the DR, there is a clear peak of activity at sunset compared to the AWP data, in which the white-tailed deer activity remains very similar throughout the day. Likewise, in these same years, the activity at sunset is lower in DR sites compared to AWP. In 2021, contrary to what was observed in 2019 and 2021, we observed that there is no peak of activity in the DR during sunrise, but it occurs at sunset. In the case of the AWP, it is observed that the activity is maintained mainly during the day and night, but although small peaks are observed at sunrise and sunset, these are not as marked as those that occur in the DR.
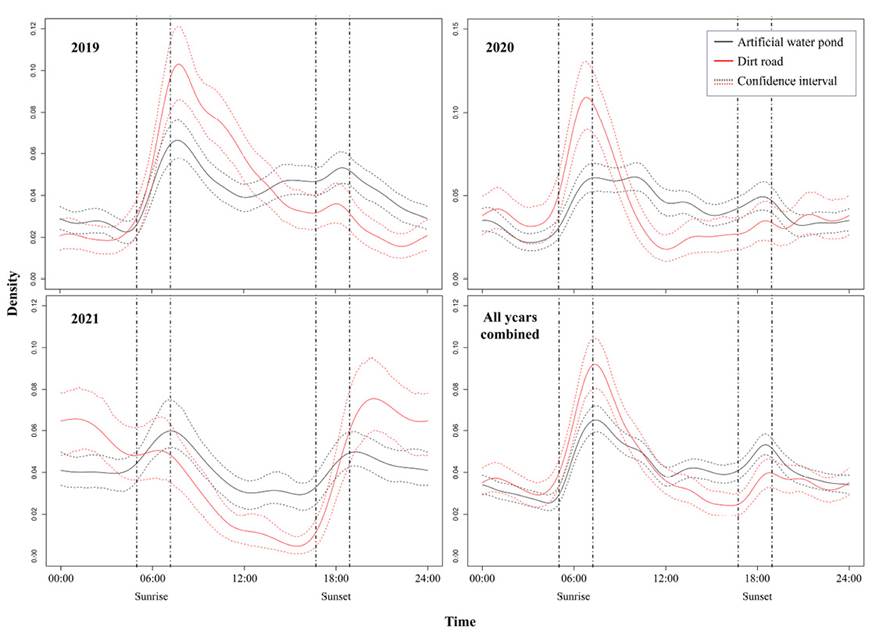
Figure 3 Activity patterns of white-tailed deer recorded in Artificial Water Ponds and Pathways/Roads during the 2019, 2020, and 2021 dry seasons in the UMA Nicte Ha, Campeche, México. Pairwise comparisons of activity overlap and 95 % Confidence Intervals of both treatments (Artificial Water Ponds and Dirt Roads) per year are shown with the identity of each activity pattern for each treatment indicated by color. Vertical dashed lines indicate sunrise (±1 h) and sunset (±1 h).
Discussion
The results show that the visitation rate of AWP in Nicte Ha has important effects on the presence of white-tailed deer during the dry season because we observed a higher RAI in the sites where there is AWP compared to the DR sites. In the case of activity patterns, white-tailed deer maintain constant activity throughout the day in AWP compared to DR, where activity peaks associated with sunrise and sunset occur.
Montalvo et al. (2019), in the tropical dry forest of Santa Rosa, Costa Rica, found that white-tailed deer during the dry season were found equally frequently at waterholes and roads; nevertheless, the species showed some dependency on waterholes. Contrary to this finding, in our study, over the three studied years, white-tailed deer RAI was higher in AWP compared to DR. In Santa Rosa, the lack of selection for waterholes was attributed to a strategy to avoid predation (Montalvo et al. 2019). In Nicte Ha, potential white-tailed deer predators such as jaguars are common (Hidalgo-Mihart et al. 2019). It is possible that higher temperatures that occur in Nicte Ha (higher than 40°C; INEGI 2013) compared with temperatures occurring in Santa Rosa (37°C: Montalvo et al. 2019) could promote dehydration and the need for more water intake (Fuller et al. 2014), and therefore more dependency on free water for the white-tailed deer in Nicte Ha.
The white-tailed deer in Nicte Ha were followed as part of a study of home ranges, showing an important association with the AWPs during the dry season. In those cases where the AWPs dried up, they made long exploratory trips to natural water bodies such as the Rio del Este, which usually take one or two days and immediately return to their home ranges (Contreras-Moreno et al. 2019a; 2021). This observation would indicate that it is likely that the visitation rate recorded in the AWP areas would be higher than in other sites, as was the case with our results. However, as reported by Contreras-Moreno et al. (2021), the capture of the radio-tracked deer was in the AWP, so many of the results reported by them are possible, and the link of the deer to the watering holes could be an effect of the territoriality of the captured organisms and overestimate the importance of AWP on the habitat use by the species. The present study, where we obtain higher RAI in the AWP compared with the DR, supports the idea that white-tailed deer use AWP much more than other areas of the UMA, showing the importance that this type of structure has for the management of the species during the dry season of the year in Nicte Ha.
We observed a significant overlap in the activity pattern between deer in the AWP and DR (between 70 and 85 %, depending on the year). Likewise, we observed that at least in 2019 and 2020, despite the high level of overlap, there was a peak of activity near dawn. It has been observed that, in general, deer in dry and tropical areas of México tend to be more active in the morning than in the afternoon (Mandujano and Hernández 2019; Hernández-SaintMartín et al. 2013), derived from the fact that it is at this time that temperatures are lower compared to the temperatures that could appear during the day and particularly during the afternoon. Thus, the deer found in the DRs could be repeating this pattern to reduce the negative impacts of high daytime temperatures, reducing the need for water intake, which must be streamlined since it does not have an available free water source. In the case of the AWP, it is noticeable that there were no clear activity peaks, and the deer could drink water throughout the day regardless of the hour of the day and the thermal conditions. The presence of a free water source probably reduces the stress associated with water loss from high temperatures, reducing the need to adjust their activity patterns to the coolest hours of the day, such as dawn and morning.
We observed that there are differences between years in the daily activity of white-tailed deer throughout periods in which the day was divided (nocturnal, diurnal, and sunset) in the DR, while in the case of the AWP, the percentages of activity were similar throughout the three years studied. This result indicates the importance that the AWP can have since the annual environmental variations of temperature and drought that affect the deer in the DR, modifying their activities, do not affect them like those inhabiting the AWP, owing to more stable conditions. This result is important in understanding how the white-tailed deer could be affected by changes already occurring in the region. The Nicte-Ha area is anticipated to suffer a general decrease in mean precipitation and an increase in drought (Imbach et al. 2012; Chiabai 2015). This will probably severely impact the white-tailed deer, as has already been detected in ungulates from the Amazon basin, where extreme drought and flooding are already occurring (Bodmer et al. 2014). In this sense, the results of stability during the three studied years in the activity patterns derived from the presence of AWP could reduce the effect of anticipated dry conditions that could affect Nicte Ha in the future.
AWPs are one of the most common strategies made in Campeche to improve wildlife habitat along with reforestation, vigilance, wildlife monitoring, signalization, and opening fire breaks (García-Marmolejo et al. 2008). The results of this work are the first evaluation of the ecological effects of AWP in this area of southern México and indicate the significant effects of these structures on aspects such as visitation rate and activity patterns. However, it is essential to evaluate the long-term effects of these structures on white-tailed deer and other species, especially under the controversy surrounding the importance and impact of these AWP in supporting wildlife populations (Simpson et al. 2011). Undesirable effects such as the wildlife distribution patterns constriction produced by the availability of free water causing local wildlife population declines due to forage scarcity associated with over-grazing, increased predation on specific species, or reduction in water quality due to excess of organic waste, and the increased risk of disease transmission should be carefully monitored (Marshal et al. 2006; Simpson et al. 2011; Griffis‐Kyle et al. 2014).











 nueva página del texto (beta)
nueva página del texto (beta)