Introduction
The demand for water resources has increased worldwide due to dynamic growth in human populations, urbanization, irrigation, and industry. Consequently, water shortages and declining water quality threaten the sustainable development of many countries and the goal of ensuring the food security of communities (WHO, 2017). Water scarcity is especially serious in arid and semiarid areas with low levels of rainfall, and decreasing availability of surface and groundwater (Dai, 2012). The problem can be devastating, with the erratic and unpredictable occurrence of droughts (Villazón-Bustillos et al., 2016).
Most water resources in northern Mexico are provided through groundwater, which is currently being depleted, and with high concentrations of toxic elements like heavy metals and metalloids (Valles-Aragon, Ojeda-Barrios, Guerrero-Prieto, Prieto-Amparan, & Sanchez-Chavez, 2017). Between 1997 and 2009, Olmos (2011) assessed 1 780 samples of water from wells in Chihuahua State in northern Mexico and found levels of heavy metals above recommended international and Mexican standards (NOM, 1994). Also, 47 of the 67 municipalities in Chihuahua State have problems with contaminated water. In another study, by Rubio, Balderrama, Burrola, Aguilar, and Saucedo (2015) documented that potable water in the Municipality of Ascension in Chihuahua, Mexico contained levels of As, Cr, Fe, Ni, Se, and Zn above the levels established by the Mexican regulation (NOM, 1996) and according to international standards (USEPA, 2013; WHO, 2012). These authors recommended developing low-cost technologies using regionally and available materials like zeolite to remove contaminants from water.
Many physical, chemical, and biological techniques have been developed to decontaminate water, among them organic and inorganic coagulants, electrodialysis, fluidization, sedimentation, microfiltration, membrane nano-filtration, and inverse osmosis (Demirel, Yenigun, & Onay, 2005; Kushwaha, Srivastava, & Deo-Mall, 2011; Villalobos-Rodríguez, Montero-Cabrera, Esparza, Herrera-Peraza, & Ballinas, 2012; Porwal, Mane, & Velhal, 2015; Chowdhury, Mazumder, Al-Attas, & Husain, 2016; Reddy & Yun, 2016; Prathna, Kumar, & Kennedy, 2018). Andrejkovičová et al. (2016) studied the use of natural zeolite to remove heavy metals from water. They found that zeolite removed about 75 % of heavy metals in the following order: Pb2+ > Cd2+ > Zn2+ > Cu2+ > Cr3+. In another study, Zanin et al. (2017) found that natural zeolite removed Fe (95.4 %), Cu (96 %), and Cr (85.1 %) from water. Several studies have reported that natural zeolites represent an excellent alternative to remove contaminants from water, especially heavy metals and metalloids (Andrejkovičová et al., 2016; Chowdhury et al., 2016). It is well documented that because of its porosity and high negative charge, zeolite has a comparative advantage of low cost to remove ions (Wibowo, Mamat-Rokhmata, Mumiatia, Khairumjala, & Abdullaha, 2017; Rouquerol, Rouquerol, Sing, Llewellyn, & Maurin, 2014). Nevertheless, few studies have analyzed the removal capacity of zeolite-based on grain size. Therefore, the objective of this study was to evaluate the removal of heavy metals and metalloids from natural water using filters packed with natural zeolites of different sizes. This information will be useful for designing improved filters to remove contaminating ions for the reuse of water by downstream communities, thus contributing to the sustainability of water resources and consequently improving the availability and quality of this natural resource.
Methods and materials
The study was carried out at “La Campana” Experimental Research Station of the National Research Institute for Forestry, Agriculture and Animal Production (INIFAP-Mexico), located in the city of Aldama, Chihuahua State, Mexico. The experimental units were 56-cm long filters packed with 1 kg of natural zeolite. Four treatments were evaluated based on different sizes of zeolite particles: 70 mm in treatment 1 (T1-large); 30 mm in treatment 2 (T2-medium); 500 μm in treatment 3 (T3-small); while treatment 4 (T4) was packed with a combination of the three sizes (large, medium and small). The particles in the T4 filter were packed according to size, the largest at the bottom, then the intermediate-sized particles, and finally the smallest particles at the top. There were two replicates of every treatment, so eight filters were built and packed. Before packing the filters, the zeolite was sieved according to the size required for each treatment.
The zeolite used in this research was obtained from a mining company near Aldama, Mexico. The mineral was crushed and sieved to obtain specific particle sizes. A zeolite sample was chemically characterized through X-Ray Diffraction (XRD) - mineralogical composition at the Mexican Geologic Service Laboratory (SGM-Mexico). In addition, the petrographic analysis and the X-Ray were used to estimate porosity and permeability in the SLE Laboratories (CICESE). The water filtered through the treatments was obtained from thermal water facilities in San Diego de Alcala, in the central part of Chihuahua State, Mexico (28° 35´13.83´´ N and 105° 32´48.50´´ W, 1119 m.a.s.l.). More information about San Diego de Alcala can be obtained in Villalba et al. (2015).
The water samples were transported to the laboratory and kept at a low temperature (4° C) until further analysis. The following heavy metals and metalloids were quantified in two samples of influent water, using Inductively Coupled Plasma-Mass Spectrometer (ICP-MS) Perkin-Elmer model ELAN 6100®: silver (Ag), aluminum (Al), arsenic (As), gold (Au), boron (B), barium (Ba), beryllium (Be), bismuth (Bi), calcium (Ca), cadmium (Cd), cobalt (Co), chromium (Cr), cooper (Cu), iron (Fe), mercury (Hg), potassium (K), lithium (Li), magnesium (Mg), molybdenum (Mo), sodium (Na), nickel (Ni), phosphorous (P), lead (Pb), antimony (Sb), selenium (Se), tin (Sn), strontium (Sr), tellurium (Tu), titanium (Ti), thallium (Tl), vanadium (V), tungsten (W) and zinc (Zn). In addition, hydrogen potential (pH), total dissolved solids (TDS), and electrical conductivity (EC) were determined with a portable multiparameter meter Hanna Instruments model HI 98130®. The influent water was passed through the filters three times, after which the effluent water was collected and analyzed to quantify the same parameters as had been quantified in influent water to determine changes in the parameters (% of removal).
Statistical analysis
In the first step, a database was created using Minitab software, after which the parameters were tested individually with an ANOVA using a factorial arrangement of treatments 4×3. Factor A was particle size, with four levels: large (T1), intermediate (T2), small (T3), and mixed (T4). Factor B was the number of times the water passed through the filters, with three levels: once, twice, and three times. With this analysis, it was possible to detect differences in effects of particle sizes (factor A), number of filtrations (factor B), and the interaction between the two factors. All statistical analyses considered a significance level of 95 %, hence α=0.05. If an analysis showed an interaction to be significant, an interaction graph was developed. Graphs were also developed to showcases where there was no interaction, but the effect of a factor was significant (Rubio & Jimenez, 2012).
Results
Table 1 shows the chemical composition of the zeolite used in this study. The zeolite was a heulandite type (CaAl2Si7O18•6H2O), with 61.43 % purity, with several minor constituents like albite, orthoclase, quartz, cristobalite, muscovite, calcite, magnetite, and hematite. According to the results of CICESE’s laboratory, the total porosity (percentage) was 0.29392 % and permeability was 8.4263 e-06 (0.05 mD).
Table 1 Chemical characterization of zeolite used in the water decontamination process (Mexican Geologic Service).
| Mineral Species | Formule | Percentage (%) |
|---|---|---|
| Heulandite | CaAl2Si7O18•6H20 | 61.43 |
| Albite | NaAlSi3O8 | 10.16 |
| Orthoclase | KAlSi3O8 | 9.78 |
| Quartz | Α-SiO2 | 6.77 |
| Christobalite | SiO2 | 6.55 |
| Muscovite | KAl2Si3O10(OH)2 | 3.90 |
| Calcite | CaCO3 | 1.40 |
| Magnetite | Fe3O4 | Trace |
| Hematite | Fe2O3 | Trace |
Table 2 shows the analysis of two replicates of the influent water used in this study. Of a total of 33 elements, the following 13 were not detected: Ag, Al, Au, Be, Bi, Cd, Co, Cr, Fe, Hg, Sn, Te, and Tl, and consequently are not discussed in this study. The elements As, B, Ca, Li, K, Mg, and Na were found in high concentrations and are therefore analyzed and discussed individually. The elements Ba, Cu, Mo, Ni, P, Pb, Sb, Se, Sr, Ti, V, W, and Zn were found in low concentrations and are addressed as a group.
Table 2 Heavy metals and metalloids were quantified in two samples of influent water from San Diego de Alcala, Chihuahua, Mexico.
| Element | Sample 1 | Sample 2 | Average | Element | Sample 1 | Sample 2 | Average |
|---|---|---|---|---|---|---|---|
| mg l-1 | mg l-1 | mg l-1 | mg l-1 | mg l-1 | mg l-1 | ||
| Ag | N.D. | N.D. | N.D. | Mg | 18.912 | 17.433 | 18.172 |
| Al | N.D. | N.D. | N.D. | Mo | 0.054 | 0.050 | 0.052 |
| As | 0.190 | 0.180 | 0.189 | Na | 473.167 | 431.194 | 452.180 |
| Au | N.D. | N.D. | N.D. | Ni | 0.003 | 0.002 | 0.002 |
| B | 1.344 | 1.334 | 1.339 | P | 0.560 | 0.550 | 0.555 |
| Ba | 0.025 | 0.025 | 0.025 | Pb | 0.004 | 0.004 | 0.004 |
| Be | N.D. | N.D. | N.D. | Sb | 0.001 | 0.001 | 0.001 |
| Bi | N.D. | N.D. | N.D. | Se | 0.002 | 0.001 | 0.001 |
| Ca | 22.659 | 25.144 | 23.901 | Sn | N.D. | N.D. | N.D. |
| Cd | N.D. | N.D. | N.D. | Sr | 1.180 | 1.223 | 1.201 |
| Co | N.D. | N.D. | N.D. | Te | N.D. | N.D. | N.D. |
| Cr | N.D. | N.D. | N.D. | Ti | 0.041 | 0.046 | 0.043 |
| Cu | 0.005 | 0.004 | 0.004 | Tl | N.D. | N.D. | N.D. |
| Fe | N.D. | N.D. | N.D. | V | 0.007 | 0.008 | 0.007 |
| Hg | N.D. | N.D. | N.D. | W | 0.004 | 0.003 | 0.003 |
| K | 20.918 | 19.271 | 20.094 | Zn | 0.009 | 0.005 | 0.007 |
| Li | 0.447 | 0.369 | 0.408 |
N.D. = Not Detected.
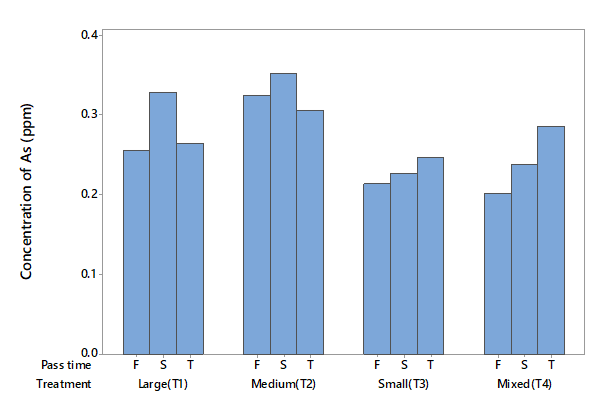
Arsenic (As)
The level of As in the influent water was 0.189 mg l-1 (Table 2), which is higher than the levels recommended for potable water by the Mexican government (0.025 mg l-1), the World Health Organization (0.01 mg l-1), and other international bodies (0.020 mg l-1). This level is also higher than the level of 0.1 mg l-1 recommended by the Mexican government for irrigation purposes (DOF, 1989). Because As contains metallic and non-metallic elements, it is considered a metalloid. The ANOVA detected statistical differences due to factor A (P < 0.05), but no differences for factor B (P > 0.05) or the interaction (P > 0.05). It can be seen from Figure 1 that treatments T3 and T4 performed better than the other two in eliminating this metalloid. T3 removed 48 % of As in the first filtration, while T4 removed around 42 %. T3 and T4 also outperformed the other treatments in the second filtration, but the capacity of zeolite to remove As decreased based on comparison with the percentage removed in the first trial (Figure 1).
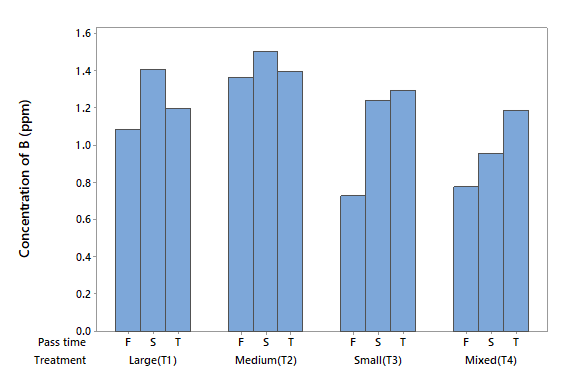
Boron (B)
The B concentration in the influent was 1.339 mg l-1 (Table 2). This metal is abundant in the Earth´s crust (10 mg kg-1), so its presence in water can be from natural sources. However, inputs from anthropogenic sources have increased in recent decades. The ANOVA did not detect statistical differences in factor A (P > 0.05), factor B (P > 0.05) or the interaction between them (P > 0.05). All four treatments removed B. T3 removed 73 % in the first filtration, while T4 removed 72 %. Figure 2 shows the levels of B with the four treatments and the three filtrations, and it can be noted that the capacity of zeolite to adsorb B decreased with successive filtrations. B is found in surface waters like river systems in the concentration of approximately 10 ppb (Lenntech, 2017a).
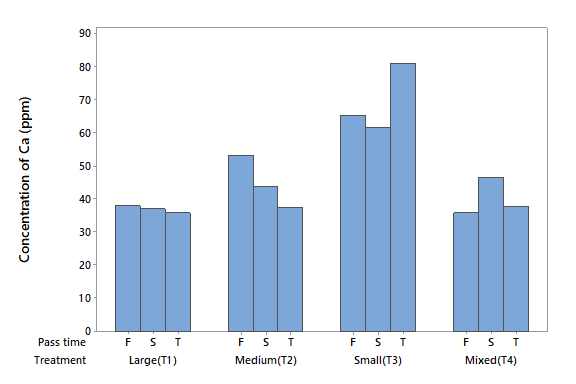
Calcium (Ca)
The level of Ca in the influent water was 23.901 mg l-1 (Table 2). The ANOVA detected statistical differences due to factor A (P < 0.05), but no differences were noted for factor B (P > 0.05) or the interaction between the two (P > 0.05). The T1 and T4 were the most effective in removing Ca from water. It is also noticeable that in T1, the zeolite did not lose the capacity to remove Ca ions, with 21 % of Ca in the first filtration, 23 % in the second, and 26 % in the third. The same effect was observed in T3 (Figure 3).
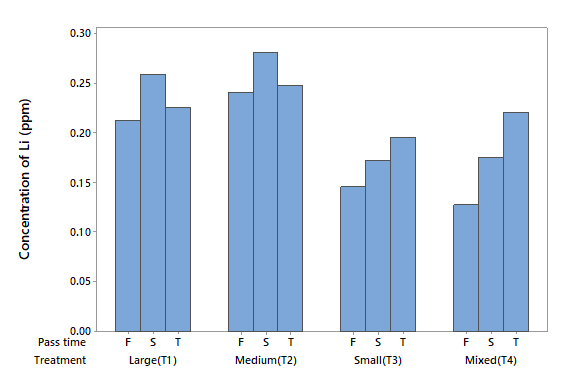
Lithium (Li)
The Li concentration in the influent water was 0.408 mg l-1 (Table 2). The ANOVA detected statistical differences due to factor A (P < 0.05) but no differences were found for factor B (P > 0.05) or the interaction (P > 0.05). All the treatments lowered the Li concentration in influent water, but T3 and T4 were the most effective. This can be noted in Figure 4, which shows that T3 and T4, respectively reduced Li by 82 and 84 %, respectivly, in the first filtration.
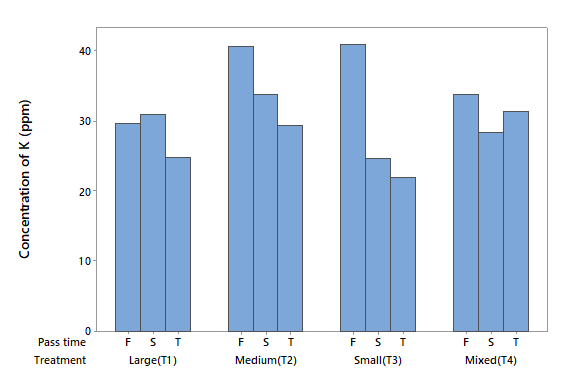
Potassium (K)
The K concentration in the influent water was 20.094 mg l-1 (Table 2). The ANOVA did not detect statistical differences due to factor A (P > 0.05), or the interaction (P > 0.05), but there were differences due to factor B (P < 0.05). Figure 5 shows that in the first filtration, T1 removed 27 % of K and T4 removed 16 %, but T2 and T3 removed 0 %. It can be also noted that the removal capacities of T2 and T3 were higher in the second and third filtrations than in the first.
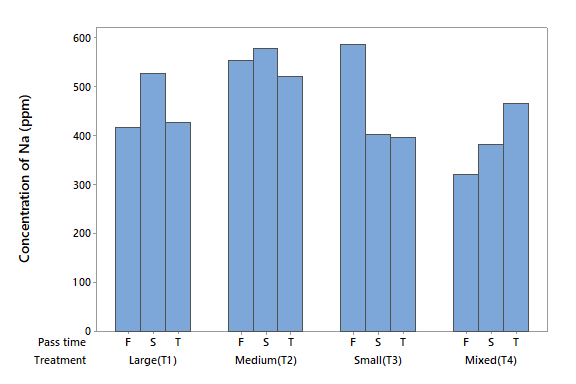
Sodium (Na)
The Na concentration in the influent was 452.18 mg l-1, which is higher than the 200 mg l-1 limit specified under Mexican law for safe potable water (NOM, 1993). The ANOVA did not detect statistical differences for either factor (P > 0.05) or the interaction (P > 0.05). However, the four treatments removed significant levels of Na. With the first filtration, T1 removed 54 %, T2 40 %, T3 36 % and T4 65 %. Figure 6 is showing that T4 had the highest Na removal rate.
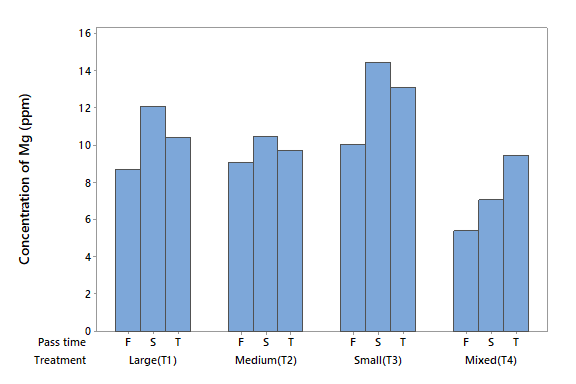
Magnesium (Mg)
The concentration of Mg in the influent water was 18.17 mg l-1 and was in the form of Mg2+. The ANOVA did not detect statistical differences for either factor (P > 0.05) or the interaction between them (P > 0.05). All four treatments removed substantial Mg ions, but the most effective was T4, which removed 86 % of Mg with the first filtration, 81 % with the second, and 74 % with the third (Figure 7). These results clearly show that zeolite loses the capacity to adsorb Mg in subsequent filtrations. The Mg adsorption rates of T1, T2, and T3 were similar to that of T4. In the first filtrations, T1 adsorbed 76 %, T2 approximately 75 %, and T2 approximately 72 %.
Discussion
The International Mineralogical Association (IMA) recognizes the following five heulandite sub-types: 1) Heulandite-Ba-NaBa4(Si27Al9)O72•24H2O; 2) Heulandite-Ca-NaCa4(Si27Al9)O72•24H2O; 3) Heulandite-K-KCa4(Si27Al9)O72•24H20, 4) Heulandite-Na-(Na,Ca)6(Si,Al)36O72•24H2O and 5) Heulandite-Sr- NaSr4(Si27Al9)O72•24H2O. Heulandite type zeolite is closely related to the clinoptilolite type and represents five minerals of the group termed tectosilicates.
According with the results, the percentage removal (%) of metals in the four treatments evaluated in this study, indicated that this type of zeolite can be used to decontaminate preferentially the elements Li, Mg, and B. This statement is based due to the following levels of removal in the four treatments:
T1 Large = Li > Mg > B > Na > K > Ca > As
T2 Medium = Mg > Li > B > Na > K > As > Ca
T3 Small = Li > Mg > B > Na > K > As > Ca
T4 Mixed = Mg > Li > B > Na > K > As > Ca
It can be noted the consistency of the elements that were removed in the water tested. In addition, it can be observed that this type of zeolite cannot be used efficiently to decontaminate the elements K, As, and Ca. In general, the zeolite did not lose the removal capacity after using it in three filtration processes.
Arsenic (As)
The capacity of T3 and T4 to remove As continued to decline with the third filtration. It is important to note that T3 and T4 were consistently more effective than T1 and T2 in removing As, which was particularly notable in the first filtration. Barnaby, Liefel, Jackson, Hampton, and Stanton (2017) evaluated five types of filters to remove As in water with concentrations of 10 µg l-1 and 100 µg l-1 and reported that the commercial filter ZeroWater reduced As to less than 0.05 µg l-1.
The As in water may originate from natural sources, according to the geology of a given area. Other sources of As in water are anthropogenic, such as pesticides and herbicides, wood preservatives, runoff from mining operations, urban and industrial wastes of different magnitude, and others. Arsenic is highly toxic to humans in even small concentrations and has been identified as the “poison of kings and the king of poisons”. Independent of the source of As in a water supply, various technologies are being implemented to remove it, such as coagulation (Song & Gallegos-García, 2014), adsorption in nanoparticles (Martinez-Vargas et al., 2018; Ivanets et al., 2018), sedimentation-filtration (Terracciano, Ge, & Meng, 2015), membrane filtration and inverse osmosis (Abejon, Garea, & Irabien, 2015), among others. Abdolahnejad, Jafari, Ebrahimi, Mohammadi, and Farrokhzadeh (2017) evaluated different types of filters to remove As among them a zeolite treatment with a particle size of 0.15-0.35 mm and a density of 1 538.4 kg m-3. The capacity of the zeolite to remove As varied according to the initial concentration. The zeolite filter removed 93.1 % of A when the initial concentration was 0.073 mg l-1, but the removal rate went down to 15 % when the initial concentration was 0.33 mg l-1. The researchers recommended using zeolite in rural and small communities because of its low cost and easy implementation. All removal techniques seek to reach the As level of 0.01 mg l-1 recommended by the WHO for potable water. Izhar, Shah, and Yuan (2014) found that zeolite (clinoptilolite) can remove 66 % of AS from residual water from petrochemical plants. The authors recommended a pH level of 8.0 and 240 min of exchange time for optimal results.
Boron (B)
The B concentration of 1.339 mg l-1 in the influent used in this study (Table 2) was assumed to originate from natural sources. This concentration is high for potable water, given that the World Health Organization and the Mexican government recommend that B levels in potable water be no higher than 0.05 mg l-1 (WHO, 1998) and less than 1.3 mg l-1 in water for irrigation (FAO, 1998). B is found in surface waters like river systems in the concentration of approximately 10 ppb (Lenntech, 2017a). Conservative water treatments like coagulation, sedimentation, and filtration do not remove B (WHO, 1998). Skoczko, Piekutin, Szatylowics and Niedzwiecka (2016) studied removing B from water; with one treatment using zeolite with particle sizes of 0.3 to 1.5 mm. The influent water in that study had a B concentration of 2.1 mg l-1. The zeolite treatment reduced B to a level of 0.05 mg l-1, which is similar to the results of our study. B removal rates from water depend on factors like particle size, porosity, and filtration intensity, among others.
Kluczka, Korolewicz, Zolotajkin, Simka, and Raczek (2013) evaluated an adsorbent product containing natural zeolite (clinoptilolite) and zirconium dioxide (ZrO2) to remove B from water. The authors found that removal rates were higher when the adsorbent was upper and with a pH of 8.0. They reported a maximum B removal rate of 75 %. In another study, Izhar et al. (2014) evaluated the adsorption capacity of the zeolite (clinoptilolite) in removing B from petrochemical wastewater. They obtained a B removal rate of 52 %, which was considered acceptable. Therefore, the authors concluded that zeolite can be used in treatment plants for this type of industry.
Calcium (Ca)
Le-van-Mao, Thanh-Vu, Xiao, and Ramsaran (1994) found that the cation exchange capacity (CEC) and zeolite pore size were the two main factors that determine Ca removal rates from aqueous solutions. The Ca (Ca2+) used in the present study was from a natural source, with a concentration of 23.901 mg l-1. This level is considered high, although concentrations as high as 100 ppm can be found in water in some geological areas. In combination with the Mg ion, the Ca ion is responsible for water hardness and can be removed with water softeners because it is a non-volatile salt. Cinar and Beler-Baykal (2005) assessed the capacity of natural zeolite to remove Ca to reduce water hardness and concluded that zeolite is an excellent alternative because of its capacity to adsorb 11 mg of Ca per gram of clinoptilolite.
The influent water tested in this study can be classified as slightly hard because the US Department of the Interior and the Water Quality Association established a range of 17.1 to 60 mg l-1 for this classification. All zeolite treatments in this study reduced the water classification from slightly hard to light, which is in a range of 0 to 17.1 mg l-1. Interestingly, the level of Ca in T3 increased in the third filtration, while this effect was noted in the second filtration with T4. This could be due to the interchange capacity among ions, which is often temporary. For example, Ca and Mg ions can be replaced by Na ions.
Lithium (Li)
It can also be noted that the capacity of T3 and T4 to adsorb Li decreased with the second and third filtrations. Li concentrations in water are generally in a range of 0.02 to 150 mg l-1, however, it is possible to find levels of 500 mg l-1 or even higher in some parts of the world (Concha et al., 2010). A study conducted in nine Lithuanian cities, Liaugaudaite, Mickuviene, Raskauskiene, Naginiene, and Sher (2017) found Li levels in water ranging from 1.24 ± 0.67 to 28.68 ± 9.68 mg l-1. Li is considered a trace element in humans and a maximum intake of 1 mg d-1 is recommended (Schrauzer, 2002). The importance of Li lies in the fact that high Li levels in water have been associated with lower suicide rates internationally (Liaugaudaite et al., 2017), in particular considering that the World Health Organization estimates that there are approximately 800 000 suicides worldwide every year (WHO, 2015).
Li is highly mobile, because it can be easily removed from rocks, and once in the water, it is monovalent (Li+). Therefore, it is common that concentrations vary with the seasons, particularly in regions where seasons are well-differentiated, as in the case of the area where our study was carried out. Hoyer, Kummer, and Merkel (2015) evaluated a Turkish zeolite (clinoptilolite) and compared its capacity to adsorb Li to the capacities of kaolinite and bentonite. They concluded that zeolite has more capacity to adsorb Li (about 65 %) than kaolinite (20 %) or bentonite (55 %). In the first filtration in our study, T1 removed 76 % of Li and T2 removed 70 %. All the treatments removed Li, but their capacity to do so decrease notably with the subsequent filtrations.
Potassium (K)
River water typically has concentrations of the monovalent ion K+ in the range of 2-3 mg l-1, while seawater contains about 400 mg l-1 (Lenntech, 2017b). Therefore, the natural water used in this study had high K levels compared to normal levels in rivers. It is well documented that K+, Mg2+, Ca2+, and Na+ ions have dynamic cation exchange capacities, meaning they can be removed or even replaced in aqueous solutions. The cationic exchange capacity (CEC) is a measure of the number of cations available for exchange per unit of weight. This effect may have affected the K+ adsorption capacity of the treatments in this study. Ames (1960) evaluated the cationic exchange capacity of K+, Mg2+, Ca2+, Na+ and NH4+ using clinoptilolite, and reported the following order K+ > NH4+ > Na+ > Ca2+ > Mg2+, which may affect absorption rates. Another study by McVeigh and Weatherley (1999) demonstrated that the K+ ion inhibits NH4+ adsorption.
Sodium (Na)
Vance, Zhao, Urynowics, Ganjegunte, and Gregory (2007) evaluated the capacity of two types of zeolite to remove Na from water: One obtained near Winston, New Mexico, and the other from Preston, Idaho, both in the United States. They documented that a metric ton (1 000 kg) of either type of zeolite decontaminates 16 000 to 60 000 l of water, with the advantage of lowering the sodium adsorption ratio (SAR) from 30 mmol l-1 to 10 mmol l-1, which is an acceptable level for use in irrigation. Nevertheless, the zeolite from Idaho performed better in removing Na than the zeolite from New Mexico. The influent used for this study contained 411 mg l-1, which is similar to the Na concentration in the influent used in the present study.
High Na+ concentrations in irrigation water can result in soils with high levels of interchangeable Na, which affects soil structure and reduces the hydraulic capacity of the soil. Salinity and sodicity are the main concerns about water quality in arid and semi-arid regions. A salinity laboratory in the United States developed the following equation to obtain the sodium adsorption ratio (SAR), which is useful for soil and water analysis:
This type of analysis is important because some cultivars cannot resist high levels of Na in water. For example, avocados and citrus fruit do not resist water with SAR levels higher than 3.0, while other cultivars can resist SAR levels higher than 46 (Bouwer & Idelovitch, 1987). A SAR above 4.0 is unsuitable for irrigation because that value indicates water high in Na (Gibb, Dynes, & Chang, 2017). It is also important to point out that Na levels of the influent water in this study were reduced to a level for being used for irrigation purposes according to the US EPA norms of < 69 mg l-1.
Magnesium (Mg)
Tomic, Rajic, Hrenovic and Povrenovic (2012) evaluated natural clinoptilolite to eliminate Mg from spring water using different initial Mg concentrations and different pH levels (from 5 to 9). They found that the Mg adsorption rate was higher with higher Mg concentrations. They also found that pH in a range of 5 to 7 does not influence Mg uptake, but the adsorption rate increased by 18 % with a pH of 8, and there was a similar effect with a pH of 9. The authors concluded that the clinoptilolite used in their study has a low Mg adsorption capacity. The water pH level in our study ranged from 8.0 to 8.5 in influent and effluent water.
Other metals
It was difficult to evaluate the effectiveness of zeolite in eliminating these elements (Ba, Cu, Mo, Ni, P, Pb, Sb, Se, Sr, Ti, V, W, and Zn) given their low concentrations. However, most elements were not detected after the filtrations, passing so it is assumed that the zeolite treatments eliminated some levels of these elements. For example, Ciosek and Luk (2018) found that zeolite was more effective in removing Pb2+ than Fe3+, Cu2+, Zn2+and Ni2+.
Potential hydrogen (pH) and electrical conductivity (EC)
The pH level plays an essential role in the capacity of zeolite to adsorb different elements and the role of its concentration in adsorbing some elements is controversial. In our study, the pH did not vary due to the treatments either the filtration process, showing a range of 8.0 to 8.5. For instance, the As adsorption rate is higher in a solution with a pH below 8.0 than in a solution with a higher pH level (Velazquez-Peña, Solache-Rios, Olguina, & Fall, 2019). In another study, Motsi, Rowson, and Simmons (2009) reported that a pH lower than 4.5 reduces the capacity of zeolite to adsorb Cu2+, Zn2+ and Mn2+, which is supported by the studies of Inglezakis, Loizidou and Grigoropoulo (2003), and Wingenfelder, Hansen, Furrer and Schulin (2005).
Concerning the parameter EC, in our study the concentration of this variable did not vary in the evaluated treatments, having a range of 1.10 to 1.30 mS cm-2. In a study that evaluated sand, activated carbon, and zeolite as filtering materials to remove diverse elements from water, Vera, Rojas, Chávez, and Arriaza (2016) found that zeolite can reduce EC levels in the water. The authors concluded that zeolite can reduce salts in water because EC was reduced to 20 % without affecting the other parameters or reaching a saturation level. These results are important given that salt commonly affects soils in arid and semiarid zones. The Food and Agriculture Organization (FAO) of the United Nations indicates that nearly 50 % of irrigated lands in the arid and semiarid area have problems of salinization (FAO, 1998). Salt has a negative effect on soils, with reduced agricultural output owing to diminished microbiological activity and consequent effects on crop yields.
Conclusion
According to the results of the present study, three conclusions can be specified: 1) the zeolite can be used to decontaminate alkaline earth metals in natural runoff water, particularly in the following order Li > Mg > B > Na > K > Ca > As; 2) the small particle and the mixed particle were better than large and medium particle sizes, therefore those sizes can be utilized to build commercial filters; 3) the three filtration processes did not affect the removal capacity of this zeolite used in this study. It is highly recommended to evaluate other types of natural zeolites to generate information that can be utilized for practical applications in different sectors.











 texto en
texto en