Artículos
Biomass and carbon storage in a continental wetland in Cuitzeo, Michoacán, Mexico
Paredes-García, Sally S.1
 http://orcid.org/0000-0003-2223-7490
http://orcid.org/0000-0003-2223-7490
Moreno-Casasola, Patricia2
 http://orcid.org/0000-0003-0468-0851
http://orcid.org/0000-0003-0468-0851
Barrera, Erick de la3
 http://orcid.org/0000-0002-0073-3410
http://orcid.org/0000-0002-0073-3410
García-Oliva, Felipe4
 http://orcid.org/0000-0003-4138-1850
http://orcid.org/0000-0003-4138-1850
Lindig-Cisneros, Roberto5
 http://orcid.org/0000-0003-2542-7038
http://orcid.org/0000-0003-2542-7038
 http://orcid.org/0000-0003-2223-7490
http://orcid.org/0000-0003-2223-7490Moreno-Casasola, Patricia2
 http://orcid.org/0000-0003-0468-0851
http://orcid.org/0000-0003-0468-0851Barrera, Erick de la3
 http://orcid.org/0000-0002-0073-3410
http://orcid.org/0000-0002-0073-3410García-Oliva, Felipe4
 http://orcid.org/0000-0003-4138-1850
http://orcid.org/0000-0003-4138-1850Lindig-Cisneros, Roberto5
 http://orcid.org/0000-0003-2542-7038
http://orcid.org/0000-0003-2542-7038-
Publication dates-
June 26, 2025
Mar-Apr , 2021
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
Abstract
Wetlands provide multiple ecosystem services, such as carbon sequestration both in living biomass and in the soil. In México, most studies have been done in coastal wetlands, mainly mangroves, and for this reason information on continental wetlands is scarce. Two research questions were answered: how much carbon is stored in the biomass and in the soil of a continental wetland; and which is the spatial structure and distribution of the dominant plant species. The study site is a floating wetland in the eastern part of lake Cuitzeo where field work was carried out during 2017 and 2018. Following a transect, six points were selected and aerial biomass was harvested in one-square-meter plots and a soil profile was excavated. Carbon content was analyzed from samples of the dominant species and apparent density and carbon stored in the soil was determined. Satellite imagery was analyzed to obtain the total area of the wetland during the period 2003-2017 and the area damaged by fires was calculated. Aerial biomass of the dominant species ranged from 5 770 kg/m² to 2 495 kg/m². The substrate contains 92 617 tons of carbon. The cover of invasive species Phragmites australis increased by 10 % with respect to the total wetland area from 2010 to 2017. Two major fires were detected, the largest in 2016 that covered 242 ha of the total of 535 ha.
Keywords::
Carbon sequestration, ecosystem services, hydrophytes, cattail, sedges, fire, conservation, floating wetland
Introduction
Wetlands are intermediate ecosystems between land and aquatic environments, distinguished by soil saturated by either fresh or salt water. These soils are dominated by anaerobic processes and biota made of vascular plants which are adapted to these environments, among which macrophytes stand out (Reddy & De-Laune, 2008; Landgrave & Moreno-Casasola, 2012; van der Valk, 2012; Smardon, 2014). Their characteristics are mainly determined by water amount, source and duration (hydrology) and by wetland landform (geomorphology) (van der Valk, 2012). Flooding slows the oxygen diffusion rate and modifies chemical and biological processes from an aerobic and oxidation state to an anaerobic and reduced state (Fiedler, Vepraskas, & Richardson, 2007). This favors carbon accumulation due to the changes brought about in microorganisms as the quantity of aerobic microbes and the propagation of facultative organisms and obligate anaerobes is reduced (Reddy & De-Laune, 2008)
-
Reddy & De-Laune, 2008Biogeochemistry of wetlands: Science and applications, 2008
-
Landgrave & Moreno-Casasola, 2012Evaluación cuantitativa de la pérdida de humedales en MéxicoInvestigación Ambiental, Ciencia y Política Pública, 2012
-
van der Valk, 2012The biology of freshwater wetlands, 2012
-
Smardon, 2014Wetland ecology principles and conservationWater, 2014
-
van der Valk, 2012The biology of freshwater wetlands, 2012
-
Fiedler, Vepraskas, & Richardson, 2007Soil redox potential: Importance, field measurements, and observationsAdvances in Agronomy, 2007
-
Reddy & De-Laune, 2008Biogeochemistry of wetlands: Science and applications, 2008
Wetlands are excellent for carbon sequestration due to their high accumulation and low decomposition biomass rates. According to Mitsch and Gosselink (2015) wetlands store an estimated 530 Pg of carbon worldwide, approximately 75 % of the carbon in the atmosphere. Among the various kinds of wetlands, peat bogs store the greatest proportion, about 89 Pg due to the organic matter stored as peat in the soil (Paige & Baird, 2016). In the face of the current environmental crisis, the amount of carbon stored in different types of wetlands should be determined, given their significant contribution to carbon sequestration.
-
Mitsch and Gosselink (2015)Wetlands, 2015
-
Paige & Baird, 2016Peatlands and global change: Response and ResilienceAnnual Review of Environment and Resources, 2016
Even though wetlands cover a relatively small area of the continental mass, the primary productivity of these systems is nearly as high as that of tropical forests (Mitsch et al., 2013; Reddy & De-Laune, 2008). Nevertheless, wetlands are subject to different types of disturbances brought about by increasing urbanization, changes in land use for different agricultural activities and the propagation of invasive species for commercial purposes (Brinson & Malvárez, 2002; van Asselen, Verburg, Vermaat, & Janse, 2013). This has caused the loss of more than 60 % of wetlands worldwide (Davidson, 2014) and, for Mexico in particular, Landgrave and Moreno-Casasola (2012) reported the loss of 62 % of domestic wetlands.
-
Mitsch et al., 2013Wetlands, carbon, and climate changeLandscape Ecology, 2013
-
Reddy & De-Laune, 2008Biogeochemistry of wetlands: Science and applications, 2008
-
Brinson & Malvárez, 2002Temperate freshwater wetlands: Types, status, and threatsEnvironmental Conservation, 2002
-
van Asselen, Verburg, Vermaat, & Janse, 2013Drivers of wetland conversion: A global meta-analysisPLoS ONE, 2013
-
Davidson, 2014How much wetland has the world lost? Long-term and recent trends in global wetland areaMarine and Freshwater Research, 2014
-
Landgrave and Moreno-Casasola (2012)Evaluación cuantitativa de la pérdida de humedales en MéxicoInvestigación Ambiental, Ciencia y Política Pública, 2012
Specifically, in lake Cuitzeo, our study site, in addition to grazing there are other more severe kinds of disturbances such as natural or induced fires. Although fire is a tool to eliminate undesirable vegetation (Nyman & Chabreck, 1995), it causes hypoxia (Rolletschek, Rolletschek, Hartzendorf, & Kohl, 2000), modifies community structure and diversity (Martin & Kirkman, 2009; Watts, Kobsiar, & Snyder, 2012; Kotze, 2013) and nutrient dynamics of the ecosystem (Schmalzer & Hinkle, 1992; Laubhan, 1995; Kotze, 2013).
-
Nyman & Chabreck, 1995Fire in coastal marshes: History and recent concernsFire in wetlands: A management perspective. Proceedings of the Tall Timbers Fire Ecology Conference, 1995
-
Rolletschek, Rolletschek, Hartzendorf, & Kohl, 2000Physiological consequences of mowing and burning of Phragmites australisstands for rhizome ventilation and amino acid metabolismWetlands Ecology and Management, 2000
-
Martin & Kirkman, 2009Management of ecological thresholds to re-establish disturbance-maintained herbaceous wetlands of the south-eastern USAJournal of Applied Ecology, 2009
-
Watts, Kobsiar, & Snyder, 2012Fire reinforces structure of pond cypress (Taxodium distichum var. imbricarium) domes in a wetland landscapeWetlands, 2012
-
Kotze, 2013The effects of fire on wetland structure and functioningAfrican Journal of Aquatic Science, 2013
-
Schmalzer & Hinkle, 1992Soil dynamics following fire in Juncus and Spartina marshesWetlands, 1992
-
Laubhan, 1995Effects of prescribed fi re on moist-soil vegetation and soil macronutrientsWetlands, 1995
-
Kotze, 2013The effects of fire on wetland structure and functioningAfrican Journal of Aquatic Science, 2013
Most wetlands in Mexico are coastal due to biogeographical reasons (Olmsted, 1993). Different studies have been conducted in recent years on the plant structure of wetlands and forest wetlands on the Mexican coast, and their value as regards carbon sequestration. These papers have focused mainly on mangroves. This is the first study undertaken to quantify plant biomass production in a floating continental wetland in Mexico, conducted to measure carbon deposits. To begin, the wetland plant species were identified and the area, in percentage, covered by them was estimated. Estimates were also made of the amount of carbon contributed by the aerial biomass of the plant community and of the carbon sequestered in this biomass. Moreover, the fires occurring in the wetlands from 2003 to 2017 were identified and the fluctuations in total wetland area during this period were also measured.
-
Olmsted, 1993Wetlands of MexicoWetlands of the world I: inventory, ecology and management. Handbook of vegetation science, 1993
Materials and methods
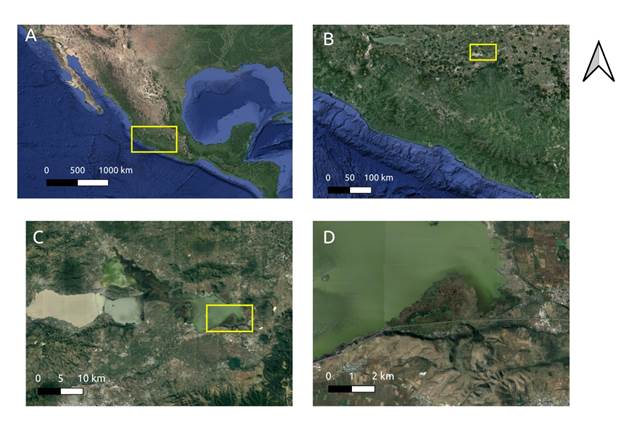
The study was conducted in the largest wetland area, that is also in the best condition of conservation, located to the east of lake Cuitzeo (Figure 1). The lake is located 34 km to the north of the city of Morelia at 19° 53’ 15’’ north and 100° 50’ 20’’ east, and 1820 masl, with sub-humid temperate climate and summer rainfalls. It is the second larges lake in Mexico, and covers approximately 420 kg2 with an average depth of 0.67 m. The lake is fed by surface flows from the Río Grande, Queréndaro and Zinapécuaro rivers, and also by rainfall and springs located within and around the lake. A few decades ago the lake was one continuous body of water but is now fragmented into three water masses as a consequence of the construction of federal highway 43, the Morelia-Salamanca toll-road and an embankment of 2 500 ha which was built to prevent flooding and to be used for agricultural purposes. This has caused the central-eastern section of the lake to shrink and resulted in increased concentrations of nutrients that flow into it with the waste waters from the Río Grande which flows in from the city of Morelia (Cram, Galicia, & Alcántara, 2010). The predominant hydrophyte emerging species belong to the Typha, Scirpus, Cyperus y Eleocharis species (Rojas & Novelo, 1995).
-
Cram, Galicia, & Alcántara, 2010Atlas de la cuenca del lago de Cuitzeo: análisis de su geografía y su entorno socioambiental, 2010
-
Rojas & Novelo, 1995Flora y vegetación acuáticas del lago de Cuitzeo, Michoacán, MéxicoActa Botánica Mexicana, 1995
Thumbnail
Figure 1
A) State of Michoacán, Mexico B) Location of the Cuitzeo lake in the western highland C) The lake is divided into three water masses by federal highway 43, the Morelia-Salamanca toll road and a 2 500 ha embankment used for agricultural purposes; D) the floating wetland in the best condition of conservation on the Cuitzeo lake.
A) State of Michoacán, Mexico B) Location of the Cuitzeo lake in the western highland C) The lake is divided into three water masses by federal highway 43, the Morelia-Salamanca toll road and a 2 500 ha embankment used for agricultural purposes; D) the floating wetland in the best condition of conservation on the Cuitzeo lake.
To determine field samples, points were selected on an East-West transect parallel to the Atlacomulco-Morelia road in the southern most section of the wetland (Figure 1). A baseline was drawn between 50 and 100 m from the road into the wetland to establish six sampling points approximately every 300 m along this transect to identify the plants. Aerial plant biomass samples were collected from one square meter at each point in the month of June 2017. The substrate profile down to the water below the floating mass was also determined in January 2018, and the plants present were collected in May 2018. Dry weight and total carbon percentage for each aerial biomass sample was measured as described below. Substrate samples were measured to obtain apparent density, organic-matter content and sequestered carbon.
Substrate carbon sequestration was defined as the amount of organic carbon stored in an area unit and known depth multiplied by apparent density. Core samples of 1000 cm3 were obtained from the floating substrate every 10 cm. Each core was left to dry at ambient temperature to eliminate excess water and was then dried in an oven at 80 °C until constant weight was achieved. Afterwards apparent density was calculated as follows:
e2
D
A
p
g
c
m
3
=
d
r
y
s
o
i
l
w
e
i
g
h
t
g
x
100
c
i
l
i
n
d
e
r
v
o
l
u
m
e
c
m
3
The percentage of organic matter (OM) was measured with a muffle furnace (Thermolyne, Thermoscientific Co., USA). Six 20 g samples were taken from the soil and placed in the furnace for 4 hours at 550 °C (Hoogsteen, Lantinga, Bakker, Groot, & Tittonell, 2015).
-
Hoogsteen, Lantinga, Bakker, Groot, & Tittonell, 2015Estimating soil organic carbon through loss on ignition: Effects of ignition conditions and structural water lossEuropean Journal of Soil Science, 2015
The carbon in the substrate was determined by combustion and colorimetric detection using a total carbon analyzer (UIC-COULOMETRICS mod. 50120, Chicago, USA) (Huffman, 1997). Carbon storage was calculated by multiplying CC by AD and substrate volume from the wetland.
-
Huffman, 1997Performance of a new automatic carbon dioxide coulometerMicrochemial Journal, 1997
Using the same colorimetric technique, total carbon (TC) for each plant species was measured from three samples. Dry aerial biomass carbon content was estimated for each dominant species on the basis of the dry biomass per square meter multiplied, for P. australis, by the area covered by this species. Since Typha spp.and S. americanus intermingle in different proportions in the wetlands, biomass was estimated by genus, assuming that the total area not covered by P australis was occupied by individuals of either one of these two genera, thus obtaining two estimates for total carbon.
Wetland area changes and fire-affected areas were recorded and quantified from satellite images by photointerpretation using the Google Earth Pro software, which includes images for the study site from 2003 until 2017, at different seasons of the year. Thus, the incidence and extent of fires may be underestimated in this work. Polygonal sections of the total wetland area and of the areas we were able to identify as affected by fire were measured in the images. Dominant plant species in areas not affected by fire were identified in the images, and this was corroborated by field trips using geo-referenced checkpoints.
Results
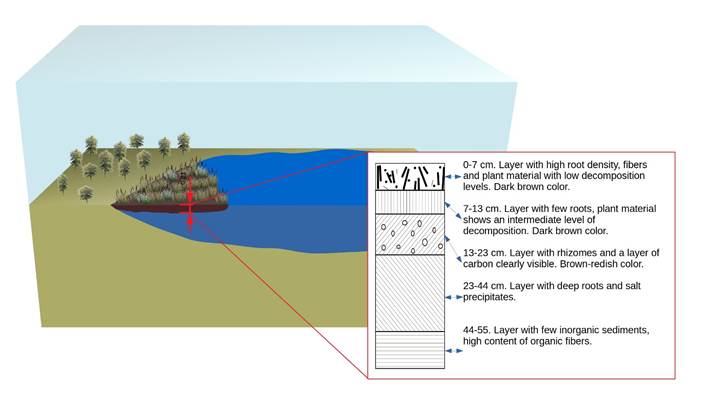
Our results show that the Cuitzeo lake wetlands are a surface made up of a substrate of organic material floating on a body of water approximately one meter in depth. The organic material of the substrate is in different phases of decay and the substrate is not in contact with any parent rock; therefore, it has no mineral components. From the cores taken it was determined that the substrate profile presented various characteristics at different depths, from the surface (0 cm) to the deepest section (55 cm; Figure 2). The greatest number of thin roots and other greenery is found in the first 7 cm. From 7-13 cm there is a reduction in thin roots, but this layer contains a high proportion of rhizomes and, on visual examination, the organic matter of this section was in a state of greater decay. From 23-55 cm deep, the degree of decomposition of roots and organic material was the greatest. It is not possible to determine the origin of this material due to its level of decay. There are no roots in the area adjacent to the water and this section has a clay-like texture. All live plant species thrive on this wetland substrate and contribute continuously to its formation.
Thumbnail
Figure 2
Morphological description of the profile of the floating wetland substrate showing a composition of organic material at different stages of decay.
Morphological description of the profile of the floating wetland substrate showing a composition of organic material at different stages of decay.
On the average, 96 % of the Cuitzeo lake wetland substrate is made up of organic material as shown by ignition analysis. Average apparent density was 0.13 ± 0.04 g/cm3. Average organic carbon content was 26 ± 0.75 kg C/m-2 at a depth of 50 cm. For the entire 552 ha wetland, a total of 92,617 tons of carbon in the substrate was calculated. (Table 1).
Table 1
Aerial biomass and carbon values of the dominant species as well as carbon content in the substrate.
Aerial biomass and carbon values of the dominant species as well as carbon content in the substrate.
| Average carbon (%) | Dry aerial biomass (kg/ha) | Aerial biomass carbon (kg/ha) | Dry aerial biomass in the wetland (ton) | Carbon in the wetland (ton) | |
|---|---|---|---|---|---|
| P. australis | 42 | 5770 | 2423 | 280 | 118* |
| Typha spp. | 39 | 3840 | 1498 | 1933 | 754* |
| S. americanus | 40 | 2495 | 998 | 1256 | 502* |
| Soil | 26 | 92617 | |||
| Maximum total (Typha spp.) | 93489 | ||||
| Miminum total (S. americanus) | 93237 |
The four dominant species were Typha domingensis, Typha latifolia, Schoenoplectus americanus and Phragmites australis. Biomass for the first two species was calculated jointly because their leaves cannot be distinguished from one another. P. australis was the species with the driest biomass per hectare, accounting for 5770 kg, followed by Typha spp. with 3840 kg and S. americanus with 2495 kg. Carbon concentration was similar among the species, 42, 39 and 40 %, respectively.
The substrate contains 26 % of carbon, that the substrate has a density of 33.6 kg/m³, therefore, there are 92 617 tons or carbon in the entire wetland substrate. With respect to the total of carbon, taking into account dry aerial biomass, we were able to obtain two estimates. Carbon content both in the substrate and in P. australis was the same in the two estimates. A maximum estimate of 93 489 tons can be made for Typha spp. (given that the individuals of this genus accumulate more biomass per area unit than those of S. americanus) and a minimum of 93 237 tons.
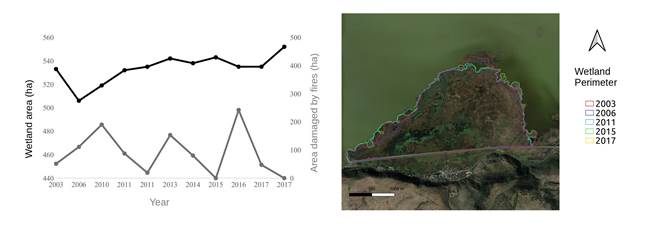
Fluctuations in the total area of the wetlands were detected from 2003 to 2017. According to historical records in 2006 the wetland area was nearly 10 % smaller than the one recorded in November 2017. Nevertheless, an analysis through time shows a slight upward trend in the size of the wetland area which went from 533 ha to 552 ha during this period (Figure 3). Moreover, eight fire events were identified for the nine years for which records for fires exist. The fire in 2010 caused damage to 35 % of the area. However, the largest fire recorded in 2016 covered an area of 45 %. Photointerpretation of satellite images also allowed us to identify that almost 10 % of the wetland area is covered by Phragmites australis, and between 2010 and 2017 the area covered by this species increased from only 3 ha to more than 70 ha.
Thumbnail
Figure 3
Total area and area damaged by fire in the eastern section of the Cuitzeo lake wetlands from 2003-2017
Total area and area damaged by fire in the eastern section of the Cuitzeo lake wetlands from 2003-2017
As regards the floristic description of the wetland, four dominant species of emerging hydrophytes were identified: Typha dominguensis Pers., Typha latifolia L., Schoenoplectus americanus Pers. and Phragmites australis (Cav.) Trin & Steud., either in association or by themselves. In addition, three subordinate species of lesser dominance were identified which are found in association with the ones mentioned above. The latter are an emerging hydrophyte, Hydrocotyle verticilata Thunb. var. Triradiata (Rich.), and two tolerant subacquatic species, Eleocharys montevidensis Kunth. andJuncus arcticus Willd.
Discussion
From the study conducted on the Cuitzeo lake wetland it was found that the live plants grow on a substrate of organic matter in different stages of decomposition, which floats freely and entirely on a body of water. The general structure of the wetland consists of a surface layer of intertwined roots next to a peat layer the depth of which is determined by the depth of the roots adjacent to the water. It is in this layer where, according to different studies, most of the processes relating to microorganisms such as nutrient and metal uptake, increased denitrification rates and plant phosphorous absorption occur (Jacobs & Harrison, 2014; Masters, 2012; Yeh, Yeh, & Chang, 2015). This is important because lake Cuitzeo is fed continuously by the Río Grande, which carries sewage from the city of Morelia, therefore continuously pouring phosphates and nitrates which are byproducts of agricultural, urban and industrial wastes (Conagua & CEAC, 2009).
-
Jacobs & Harrison, 2014Effects of floating vegetation on denitrification, nitrogen retention, and greenhouse gas production in wetland microcosmsBiogeochemistry, 2014
-
Masters, 2012The ability of vegetated floating Islands to improve water quality in natural and constructed wetlands: A reviewWater Practice and Technology, 2012
-
Yeh, Yeh, & Chang, 2015Artificial floating islands for environmental improvementRenewable and Sustainable Energy Reviews, 2015
-
Conagua & CEAC, 2009Plan de Gestión Integral de los Recursos Naturales de la Cuenca del Lago de Cuitzeo, 2009
The slight expansion of the wetlands into the lake shown by the examination of the images may be a consequence of the nutrient-rich effluents of the Río Grande entering the lake and from rainfall, since the contraction and expansion of wetlands is a product of fluctuations in the hydroperiod (Mitsch & Gosselink, 2015). The construction of the two roads crossing the lake, the river effluents and the dumping into the lake of different types of waste has resulted in creating different conditions in the three sections it is divided. The central-eastern section where the wetlands are found receives effluents that are controlled by the weirs of the Atapaneo water-treatment plant and therefore the level is maintained with no significant changes. However, on regular fieldtrips to the lake in the months of November and December 2017 and January 2018 we were unable to enter the wetland because the water level had risen as a result of the heavy and unusual rains which occurred during these months.
-
Mitsch & Gosselink, 2015Wetlands, 2015
We found seven species in the wetland, in contrast with 12 described by Rojas and Novelo (1995). This may be due to the increasing problem brought about by polluted waters that feed the lake and the expansion of the human settlements on its shores. Another relevant finding was the expansion of P. australis which in 2010 covered 3 ha and in 2017 covered 50 ha. Proliferation of this species has been well documented for both coastal and continental wetlands as a response to various disturbances, including greater concentrations of nutrients (Chambers, Meyerson, & Saltonstall, 1999; Uddin & Robbinson, 2018), with the concomitant losses in biodiversity (Chambers et al., 1999; Minchinton, Simpson, & Bertness, 2006). This study serves to highlight the importance of understanding how these floating wetlands are formed in the highland lakes of Mexico, since they play other important roles such as in carbon sequestration, even though their biodiversity may be low.
-
Rojas and Novelo (1995)Flora y vegetación acuáticas del lago de Cuitzeo, Michoacán, MéxicoActa Botánica Mexicana, 1995
-
Chambers, Meyerson, & Saltonstall, 1999Expansion of Phragmites australis into tidal wetlands of North AmericaAquatic Botany, 1999
-
Uddin & Robbinson, 2018Can nutrient enrichment influence the invasion of Phragmites australis?Science of the Total Environment, 2018
-
Chambers et al., 1999Expansion of Phragmites australis into tidal wetlands of North AmericaAquatic Botany, 1999
-
Minchinton, Simpson, & Bertness, 2006Mechanisms of exclusion of native coastal marsh plants by an invasive grassJournal of Ecology, 2006
The examination of satellite images provided, on the one hand, information on the behavior of native and invasive species; and on the other, the fire images may provide an insight into the resilience of the system. Even though the origin of the fires, whether natural or induced, is unknown, the fact that there were signs of fire on almost all the satellite images provides evidence that these are a common occurrence at the site. The study conducted by Escutia-Lara, Lara-Cabrera and Lindig-Cisneros (2009), which assessed the effect of fires on hydrophyte dynamics in La Mintzita, Michoacán, is an important precedent regarding the effect of fire on the structure and composition of wetlands, suggesting that such events have a positive effect by reducing the cover of the dominant species such as T. dominguensis and S. americanus and reducing competition, which in turn foster the expansion and proliferation of native species and as a consequence promote diversity. It should be noted that this phenomenon has been recorded for various cattail wetlands such as those in Chiapas (Rincón, 2014) and Veracruz (Moreno-Casasola, pers. comm.)
-
Escutia-Lara, Lara-Cabrera and Lindig-Cisneros (2009)Efecto del fuego y dinámica de las hidrófitas emergentes en el humedal de la Mintzita, Michoacán, MéxicoRevista Mexicana de Biodiversidad, 2009
-
Rincón, 2014Bosques de zapotonales (Pachira aquatica) en la Reserva de la Biosfera la Encrucijada, Chiapas, México, 2014
In this study, apparent density (0.13 g/cm3) and organic matter (96 %) values in the wetland substrate are similar to those reported for wetlands with organic soils. According to Mitsch and Gosselink (2015) the apparent density of organic soils ranges from 0.2-0.3 g/cm3, but peat bogs may have lower values due to their high porousness. As regards carbon sequestration in the substrate, the values obtained of 168 ± 10 Mg C/ha-1, are similar to the highest one reported by Hernández and Moreno-Casasola (2018) in the soil of tropical forests (150-650 Mg C ha-1). These results fall within the ranges of the sequestered carbon reported in studies conducted in tropical peat bogs in Southeast Asia with values 151.6 ± 36 and below those reported in swamps in Indonesia of 1752 ± 401 Mg-C ha-1 (Hergoualc'h & Verchot, 2011; Saragi-Sasmito, Murdiyarso, June, & Sasmito, 2019). As alredy mentioned, unlike the carbon stored in the substrate, carbon stored in the vegetation is considerably less, but still important because of its role in carbon wetland dynamics (Bedard-Haughn et al., 2006).
-
Mitsch and Gosselink (2015)Wetlands, 2015
-
Hernández and Moreno-Casasola (2018)Almacenes y flujos de carbono en humedales de agua dulce en MéxicoMadera y Bosques, 2018
-
Hergoualc'h & Verchot, 2011Stocks and fluxes of carbon associated with land use change in Southeast Asian tropical peatlands: A reviewGlobal Biogeochemical Cycles, 2011
-
Saragi-Sasmito, Murdiyarso, June, & Sasmito, 2019Carbon stocks, emissions, and aboveground productivity in restored secondary tropical peat swamp forestsMitigation and Adaptation Strategies for Global Change, 2019
-
Bedard-Haughn et al., 2006The effects of erosional and management history on soil organic carbon stores in ephemeral wetlands of hummocky agricultural landscapesGeoderma, 2006
It is well known that great amounts of carbon worldwide, approximately 530 pgC, are sequestered in wetlands (Mitsch & Gosselink 2015) and a large amount is found on the surface or soils of peat bogs (Saragi-Sasmito et al., 2019). This study is the first of its kind for the western region of Mexico and provides evidence of the potential of continental wetlands to store carbon, mainly in the soil Hernández y Moreno-Casasola (2018). However, there are various factors which may turn a wetland from a carbon sink into a source of carbon, such as changes in soil temperature (Christensen et al., 2003), the depth of the body of water (Moore, Roulet, & Waddington, 1998; Hirano et al., 2007) and the chemical composition and amount of the organic matter (Christensen et al., 2003).
-
Mitsch & Gosselink 2015Wetlands, 2015
-
Saragi-Sasmito et al., 2019Carbon stocks, emissions, and aboveground productivity in restored secondary tropical peat swamp forestsMitigation and Adaptation Strategies for Global Change, 2019
-
Hernández y Moreno-Casasola (2018)Almacenes y flujos de carbono en humedales de agua dulce en MéxicoMadera y Bosques, 2018
-
Christensen et al., 2003Factors controlling large scale variations in methane emissions from wetlandsGeophysical Research Letters, 2003
-
Moore, Roulet, & Waddington, 1998Uncertainty in predicting the effect of climatic change on the carbon cycling of Canadian peatlandsClimate Change, 1998
-
Hirano et al., 2007Carbon dioxide balance of a tropical peat swamp forest in Kalimantan, IndonesiaGlobal Change Biology, 2007
-
Christensen et al., 2003Factors controlling large scale variations in methane emissions from wetlandsGeophysical Research Letters, 2003
Recently great importance is being given, in Mexico and worldwide, to blue carbon, which is the one stored in coastal and marine ecosystems such as mangroves and seagrass beds. However, there are large bogs in both highland and coastal regions where freshwater wetlands dominate. This paper contributes to highlight the importance of increasing research focusing on freshwater wetlands. In the particular case of the wetland in our study, conservation measures should be taken to preserve its hydrophyte vegetation, reduce induced fires, ensure proper management as regards the hydrological dynamics of the lake and the quality of the water flowing into it. The Cuitzeo lake should be proposed as a RAMSAR site in order to contribute to its sustainable management and conservation.
Acknowledgments
To the Posgrado en Ciencias Biológicas, to the National Science and Technology Council for the scholarship awarded to S. P-G, to Rodrigo Velázquez-Durán, M.Sc. and to the Laboratorio de Biogeoquímica de Suelos, IIES, UNAM for carbon analyses. This project received funding from institutional resources of the IIES-UNAM.
Referencias
- Bedard-Haughn, A., Jongbloed, F., Akkerman, J., Uijl, A., Jong, E., Yates, T., & Pennock, D. (2006). The effects of erosional and management history on soil organic carbon stores in ephemeral wetlands of hummocky agricultural landscapes. Geoderma, 135, 296-306. DOI: https://doi.org/10.1016/j.geoderma.2006.01.004 Links
- Brinson, M. M., & Malvárez, A. I. (2002). Temperate freshwater wetlands: Types, status, and threats.Environmental Conservation, 29,115-133. DOI: https://doi.org/10.1017/S0376892902000085 Links
- Conagua & CEAC, Comisión Nacional del Agua & Comisión Estatal del Agua y Gestión de Cuencas. (2009). Plan de Gestión Integral de los Recursos Naturales de la Cuenca del Lago de Cuitzeo. Morelia, México: Comisión Nacional del Agua & Comisión Estatal del Agua y Gestión de Cuencas. Links
- Cram, S., Galicia, L., & Alcántara, I. I. (2010). Atlas de la cuenca del lago de Cuitzeo: análisis de su geografía y su entorno socioambiental. Instituto de Geografía-UNAM/Universidad Michoacana de San Nicolás de Hidalgo, México. Recuperado de http://www.publicaciones.igg.unam.mx/index.php/ig/catalog/book/132 Links
- Chambers, R. M., Meyerson, L. A., & Saltonstall, K. (1999). Expansion of Phragmites australis into tidal wetlands of North America.Aquatic Botany, 64, 261-273. DOI: https://doi.org/1016/S0304-3770(99)00055-8 Links
- Christensen, T. R., Ekberg, A., Ström, L., Mastepanov, M., Panikov, N., Oquist, M., Svenson, B. H., Nykanen, H., Martikainen, P. J., & Oskarsson, H. (2003). Factors controlling large scale variations in methane emissions from wetlands. Geophysical Research Letters, 30, 1-67. DOI: http://doi.org/10.1029/2002GL016848 Links
- Davidson, N. C. (2014). How much wetland has the world lost? Long-term and recent trends in global wetland area.Marine and Freshwater Research, 65, 934-941. DOI: https://doi.org/10.101017/MF14173 Links
- Escutia-Lara, Y., Lara-Cabrera, S., & Lindig-Cisneros, R. (2009). Efecto del fuego y dinámica de las hidrófitas emergentes en el humedal de la Mintzita, Michoacán, México.Revista Mexicana de Biodiversidad, 80, 771-778. Links
- Fiedler, S., Vepraskas, M. J., & Richardson, J. L. (2007). Soil redox potential: Importance, field measurements, and observations.Advances in Agronomy, 94, 1-54. DOI: https://doi.org/10.1016/S0065-2113(06)94001-2 Links
- Hernández, M. E., & Moreno-Casasola, P. (2018). Almacenes y flujos de carbono en humedales de agua dulce en México.Madera y Bosques, 24. DOI: https://doi.org/10.21829/myb.2018.2401881 Links
- Hergoualc'h, K., & Verchot, L. V. (2011). Stocks and fluxes of carbon associated with land use change in Southeast Asian tropical peatlands: A review. Global Biogeochemical Cycles, 25. DOI: https://doi.org/10.1029/2009GB003718 Links
- Hirano, T., Segah, H., Harada, T., Limin, S., June, T., Hirata, R., & Osaki, M. (2007). Carbon dioxide balance of a tropical peat swamp forest in Kalimantan, Indonesia. Global Change Biology, 13, 412-425. DOI: https://doi.org/10.1111/j.1365-2486.2006.01301.x Links
- Hoogsteen, M. J. J., Lantinga, E. A., Bakker, E. J., Groot, J. C. J., & Tittonell, P. A. (2015). Estimating soil organic carbon through loss on ignition: Effects of ignition conditions and structural water loss.European Journal of Soil Science, 66, 320-328. DOI: https://doi.org/10.1111/ejss.12224 Links
- Huffman, E. N. (1997). Performance of a new automatic carbon dioxide coulometer.Microchemial Journal, 2, 567-573. DOI: https://doi.org/10.1016/0026-265X(77)90128-X Links
- Jacobs, A. E., & Harrison, J. A. (2014). Effects of floating vegetation on denitrification, nitrogen retention, and greenhouse gas production in wetland microcosms.Biogeochemistry, 119, 51-66. DOI: https://doi.org/10.1007/s10533-013-9947-9 Links
- Kotze, D. C. (2013). The effects of fire on wetland structure and functioning.African Journal of Aquatic Science, 38, 237-247. DOI: https://doi.org/10.2989/16085914.2013.828008 Links
- Landgrave, R., & Moreno-Casasola, P. (2012). Evaluación cuantitativa de la pérdida de humedales en México.Investigación Ambiental, Ciencia y Política Pública, 4. Links
- Laubhan, M. K. (1995). Effects of prescribed fi re on moist-soil vegetation and soil macronutrients. Wetlands, 15, 159-166. DOI: https://doi.org/10.1007/BF03160669 Links
- Martin, K. L., & Kirkman, L. K. (2009). Management of ecological thresholds to re-establish disturbance-maintained herbaceous wetlands of the south-eastern USA. Journal of Applied Ecology, 46, 906-914. DOI: https://doi.org/10.1111/j.1365-2664.2009.01659.x Links
- Masters, B. (2012). The ability of vegetated floating Islands to improve water quality in natural and constructed wetlands: A review.Water Practice and Technology, 7. DOI: https://doi.org/10.2166/wpt.2012.022 Links
- Minchinton, T. E., Simpson, J. C., & Bertness, M. D. (2006). Mechanisms of exclusion of native coastal marsh plants by an invasive grass. Journal of Ecology, 94, 342-354. DOI: https://doi.org/10.1111/j.1365-2745.2006.01099.x Links
- Mitsch, W. J., Bernal, B., Nahlik, A. M., Mander, Ü., Zhang, L., Anderson, C. J., & Brix, H. (2013). Wetlands, carbon, and climate change.Landscape Ecology, 28, 583-597. DOI: https://doi.org/10.1007/s10980-012-9758-8 Links
- Mitsch, W. J., & Gosselink, J. (2015). Wetlands (5th ed.). New York, USA: John Wiley and Sons Inc. Links
- Moore, T. R., Roulet, N. T., & Waddington, J. M. (1998). Uncertainty in predicting the effect of climatic change on the carbon cycling of Canadian peatlands. Climate Change, 40, 229-245. DOI: https://doi.org/10.1023/A:1005408719297 Links
- Nyman, J. A., & Chabreck, R. H. (1995). Fire in coastal marshes: History and recent concerns. In:Fire in wetlands: A management perspective. Proceedings of the Tall Timbers Fire Ecology Conference. Recuperado de http://talltimbers.org/wp-content/uploads/2014/03/NymanandChabreck1995_op.pdf Links
- Olmsted, I. (1993). Wetlands of Mexico. In: Whigham, D. F., Dykyjová, D., & Hejnÿ, S. (eds.) Wetlands of the world I: inventory, ecology and management. Handbook of vegetation science. Dordrecht, The Netherlandas: Kluwer Academic Publishers. DOI: https://doi.org/10.1007/978-94-015-8212-4 Links
- Paige, S. E., & Baird, A. J. (2016). Peatlands and global change: Response and Resilience.Annual Review of Environment and Resources, 41, 35-57. DOI: https://doi.org/10.1146/annurev-environ-110615-085520 Links
- Reddy, K. R., & De Laune, R. D. (2008).Biogeochemistry of wetlands: Science and applications. CRC press, Boca Raton, USA. Links
- Rincón, P. M. (2014). Bosques de zapotonales (Pachira aquatica) en la Reserva de la Biosfera la Encrucijada, Chiapas, México (tesis de maestría en ciencias biológicas). Universidad Nacional Autónoma de México, México. Links
- Rojas, M. J., & Novelo, R. A. (1995). Flora y vegetación acuáticas del lago de Cuitzeo, Michoacán, México.Acta Botánica Mexicana, 31, 1-17. Links
- Rolletschek, H., Rolletschek, A., Hartzendorf, T., & Kohl, J. G. (2000). Physiological consequences of mowing and burning of Phragmites australisstands for rhizome ventilation and amino acid metabolism.Wetlands Ecology and Management, 8, 425-433. DOI: https://doi.org/10.1023/A:1026562002388 Links
- Smardon, R. (2014). Wetland ecology principles and conservation.Water, 6, 813-817. DOI: https://doi.org/10.3390/w6040813 Links
- Saragi-Sasmito, M. F., Murdiyarso, D., June, T., & Sasmito, S. D. (2019). Carbon stocks, emissions, and aboveground productivity in restored secondary tropical peat swamp forests.Mitigation and Adaptation Strategies for Global Change, 24, 521-533. DOI: https://doi.org/10.1007/s11027-018-9793-0 Links
- Schmalzer, P. A., & Hinkle, C. R. (1992). Soil dynamics following fire in Juncus and Spartina marshes. Wetlands, 12, 8-21. DOI: https://doi.org/10.1007/BF03160539 Links
- Uddin, M. N., & Robinson, R. W. (2018). Can nutrient enrichment influence the invasion of Phragmites australis?Science of the Total Environment, 613, 1449-1459. DOI: https://doi.org/10.1016/j.scitotenv.2017.06.131 Links
- van Asselen, S., Verburg, P. H., Vermaat, J. E., & Janse, J. (2013). Drivers of wetland conversion: A global meta-analysis.PLoS ONE, 8. DOI: https://doi.org/10.1371/journal.pone.0081292 Links
- van der Valk, A. (2012).The biology of freshwater wetlands. Oxford, UK: Oxford University Press. Links
- Watts, A. C., Kobsiar, L. N., & Snyder, J. R. (2012). Fire reinforces structure of pond cypress (Taxodium distichum var. imbricarium) domes in a wetland landscape. Wetlands, 32, 439-448. DOI: https://doi.org/10.1007/s13157-012-0277-9 Links
- Yeh, N., Yeh, P., & Chang, Y. H. (2015). Artificial floating islands for environmental improvement.Renewable and Sustainable Energy Reviews, 47, 616-622. DOI: https://doi.org/10.1016/j.rser.2015.03.090 Links