Artículos
Chemical oxygen demand with APHA/AWWA/WEF 5220 D procedure for high concentration adapted to micro-volume
Morales-Mejía, Julio César1
 http://orcid.org/0000-0002-0230-1953
http://orcid.org/0000-0002-0230-1953
Vargas-Martínez, María Gabriela2
Medina-Camps, Javier3
 http://orcid.org/0000-0002-0230-1953
http://orcid.org/0000-0002-0230-1953Vargas-Martínez, María Gabriela2
Medina-Camps, Javier3
-
Publication dates-
June 26, 2025
Jan-Feb , 2021
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
Abstract
In this study, linearity and precision tests were performed for the determination of the Chemical Oxygen Demand (COD) in water, for a micro-volume adaptation of the closed reflux method for small volume for high range of APHA/AWWA/WEF. The main objective of the work was to demonstrate that it is possible to obtain adequate measurements by adapting the APHA/AWWA/WEF method 5220 D, a widely used international reference for determining COD in water, on a micro-volume adaptation. The micro-volume adapted method required only changes in the concentration of each substance in the digestion solution, but not in the silver sulfate catalyst solution, so that the concentration of each of these substances in the tubes digestion was almost equal to that corresponding to the original method (except for mercury sulfate, which was deliberately reduced). In the micro-volume adaptation presented here, the dosed volumes of all liquid reagents were measured using class A volumetric pipettes, improving the precision of measurements with respect to the use of both graduated and plunger pipettes (necessary to carry out the original method). The results obtained for the micro-volume adaptation demonstrated excellent linear fitting and precision of the system.
Keywords::
COD, micro-volume, linearity, precision, water, Standard Methods
Introduction
In the treatment of water and wastewater it is necessary to determine impurities to establish the most appropriate purification technologies. The contaminants present in the water are very varied in nature and include, among many others, organic materials. Global analyses of organic substances in water, Biochemical Oxygen Demand (BOD), Chemical Oxygen Demand (COD) and Total Organic Carbon (TOC) provide an adequate approximation of the sum of organic pollutants that share some common property. These three parameters, based on oxidation processes, are indirect quantifications of organic constituents of the water, whose determination is usually a routine in Wastewater Treatment Plants, as well as in many certified laboratories.
According to Metcalf & Eddy Inc. (2004), the total COD is the oxygen equivalent to the organic materials in the water sample that can be chemically oxidized by means of a dichromate solution in acidic medium (dichromate is the most common oxidant), along with a catalyst and interference inhibitors. Meanwhile, APHA, AWWA, and WEF (1985) define COD more generally, indicating that it is the measurement of oxygen equivalent to the organic matter content of a sample that is susceptible to chemical oxidation by a strong chemical oxidant (not necessarily dichromate).
-
Metcalf & Eddy Inc. (2004)Wastewater engineering, 2004
-
APHA, AWWA, and WEF (1985)Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
In the work of APHA, AWWA, and WEF (1985) it is established that, during the quantification of COD, the oxidation with dichromate manages to oxidize between 95 and 100% of organic substances, resisting this oxidation only the pyridine compounds and volatile organic compounds (VOCs), the former because they are chemically stable under the proposed reaction conditions and the latter because they can only be oxidized by being in the liquid phase in contact with the oxidant and, being at reflux, they are easily transferred to the gas phase.
-
APHA, AWWA, and WEF (1985)Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
Among the available methods for determining COD, small-scale closed reflux colorimetry is widely used (as in the NMX-AA-030/2-SCFI-2011 standard) (DOF, 2011) and its importance is increasing. This is because it requires only a visible spectrophotometer and the sample volumes and reagents are much lower than in the case of open reflux, generating less hazardous waste (with environmental toxicity by chromium and/or mercury species, which are highly reactive because they are strongly acidic media and due to the presence of the strong oxidant dichromate): 50 to 150 ml with open reflux compared to 5 to 7.5 ml with conventional closed reflux methods.
-
DOF, 2011Norma mexicana NMX-AA-030/2-SCFI-2011, Análisis de agua - Determinación de la demanda química de oxígeno en aguas naturales, residuales y residuales tratadas. Método de prueba. Parte 2, 2011
In general, Equation (1) shows the reaction that takes place during the COD analysis (considering organic nitrogen species where the nitrogen valence is -3, there are nitrites in a significant amount (greater than 2 mg of nitrite nitrogen per liter), and there is no significant concentration of reduced inorganic species such as Fe2+, S2-, Mn2+, etc. (APHA, AWWA, & WEF, 1985; APHA, AWWA, & WEF, 2017; ASTM, 2012). Potassium biphthalate as organic material to be oxidized (calibration curve), the reaction in the reflux system will be that of Equation (2) (ASTM, 2012). Finally, in Equation (3) we have the chemical reaction if the oxidation of the Potassium biphthalate with molecular oxygen (ASTM) Thus, according to reactions (2) and (3), one mole of dichromate has the same oxidation capacity as 1.5 moles of molecular oxygen and, based on the reaction (3), the theoretical COD of biphthalate is 1.175 g of oxygen per each gram of potassium bipthalate:
-
APHA, AWWA, & WEF, 1985Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
-
APHA, AWWA, & WEF, 2017; ASTM, 2012Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
-
ASTM, 2012Standard Test Methods for chemical oxygen demand (dichromate oxygen demand) of water, D 1252-06, 2012
e4
C
n
H
a
O
b
H
c
+
e
N
O
2
-
+
d
C
r
2
O
7
2
-
+
8
d
+
c
H
+
→
n
C
O
2
+
f
H
2
O
+
c
N
H
4
+
+
2
d
C
r
3
+
+
e
N
O
3
-
(1)
e5
41
H
2
S
O
4
+
10
K
2
C
r
2
O
7
+
2
K
C
8
H
5
O
4
→
10
C
r
2
(
S
O
4
)
3
+
11
K
2
S
O
4
+
16
C
O
2
+
46
H
2
O
(2)
e6
2
K
C
8
H
5
O
4
+
15
O
2
+
H
2
S
O
4
→
K
2
S
O
4
+
16
C
O
2
+
6
H
2
O
(3)
On the other hand, the main interferences in the determination of COD are: presence in significant concentrations of linear aliphatic volatile organic compounds, halogens, nitrites and reduced inorganic species (Fe2+, S2-, Mn2+, etc.). For the original 5220 D method, it is established that, in samples with low presence of nitrites and chemical interferences (the chlorides are the only ones that are usually present in most of the samples), the required reagents are only two: the digestion reagent (potassium dichromate) and the catalytic reagent (silver sulfate in concentrated sulfuric acid). If the amount of chlorides is of importance, then the addition of mercury sulfate to the dichromate reagent is necessary). In the case of the original 5220 D method, the recommended volume of the sample and the other reagents is presented in Table 508:I of APHA, AWWA, and WEF (1985), where it is also established that other volumes of these reagents and sample can be used, as long as the ratio between them is kept at 1: 0.6: 1.4 (sample:digestion solution:catalyst solution), with the concentrations indicated in the same reference and in subsequent editions.
-
APHA, AWWA, and WEF (1985)Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
Materials and methods
In this work, the partial validation of a 5220 D closed reflux (APHA, AWWA, & WEF, 1985) COD analysis method was developed, which was adapted to a micro-volume. For this, the linearity of the system and the precision were analyzed. Solutions were made with a primary standard, high purity potassium biphthalate (Sigma-Aldrich). The standard was dried at 105 °C for 60 minutes and then dissolved in tri-distilled water (Ventas Químicas, SA), to have solutions with concentration of 0, 200, 400, 600 and 800 mg/l of the compound (equivalent to COD of 0, 235, 470, 705 and 940 mg O2 /l, respectively).
-
APHA, AWWA, & WEF, 1985Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
The borosilicate glass vials were first washed with a 1:10 (volume basis) solution of concentrated sulfuric acid in tri-distilled water, rinsed with water and air-dried. The catalyst and digestion reagents were prepared in such a way that the same concentration of dichromate, silver sulfate and sulfuric acid was maintained in the glass vials, just before the digestion began, in the micro-volume adaptation as in the original 5220-D method (except for the initial concentration of mercury sulfate, which was reduced when considering that the micro-volume method will be applied to municipal wastewater with low concentrations of chlorides, in order to reduce the danger and environmental impact of the waste generated in the COD analysis).
Subsequently, the reagents (Table 1) and then the samples (potassium biphthalate solutions) were added to the glass vials, as indicated in Table 2; the vials were tightly capped, slowly shaken and placed in the digester block (HANNA Instruments, HI839800) preheated to 150 ° C. There, the standard solutions were digested at 150 ° C for 2 hours (closed reflux); after the digestion was complete, the vials were slowly air-cooled to room temperature and gently shaken without inverting them; the absorbance of these digested and cooled solutions were read in the spectrophotometer (Shimadzu UV1601) in a rectangular glass cuvette with an optical path length of 1 cm, using tri-distilled water as blank (or reference) of the spectrophotometer, measuring the absorbance at 600 nm (due to the generation of chromium III). For this micro-volume adaptation, tri-distilled water was used as a blank instead of the undigested 0 mg O2 /l solution established in APHA, AWWA, & WEF (2017) to determine the performance of the method with a non-specific blank for parameter. The pure water blank is indicated for measurement at 420 nm by APHA, AWWA and WEF (2017), so that its use as a reference at 600 nm constitutes an option of practical interest in laboratories of water quality analysis.
-
APHA, AWWA, & WEF (2017)Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
-
APHA, AWWA and WEF (2017)Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
Table 1
Reagents for Method 5220 D and its modification to micro-volume (Based on APHA, AWWA, & WEF, 2017).
Reagents for Method 5220 D and its modification to micro-volume (Based on APHA, AWWA, & WEF, 2017).
| Original method | Method adapted to micro-volume | |
|---|---|---|
| Catalytic reagent (Ag2SO4) | 5.5 g of Ag2SO4 for each kilogram of concentrated H2SO4 | Commercial solution, 10 g/l of Ag2SO4 in concentrated H2SO4 |
| Digestion reagent (Cr2O72-) | 10.216 g of K2Cr2O7, 33.3 g of HgSO4 (dried at 105 °C during 2 hours); 400 ml of tri-distilled water; 167 ml of concentrated H2SO4 (slowly added), measure to 1 000 ml with tri-distillated water; in this sequence. | 10.22 g of K2Cr2O7, 0.75g of HgSO4 (dried at 105°C during 2 hours); 400 ml of tri-distillated water; 500 ml of concentrated H2SO4 (slowly added and externally ice-cooled to room temperature); measure to 1000 ml with tri-distillated water; in this sequence. |
-
APHA, AWWA, & WEF, 2017Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
Table 2
Dosage of sample and reagents in the reaction vial for Method 5220-D. Based on APHA, AWWA, & WEF (2017).
Dosage of sample and reagents in the reaction vial for Method 5220-D. Based on APHA, AWWA, & WEF (2017).
| Original method | Method adapted to micro-volume | |
|---|---|---|
| Water sample | 2.5 ml | 1.0 ml |
| Catalytic reagent (Ag2SO4) | 3.5 ml | 1.0 ml |
| Digestion reagent (Cr2O72-) | 1.5 ml | 0.5 ml |
| Total volume inside digestion vial (ml) | 7.5 | 2.5 |
-
APHA, AWWA, & WEF (2017)Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
In some samples, a white precipitate was produced, which was deposited in the mouth of the vials; in those cases, it was mechanically removed before reading the samples on the spectrophotometer. The data were analyzed using Origin Pro v. 8 and Microsoft Excel.
For linearity, the calibration curve was measured in triplicate for 5 concentration levels, and for the precision of the method, six-fold digestions were made of samples of 3 concentration levels, all in duplicate. With the determined COD values, the coefficient of determination (r2) was calculated to evaluate the fit to the linear model, and the coefficient of variation (CV) that was used to evaluate the precision.
In the 5220 D method adapted to micro-volume, care was taken that the additions of reagents and samples were made with class A volumetric pipettes and not with piston or graduated pipettes, as it would be necessary in the original 5220 D method or in the Mexican technical standard NMX-AA-030/2-SCFI-2011. Table 3 shows the concentrations of the reagents in the vials just before starting the digestion of the samples. In order to comply with equality of initial concentrations of both methods (the original and that adapted to the micro-scale), the amount of H2SO4 in the digestion solution was adapted in the proposed micro-volume method.
Table 3
Concentration of chemical species of interest for Method 5220-D just before starting digestion. Based on APHA, AWWA and WEF (2017).
Concentration of chemical species of interest for Method 5220-D just before starting digestion. Based on APHA, AWWA and WEF (2017).
| Original method | Method adapted to micro-volume | |
|---|---|---|
| Ag2SO4 | 4.697 g/l | 4.000 g/l |
| K2Cr2O7 | 2.043 g/l | 2.044 g/l |
| H2SO4 | 901.720 g/l | 901.600 g/l |
| HgSO4 | 6.660 g/l | 0.150 g/l |
-
APHA, AWWA and WEF (2017)Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
It is important to establish that the concentration of HgSO4 in the digestion solution was drastically reduced in the micro-volume adaptation, since in the majority of municipal wastewater and treated wastewater samples high concentrations of chlorides are not expected. For this study, a maximum concentration of chlorides in water of 4.5 mg Cl-/l (7.4 mg NaCl/l) was considered. Additionally, in the original method it is established that the amount of mercury sulfate can be modified according to the expected chlorides in the sample to be analyzed. Chlorides interference is considered properly eliminated with mercury concentration in the digestion vial 10 times higher than that of chlorides (APHA, AWWA, & WEF, 2017).
-
APHA, AWWA, & WEF, 2017Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater, 2017
Results and discussion
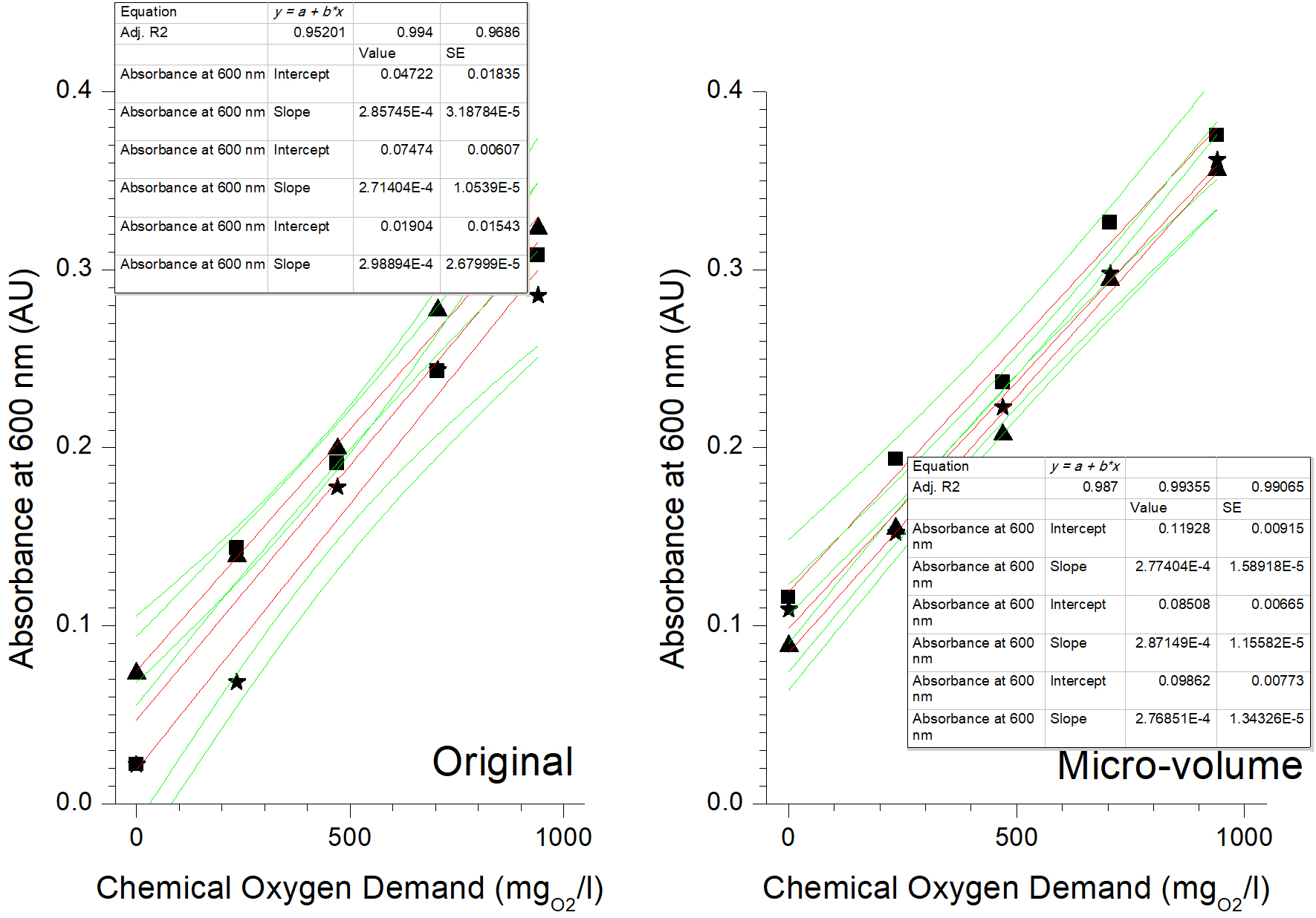
After performing the digestion of the standard samples in triplicate, the calibration curves were plotted, both for the results of the analysis with the original (conventional) method, and for those obtained from the adapted micro-volume (Figure 1). In this second case, all the resulting plots had a coefficient of determination (r 2) greater than 0.98. Therefore, according to the Colegio Nacional de Químicos Farmacéuticos Biólogos México, A.C. (2002), the micro-volume method is valid for linearity of the system. The original 5220-D method has already been widely validated and its validation was not repeated in this work.
-
Colegio Nacional de Químicos Farmacéuticos Biólogos México, A.C. (2002)Guía de Validación de Métodos Analíticos, 2002
Thumbnail
Figure 1
Linearity of Methods APHA/AWWA/WEF 5220 D original and of its adaptation to micro-volume.
Linearity of Methods APHA/AWWA/WEF 5220 D original and of its adaptation to micro-volume.
Table 4 presents the coefficients of determination (r 2) for the linear fit of both COD methods at closed reflux. It is important to note that the fit to the model (r 2) is more repeatable for the micro-volume adaptation than for the original method. An important aspect is the modification of the proportions of the reagents with respect to the original method, which is due to the fact that the 5220 D procedure requires additions of volumes for which there is no exact volumetric (bulb) pipette, which implies using at least 2 pipettes, which increases the error in the procedure (particularly if air displacement micropipettes are used without their own calibration). Using 0.5 and 1 ml of the reagents allows each dosage to be carried out with a single volumetric pipette and in a single addition, reducing the error associated with taking the aliquot.
Table 4
Experimental coefficients of determination of linear fitting (r2) for Method 5220-D.
Experimental coefficients of determination of linear fitting (r2) for Method 5220-D.
| Original | Method adapted to micro-volume | |
|---|---|---|
| Repetition 1 | 0.9520 | 0.9870 |
| Repetition 2 | 0.9940 | 0.9933 |
| Repetition 3 | 0.9686 | 0.9906 |
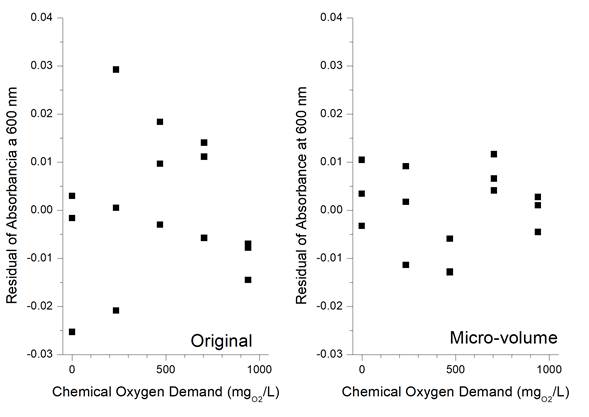
For the precision of the method adapted to micro-volume (Table 5), the samples showed good repeatability, so that the value of the Coefficient of Variation (CV) was always less than 3%. This CV value indicates that the Method 5220-D adapted to micro-volume is precise enough to be used in a routine analysis. In fact, in the same original method 5220 D (APHA, AWWA, & WEF, 1985) it is established that 48 samples were analyzed in 5 laboratories and that the CV was 8.7% for a mean COD of 193 mg O2 /l in the absence of chlorides, and 9.6% for an average COD of 212 mg O2 /l in the presence of 100 mg/l of chlorides, so the results obtained here have a lower CV value (Figure 2).
-
APHA, AWWA, & WEF, 1985Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater, 1985
Table 5
Precision for Method 5220-D adapted to micro-volume.
Precision for Method 5220-D adapted to micro-volume.
| Repetition | Average DQO (mg O2/l) | Standard Deviation S (mg O2/l) | Precision (CV) |
|---|---|---|---|
| 1-a | 225.86111 | 5.30512 | 2.34884 |
| 2-a | 563.80417 | 10.20365 | 1.80978 |
| 3-a | 800.24028 | 22.61028 | 2.82544 |
| 1-b | 225.33889 | 4.33083 | 1.92191 |
| 2-b | 523.52778 | 13.43289 | 2.56584 |
| 3-b | 774.71667 | 19.37226 | 2.50056 |
Thumbnail
Figura 2
Residuals for linearity of original Method 5220-D (left) and adapted to micro-volumen (right).
Residuals for linearity of original Method 5220-D (left) and adapted to micro-volumen (right).
Conclusions
-
The adaptation to micro-volume of Method 5220-D allowed quantifying the chemical oxygen demand in water in an analogous way to its original version.
-
The Method 5220-D adapted to micro-volume was successfully described by a simple linear model in the COD range from 0 to 940 mg O2 /l, based on the successful determination of linearity (r2> 0.98).
-
The Method 5220-D adapted to micro-volume was precise, with a coefficient of variation of less than 3% in all tests (analysis for 3 concentration levels).
-
The hazardous waste generated after implementing the water analysis with the micro-scale adaptation (2.5 ml per analysis), has been reduced by up to 50% by volume compared to the analysis method of the Mexican standard NMX-AA-030/2-SCFI-2011 (5.2 ml of residue per test) and up to 75% by volume with respect to analyzes based on the original 5220-D method (7.5 ml of residue per determination).
Acknowledgements
Authors thank to DGAPA-UNAM by financial support through the projects PE214517 “Microescala en Fotocatálisis Heterogénea Aplicada al Tratamiento de Aguas” and IA106318 “Depositado de TiO2 sobre soportes densos para la oxidación fotocatalítica solar de contaminantes de preocupación emergente en agua”.
Referencias
- APHA, AWWA, & WEF, American Public Health Association, American Water Works Association, & Water Environment Federation. (1985). Method 5220 D, chemical oxygen demand. Standard methods for the examination water and wastewater. Washington DC, Denver, & Alexandria, USA: American Public Health Association, American Water Works Association, Water Environment Federation. Links
- APHA, AWWA, & WEF, American Public Health Association, American Water Works Association, & Water Environment Federation. (2017). Method 5220 D, Chemical Oxygen Demand. Standard Methods for the Examination Water and Wastewater. Washington DC, Denver, & Alexandria, USA: American Public Health Association, American Water Works Association, Water Environment Federation . Links
- ASTM, American Society for Testing and Materials. (2012). Standard Test Methods for chemical oxygen demand (dichromate oxygen demand) of water, D 1252-06. Pennsylvania, USA: American Society for Testing and Materials. Links
- Colegio Nacional de Químicos Farmacéuticos Biólogos México, A.C. (2002). Guía de Validación de Métodos Analíticos. México, DF, México: Editorial Colegio Nacional de Químicos Farmacéuticos Biólogos México, A.C. Links
- DOF, Diario Oficial de la Federación. (2011). Norma mexicana NMX-AA-030/2-SCFI-2011, Análisis de agua - Determinación de la demanda química de oxígeno en aguas naturales, residuales y residuales tratadas. Método de prueba. Parte 2. México, DF, México: Diario Oficial de la Federación. Links
- Metcalf & Eddy Inc. (2004). Wastewater engineering. Boston, USA: McGraw-Hill. Links