Introduction
Gastrointestinal cellular development, physiology, microbiota, and immunity adjust their functions according to the animal requirements, to nurture and confer protection against potentially pathogen microorganisms that could compromise the survival of the early weaned piglets, this due to the immaturity of their digestive and immunological activity1,2. Weaning of piglets is associated with an imbalance between immune and inflammation responses, leading to the development of pathological diseases by the secretion of pro-inflammatory cytokines3. Cytokines are a group of proteins and glycoproteins synthesized by different cell lines, responsible for the regulation of inflammatory and immune responses that act at the systemic level by modulating cellular activity through interaction with specific membrane receptors that trigger a chemical reaction downstream. The synthesis and release of cytokines can be stimulated by inappropriate inflammatory activation caused by feed, environment, bacteria, and metabolites, as well as the presence of other proinflammatory cytokines, including interleukin-1 (IL-1), tumor necrosis factor alpha (TNF-(), and interleukin-6 (IL-6), which induce the production of acute phase proteins4,5. The proinflammatory cytokine TNF-( acts as an acute inflammation mediator that increases the production of IL-1 and IL-66.
The gut microbiota is a complex population with hundreds of diverse microorganisms that contribute to the breakdown of nutrients and energetic metabolism, affecting the immune system and homeostasis7. Gut microorganisms in the large intestine are responsible for the fermentation of indigestible carbohydrates and protein in piglets diets to produce a series of metabolites denominated volatile fatty acids (VFA). Among the various metabolites, short chain fatty acids (SCFA) have received extensive attention because of their positive effects on health8.
Antibiotics have been widely used in the swine industry as growth promoters, which has led to bacterial resistance development and to the presence of antibiotic residue in animal products9. To face this problematic, the need to identify alternatives that replace the use of antibiotics while maintaining growth productive parameters has arisen. Potato protein concentrate has been considered as a valuable essential amino acid source that could replace the inclusion of animal protein in the piglet starter diets due to the presence of antimicrobial peptides (AMP) and the content of high digestible protein10,11. The antibacterial activity of PPC AMP is based primarily on the interaction of positively charged peptides with negatively charged components of the bacterial membrane such as phospholipids and teichoic acids of gram-positive bacteria or lipopolysaccharide of gram-negative bacteria, which leads to pore formation, membrane permeabilization, and cell lysis after re-localization in the cytosolic membrane12.
PPC antimicrobial activity could be an interesting alternative to the use of antibiotic as growth promoters, altering the composition of the microbiota to reduce competition for nutrients, reduce pathogen, and control mucus, however the level of inclusion has not yet been determined for piglets nutrition, therefore, it is necessary to evaluate the effect of different levels of inclusion.
This study aim was to establish the optimum level of PPC inclusion to decrease the post weaning inflammatory process, defined by the presence of pro inflammatory markers, as well as VFA profile.
Material and methods
Animal management
All animal experimental procedures were conducted according to the guidelines of the Mexican Official Norm NOM-062-ZOO-1999 for production, care and use of animals for experimentation13 and guidelines of the International Guiding Principles for Biomedical Research Involving Animals14. The experimental design and procedures in this study were reviewed and approved by the Bioethical Committee of the Faculty of Natural Sciences of the Autonomous University of Queretaro (approval number: 96FCN2021).
Ninety (90) hybrid piglets (Large white x Landrace) x PIC337 weaned at 21 ± 2 d and weighing 6.85 ± 0.93 kg were utilized in this study and assigned to three experimental diets according to litter origin and bodyweight, and were housed in six pens per diet, each with five piglets, making a total of 30 piglets per diet. Three experimental diets were formulated with a basal diet without antibiotics or potato protein concentrate (C) subsequently 6 % and 8 % of potato protein concentrate was added to the basal diet to establish the two other diets, namely PPC 6% and PPC 8% respectively, as described in Table 1. Pens were equipped with a feeder with six spaces and nipple drinkers in an environmentally controlled weaning room (32 ºC with -2 ºC during the first and second post weaning weeks).
Table 1 Ingredients and chemical percentage composition of the experimental diets
| Ingredients (%) | Experimental diets | ||
|---|---|---|---|
| C | PPC 6% | PPC 8% | |
| Maize | 40.63 | 39.69 | 39.36 |
| Soybean meal | 15.00 | 15.00 | 15.00 |
| Soybean protein isolate | 11.55 | 6.98 | 5.46 |
| Potato protein concentrate | - | 6.00 | 8.00 |
| Sweet whey powder | 24.69 | 24.69 | 24.69 |
| Maize oil | 3.29 | 3.06 | 2.98 |
| Lysine | 0.41 | 0.29 | 0.24 |
| AminoGut®* | 0.80 | 0.80 | 0.80 |
| Threonine | 0.11 | 0.01 | - |
| Methionine | 0.22 | 0.17 | 0.15 |
| Tryptophan | 0.01 | - | - |
| Sodium | 0.50 | 0.50 | 0.50 |
| Calcium carbonate | 0.54 | 0.48 | 0.45 |
| Dicalcium phosphate | 1.64 | 1.71 | 1.74 |
| Titanium dioxide | 0.30 | 0.30 | 0.30 |
| Vitamins** | 0.07 | 0.07 | 0.07 |
| Coline | 0.14 | 0.14 | 0.14 |
| Minerals*** | 0.10 | 0.10 | 0.10 |
| Dry matter, %a | 92.73 | 92.85 | 92.55 |
| Crude protein, %a | 23.12 | 22.99 | 23.45 |
| ME, kcal/kgb | 3,400 | 3,400 | 3,400 |
*L-glutamine, L-glutamic acid (1:1).**vitamins per kg of diet, A: 13,000 UI, E: 160 mg, K: 9 mg, thiamine: 4 mg, riboflavine: 12 mg, pyridoxine:6 mg, cianocobalamine: 0.07 mg, niacin:66 mg, panthotenate: 46 mg, folate: 5 mg, biotin: 0.67 mg, C: 266 mg. ***minerals per kg of diet, manganese: 32 mg, zinc: 120 mg, iron: 100 mg, copper: 12 mg, iodine 0.8 mg, selenium: 0.25 mg, cobaltum: 0.6 mg.
a Analyzed value, b Calculated value.
Samples collection
At 15 d post weaning, six piglets per diet group were euthanized for posterior sampling. Animals were tranquilized with 20 mg/kg azaperone (Sural® Chinoin, Mexico City, Mexico) and then euthanized with an overdose of sodium pentobarbital (Pisabental ®, PiSA Agropecuaria, Hidalgo, Mexico). Ileal tissue sections (10 cm) were sampled from 5 cm before the ileocecal valve, gently washed with distilled water, and then dissected and fixed in 10 % buffered formalin solution. Ileal sections were embedded in paraffin and cut into 5 µm slices. Colonical digest content was collected, frozen and stored for later analysis.
Cytokines analysis
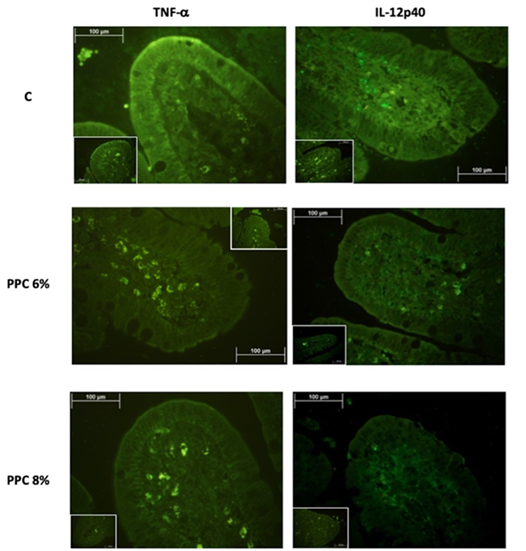
The 5 µm slices were used to quantify TNF-( and IL-12p40 using an immunofluorescence technique adapted from that of Bautista-Marín et al15. Ileal slices were deparaffinized for 24 h at 60 °C in a dry-heat oven and then rehydrated in 100% xylol (10 min), 100% ethanol (5 min), 96% ethanol (5 min) and deionized water (10 min), in this order. Ileal slices were then soaked in saline sodium citrate buffer concentrate (Sigma Aldrich, USA) in a water bath at 80 ºC for 25 min .After that, ileal slices were blocked in 1% non-fat milk for 1 h, washed three times for 5 min each with 0.05% TRIS buffered saline plus Tween (TBST), and incubated for 2 h at 4 ºC with the following antibodies diluted in TBST: i) rabbit anti TNF-( (ab 6671) (Abcam, Cambridge, MA, USA) at 1:200 dilution, ii) rabbit anti IL-12p40 (ab 106270) (Abcam, Cambridge, MA, USA) at 1:100 dilution and incubated for 16 h .Ileal slices were then washed three times for 5 min each with 0.05% TBST and incubated for 2 h with the secondary antibody Alexa Fluor 488 chicken anti-rabbit IgG (H+L) (A-21441) (Invitrogen Molecular Probes Inc., Eugene, OR, USA) at 1:500 dilution. The intensity of the fluorescence (arbitrary fluorescence units (AFU)) was evaluated with the positive (primary and secondary antibodies) and negative (secondary antibodies only) controls considering the same size of area in ileal tissue under both conditions. For the analysis, fluorescence was visualized and measured with a fluorescence microscope (Eclipse E600, Nikon) and Image-Pro Plus version 6.1 in 10 intestinal villi per piglet and photographed at 40x magnification and then 10 sections from each intestinal villi were selected per photo to obtain 100 measurements per piglet.
Volatile fatty acid analysis
Approximately 2 g of colonical digest content were weighted and placed in previously identified centrifuge tubes, after that 3 ml of HPLC- grade water were added to the tubes and mixed with a vortex for 30 sec. Tubes were then placed in a Beckman Coulter centrifuge at 4 ºC for 30 min. With the assistance of a 3 ml syringe supernatant was extracted and emptied in an amber tube after passing through a 0.2-μm syringe filter. Concentrations of acetic, propionic, butyric, and valeric acids (SCFA); isobutyric, isovaleric, and isocaproic acids (BFA) were analyzed on an Agilent 6890 gas chromatograph equipped with a flame-ionization detector and DB-FFAP column (30 m × 0.25 mm × 0.25 μm; Agilent Technologies, Wilmington, DE). The following gas chromatograph parameters were used: split mode, 20:1; inlet temperature, 220 °C; initial inlet pressure, 168 kPa; injection volume, 1 μL; constant column flow (He), 1.4 mL/min; and detector temperature, 250 °C.
Statistical analysis
Inflammatory marker concentration as well as volatile fatty acid concentration were analyzed using a completely randomized design, each piglet was considered an experimental unit16. ANOVA was utilized to analyze the differences between diets groups. Means were compared with the Tukey test using the GLM procedure of SAS and differences were considered statistically significant at P<0.0517.
Results
Inflammatory cytokines concentration
The concentrations of TNF-( and IL-12p40 in the ileal villi of weaned piglets were affected (P<0.0001) by diet (Table 2). The piglets fed the C diet had the highest concentrations of all inflammatory markers. Animals fed the PPC 6% diet showed intermediate concentrations, and the PPC 8% group had the lowest concentrations (P<0.0001). There was the highest AFU signal for IL-12p40 compared with TNF-( in piglets fed all experimental diets, but particularly in those fed the C diet (Figure 1).
Table 2 Effect of potato protein concentrate (PPC) level of inclusion on pro inflammatory cytokines ileal concentration
| Cytokines (AFU) | Experimental diets | P-value | SEM | ||
|---|---|---|---|---|---|
| C | PPC 6% | PPC 8% | |||
| TNF-( | 6.26 a | 4.33 b | 3.55 c | *** | 0.0326 |
| IL-12p40 | 20.65 a | 12.54 b | 8.17 c | *** | 0.0058 |
AFU= arbitrary fluorescence units; PPC= potato protein concentrate; C= diet without antibiotics nor PPC; PPC 6%= basal diet with 6% PPC; PPC 8%= basal diet with 8% PPC; P= probability. SEM= standard error of mean
abc Row means with different superscripts differ significantly at P<0.05. *** P<0.0001.
Volatile fatty acid concentration
Fermentation profiles in the colon were affected by diet (Table 3). Piglets fed C diet had lower concentration of total VFA (P<0.0001) than piglets fed diets with PPC at 6 and 8%. Consequently, the inclusion of PPC in both levels in the diet increased SCFA and branched fatty acids (BFA) production in the colon compared to the C diet (P<0.0001). Animals fed the PPC 8% diet had higher concentration of SCFA (P<0.0001) than animals fed the PPC 6% or C diet. Acetic was the most abundant SCFA in the digesta of all experimental diets, followed by butyric and propionic. Higher concentrations of BCFA were observed in the digesta of animals fed the PPC 8% diet than in those fed the PPC 6% and C diets (P<0.0001).
Table 3 PPC level of inclusion effect on colonic volatile fatty acid production
| VFA (μmol/g) | Experimental diets | P-value | SEM | ||
|---|---|---|---|---|---|
| C | PPC 6% | PPC 8% | |||
| Acetic | 77.86 c | 95.38 b | 124.68 a | *** | 0.5415 |
| Propionic | 22.96 c | 31.88 b | 55.46 a | *** | 0.2110 |
| Butyric | 32.13 c | 36.45 b | 41.00 a | *** | 0.2433 |
| Isobutyric | 1.33 c | 1.66 b | 2.26 a | *** | 0.0187 |
| Valeric | 1.85 c | 2.41 b | 2.56 a | *** | 0.0106 |
| Isovaleric | 1.20 c | 1.56 b | 1.86 a | *** | 0.0125 |
| Isocaproic | 0.50 c | 0.71b | 0.90 a | *** | 0.0060 |
| BFA | 3.01 c | 3.91b | 5.02 a | *** | 0.0271 |
| SCFA | 134.81 c | 166.13 b | 223.70 a | *** | 0.7813 |
| Total | 137.83 c | 170.03 b | 228.70 a | *** | 0.7787 |
PPC= potato protein concentrate; C= basal diet without antibiotic nor PPC; PPC 6%= basal diet with 6% PPC= PPC 8%= basal diet with 8% PPC; BFA= branched fatty acids; SCFA= short chain fatty acids.
P= statistical significance; SEM= standard error of mean.
abc Row means with different superscripts differ significantly at P<0.05. ***P<0.0001.
Discussion
Previous work had suggested that the results from the immunological markers in the ileum confirmed that cytokines play a central role in cell immunity and participate in maintaining tissue integrity. Alterations in intestinal cytokines concentrations were expected for weaning piglets due to severe modifications in dietary elements and environmental stressful factors during this period lead to important morphophysiological adaptations in the gastrointestinal tract1,4,5. Microbial surface antigens in the intestinal lumen generate an early inflammatory response by modulating proinflammatory cytokines, which activate a signaling pathway, which leads to the release and nuclear translocation of nuclear factor-κB (NF-κB), serving as a transcription factor for the synthesis of IL-6 and TNF-18-20.
Interleukin 12 subunit p40 (IL-12p40) concentration arises when a chronic inflammation process takes place in the gastrointestinal tract21. The inflammation modulated by IL-12p40 is triggered through the Janus kinase/signal transducers and activators of transcription (JAK/STAT), in which it is dimerized and phosphorylated, translocating to the nucleus where IL-12p40 is transcribed22.
The cross-talk between intestinal epithelial cells and underlying lamina propria cells transfers immune-related signals to the local adaptive immunity, which subsequently help to maintain gut immune homeostasis23.
The disruption in the integrity of the intestinal mucosa generates an inflammatory process that activates NF-κB and subsequent production of proinflammatory cytokines6. The proinflammatory response is exacerbated when early weaned piglets are fed antibiotic-free diets24.
Dietary supplementation of potato protein concentrate helped to regulate the synthesis of inflammatory markers TNF-( and IL-12p40, which concentration decreased of 43.31 % and 60.45 % respectively in PPC 8% diet in compare with C diet. The downregulation of cytokines by PPC antimicrobial peptides was confirmed by the current results, since TNF-( and IL-12p40 concentration decreased in piglets fed the PPC 6% and PPC 8% diet. The antimicrobial effect on the colon bacterial population probably downregulated the signaling pathway that induces the release of NF-κB19, which was highly marked by the primary antibody used in the current study for immunofluorescence staining (Figure 1). LPS is a major component of Gram-negative bacterial outer membranes and can be recognized by host toll like receptor-4 (TLR4) and activates production of pro-inflammatory cytokines by immune cells via TLR4 signaling25. The anti-inflammatory functions of PPC are mainly due to LPS-neutralizing activity, which suppresses downstream TLR4 signaling pathways, such as mitogen-activated protein kinase [MAPK] and NF-κB signaling26. Furthermore, the PPC inclusion in the diet decreased IL-12p40 synthesis, and thus concentration, these attributed to the lack of stimulus generated by pathogen microorganisms and to the inhibition of the pathway mediated by JAK/STAT21,22. Therefore, dietary PPC supplementation regulates inflammatory response and indirectly protects the intestinal mucosa, decreasing the detrimental effects of weaning on the intestinal cells27,28.
Volatile fatty acids also have a key role in the regulation of inflammatory mechanisms that confer a protector or causal effect by the modulation of inflammatory cytokines release and the recruitment of immune cells through surface receptors or by the enzymatic activity inhibition29. SCFAs are defined as groups of volatile fatty acids comprising less than six carbons, mainly acetate, propionate, and butyrate, which are known to be the main fuel source for colonocytes and are essential for maintaining the normal metabolism of colon mucosa, including colonocyte growth and proliferation2. These acids make possible the regulation of the pH of the colonic content, inhibiting the growth and development of potentially pathogen bacteria such as E. coli, Clostridium and Salmonella. VFA stimulate bicarbonate luminal secretion, which has an important buffer effect upon the luminal pH regulation while induces the absorption of sodium and water at the mucosal level30.
The increase in SCFAs observed in piglets fed with PPC 6% and PPC 8%, compared to those produced by piglets fed a diet without antibiotic nor PPC, may probably be the outcome of intestinal environmental alterations favoring the establishment of beneficial bacteria and limiting the growth of pathogenic microbes31. In turn, acetic acid can be metabolized to butyric acid, which contributes between 70 % and 90 % of the energy necessary by colonocytes metabolism31. These changes may reduce the incidence and severity of postweaning diarrhea and improve piglet’s growth26. SCFA promote an eubiotic intestinal environment by maintaining an acidic pH, which suppresses the growth of potentially pathogenic bacteria that require an alkaline pH to survive32. Beneficial microbiota proliferates through competitive exclusion, occupying binding sites on the intestinal mucosa that could otherwise be employed by pathogenic bacteria33. Establishment of beneficial microorganisms modulates pathogenic bacteria development through bacteriocin secretion, which destroys the bacterial cell wall mimicking an antibiotics mechanism32,33. SCFAs could act as histone deacetylase (HDAC) inhibitors and therefore attenuate inflammatory responses by blocking nuclear factor-κB (NF-κB) signaling34. In addition, SCFA also have a direct effect on microbiota-derived aryl hydrocarbon receptor (AHR) ligands which enhance regulatory T cells’ (Tregs) immunosuppressive activities during inflammation, and indirectly influence neutrophil recruitment and activation23. Increased production of acetic, propionic and butyric acids are most likely related to the modulation of the intestinal microbial population through PPC antimicrobial activity.
The influence of dietary factors, through dietary interventions such as butyric acid and acid-producing bacteria, can increase the SCFA in the intestine, however, excessive SCFA in the hindgut could promote the development of metabolic syndrome via the gut microbiota-brain-β-cell axis34. An appropriate amount of SCFA in the intestine may be beneficial to early weaned piglet gut health. The content and ratio of VFA in the intestine are affected by the characteristics of proteins, like solubility and fermentability35. As for protein fermentation, proteolysis is the first step in the utilization of protein by bacteria, followed by deamination and decarboxylation of amino acids which limits their availability to the host and yields several putrefactive compounds including ammonia, amines, branched fatty acids, indoles, phenols and sulfur-containing compounds36. These compounds may have toxic effects on the piglets and can affect function and diversity of the gut microbiota37. Therefore, there may be a threshold for SCFA and BFA in the intestine, even though PPC 8% had the highest levels of total VFA, particularly SCFA, it was also the diet with the highest level of BFA, suggesting that PPC 6% may be the optimum level of inclusion, in order to prevent harmful effects upon gut health, maintaining the immunomodulatory and anti-inflammatory properties of PPC inclusion.
The current results support the hypothesis that potato protein concentrate can modulate the levels of intestinal cytokines and increase the SCFA production. Piglets fed diets supplemented PPC had the lowest levels of inflammatory markers that can be comparable with those of piglets fed diets with antibiotics15. The immunomodulatory effect of PPC at the intestinal level is probably due to the inhibition of signaling pathways like NF-κB and JAK/STAT complex suppressing the synthesis of proinflammatory cytokines38. An additional benefit observed with the inclusion of PPC to antibiotic-free diets was the control of chronic inflammation associated with the increase in IL-12p4039. The efficiency of PPC observed in the present study had a positive effect in controlling acute and chronic intestinal inflammation.
Conclusions and implications
Inclusion of potato protein concentrate in antibiotic-free diets can improve intestinal health in the post-weaning period. Furthermore PPC 6% and 8% helped to decrease the inflammatory response of the measured pro-inflammatory markers, mainly in the IL-12p40 concentration, as well as it probably helped establishing an optimum intestinal environment for beneficial microbial fermentation and SCFA production having a potential influence in the proliferation of intestinal bacteria. However, PPC 6% may be the optimum level of inclusion, preventing BFA harmful effects upon gut health, maintaining the immunomodulatory and anti-inflammatory properties of PPC inclusion. Therefore, the inclusion of potato protein concentrate can be used as an effective feed additive in antibiotic-free diets. Economic considerations should be considered for the selection of the optimal level of inclusion of PPC in the piglet diet by the producer. The broad-spectrum antibacterial activities of PPC have been indirectly demonstrated, making them promising alternatives to antibiotics. The immunomodulatory properties of PPC mean they likely have similar performance compared to antibiotics in the swine industry. Future research is needed to elucidate its precise effect upon anti-inflammatory cytokines concentration and microbial intestinal population diversity to fully understand its action mechanism.











 texto en
texto en