Introduction
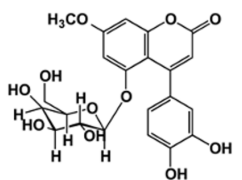
A metabolic syndrome with manifestations including alterations in the metabolism of macronutrients such as carbohydrates, proteins, and fats, diabetes mellitus is characterized by a state of hyperglycemia, namely increased blood glucose concentration, and glycosuria, namely the presence of glucose in the urine,10 both of which are attributed to deficiencies in insulin secretion and action. While there are three main types of diabetes mellitus-type 1 diabetes mellitus (insulin-dependent), type 2 diabetes mellitus (non-insulin-dependent), and gestational diabetes, there are also other specific types such as those produced or induced by genetic β cell and insulin action defects, endocrinopathies, drug use, and chemical agents. 11,12 From the outset, the treatment of this disease is highly complex, wherein it has to take into account any complications that may arise in individual cases, specifically considering the potential changes to eating habits and sedentary lifestyles and, of course, the appropriate pharmacological treatment.12 The pharmacological treatment options for type 2 diabetes include such drugs as insulin sensitizers (biguanides/metformin), insulin secretagogues (sulfonylureas/glibenclamide), incretin effect enhancers (GLP-1 analogues/albiglutide), and intestinal α-glucosidase inhibitors (acarbose and miglitol) . 13 With medicinal plants used since ancient times for the treatment of various diseases, type 2 diabetes mellitus has traditionally been treated with copalchi, 3,14 a herbal complex which Hintonia latiflora, a plant of the Rubiaceae family whose antihyperglycemic action is related to the secondary metabolites with antidiabetic properties, mainly 4-phenylcoumarins, found in its leaves. 14 One of the metabolites with hypoglycemic activity is 5-O-β-Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (4-FC) (Fig. 1), which consists of an amorphous solid of beige or yellow color and has a molecular weight of 462 g/mol. 15 The present study used high-performance liquid chromatography-ultraviolet (HPLC-UV) to develop and validate an analytical method for performing both the cytotoxicity (IC50) and permeability studies in the Madin-Darby canine kidney (MDCK) cells of 4-FC.
Materials and reagents
The 5-O-β-D-galactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (Fig. 1) used by the present study was provided by Dr. Rachel Mata Essayag from Laboratory 123 of Group E at the Faculty of Chemistry, Universidad Nacional Autónoma de México (UNAM or National Autonomous University of Mexico), while Lucifer yellow potassium salt (Sigma Aldrich), cisplatin (Sigma, P4394), ranitidine hydrochloride (Ran) and USP grade propranolol hydrochloride (Pro) were used as low and high permeability standards. Costar sterile 96-Well ELISA plates (12 12-mm-diameter inserts), collagen-coated polytetrafluoroethylene (PTFE) membrane, 0.4 μm pore size and 1.12 cm2 growth surface area, laminar flow hood, Class II, Type A2, Nuaire, Model UN-425-300, centrifuge Hettich, Model TYP 1706-01, Olympus light microscope, Model CKX41, Daigger ELISA reader, Model ELX800GDGR, EVOM2 epithelial voltmeter (STX2 electrode), BioTek Synergy HT Fluorometer and Agilent 1100 High Performance Liquid Chromatograph (HPLC) with: Detector Model G1315B, Model G1316A Thermostat, Model G1322A Degasser, Model G1311A Pump, and Model G1313A Injection System. Three types of chromatographic columns were used in the present study: a Symmetry C18 chromatographic column (4.6 x 150 mm, 5 μm); an Agilent Zorbax SB-C18 chromatographic column (4.6 x 150 mm, 3.5 μm); and an Agilent Zorbax Eclipse Plus SB-C18 chromatographic column (4.6 x 150 mm, 3.5 μm). J.T. Baker HPLC grade methanol, Milli-Q deionized water, J.T Baker monobasic sodium phosphate monohydrate, J.T. Baker, HPLC grade water, ATCC fetal bovine serum (FBS), ATCC dimethyl sulfoxide (DMSO), Costar 96-well ELISA sterile plates, Gibco DMEM culture medium, Gibco Hank's solution, J.T.Baker ethyl alcohol, daigger ELISA plate reader model ELX800GDGR, Aldrich Chemistry sulforhodamine, Gibco tris base solution, J.T.Baker trochloroacetic acid, J.T. Baker, trifluoroacetic acid (sigma aldrich), 0.45 µm Milipore type GVPL filters), hydrochloric acid (J.T. Baker), phosphoric acid (J.T Baker), and sodium hydroxide (Meyer).
Methodology
HPLC-UV validation of the analytical method for quantifying 4-phenylcoumarin in Hank's solution
Parameters assessed: linearity; repeatability; reproducibility; tolerance; sample stability; and absolute recovery
The validation of the method via HPLC-UV took into account guide criteria, such as those emitted by the U.S. Food and Drug Administration (FDA) in 2001 and the Official Mexican Standard NOM.177-SSA1-20138,9. The linearity, repeatability and reproducibility, tolerance, sample stability (ambient temperature, refrigeration, and freeze-thaw cycles), and absolute recovery were determined in Hank's solution for the apparent permeability coefficient (Papp) tests to be conducted on the MDCK cell line.
The corrected calibration curve was prepared, in triplicate and in a range of 0.5 to 40 µg/mL, in Hank's solution, at pH 7.4, from the 100 µg/mL phytopharmaceutical 5-O-β-Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin stock solution. The general acceptance criterion applied was an r2 > 0.98 coefficient of determination, while accuracy for quality control corresponded to a mean concentration within 15 % of the nominal values. The control samples were classified as low-quality control (LQC), mid-quality control (MQC), and high-quality control (HQC).
Concentrations prepared for conducting six-fold quality control-LQC (2 µg/mL), MQC (8 µg/mL), and HQC (25 µg/mL)-were used in Hank's solution and titrated under the same analytical conditions as those applied on Day 1 of the experiment to assess the precision (repeatability) of the analytical method proposed. To assess reproducibility, the same three quality control concentrations were closely monitored, via HPLC-UV, on two consecutive days under the same analytical conditions. The acceptance criteria applied on each quality control sample was ≤ 15 % relative standard deviation (RSD). The accuracy of the analytical method was reported as a percentage of the nominal value of the three quality control samples (LQC-2 µg/mL, MQC-8 µg/mL, and HQC-25 µg/mL). To ascertain sample stability, the LQC, MQC, and HQC control samples were subjected, in triplicate, to two freeze-thaw cycles at -20 °C, kept at ambient temperature for 24 hrs, and then refrigerated at 3 °C. Absolute recovery was determined using the formula (concentration of the analyte in the analytical matrix / analyte concentration in solution) *100. The tolerance of the analytical method was evaluated using the changes in the proportion of the mobile phase (FM: methanol: H2O acidified with ATF 0.1 % (55:45 and 45:55)).
Chromatographic conditions for the quantification of ranitidine hydrochloride, propranolol hydrochloride, and 4-phenylcoumarin in Hank's solution
The Pro was quantified from a stock solution of 100 µg/mL in Hank's solution, with the calibration curve prepared in a range of 0.5 to 20 µg/mL and the quality control (LQC, MQC, and HQC) samples set at 2, 9, and 19 µg/mL. An 8 µg/mL concentration (in triplicate) in Hank's solution was used for the permeability test on the MDCK cell line. The chromatographic conditions applied for the permeability assay were an Agilent Zorbax Eclipse Plus SB-C18 column, at 4.6x150mm and 3.5μm at room temperature, while the methanol:buffer phosphate mobile phase comprised 0.01M pH 7.6 (70:30) and was applied at a flow rate of 0.5 ml/min, a wavelength of 322 nm, an injection volume of 25 μL, a retention time of 3.25 min ± 0.5 min, and a run time of 4.5 min.
The Ra was quantified from a stock solution of 100 µg/mL in Hank's solution, with the calibration curve prepared in a range of 0.5 to 18 µg/mL and the quality control (LQC, MQC, HQC) samples set at 1, 9, and 17 µg/mL. An 8 µg/mL concentration (in triplicate) in Hank's solution was used for the permeability test on the MDCK cell line. The chromatographic conditions applied for the permeability assay were an Agilent Zorbax SB-C18 column, at 4.6x150 mm and 3.5μm at room temperature, while the methanol:buffer phosphate mobile phase comprised 0.01M pH 7.6 (60:40) and was applied at a flow rate of 1.0 mL/ min, a wavelength of 290 nm, an injection volume of 25 μL, a retention time of 2.45 min ± 0.5 min, and a run time of 3.5 min.
The 5-O-β-Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (4-FC) was quantified from a stock solution of 100 µg/mL in Hank's solution, with the calibration curve prepared in a range of 0.5 to 40 µg/mL and the quality control (LQC, MQC, HQC) samples set at 2, 8, and 25 µg/mL. Concentrations of 4, 8, and 16 µg/mL (in triplicate) were used, in Hank's solution at pH 7.4, to evaluate permeability in MDCK cells. The chromatographic conditions applied for the permeability assay were a Symmetry-C18 column, at 4.6x150mm and 5 μm at a temperature of 25 °C, while the methanol:water acidified with 0.1 % ATF (50:50) mobile phase was applied at a flow rate of 1.2 mL/min, a wavelength of 328 nm, an injection volume of 20 μL, a retention time of 2.0 min ± 0.5 min, and a run time of 5.0 min.
Cytotoxicity study on the MDCK cell line for 4-phenylcoomanine
Cytotoxicity was assessed by applying the sulforhodamine B (SRB) colorimetric method on ELISA-type plates. 16 Each plate was divided into quadrants to test, six-fold, concentrations of 100, 10, 1, and 0.1 µg/mL for 5-O-β-Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (4-phenylcoumarin), while cis-platinum was used as a positive control at concentrations of 10, 1, 0.1, and 0.01 µg/mL. Twenty-thousand MDCK cells were seeded per well, giving a total volume of 100 μl of supplemented DMEM medium, and then incubated at 37 °C, with 5 % CO2 and 95 % of Relative Humidity, for 24 hours, with the medium then removed and 90 μL of fresh medium was added. The treatment was conducted for 24 hours at 37 °C, in an atmosphere of 5 % CO2 and 95 % RH, at the end of which the medium was removed and the cells fixed with 100 μL 10 % TCA solution for one hour. The TCA was then discarded, the cells were washed with water, and the plates were left to dry. Once dry, the cells were stained with 50 μL 0.4 % SRB solution in acetic acid and the plate was read at a length of 564 nm in a Daigger ELISA reader. The viability or proliferation of cells was determined by the degree of staining as corresponding to the absorbance of the wells, which are directly proportional, wherein the higher the number of live cells, the greater the level of staining and, therefore, the greater the absorbance. 17,18
Apparent permeability coefficient test on the MDCK cell line for the phytopharmaceutical of interest
The Papp study was performed on MDCK cells following previously explained methodologies.19,20 The formation of the cell monolayer took place in transwell cells, with 500 μL of supplemented DMEM medium added to the apical zone (upper compartment) and 1500 μL of supplemented DMEM medium to the basolateral zone (lower compartment), which were then incubated at 37 °C, 5 % CO2, and 95 % RH for one hour. Then, 67,200 cells were incubated in the AP chamber at 37 °C, 5 % CO2, and 95 % RH, with the DMEM medium then changed every 24 hours until the cell monolayer had formed, which was observed through the optical microscope after the transwell cells had been monitored daily for approximately three days.
For the measurement of transepithelial electrical resistance (TEER), the monolayer was observed under a microscope with a 10x objective, which required the DMEM culture medium to be immediately removed by decantation and replaced with 2 mL of sterile Hank's solution (pH 7.45) at a temperature of 37 °C. The TEER level (Ω•cm2) was measured in triplicate using an EVOM2 epithelial voltmeter over a range of 180 to 250 Ω•cm2 until the signal was stable, given that a stable TEER is a sign of monolayer formation. Once the readings had been taken, the Hank's solution was replaced with supplemented DMEM medium and, finally, incubated to continue with the permeability study.
The permeability of the apical-basolateral (AP-BS) and basolateral-apical (BS-AP) transport in both directions was measured by removing the DMEM culture medium and then adding, in triplicate, 500 μL to the apical zone (upper chamber) of the working solutions, corresponding to 4, 8, and 16 µg/mL for the phytopharmaceutical of interest and 8 µg/mL for the high and low permeability standards (Pro and Ran). Next, 1500 μL of Hanks' solution (pH 7.45) was added to the basolateral chamber (lower chamber) at a temperature of 37 °C, with the plate then subjected at 80 rpm and 37 °C. Samples of 200 μL were then taken from the basolateral chamber at 15, 30, 45, 60, 75, 90, and 120 minutes, with the medium replaced after each sample collection, to maintain osmolality.
Finally, the Papp was calculated using the following equation:
where Papp is measured in cm/s (dQ/dt), the transport velocity of the (phytopharmaceutical) analyte of interest is measured in µg/s, Co is the initial concentration in the donor chamber and measured in µg/mL, and A is the surface area of the PTFE membrane expressed in cm2.
Results and discussion
Validation, by HPLC-UV, of the analytical method to quantify 5-O-β- Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (4-FC) in Hank's solution
Both the analytical method for quantifying the phytopharmaceutical of interest in Hank's solution and the implementation of the permeability study was carried out following the validation criteria set out in the FDA bioanalytical method (2001),8 titled Guidance for Industry: Bioanalytical Method Validation (2001), and NOM-177-SSA1-2013.9
As shown in Fig. 2, no interferences were observed on the retention time for the analyte of interest. Notably, the chromatograms corresponding to the high and low permeability standards (Pro and Ran), as shown in Fig. 3, were analyzed under different chromatographic conditions to those applied for 4-FC. Although different methodologies were applied, no interferences were observed on the retention times for either Ran or Pro, both of which were kept in Hank's solution.
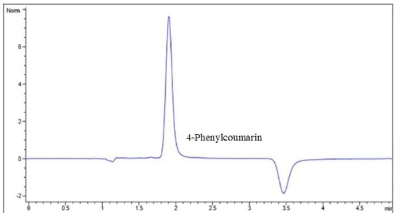
Fig. 2 Shows the chromatogram that demonstrates the selectivity of the analytical method used for 4-FC in Hank's solution (Tr=1.8 min).
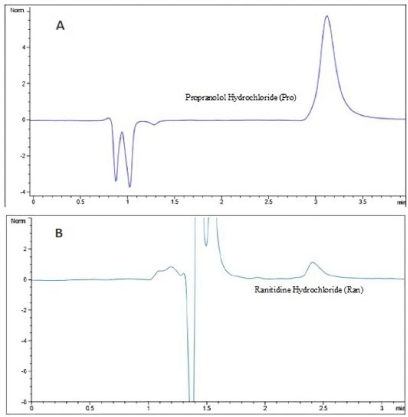
Fig. 3 Chromatographic signals of the high and low permeability standards. (A) Propranolol, Tr=3.3 min (B) Ranitidine, Tr=2.4 min. Assay conducted in the AP-BS direction, concentration 8 μg/mL, in Hank's solution.
The main results obtained for the validation of the analytical method, as shown in Table 1, are also presented below for the quantification of 4-FC in Hank's solution.
Table 1 Summary of the validation process for the 5-O-β- Dgalactopyranosyl-7-methoxy-3',4'-dihydroxy-4-phenylcoumarin (4-FC).
| Linearity of the system | Acceptance criteria | Result | ||
|---|---|---|---|---|
| r2 should be ≥0.99 | r2=0.9998 | |||
| Linearity of the method | r2= ≥ 0.99 (each level should be ≤ 15 RSD (%) and no more than 20 RSD (%)) | r2=0.9993. the RSDs obtained were ≤ 15 % for each level of the calibration curve | ||
| LQC | MQC | HQC | ||
| Intra-day precision (RSD%) | RSD ≤15 % | 3.24 | 2.88 | 0.96 |
| Inter-day precision (RSD%) | RSD ≤15 % | 5.99 | 0.52 | 1.02 |
| Accuracy (%) | Mean concentration must be within 15 % of the nominal values | 5.81 | 3.24 | 3.02 |
| Absolute recovery (%) | Recovery does not necessarily need to be 100%, but should be reproducible at each concentration level | 93.07 | 96.88 | 98.26 |
| Stability | ||||
| Room temperature (24 h) | RSD ≤15 % | 2.7 | 4.37 | 2.97 |
| Cooling temperature (3 °C) | 1.78 | 0.66 | 3.00 | |
| Freeze-thaw cycles (-20 °C) | 7.43 | 3.94 | 3.01 | |
| Tolerance | ||||
| MeoH:water with ATF 0.1% (55:45) | RSD ≤15 % | 6.52 | ||
| MeoH:water with ATF 0.1% (45:55) | 6.01 | |||
| Limit of quantification (RSD%) | RSD ≤20 % | The concentration of the limit of quantification (LOQ)=1.12µg/mL,RSD=0.83 | ||
Accuracy is reported as a percentage of the nominal value (%). RSD (relative standard deviation); LQC (low-quality control, 2 µg/mL); MQC (mid-quality control, 8 µg/mL); and HQC (high-quality control, 25 µg/mL).
As can be seen in Table 1, the HPLC-UV-validated analytical method for quantifying 4- FC meets all the criteria in order to be considered as a reproducible and reliable method. Therefore, the method can be used to perform the Papp assay on the MDCK cell line.
Cytotoxicity study for 4-phenylcoumarin in MDCK cells
It was determined that, using the SRB colorimetric method, the cytotoxic effect is obtained for 4-phenylcoumarin in the MDCK cell line after 24 hours of treatment. Fig. 4 shows the cytotoxicity results obtained at different 4-FC concentrations together with the positive control in the MCDK cell line, namely cis-platinum.
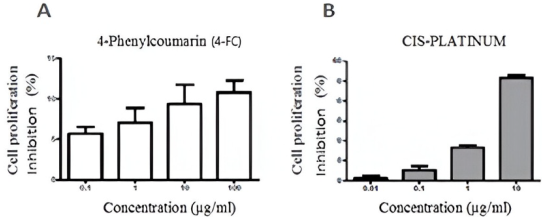
Fig. 4 Cell growth inhibition. Viability of MDCK cells due to the effect of 4-FC (A) and cis-platinum, positive control (B), n=6.
As can be seen in Fig. 4, the cytotoxic effect of the phytopharmaceutical of interest became apparent when it was barely able to exceed a 10 % inhibition of cell proliferation at a concentration of 100 μg/mL. In contrast, its positive control inhibited proliferation by slightly higher than 50 %, at a concentration that was ten times lower. The results obtained show that 4 -phenylcoumarin is a phytopharmaceutical with a wide concentration range, 0.1 to 100 μg/mL, within which it presents very low cytotoxicity; therefore, the concentrations used in the permeability study (4, 8, and 16 μg/mL) do not represent an imminent risk to the integrity of the MDCK cell monolayer.
Papp test of 4-phenylcoumarin in the MDCK cell line
The permeability test on the MDCK cell line, to which the phytopharmaceutical of interest also corresponds, was conducted in order to ascertain the permeation rate and the mechanism via which it is absorbed through the cell membrane. Table 2 shows the average transport velocities (dQ/dt) obtained for 4FC and the permeability standards (n=3) for the different concentrations applied in both directions, which correspond to the slope obtained from plotting the cumulative amount of each compound as a function of sampling time.
The data presented in Table 2 shows that the transport velocity, in both directions, of the three molecules of interest (4-FC, Pro, and Ran) was directly proportional to the concentration of the solutions, wherein the higher the concentration, the higher the velocity of transport. Furthermore, the values obtained reveal that, when 4-FC passes through the MDCK cell monolayer in the absorption direction (AP-BS), it did so at a rate similar to the high permeability standard.
The results shown in Table 3 indicate that the passage of 4-FC through the monolayer was not faster than the high permeability standard (Pro), indicating that it presents a moderate permeation rate. The Papps obtained for both the AP-BS and BS-AP directions for 4-FC and Ran did not show significant variation, indicating that both compounds were passively diffused through the MDCK cell monolayer. The average Papp obtained for Pro was slightly higher in the AP- BS direction, indicating that the input of the compound was greater than the output, thereby ruling out the involvement of efflux transporters. 21,22 The Papp ratio observed elucidates the type of transport mechanism to which the molecule of interest may be subject. Compounds transported by solely passive diffusion present similar permeability values in both directions (AP-BS and BS-AP), directional permeability ratio values that should be around one.23
Table 3 Average Papp values in the AP-BS and BS-AP directions (n=3), both moving through the MDCK cell monolayers and corresponding to the ratio of permeability coefficients.
| Propranolol hydrochloride (Pro) | 4-Phenylcoumarin (4-FC) | Ranitidine hydrochloride (Ran) | ||||
|---|---|---|---|---|---|---|
| Concentration (µg/mL) | 8.00 | 4.00 | 8.00 | 16.00 | 8.00 | |
| Papp AP-BS (X 10-5 cm/s) | 20.80 | 18.05 | 13.05 | 9.52 | 2.56 | |
| SD (X10-5) | ±3.56 | ±1.35 | ±0.87 | ±1.96 | ±0.10 | |
| Papp BS-AP (X 10-5 cm/s) | 13.90 | 10.88 | 11.73 | 8.22 | 1.56 | |
| SD (X10-5) | ±1.38 | ±1.60 | ±1.05 | ±0.32 | ±0.67 | |
| Relationship Papp | (AP-BS)/(BS-AP) | 1.50 | 1.70 | 1.10 | 1.20 | 1.60 |
| (BS-AP)/(AP-Bs) | 0.70 | 0.60 | 0.90 | 0.90 | 0.60 | |
Papp= Apparent permeability coefficient, SD= Standard deviation, (AP-BS)= Apical-basolateral direction, (BS-AP)=Basolateral-apical direction
While, for drugs that have affinity for cellular efflux pumps and are actively transported out of cells, the Papp ratio must be greater than or equal to two, as the permeability observed in the BS-AP direction was higher than that observed for the AP-BS direction. As this condition was not met for all the analytes of interest (both the controls and the 4-FC itself), it can be concluded that they passed through the membranes by simple diffusion. Therefore, it can be clearly observed that 4-FC, at concentrations of 8 and 16 μg/mL, presents a ratio no greater than two, as it has been reported that drugs with affinity for efflux transporters present a ratio equal to or greater than two.24
Fig. 5 and Table 3 show that the transport velocity, in most cases, was slightly higher in the AP-BS direction, which indicates the lack of efflux mechanism transporters. At concentrations of 4, 8, and 16 μg/mL, 4-FC presented higher permeability values than those found for Ran and values lower than those found for Pro, with a markedly different value found in the AP-BS direction, which suggests a moderate permeability for the phytopharmaceutical of interest. According to the results obtained by the present study indicate that 4FC was transported by apparent passive diffusion through the MDCK cells, as transport in the apical chamber was not observed to be saturated.
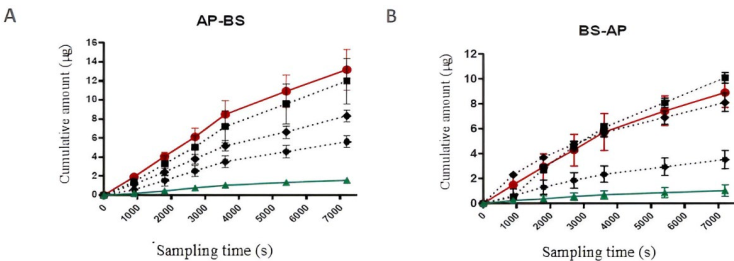
Fig. 5 Permeation rate. The average accumulated amount of the phytopharmaceutical of interest and permeation standards (n=3) in the receptor chamber, as a function of time (seconds). Ingress of solutions of 4 μg/mL (••ӿ••), 8 μg/mL (•••••), and 16 μg/mL of 4-phenylcoumarin (••■••) and of 8 μg/mL propranolol hydrochloride (―●―) and ranitidine hydrochloride (―▲―) into the receiving chamber of the measurement system.
Conclusions
The present study fully validated the analytical HPLC-UV methodology used to quantify 4-FC, including the parameters of linearity, repeatability, reproducibility, tolerance, sample stability, and absolute recovery. Moreover, the method complied with public health criteria under the legislation NOM.177-SSA1-2013. The cytotoxicity results found show that 4-FC has a low cytotoxic effect, in a range of between 0.1 and 100 µg/mL, revealing that the doses utilized in the permeability study were found not to be toxic in MDCK cells, a finding confirmed by the viability and integrity of the cell monolayer observed.
The apparent permeability coefficient (Papp) of 4-FC observed for the MDCK cell line showed a moderate permeation rate at concentrations of 4, 8, and 16 µg/mL, when compared to the high and low permeability standards (Pro and Ran, 8 µg/mL). 25,26 The apparent permeability coefficients (Papp* AP-BS cm/s) obtained for the Pro and Ran standards for a 8 µg/mL dose were as follows: Pro-20,803 ± 3,567 x 10-5 cm/s (AP-BS direction) and 13,903 ± 1,383 x 10-5 cm/s (BS-AP direction); and Ran-2,560 ± 0.101 x 10-5 cm/s (AP-BS direction) and 1,565 ± 0.674 x 10-5 cm/s (BS-AP direction). It appeared that the 4-FC crossed the membrane monolayer via a simple diffusion mechanism, as the permeability values obtained for the 4, 8, and 16 µg/mL doses-respectively, 18.051 ± 1.355, 13.058 ± 0.872, 9.524 ± 1.966 x 10-5 cm/s (AP-BS direction) and 10.885 ± 1.604, 11.730 ± 1.054, 8.222 ± 0.327 x 10-5 cm/s (BS-AP direction)-were similar in both directions (AP-BS and BS-AP). 27,28 The results obtained revealed that the Papp for both the standards and 4-FC presented higher speeds in the AP-BS direction, showing that the input of the compounds was greater than their output, therefore ruling out the involvement of efflux transporters and confirming the involvement of mainly passive transport mechanisms.
The results found by the present study represent an important contribution to understanding the biopharmaceutical characteristics of 4-FC from a preclinical perspective. Further experiments may find these observations useful for the design of clinical studies on its use as an auxiliary herbal option in the treatment of diabetes mellitus.











 nueva página del texto (beta)
nueva página del texto (beta)