Introduction
Lamiaceae is a family of flowering plants with about 236 genera and over 7173 species 1. Plants of this family are mostly aromatic herbs or small shrubs with young stems, often four-angled. Many lamiaceous herbs are used as spices and for medicinal purposes, such as balm (Melissa officinalis L.), basil (Ocimum basilicum L.), lavender (Lavandula angustifolia Mill.), marjoram (Origanum majorana L.), mint (Mentha spp.), oregano (Origanum vulgare L.), perilla ( Perilla frutescens (L.) Britton), rosemary (Rosmarinus officinalis L.), sage ( Salvia officinalis L.), savory (Satureja spp.), and thyme (Thymus vulgaris L.). Some dried herbs or spices in Europe and Costa Rica are used in commercial blends named Herbes de Provence and Italian Seasoning2.
Salvia (tribe Mentheae) is a cosmopolitan genus of about 900 to 1000 species distributed worldwide and predominantly occurring in the American continent (Central and South America, ca. 500 species), the Mediterranean basin, and Central Asia (ca. 250 species) and Eastern Asia (ca. 90 species) 3. The characteristics of this genus are the flowers with only two stamens and the secreting glandular trichomes that accumulate complex essential oils composed mainly of terpenoids, which are stored in the subcuticular space within these anatomical structures 4-6. The genus Salvia has been the subject of numerous chemical studies. It is a rich source of terpenoids, steroids, and polyphenolics 7-9.
Salvia is the only genus of Lamiaceae in Costa Rica (apart from the cultivated Rosmarinus officinalis, native to the Mediterranean region) whose flowers have a bilabial calyx and only two stamens. In Costa Rica, the genus Prunella is the only one that shares these morphological traits. Salvia polystachia Cav. is a perennial herb or subshrub, up to 2 m high, with a distributional range from Mexico to Honduras, Costa Rica, and Panama 10. In Costa Rica, it is distributed in several habitats from 850 to ca. 2500 m of altitude, in tropical rainforests, tropical montane forests, oak forests, and disturbed areas where the plant is commonly known as “chan,” “chirrite” and “jalacate”. This species is recognized by its petiolate and ovate-lanceolate leaves up to 13 cm long and 7 cm wide with rounded or slightly cordate base, acuminate apex, and characteristic densely spiciform inflorescences with blue flowers 11. Salvia polystachia belongs to subgenus Calosphace, containing ca. 500 species endemic to North, Central, and South America 12 and represents a well-defined natural group, strongly supported as monophyletic 3.
In Mexico, S. polystachia is popularly known as “chía” with S . hispanica L., S. columbria Benth., and another Salvia spp. Since pre- Hispanic times, the rural population has used the “chía” nutlets as food 13,14. Salvia polystachia is used in traditional medicine as a purgative drug to treat dysentery and to stop diarrhea 11.
Phytochemical studies of S. polystachia from Mexico have been directed primarily to the extraction, isolation, and structural elucidation of neo-clerodane diterpenes (polystachynes) by spectroscopic methods 15-17. Calzada and collaborators 18,19 studied the antiamoebic and antigiardial activities of crude acetone extract of S. polystachia and of four of its diterpenoid constituents, resulting in linearolactone (=linearifoline) was the most active clerodane against both Entamoeba histolytica and Giardia lamblia trophozoites.
Several exotic species of sage are occasionally cultivated as ornamentals in Costa Rica, such as Salvia leucantha Cav. (“bandera”), and S. microphylla natives of Mexico. Salvia microphylla Kunth is an introduced subshrubby species with bright red flowers grown in the gardens of the University of Costa Rica campus. This species is native to Mexico, where it is commonly known as “mirto or toronjil” and it is also part of the subgenus Calosphace, used traditionally to relieve gastrointestinal troubles (stomachache, diarrhea, dysentery, cramps, and colic) and insomnia; and is used externally to treat rheumatism and skin problems 12. Phytochemical studies of S. microphylla revealed the presence of sesquiterpenoids, diterpenoids, triterpenoids, and phenolic compounds 20-23.
In the literature, there are many studies on the chemical composition of essential oils of diverse species of Salvia growing in different countries 24-34. The composition of the essential oils of S. microphylla cultivated in Italy (Giardini Botanici Hanbury, Capo Mortola, Ventimiglia) 35, Brazil (the Garden of Medicinal Plants of the Federal University, Lavras, Minas Gerais) 36, and Algeria (Blida, southwest of Algiers) 37 have been reported. Recently, two reports on the volatile chemical composition of S. microphylla cv. Hot Lips (with striking bicolor flowers with half- white and half-red petals that resemble crimson-red lips) cultivated in Italy (Sanremo) 38 and in the USA (Grover Beach, California) 39 were published.
To the best of our knowledge, there are no reports in the literature about the chemical composition of the essential oils from S. polystachia. The present paper is intended to contribute to the chemical knowledge of the essential oil from aerial parts of this species growing wild in Costa Rica. In this study, we also determine the chemical composition of the essential oils of S. microphylla, an introduced species cultivated in a garden in Costa Rica.
Experimental
Plant material
Aerial parts of a representative sample of a population of Salvia polystachia were collected in May 2015 in El Tirol, Province of Heredia (10°04’11’’N, 84°05’07” W), at an elevation of 1850 m. A voucher specimen was deposited in the Herbarium of the University of Costa Rica (USJ 105600) (See (A), Fig. 1).

Fig. 1 (A) Salvia polystachia and, (B) S. microphylla blooming in Costa Rica. (Photografies by J. J. Araya and C. Chaverri).
Aerial parts of Salvia microphylla were collected in August 2016 in the University of Costa Rica Campus, locality of Mercedes de Montes de Oca, Province of San José (9°56’16’’N, 84°02’57” W) at an elevation of 1205 m. A voucher specimen was deposited in the Herbarium of the University of Costa Rica (USJ 111225). (See (B), Fig. 1).
Essential oil extraction
Fresh aerial parts of Salvia polystachia (520 g) and S. microphylla (240 g) were submitted to hydrodistillation at atmospheric pressure using an all-glass Clevenger-type apparatus for 3 h. The distilled essential oils were collected and dried over sodium sulfate (Na2SO4, Merck KGaA, anhydrous GR for analysis), filtered, and stored at 0-10 °C in the dark for further analyses.
Gas chromatographic analysis (GC-FID)
The essential oils of S. polystachia and S. microphylla were analyzed by capillary gas chromatography with a flame ionization detector (GC-FID) using a Shimadzu GC-2014 gas chromatograph. The data were obtained on a 5 % diphenyl-95 %-dimethylpolysiloxane fused silica capillary column (30 m x 0.25 mm; film thickness 0.25 μm; MDN-5S, Supelco). The GC integrations were performed with a LabSolutions, Shimadzu GCsolution™ Chromatography Data System software, version 2.3. Operating conditions used were carrier gas N2, flow 1.0 mL/min; oven temperature program: 60 to 280 °C at 3 °C/min, 280 °C (2 min); sample injection port temperature 250 °C; detector temperature 280 °C; the split ratio was adjusted to 1:60.
Gas chromatography-Quadrupole mass spectrometry analysis (GC-MS)
GC-MS analyses were conducted with a Shimadzu GC-17A gas chromatograph coupled with a GCMS-QP5000 apparatus and GCMSsolution™ software (version 1.21), with Wiley 139 and NIST computerized databases. The data were obtained with the same column described above. Operating conditions were carrier gas He, flow 1.0 mL/min; oven temperature program: 60-280 °C at 3 °C/min; sample injection port temperature 250 °C; transfer line temperature 260 °C; ionization voltage: 70 eV; ionization current 60 μA; scanning speed 0.5 s over m/z 38 to 400 Da range; the split ratio was adjusted to 1:70.
Compound identification
Identification of the constituents of the oils was performed using the retention indices, which were calculated employing a homologous series of n-alkanes on a 5 % diphenyl-95 % dimethylpolysiloxane type column 40, and by comparison of their mass spectra with those published in the literature 41 or those of our own homemade MS spectra library or comparing their mass spectra with those available in the NIST 107 and Wiley 139 computerized databases or in a web source 42. To obtain the retention indices for each peak, 0.1 μL of the n-alkane mixture (Sigma, C8-C32 standard mixture) was co-injected under the same experimental conditions reported above. Integration of the total chromatogram (GC-FID), expressed as area percent, without correction factors, has been used to obtain quantitative compositional data.
Results and discussion
The hydrodistillation of fresh aerial parts of S. polystachia from Costa Rica produced a colorless oil yielding 0.02 % (v/w). The essential oil contained 123 identified compounds, accounting for 91.9 % of the total composition (see the Sp column, Table 1). The essential oil was found to be a complex mixture consisting largely of sesquiterpene hydrocarbons (64.0 %) and oxygenated sesquiterpenes (20.3 %) with minor quantities of monoterpenoids, aliphatic compounds, and diterpenoids. The major constituents were germacrene D (25.8 %), (E) -caryophyllene (15.9 %), bicyclogermacrene (7.2 %), caryophyllene oxide (5.7 %), palustrol (3.2 %) and β-elemene (2.8 %) (see the Total Ion Chromatogram -TIC- in Fig. 2).
Table 1 Chemical composition of aerial parts essential oil of Salvia polystachia (Sp) and S. microphylla (Sm) from Costa Rica.
| No. | aCompound | RIb | RILit.c | Class | Sp (%) | Sm (%) | IMd |
|---|---|---|---|---|---|---|---|
| 1 | (Z)-Hex-3-en-1-ol | 850 | 850 | A | 0.3 | 1,2 | |
| 2 | (E)-Hex-2-en-1-ol | 854 | 854 | A | tr | 1,2 | |
| 3 | Hexan-1-ol | 863 | 863 | A | tr | 1,2 | |
| 4 | α-Thujene | 925 | 924 | M | tr | 0.1 | 1,2 |
| 5 | α-Pinene | 932 | 932 | M | 0.1 | 0.5 | 1,2,3 |
| 6 | Camphene | 946 | 949 | M | 0.5 | 1,2 | |
| 7 | Benzaldehyde | 956 | 952 | A | tr | 1,2 | |
| 8 | Sabinene | 972 | 969 | M | 0.7 | 0.2 | 1,2 |
| 9 | β-Pinene | 977 | 974 | M | 0.2 | 3.2 | 1,2,3 |
| 10 | Oct-1-en-3-ol | 978 | 974 | A | tr | 1,2 | |
| 11 | Octan-3-one | 988 | 979 | A | tr | tr | 1,2 |
| 12 | Myrcene | 989 | 988 | M | tr | 0.2 | 1,2 |
| 13 | Octan-3-ol | 996 | 988 | A | tr | tr | 1,2 |
| 14 | Octanal | 998 | 998 | A | tr | 1,2 | |
| 15 | α-Phellandrene | 1001 | 1002 | M | tr | 1,2 | |
| 16 | (Z)-Hex-3-enyl acetate | 1004 | 1004 | A | tr | 1,2 | |
| 17 | δ-3-Carene | 1009 | 1008 | M | 0.1 | tr | 1,2 |
| 18 | α-Terpinene | 1016 | 1014 | M | tr | 0.1 | 1,2 |
| 19 | p-Cymene | 1023 | 1020 | M | tr | 0.8 | 1,2 |
| 20 | Limonene | 1027 | 1024 | M | tr | 0.1 | 1,2,3 |
| 21 | β-Phellandrene | 1030 | 1025 | M | tr | 0.1 | 1,2 |
| 22 | 1,8-Cineole | 1032 | 1026 | OM | tr | 1.5 | 1,2,3 |
| 23 | (Z)-β-Ocimene | 1033 | 1032 | M | tr | 1,2 | |
| 24 | (E)-β-Ocimene | 1044 | 1044 | M | tr | 1,2 | |
| 25 | γ-Terpinene | 1055 | 1054 | M | 0.1 | 3.2 | 1,2 |
| 26 | cis-Sabinene hydrate | 1067 | 1065 | OM | 0.2 | tr | 1,2 |
| 27 | p-Mentha-2,4(8)-diene | 1081 | 1085 | M | tr | 1,2 | |
| 28 | Terpinolene | 1082 | 1086 | M | tr | 0.1 | 1,2 |
| 29 | Linalool | 1093 | 1095 | OM | 0.3 | 1,2,3 | |
| 30 | trans-Sabinene hydrate | 1098 | 1098 | OM | 0.2 | tr | 1,2 |
| 31 | Nonanal | 1100 | 1100 | A | tr | tr | 1,2 |
| 32 | trans-Thujone | 1111 | 1112 | OM | tr | 1,2 | |
| 33 | trans-Pinene hydrate | 1115 | 1119 | OM | tr | 1,2 | |
| 34 | trans-Pinocarveol | 1140 | 1135 | OM | 0.1 | 1,2 | |
| 35 | cis-p-Menth-2-en-1-ol | 1122 | 1118 | OM | tr | 1,2 | |
| 36 | Undec-1-yne | 1127 | 1122 | A | tr | 1,2 | |
| 37 | cis-p-Mentha-2,8-dien-1-ol | 1132 | 1133 | OM | tr | 1,2 | |
| 38 | trans-p-Menth-2-en-1-ol | 1144 | 1136 | OM | tr | 1,2 | |
| 39 | Camphor | 1147 | 1141 | OM | tr | tr | 1,2,3 |
| 40 | (2E,6Z)-Nona-2,6-dienal | 1151 | 1150 | A | tr | ||
| 41 | Camphene hydrate | 1154 | 1145 | OM | tr | 1,2 | |
| 42 | (Z)-Isocitral | 1158 | 1160 | OM | 0.1 | 1,2 | |
| 43 | Pinocarvone | 1162 | 1160 | OM | tr | 1,2 | |
| 44 | Borneol | 1166 | 1165 | OM | tr | 2.2 | 1,2,3 |
| 45 | Terpinen-4-ol | 1175 | 1174 | OM | 0.1 | 0.3 | 1,2,3 |
| 46 | Thuj-3-en-10-al | 1181 | 1181 | OM | tr | 1,2 | |
| 47 | Dill ether | 1184 | 1184 | OM | tr | 1,2 | |
| 48 | α-Terpineol | 1189 | 1186 | OM | 0.2 | 0.1 | 1,2 |
| 49 | Myrtenol | 1192 | 1194 | OM | tr | 1,2 | |
| 50 | Decanal | 1201 | 1201 | A | tr | 1,2,3 | |
| 51 | trans-Piperitol | 1208 | 1207 | OM | tr | 1,2 | |
| 52 | trans-Pulegol | 1213 | 1213 | OM | tr | 1,2 | |
| 53 | β-Cyclocitral | 1218 | 1217 | OM | tr | 1,2 | |
| 54 | Cumin aldehyde | 1248 | 1238 | OM | tr | tr | 1,2 |
| 55 | Pinocamphone | 1247 | 1246 | OM | tr | 1,2 | |
| 56 | 1-Octen-3-ol butanoate | 1278 | 1280 | A | 0.1 | 1,2 | |
| 57 | Isobornyl acetate | 1283 | 1283 | OM | tr | 1,2 | |
| 58 | Bornyl acetate | 1291 | 1287 | OM | tr | 1,2 | |
| 59 | α-Terpinen-7-al | 1287 | 1285 | OM | tr | 1,2 | |
| 60 | Dihydroedulan I | 1294 | 1292 | OM | 0.2 | 1,2 | |
| 61 | (2E,4E)-Deca-2,4-dienal | 1318 | 1315 | A | tr | 1,2 | |
| 62 | Bicycloelemene | 1331 | 1330e | S | 0.1 | 1,2 | |
| 63 | δ-Elemene | 1334 | 1335 | S | 0.2 | 1,2 | |
| 64 | α-Cubebene | 1345 | 1345 | S | 0.1 | 1,2 | |
| 65 | α-Ylangene | 1367 | 1373 | S | 0.1 | 0.1 | 1,2 |
| 66 | α-Copaene | 1373 | 1374 | S | 1.6 | 1.3 | 1,2 |
| 67 | β-Bourbonene | 1381 | 1387 | S | 1.5 | 1.2 | 1,2 |
| 68 | β-Elemene | 1387 | 1389 | S | 2.8 | 0.3 | 1,2 |
| 69 | Cyperene | 1397 | 1398 | S | tr | 1,2 | |
| 70 | (Z)-Caryophyllene | 1400 | 1408 | S | 0.1 | 1,2 | |
| 71 | α-Gurjunene | 1405 | 1409 | S | 1.6 | 1,2 | |
| 72 | (E)-Caryophyllene | 1419 | 1417 | S | 15.9 | 34.5 | 1,2,3 |
| 73 | β-Copaene | 1428 | 1430 | S | 0.4 | 1,2 | |
| 74 | β-Gurjunene | 1429 | 1431 | S | 3.2 | 1,2 | |
| 75 | trans-α-Bergamotene | 1430 | 1432 | S | tr | 1,2 | |
| 76 | α-Guaine | 1432 | 1437 | S | 0.3 | 1,2 | |
| 77 | γ-Elemene | 1437 | 1439 | S | 0.6 | 1,2 | |
| 78 | Aromadendrene | 1436 | 1439 | S | tr | tr | 1,2 |
| 79 | 6,9-Guaiadiene | 1439 | 1442 | S | tr | 1,2 | |
| 80 | cis-Muurola-3,5-diene | 1442 | 1448 | S | 0.1 | 1,2 | |
| 81 | α-Humulene | 1452 | 1452 | S | 1.2 | 1.2 | 1,2,3 |
| 82 | Dehydroaromadendrene | 1457 | 1460 | S | 0.1 | 1,2 | |
| 83 | cis-Cadina-1(6),4-diene | 1460 | 1461 | S | tr | 1,2 | |
| 84 | cis-Muurola-4(14),5-diene | 1463 | 1465 | S | tr | 1,2 | |
| 85 | 9-epi-(E)-Caryophyllene | 1465 | 1464 | S | 0.4 | 1,2 | |
| 86 | 4,5-di-epi-Aristolochene | 1466 | 1471 | S | 0.5 | 1,2 | |
| 87 | γ-Gurjunene | 1473 | 1475 | S | tr | 1,2 | |
| 88 | β-Chamigrene | 1473 | 1476 | S | 0.5 | 1,2 | |
| 89 | Germacrene D | 1481 | 1484 | S | 25.8 | 8.4 | 1,2 |
| 90 | β-Selinene | 1489 | 1489 | S | tr | 1,2 | |
| 91 | trans-Muurola-4(14),5-diene | 1491 | 1493 | S | 6.5 | 1,2 | |
| 92 | γ-Amorphene | 1492 | 1495 | S | 0.5 | 1,2 | |
| 93 | Bicyclogermacrene | 1495 | 1500 | S | 7.2 | 1,2 | |
| 94 | α-Muurolene | 1500 | 1500 | S | 0.3 | 0.5 | 1,2 |
| 95 | Germacrene A | 1506 | 1508 | S | 0.7 | 1,2 | |
| 96 | γ-Cadinene | 1511 | 1513 | S | 0.7 | 1,2 | |
| 97 | (Z)-β-Bisabolene | 1513 | 1514 | S | 0.7 | 1,2 | |
| 98 | Cubebol | 1514 | 1514 | OS | 0.3 | 1,2 | |
| 99 | δ-Cadinene | 1516 | 1522 | S | 1.6 | 0.9 | 1,2,3 |
| 100 | trans-Cadina-1,4-diene | 1528 | 1533 | S | 0.2 | 1,2 | |
| 101 | 10-epi-Cubebol | 1532 | 1533 | OS | 0.2 | 1,2 | |
| 102 | α-Cadinene | 1534 | 1537 | S | 0.1 | 1,2 | |
| 103 | Germacrene B | 1560 | 1559 | S | 4.2 | 1,2 | |
| 104 | (E)-Nerolidol | 1561 | 1561 | OS | 0.3 | 1,2,3 | |
| 105 | Palustrol | 1569 | 1567 | OS | 3.2 | 1,2 | |
| 106 | Dendrolasin | 1570 | 1570 | Misc | tr | 1,2 | |
| 107 | Spathulenol | 1578 | 1577 | OS | 1.6 | 1,2 | |
| 108 | Caryophyllene oxide | 1583 | 1582 | OS | 5.7 | 4.7 | 1,2 |
| 109 | Globulol | 1590 | 1590 | OS | tr | 1,2 | |
| 110 | Viridiflorol | 1592 | 1592 | OS | 0.5 | 1,2 | |
| 111 | Salvial-4(14)-en-1-one | 1597 | 1594 | OS | 0.1 | tr | 1,2 |
| 112 | Longiborneol (Juniperol) | 1598 | 1599 | OS | tr | 1,2 | |
| 113 | Ledol | 1605 | 1602 | OS | 1.7 | 1,2 | |
| 114 | β-Oplopenone | 1605 | 1607 | OS | tr | 1,2 | |
| 115 | Humulene epoxide II | 1610 | 1608 | OS | 0.1 | 0.7 | 1,2 |
| 116 | 1,10-di-epi-Cubenol | 1615 | 1618 | OS | 0.9 | 1,2 | |
| 117 | Junenol | 1619 | 1618 | OS | 0.2 | 1,2 | |
| 118 | 1-epi-Cubenol | 1627 | 1627 | OS | 0.1 | 1,2 | |
| 119 | Muurola-4,10(14)-dien-1-β-ol | 1626 | 1630 | OS | 0.3 | 1,2 | |
| 120 | Caryophylla-4(12),8(13)-dien-5α-ol | 1632 | 1639 | OS | 0.3 | 0.3 | 1,2 |
| 121 | Caryophylla-4(12),8(13)-dien-5β-ol | 1634 | 1639 | OS | 0.8 | 1,2 | |
| 122 | epi-α-Cadinol (τ-cadinol) | 1638 | 1638 | OS | 0.3 | 1,2 | |
| 123 | epi-α-Muurolol (τ-Muurolol) | 1643 | 1640 | OS | 0.5 | tr | 1,2 |
| 124 | α-Eudesmol | 1654 | 1652 | OS | 1.2 | 1,2 | |
| 125 | α-Cadinol | 1658 | 1652 | OS | 1.3 | 1,2 | |
| 126 | Intermedeol | 1666 | 1665 | OS | 0.6 | 1,2 | |
| 127 | 14-Hydroxy-9-epi-(E)-caryophyllene | 1670 | 1668 | OS | 0.4 | 1,2 | |
| 128 | Germacra-4(15),5,10(14)-trien-1α-ol | 1685 | 1685 | OS | 1.2 | 0.6 | 1,2 |
| 129 | Eudesma-4(15),7-dien-1β-ol | 1689 | 1687 | OS | 0.8 | 0.3 | 1,2 |
| 130 | cis-Thujopsenal | 1707 | 1708 | OS | 0.1 | 1,2 | |
| 131 | Pentadecanal | 1714 | 1715 | A | 0.1 | 1,2 | |
| 132 | Mint sulfide | 1744 | 1740 | SS | tr | 1,2 | |
| 133 | Drimenol | 1770 | 1766 | OS | 0.4 | 1,2 | |
| 134 | Hexadecan-2-one | 1809 | 1809 | A | tr | 1,2 | |
| 135 | (5E,9E)-Farnesyl acetone | 1909 | 1913 | OS | tr | 1,2 | |
| 136 | Phytol | 1943 | 1942 | OD | tr | 1,2 | |
| 137 | Pimaradiene | 1953 | 1948 | D | tr | 1,2 | |
| 138 | Hexadecanoic acid | 1961 | 1959 | A | 1.8 | 1,2,3 | |
| 139 | Sandaracopimar-8(14),15-diene | 1964 | 1968 | D | 0.6 | 1,2 | |
| 140 | Manoyl oxide | 1992 | 1987 | OD | 0.3 | 1,2 | |
| 141 | Ethyl hexadecanoate | 1997 | 1992 | A | 0.1 | 1,2 | |
| 142 | (E)-Biformene (labda-8(20),12,14-triene) | 1998 | 1997 | D | 0.1 | 1,2 | |
| 143 | (E,E)-Geranyl linalool | 2021 | 2026 | OD | tr | tr | 1,2 |
| 144 | Abietatriene | 2052 | 2055 | D | 0.2 | 1,2 | |
| 145 | (Z)-Phytol | 2108 | 2114f | OD | 0.6 | tr | 1,2 |
| 146 | Nezukol | 2129 | 2132 | OD | tr | 1,2 | |
| 147 | Oleic acid | 2142 | 2141 | A | 0.8 | 1,2,3 | |
| 148 | Octadecanoic acid | 2170 | 2172 | A | 1.2 | 1,2,3 | |
| 149 | Docosane | 2200 | 2200 | A | tr | 1,2,3 | |
| 150 | 8,13-Abietadien-18-ol | 2319 | 2324 | OD | 0.2 | 1,2 | |
| 151 | trans-Ferruginol | 2328 | 2331 | OD | 0.1 | 1,2 | |
| 152 | 3-α-Acetoxy-manool | 2351 | 2359 | OD | 0.1 | 1,2 | |
| 153 | Tetracosane | 2400 | 2400 | A | tr | 1,2,3 | |
| 154 | Pentacosane | 2500 | 2500 | A | tr | 1,2,3 | |
| Chemical classes | |||||||
| Monoterpene hydrocarbons | M | 1.2 | 9.1 | ||||
| Oxygenated monoterpenes | OM | 1.1 | 7.6 | ||||
| Sesquiterpene hydrocarbons | S | 64.0 | 64.2 | ||||
| Oxygenated sesquiterpenes | OS | 20.3 | 9.4 | ||||
| Aliphatics | A | 4.0 | 0.4 | ||||
| Diterpenes | D | 0.3 | 0.6 | ||||
| Oxygenated diterpenes | OD | 1.0 | 0.3 | ||||
| Others | tr | ||||||
| Identified components (%) | 91.9 | 91.6 | |||||
aCompounds listed in order of elution from 5 % phenyl 95 % dimethylpolysiloxane type column. bRI = Retention index relative to C8-C32 n-alkanes on the 5 % phenyl 95 % dimethylpolysiloxane type column. cLit. RI = DB-5 41,42. dIMIdentification method: 1 = Experimental retention index; 2 = MS spectra; 3 = Standard. tr = Traces (<0.05 %). e43; f44. Major compounds are in boldface.
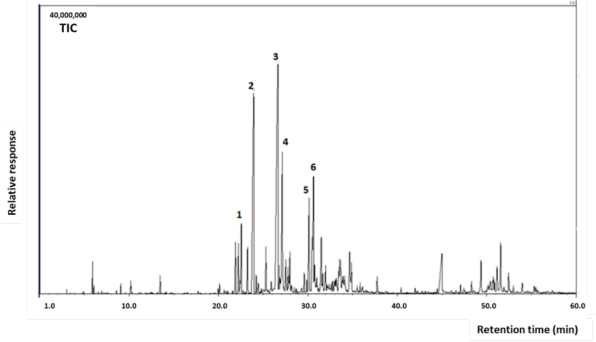
Fig. 2 GC-MS chromatogram (TIC) of Salvia polystachia oil: 1. β-elemene; 2. (E)-caryophyllene; 3. germacrene D; 4. bicyclogermacrene; 5. palustrol; and 6. caryophyllene oxide.
The hydrodistillation of fresh aerial parts of S. microphylla cultivated in Costa Rica produced a colorless oil yielding 0.13 % (v/w). The 73 identified oil compounds, accounting for 91.6 % of the whole volatiles, are summarized in Table 1 (see the Sm column). The essential oil composition consisted largely of sesquiterpene hydrocarbons (64.2 %), oxygenated sesquiterpenes (9.4 %), and monoterpene hydrocarbons (9.1 %), with minor quantities of monoterpenoids, aliphatic compounds, and diterpenoids. Seventy-three individual constituents were identified. The oil was dominated by (E)-caryophyllene (34.5 %), germacrene-D (8.4 %), trans-muurola-4(14),5-diene (6.5 %), with moderate amounts of caryophyllene oxide (4.7 %), germacrene B (4.2 %), β-pinene (3.2 %), γ-terpinene (3.2 %), β-gurjunene (3.2 %) and borneol (2.2 %), (see Fig. 3).
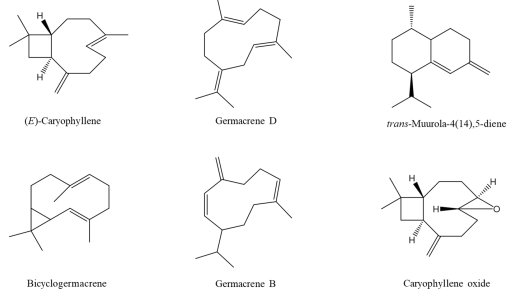
Fig. 3 Structure of the major sesquiterpenoid compounds identified in the essential oils of Salvia polystachia and S. microphylla, which occur in Costa Rica.
In the few studies carried out to date, quantitative and some qualitative differences in the chemical composition of essential oils have been observed. The oil of air-dried plants of S. microphylla cultivated in Italy 35, was rich in (E)-caryophyllene (10.8 %), bornyl acetate (9.1 %), 1,8-cineole (8.4 %), α-eudesmol (6.4 %), α-pinene (5.9 %), camphene (5.3 %), and β-eudesmol (5.3 %). The essential oil obtained from fresh leaves of plants cultivated in Brazil 36 presented as a major constituent (E)-caryophyllene (15.35 %), accompanied by α-eudesmol (14.06 %), β-eudesmol (8.74 %), γ-eudesmol (7.64 %), bicyclogermacrene (6.17 %) and isobornyl acetate (4.94 %), while the essential oil of S. microphylla from Algeria 37, contain (E)-caryophyllene (15.65-17.86 %), α-eudesmol (9.86-21.47 %), bornyl acetate (5.18-8.35 %), 1,8-cineole (1.98-7.50 %), γ-eudesmol (5.93-7.43 %), β-eudesmol (6.04-6.05 %), spathulenol (4.30-5.12 %), aromadendrene (4.90-5.39 %), and bicyclogermacrene (3.65-5.02 %). The chemical composition of the essential oil from the Costa Rican S. microphylla sample (University of Costa Rica campus) not previously studied is roughly similar to those formerly reported 35-37. The Costa Rican essential oil is somewhat different in that neither bicyclogermacrene, spathulenol, γ-eudesmol, nor β-eudesmol was found. However, germacrene D and trans-muurola-4(14),5-diene were seen as important constituents not present in previously studied samples.
There are only two reports in the literature on the chemical composition of the essential oils of the garden cultivar Hot Lips. The essential oil extracted from leaves of S. microphylla cv. Hot Lips cultivated in California contains guaiol (24.6-26.3 %) as the major constituent, accompanied by α-eudesmol (15.6-19.9 %), (E) -caryophyllene (5.5-11.5 %), camphor (3.5-10.6 %) and 1,8-cineole (1.4-6.2 %) 39. Guaiol is also present in the flower volatiles (4.0 %) of that cultivar growing in Italy (Research Centre for Vegetable and Ornamental Crops, Sanremo) 38, but in this case, the principal constituents were completely different (!): davana ether (16.3 %), hexahydrofarnesyl acetone (11.9 %), carvacrol (10.9 %), thymol (8.1 %), and (E,E) -farnesyl acetate (4.4 %). This difference could be due, on the one hand, to the fact that the flowers were specifically studied as a distinctly morphological part, and on the other hand, because an essential oil was not strictly extracted, since the volatile compounds were obtained by Headspace-Solid Phase Microextraction (HS-SPME) technique.
Although more than thirty -five compounds are shared by the Costa Rican oil of S. microphylla and the oil from cultivar Hot Lips 39, notable differences can be observed between them. In the studies carried out on S. microphylla, the major compound of the oil was (E)-caryophyllene, and the Costa Rican oil sample was the one that contained it in greater quantity (34.5 %). Some biological activities reported for this compound include analgesic, anti-inflammatory, antioxidant, neuroprotective, gastroprotective, anti-diabetic, antimicrobial, and antiproliferative effects. These properties have provided health benefits in several experimental models, such as analgesia, inflammation, anxiety disorder, depression, colitis, neurodegeneration, autoimmune diseases, metabolic ailments, osteoarthritis, and some types of cancer 45,46.
Other main constituents of the oil were germacrene-D, trans-muurola- 4(14),5-diene, caryophyllene oxide, and germacrene B. The volatiles from the cultivar Hot Lips growing in the USA and Italy contained guaiol, which appears to be a characteristic biomarker compound. The previously studied essential oils from S. microphylla do not contain guaiol 35-37, as does our oil sample of this species growing in Costa Rica.
Additionally, the essential oil of S. polystachia contained some diterpenoids (1.3 %) as minor constituents: phytol, abietatriene, (E)-biformene, 8,13 -abietadien-18-ol, trans-ferruginol, 3α-acetoxy-manool, pimaradiene, and nezukol, whereas the essential oil of S. microphylla presented sandaracopimar-8(14),15-diene and manoyl oxide. Diterpenoids isolated from Salvia spp. are the largest class of terpenoids found in this genus 9 and, in the subgenus Calosphace they are both ubiquitous and characteristic 12. Diterpenoid classes identified belong to four chemical skeletons: phytane, abietane, pimarane, and labdane (see the structures of cyclic diterpenoids identified, in Fig. 4).
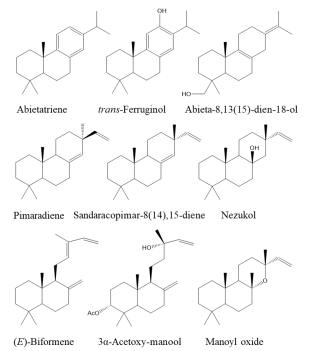
Fig. 4 Structures of cyclic diterpenoids identified in the essential oils of Salvia polystachia and S. microphylla from Costa Rica.
The essential oils of these two Salvia species from Costa Rica seem to be part of the group of Salvia oils containing mainly sesquiterpenoids with (E)-caryophyllene, germacrene D, bicyclogermacrene, and caryophyllene oxide as some of the principal constituents. Some examples are S. aethiopis L. 24,47-50, S. amplexicaulis Lam. 51, S. cardiophylla Benth. 52, S. chionantha Boiss. 53, S. chloroleuca Rech. f. & Aellen 53,54, S. compressa Vent. 55, S. grossheimii Sosn. 56, S. guaranitica A.St.-Hil. ex Benth. 57, S. leucantha Cav. 58, S. longipedicellata Hedge 51,59, S. nemorosa L. 49, S. nubicola Wall. ex Sweet 60, S. palaestina Benth. 61, S. sclareopsis Bornm. ex Hedge 62, S. verbascifolia M. Bieb. 62, S. verticillata L. 50,63, and S. xanthocheila Boiss. ex Benth. 54,64. Jassbi et al. 65 and more recently, Asgarpanah 66 classified the species of the genus Salvia growing in Iran into four categories according to their essential oil composition (GC-MS and GC-FID analyses) depending upon their major components: a) monoterpene chemotype, b) mono and sesquiterpene chemotype, c) sesquiterpene chemotype with caryophyllane type-C-skeleton as the predominant compounds, and germacrane, aromadendrane, cadinane, and other sesquiterpene classes as the other major constituents and, d) GLV (green leaf volatiles) chemotype. Most species examined in Iran are represented by the sesquiterpene chemotype which is prevalent there. The publications included in these studies indicated that the species of the Salvia genus showed that there is a large chemical polymorphism. These chemical differences could be due to biotic (genetic and biological differences) and abiotic conditions (which include all environmental factors) and those resulting from human activities, including extraction and analysis methods.
Conclusions
The chemical composition of the essential oil obtained from the aerial parts of a wild natural population of Salvia polystachia growing in Costa Rica has been determined for the first time. Sesquiterpenoids dominated the chemical composition of the oil (84.3 %), with germacrene D, (E)-caryophyllene, bicyclogermacrene, and caryophyllene oxide as principal constituents. The chemical composition of essential oils obtained from the aerial parts of cultivated S. microphylla in Costa Rica was studied for the first time. The essential oil also consists mainly of sesquiterpenoids (70.4 %). The main sesquiterpene encountered was (E)-caryophyllene. This was accompanied by lesser amounts of germacrene D, trans-muurola-4(14),5-diene, caryophyllene oxide, and germacrene B. Both species can be classified within the sages whose essential oils have sesquiterpenoids as major constituents (sesquiterpene chemotype).











 nueva página del texto (beta)
nueva página del texto (beta)



