Introduction
The search for new bioactive compounds as drug candidates is a continuous and urgent work that aims to lead to the discovery of new antibiotics. According to the World Health Organization, deaths caused by infections reached 25 % worldwide and 45 % in underdeveloped countries. There were 2.49 million deaths worldwide recorded in 2019 due to respiratory infections 1. The discovery of new molecules for the treatment of infectious diseases is ongoing and has become a public health issue, considering the growing emergence of resistance of microbial agents to commercially available antifungals and antibiotics 2. Natural products have been considered an excellent source of undiscovered molecules that may act as prototypes for the discovery of new drugs 3-6. It is estimated that over 65 % of medicines have been developed based on natural sources, with 32 % being from natural compounds or their derivatives. Over a 30 -year period (1981 to 2014), 43.5 % of the drugs approved worldwide for the treatment of infections caused by bacteria, fungi, parasites and viruses have been obtained from natural products 7. Pyrone lactone is one of these compounds, found naturally in many chemical structures of secondary metabolites. It displays diverse biological activity and is widely used as building blocks both in medicinal and synthetic chemistry 8-10. An analogous series of 2-styryl-5-hydroxy-4-pyrone derivatives were synthesized and found to have multifunctional action with the potential for the treatment of Alzheimer’s disease 11. A series of new monoprenylated and diprenylated 2 -pyrone derivatives with different halogen substituents were synthesized showing antimicrobial activity mainly against the bacteria Escherichia coli and Klebsiella pneumoniae12. Pyrones have also been used as ligands to obtain a complex containing vanadium with strong antimicrobial activity against the microorganisms Staphylococcus aureus, E. coli and Candida albicans13. Considering the biological potential of pyrone 14 and our continuous effort in the search for molecules with antimicrobial action, here we report the first biomonitored total synthesis of pyrones with an evaluation of their activities against bacteria and fungi.
Material and methods
Chemicals and reagents
4- Commercially obtained solvents were distilled (hexane and ethyl acetate) or dried over anhydrous Na2SO4 (t-butanol and dimethylformamide). For chromatographic analysis, silica gel 60 with a UV254 fluorescence indicator and silica gel with 0.063 - 0.2 mm (70-230 mesh) were used.
Instrument used
Fourier transform infrared spectra were obtained with a Varian model 640 IR-FT spectrometer. GC-MS analyses (60-260 ºC at 3 ºC/min. heating rate) were carried out in a Varian 431-GC coupled to a Varian 220- MS instrument using (CA, USA) fused-silica capillary column (30 m × 0.25 mm i.d. × 0.25 μm) coated with DB-5. MS spectra were obtained using electron impact at 70 eV with a scan interval of 0.5 s and fragments from 40 to 550 Da. The 1H and 13C Nuclear Magnetic Resonance (NMR) spectra were obtained using a Varian Mercury device - 400 or 300 and 100 or 75 MHz, respectively, using CDCl3 and C3D6O as solvents and tetramethylsilane (TMS) as the internal standard. Elemental microanalyses were performed using a EURO Vectora, EA3000 Model CHNS/O Analyzer.
Synthesis steps
Synthesis of methyl iodide
In a 500 mL flask, 179 mL of H 3PO 4 (85 %), 64 g (0.38 mol) of KI and 120 mL of methanol were added, and then heated in an oil bath at 70 °C. The reaction mixture was distilled at a temperature of 35 °C and the distillate was fractionated with 20 mL of saturated Na2S2O3 solution. The organic phase was treated with anhydrous Na2SO4, obtaining a colorless liquid (24 g).
Synthesis of acetyl-4-hydroxy-6-methyl-2H-pyran-2-one (1)
5 g (43 mmol) of methyl acetoacetate and 120 mg (1.42 mmol) of NaHCO3 were mixed and heated at 169 °C for 2 hours. Then 20 mL of dichloromethane was added, and the reaction mixture washed with H2O2 (3 x 15 mL). The organic phase was concentrated in vacuum and the product recrystallized with ethanol, obtaining 0.5 g of crystalline white solid. 53.8 % yield. Melting point: 108 °C; IR, ν, cm-1 3424 (OH), 3090 (=CH), 1723 (C=O), 1645 (C=O), 1549 (C=C), 1248 (C-O); MS (EI) m/z: 168 (M+), 153, 125; 1H NMR (300 MHz, CDCl3), δ, ppm: 2.26 (s, 3H), 2.64 (s, 3H), 5.93 (s, 1H); 13C NMR (75 MHz, CDCl3), δ, ppm: 20.7; 30.0; 99.9; 10.,4; 161.2; 169.0; 181.1; 205.2.
Synthesis of 4-hydroxy-6-methyl-2H-pyran-2-one (2)
In a flask, 0.45 g (2.67 mmol) of compound 1 was solubilized in 10 mL of H2SO4; the reaction mixture was then kept under stirring and heated under reflux at 120 °C for 1.5 h. The reaction mixture was cooled in an ice bath and the obtained solid was washed with ice -cold H2O2 to yield 0.215 g of compound 2 as a white solid, 63.7% yield. Melting point: 186 °C; IV, ν , cm-1: 3.442 (OH), 3.093 (C-H, sp2), 1.718 (C=O, cyclic), 1623 (C=C), 1.539 (C=C), 1258 (C-O); 1H NMR (400 MHz, DMSO-d6), δ, ppm: 2.15 (s, 3H), 5.20 (s, 1H), 5.95 (s, 1H), 11.58 (s, 1H); 13C NMR (100 MHz, DMSO-d6), δ, ppm: 19.4; 88.1; 100.1; 163.2; 163.88 170.5.
Synthesis of 4-methoxy-6-methyl-2H-pyran-2-one (3)
To a solution of 4-hydroxy-6-methyl-2H-pyran-2-one (51) (0.013 g/mL) was added 1.58 g (11 mmol) of K2CO3 and 0.256 mL (3.96 mmol) of methyl iodide. The reaction mixture was kept under stirring at 58 °C for 3 hours. The reaction mixture was washed with 20 mL of saturated NH4Cl solution and subjected to extraction with ethyl acetate (3 x 20 mL). The organic phase was treated with anhydrous Na2SO4, concentrated under reduced pressure and purified by column chromatography (Hexane:ethyl acetate, 7:3), to obtain 0.264 g of a yellow solid. 63 %, Melting point: 86 °C; IR, ν, cm-1: 3086 (C-H sp2), 1739 (C=O, cyclic), 1645 (C=C), 1575 (C=C), 1253 (C-O), 1146 (C-O); MS (EI) m/z: 140 (M+), 125, 112, 69; 1H NMR (400 MHz, CDCl3), δ, ppm: 2.21 (s, 3H), 3.79 (s, 3H), 5.41 (s, 1H), 5.78 (s, 1H); 13C NMR (100 MHz, CDCl3), δ, ppm: 19.7; 55.7; 87.2; 100.2; 161.9; 164.8; 171.2.
Synthesis of 3-methoxy-2-methyl-4H-pyron-4-one (4)
To a solution of 3-hydroxy-2-methyl-4H-pyran4-one (0.02 g/mL) there was added 2.46 g of K2CO3 (16.90 mmol) and 0.39 mL of methyl iodide (6.15 mmol). The reaction mixture was kept under stirring at 60 °C for 3 hours. The reaction mixture was washed with 30 mL of saturated NH4Cl solution and subjected to extraction with ethyl acetate (3 x 30 mL). The organic phase was treated with anhydrous Na2SO 4 and concentrated under reduced pressure to obtain 0.530 g of compound 4 as orange Oil. Yield: 95 %; IR, ν, cm-1: 3.071 (C-H sp2), 2929, 1639 (C=O), 1261 (C-O), 1219 (C-O), 1168 (C-O); MS (EI), m/z: 140 (M+), 122, 110, 69; 1H NMR (400 MHz, CDCl3), δ, ppm: 2.33 (s, 3H), 3.86 (s, 3H), 6.36 (d, 1H, J = 5.6 Hz), 7.63 (d,1H, J = 5.6 Hz); 13C NMR (100 MHz, CDCl3), δ, ppm: 14.5; 59.9; 117.2; 145.6; 153.3; 159.0; 174.8.
Synthesis of styrylpyrones 5-7
In a 25 mL flask with N2 atmosphere, 0.135 mL (1.42 mmol) of dry t-butanol and 0.033 g (1.42 mmol) of metallic sodium were mixed, then kept under agitation until complete dissolution of metallic sodium. Then, 0.1 g (0.71 mmol) of the pyrones (3 or 4) dissolved in 1 mL of dry dimethylformamide and slowly added to the previously prepared sodium t-butoxide. Then, 1.43 mmol of the aldehyde (benzaldehyde or 4-methoxybenzaldehyde) was transferred dropwise into the flask. The reaction mixture was placed in an ultrasound bath at room temperature for 6 hours, observing the consumption of pyrone. The reaction mixture was extracted with ethyl ether (3 x 20 mL). The organic phase was washed with saturated NaCl solution (3 x 15 mL) and distilled water (3x15 mL), then dried with anhydrous Na2SO4 and concentrated in vacuum. The products were purified by preparative plate chromatography using the hexane/ethyl acetate (7:3) solvent system as mobile phase.
5,6-Dehydrokavaine (5): Yellow solid, 53 % yield, Melting point: 136 °C, IR, ν, cm-1: 3086 (C-H sp2), 1724 (C=O, cyclic), 1624 (C=C), 1557 (C=C ), 1446 (C=C, aromatic), 1410 (C=C, aromatic), 1253 (C-O), 1140 (C-O); MS (EI) m/z: 228 (M+), 200, 185, 157, 129, 103, 77, 69; 1H NMR (400 MHz, CDCl3), δ, ppm: 3.82 (s, 3H), 5.50 (d, 1H, J4 = 2 Hz), 5.9 (d, 1H, J4 = 2 Hz) 6 .60 (d, 1H, J = 16 Hz), 7.38 (m, 3H), 7.51 (m, 3H), 7.52 (d, 1H, J = 16 Hz); 13C NMR (100 MHz, CDCl3), δ, ppm: 55.8; 88.80; 101.3; 118.6; 127.0; 128.0; 129.0; 135.70; 135.10; 158.50; 163.8; 171.0 (Supplementary Figures S1-S4).
Styrylpyrones (6): Yellow solid, 39 % yield, Melting point: 120°C, IR, ν, cm-1: 2922 (C-H sp3), 1746 (C=O), 1632 (C=C), 1454 (C=C), 1410 (C=C), 1253 (C-O), 1154 (C-O); MS (EI) m/z: 228 (M+), 213, 137, 115, 77. Anal. calcd. (%): C14H12O3: C, 73,69; H, 5.26; O, 21.05; Found: C, 73,67; H, 5.23; O, 21.10. 1H NMR (400 MHz, CDCl3), δ, ppm: 3.98 (s, 3H); 6.39 (d, 1H, J = 5.6 Hz); 7.25 (d, 1H, J = 16 Hz); 7.39 (d, 1H, J = 16 Hz); 7.57 (d, 1H); 7.71 (d, 1H, J = 5.6 Hz); 13C NMR (100 MHz, CDCl3), δ, ppm: 175.31; 155.3; 152.9; 144.5; 135.4; 135.1; 129.4; 128.8; 127.4; 117.0; 114.9; 60.6 (Supplementary Figures S5-S8).
Yangonin (7): Yellow solid, 42 % yield, Melting point 146 °C, IR, ν, cm-1: 3.079 (C-H sp2), 2929 (C-H sp3), 1724 (C=O), 1611 (C=C), 1561 (C=C), 1454 (C=C), 1410 (C=C), 1253 (C-O), 1154 (C-O); MS (EI) m/z: 258 (M+), 230, 187, 115, 69; 1H NMR (400 MHz, CDCl3), δ, ppm: 7.48 (d, 1H, J = 16 Hz); 7.46 (d, 1H J = 8 Hz); 6.90 (d, 1H J = 8 Hz); 6.47 (d, 1H, J = 16 Hz); 5.90 (d, 1H, J4 = 2.4 Hz); 5.47(d, 1H, J4 = 2.4Hz); 3.82 (s, 3H); 3.83 (s, 3H); 13C NMR (100 MHz, CDCl3), δ, ppm: 171.2; 160.7; 164.1; 159.0; 135.4; 128.0; 128.8; 116.3; 114.3; 100.4; 88.3; 55.8; 55.3 (Supplementary Figures S9-S12).
In vitro antimicrobial activity
The antimicrobial potential of ligands and complex was evaluated against the gram-positive bacteria Staphylococcus aureus (UFPEDA 02), Enterococcus faecalis (UFPEDA 138) and the gram-negative bacteria: Klebsiella pneumoniae (UFPEDA 396), Pseudomonas aeruginosa (UFPEDA 416) as well as the fungi Candida albicans and C. utilis. The bacteria and fungi came from the collection of microorganisms from the Antibiotics Department of the Federal University of Pernambuco. The suspension of microorganisms was standardized by the turbidity equivalent to a 0.5 tube on the McFarland scale in distilled water, corresponding to a concentration of approximately 108 CFU/mL for bacteria and 107 CFU/mL for fungi 15-16.
Determination of the Minimum Inhibitory Concentration (MIC)
MIC was performed using the microdilution technique in 96-well multiplates 15-16. The culture media used were Sabourand Agar (for fungus) and Muelle-Hinton Agar (for bacteria). Metronidazole and Fluconazole were used as a positive control, while ethyl alcohol was used as a negative control. Microplates were cultured at 37 ºC for 18-24 h for bacteria and 30 ºC for 48-72 h for the fungus. After the culture period, the microplates were developed with the addition of 10 μL of 0.01 % resazurin solution and incubated for 3 h. The MIC was defined as the lowest concentration of the sample that inhibited the growth of the microorganisms. Analyses were performed in triplicate and values were expressed as the mean± standard deviation.
Results and discussion
Synthesis
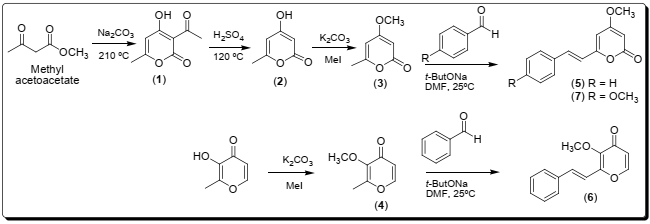
The synthesis of pyrones was carried out in four steps (Scheme 1), starting with obtaining of compound 1 through Claisen condensation between two equivalents of methyl acetoacetate at 169 °C, followed by an intramolecular cyclization yielding a white crystalline solid, yield 53.8 % and melting point of 108 °C. The deacetylation reaction of compound 1 in acid medium at 120 °C for 1.5 h provided compound 2, a white crystalline solid, with a yield of 63.7 % and melting point of 186 °C 17,18. Compound 3 was derived from the methylation reaction of compound 2 using methyl iodide at reflux at 58 °C which gave a beige crystalline solid in 63% yield and melting point of 86 °C. 3-methoxy-2-methyl-4H-pyron-4-one (4) was obtained as an orange oil in 95% yield from the methylation of the compound 3-hydroxy-2-methyl-4H-pyran-4-one with methyl iodide under basic medium at reflux at 58 °C by 3 h. Compounds 1-4 were characterized based on their IR, MS, 1H and 13C NMR spectra. The synthesis of styrylpyrones was carried out through an aldol reaction using sodium t-butoxide as a base under stirring, as previously described by Kraus and Wanninayake 19 with some modifications. The reaction time for the styrylpyrone syntheses 5-7 (Scheme 1) was optimized in 5h using ultrasound radiation with reaction yields 53 %, 39 % and 42 %, respectively.
The synthesized styrylpyrones 5 and 7 correspond to the natural compounds 5,6-dehydrokavaine and yangonin, respectively, previously isolated as kavalactones from the roots of Piper methysticum20. Compounds 5 and 7 were elucidated based on the interpretation of their IR spectra, EM 1H and 13C NMR, which are similar to their respective spectral data previously reported 21-23. Styrylpyrone 6 is an unpublished compound and isomer of 4,5- dehydrokavaine 5. Compound 6 was obtained as a yellow solid in 42 % yield and melting point 120°C. The IR spectrum of 6 showed stretching bands at 1746 cm-1 of carbonyl, at 1632 cm -1 of conjugated sp2 carbons, at 1454 and 1410 cm-1 of aromatic ring. The 1H NMR spectrum showed a singlet at δ 3.98 of the methoxyl group (H12), two doublets at δ 6.39 and 7.71 referring to the coupling of H1 with H2 (J = 5 .6 Hz). The signals of the doublets referring to the coupling of H6 with H-7 with J = 16.0 Hz in E configuration and a set of signals in δ 7.57-7.33 ppm referring to the hydrogens of the phenyl group. The 13C NMR spectrum of compound 6 showed signs of the methoxyl group (C12 δ 60.67), low field carbonyl group (δ 175.31), carbons sp2 signals of the pyrone (δ 155.31 C1, 152.92 C5, 144.53 C2, 117.00 C4) and aromatic (δ 135.10 C8, 129.45 C10 and C10’, 128.89 C11, 127.49 C9 and C9') rings. The mass spectrum showed a base peak in m/z of 228 Da referring to the molecular ion according to the molecular formula C14H12O3.
Biomonitoring of synthesis steps
The synthesis of pyrones was biomonitored by evaluating the antimicrobial activity of reaction intermediates and products against bacteria and fungi (Table 1). The microorganisms used in the present study were selected due to their clinical importance and resistant to certain antibiotics.
Table 1 Minimum inhibitory concentration at μg.mL-1 for synthesized compounds.
| Compounds | Microorganisms | |||||
|---|---|---|---|---|---|---|
| S. aureus | E. faecalis | P. aeruginosa | K. pneumoniae | C. utilis | C. albicans | |
| Methyl acetoacetate | 2500 | 2500 | 2500 | >2500 | 2500 | 625 |
| Benzaldehyde | 2500 | 2500 | 2500 | >2500 | 2500 | 1250 |
| 1 | 625 | 2500 | 625 | 2500 | 2500 | 625 |
| 2 | 2500 | 2500 | 2500 | 2500 | 2500 | 2500 |
| 3 | 2500 | 2500 | 2500 | 2500 | 2500 | 1250 |
| 4 | 2500 | 2500 | 625 | 2500 | 2500 | 1250 |
| 5 | 2500 | 2500 | 2500 | 2500 | 2500 | 2500 |
| 6 | 1250 | 2500 | 1250 | 2500 | 1250 | 1250 |
| 7 | 2500 | 2500 | 2500 | 2500 | 2500 | 1250 |
MIC results were classified as strong for MIC ≤ 100 μg/mL; moderate for MIC ˃ 100 ≤ 625 μg/mL; weak for MIC ˃ 625 μg/mL 24,25. Methyl acetoacetate, the starting material for dehydroacetic acid 1, showed moderate activity against C. albicans with a value of 625 μg/mL. The intermediate that showed the best activity was compound 1 with a MIC of 625 μg/mL against the bacteria S. aureus, P. aeruginosa and the fungus C. albicans. Compound 1 has been widely used in building blocks for obtaining a series of biologically active compounds, including those compounds with antimicrobial action against fungi, bacteria and viruses 26,27. A series of enaminopyran-2,4-diones derived from compound 50 were synthesized and showed activity against both gram positive and gram negative bacteria 28. Naphtho-γ- pyrones has been isolated of fungi as potential antibacterial agents against E. coli, P. aeruginosa and E. faecalis with MIC values in the range of 4.3-50 µg/mL 29,30. Molecular docking-based target identification of naphtho-γ-pyrones revealed bacterial enoyl-acyl carrier protein reductase as an antibacterial target 30.
Modifications to the substituents of the compound 1 ring to obtain compounds 2 and 3 resulted in reduced antimicrobial activity. The coupling of 3 with benzaldehyde to obtain 4,5-dehydrokavaine (5) did not enhance antimicrobial activity. Comparing the MIC values for styrylpyrones 5 and 7 with 6, it was observed that the γ-pyrone ring 6 was more active than the α-pyrone ring (5 and 7). Compound 5, found in certain plant species, has shown potent anti-inflammatory activity and effectiveness in preventing fulminant hepatitis in a study with mice 31,32. Yangonin (7) also showed potential for the prevention of ethanol-induced chronic liver damage in mice 33.
Conclusions
This is the first report of a biomonitored synthesis and antimicrobial activity for the synthesized compounds. The results revealed that the styrylpyrones exhibited activity against dimethylformamide gram-positive bacteria (S. aureus and E. faecalis), gram-negative bacteria (K. pneumoniae and P. aeruginosa) and fungi (C. albicans and C. utilis). The results obtained contribute significantly to knowledge of the biological potential of pyrones derived, considering that pyrone rings are widely used as building blocks in the synthesis of bioactive molecules.











 nueva página del texto (beta)
nueva página del texto (beta)