Introduction
Nosocomial infections (NI) represent a major public health problem worldwide. They are of concern in all countries because they constitute a permanent risk in healthcare, with important repercussions on hospital stays 1. It is the most frequent complication and is related to multiple factors such as low immunity of patients, contaminated objects and clothing of healthcare personnel 2,3.
The clothing worn by healthcare personnel constitutes an important source for the growth of microorganisms, since they tend to proliferate in humid environments 4. Even though there are guidelines for proper cleaning of the hospital environment, pathogens possess mechanisms to adapt and survive for prolonged periods of time. Concern for the protection of both healthcare personnel and patients is the most important thing 5.
In order to solve this problem, several research have focused on the use of antibacterial agents such as plant extracts. Because they represent a promising development to functionalize textiles, taking full advantage of the antibacterial properties they possess, since they allow the manufacture of skin and environmentally friendly products, such as dressings, gauze, cloths, or surgical garments 6. The antibacterial properties exhibited by plant extracts are mainly attributed to bioactive compounds such as phenolic terpenes and hydroxycinnamic acids, which are found in plants such as Rosmarinus officinalis7.
The Rosmarinus officinalis plant is approximately 1.5 to 2 meters tall, its origin comes from the union of the Greek words "rhos" shrub and "myrinos" aromatic 8. The bioactive compounds of Rosmarinus officinalis are concentrated in the leaves, including camphor, β-caryophyllene, α-pinene, borneol and 1,8-cineole, carnosol, rosmanol, isorosmanol, rosmarinic acid and rosmadiol, among others 9,10.
Conventional processes used to obtain these bioactive compounds have drawbacks to be taken into account, such as low extraction efficiency, poor selectivity and harsh extraction conditions. An alternative for the extraction of bioactive compounds from plants are green technologies such as ultrasound, which is used due to its advantages such as short times, low cost and high extraction yields 11,12. Ultrasound is based on the cavitation phenomenon, in which the extracting solvent diffuses through the cell wall of plant leaves due to the collapse of bubbles generated by high pressures and temperatures 13-15.
Considering the above, the use of nonwoven fabric in the manufacture of textiles with antibacterial properties will improve the final performance of the material. The nonwoven fabric is formed by directionally or randomly oriented fibers, joined by friction, cohesion and/or adhesion, by means of mechanical, chemical or thermal processes. This has magnificent properties among which are its moisture repellency, porosity, flexibility and it is ideal for manufacturing surgical garments, dressings and mouth covers 16. Among the synthetic polymers used for the manufacture of nonwoven fabrics are Nylon 6 and Polypropylene (PP), due to their characteristics 17,18.
Therefore, this work is mainly focused on the study of obtaining composite materials based on Nylon 6 and Polypropylene nonwoven fabrics with Rosmarinus officinalis extracts by ultrasound to evaluate their antibacterial properties. This is an area of great opportunity due to the need of hospitals to control or counteract nosocomial infections.
Methodology
Extraction of bioactive compounds from leaves of Rosmarinus officinalis
4 Dried leaves of Rosmarinus officinalis were used as extracting solvents: ethyl acetate (Jalmek, purity 99.5 %) and ethanol (Jalmek, purity 99.5 %), using the following treatments: magnetic agitation for 24 h and ultrasound with a frequency of 60 kHz for four h, weighing 5 g of dried leaves with 50 mL of the extracting solvent, once the treatment time had elapsed, they were decanted and left to dry at room temperature.
Characterization of Rosmarinus officinalis extracts
FTIR-ATR infrared spectroscopy
The extracts obtained were characterized by FTIR-ATR spectroscopy, using a Perkin-Elmer spectrophotometer; the samples were analyzed without previous treatment, working in a range of 4500 to 500 cm-1, Attenuated Total Reflectance (ATR) was used, with 2 cm-1 resolution and 32 acquisitions.
High performance liquid chromatography coupled to mass spectrometry HPLC-MS
The extracts of Rosmarinus officinalis obtained were analyzed by HPLC (Varian Prostar) with a diode array detector (280 nm). A total of 1.8 ml sample was filtered (0.45 µm membranes). The separation of the compounds was performed on a Grace Denali c- 18 column (5 µm, 250 x 4.6 mm) at 30 °C. The mobile phase A was methanol (wash), B acetonitrile, and 3 % acetic acid C (initial 3 % B and 97 % C, 0-5 min 9 % B and 91 % C, 5 -15 min 16 % B and 84 % C, 15 -30 min 33 % B and 67 % C, 30-33 min 90 % B and 10 % C, 33-35 min 90 % B and 10 % C, 35-42 min 3 % B and 97 % C), flow rate 1 ml/min, injection volume 10 µL. Mass analysis was performed using a Varian 500-MS ion trap, electrospray ionization (ESI), capillary voltage 90 V, negative mode ([M-H] - m/z), mass acquisition range 100-2000 m/z.
Thermogravimetric TGA Analysis
For this analysis a TA instruments Q500 thermal analyzer was used, with a heating rate of 10 °C / min and a temperature range of 30 - 700 °C.
Antioxidant capacity tests
In the antioxidant capacity tests, dilutions of the extracts were performed, weighing 3 mg of Rosmarinus officinalis extract and dissolved in 1 mL of ethanol (Jalmek, purity 99.5 %), for the DPPH (2,2-Diphenyl-1-Picrylhydrazyl) technique, 193 μL of the DPPH reagent was placed with 7 μL of the extract dilution and measured in a spectrophotometer at 519 nm, for ABTS (2,2'-azino- bis-3-ethylbenzothiazoline-6- sulfonic acid), 190 μL of the ABTS reagent was placed with 10μL of the extract dilution and measured at 734 nm, for the FRAP (ferric ion reducing antioxidant capacity) technique, 290 μL of the FRAP reagent was placed along with 10 μL of the extract dilution and measured at 595 nm, each treatment was performed in triplicate with the different techniques.
In vitro antibiosis bioassay of Rosmarinus officinalis extracts
Tablets of 0.1 g of Rosmarinus officinalis extracts were made. Subsequently, dilutions of Staphylococcus aureus (gram-positive) and Escherichia coli (gram negative) were made up to 1x10-3; from the last dilution, 300 μL were placed in the Petri dish, spread with a glass rod, and 4 tablets of the extract were placed. Each extract magnetic agitation 24 h and ultrasound for 4 h, using ethanol as solvent) was performed in triplicate; in addition, positive control of both bacteria and control with antibiotic were seeded and left incubating for 24 h, after which time the presence or absence of inhibition was observed, determined by a halo around the tablets.
Obtaining the compost Impregnation of Nylon 6 and Polypropylene nonwoven fabric with Rosmarinus officinalis extracts
The impregnation was carried out by immersion, and three different concentrations of Rosmarinus officinalis extracts were used, which were 3000, 5000, and 10000 ppm. Squares of 10 x 10 cm were cut from Nylon 6 and Polypropylene nonwoven fabrics, placed in the different solutions that were previously prepared with ethanol (Jalmek, purity 99.5 %), and left in these solutions for 12, 24, and 48 h, after which time the fabrics were removed from the solutions, placed in containers and left to dry at room temperature. The fabrics were characterized by FTIR-ATRA and TGA.
In vitro antibiosis bioassay of Nylon 6 and Polypropylene nonwoven fabrics impregnated with Rosmarinus officinalis extracts
Circles of 6 mm diameter were cut from Nylon 6 and PP nonwoven fabrics impregnated with Rosmarinus officinalis extracts, including blanks (ethanol-impregnated nonwoven fabrics). Dilutions of Staphylococcus aureus (gram positive) and Escherichia coli (gram negative) were made up to 1x10-3, placing 300 μL in the Petri dishes, spread with a glass rod, 4 circles of the fabrics were placed. Each test was performed in triplicate, an absolute control without treatment and an ethanol control was used, which were left incubating for 24 h, after which time the presence or absence of inhibition was observed, determined by a halo around the cloths.
Results and discussion
Extraction of bioactive compounds from Rosmarinus officinalis leaves
Ethanol and ethyl acetate were used as extracting solvents, in order to obtain polar compounds present in the Rosmarinus officinalis plant. Nieto et al 19 indicate that, in the order of polarity of polar solvents used in the extraction of plant compounds, ethanol is one of the most polar, besides being an environmentally friendly solvent, because it is non-toxic and allows a green extraction that does not generate waste.
Table 1 shows the percentage yields of the extractions performed using different treatments and extraction conditions. The differences in extraction yield are mainly due to the synergistic effect of the bioactive compounds present in the plant, the properties of the solvent and the affinity of the bioactive compounds of Rosmarinus officinalis with the solvent 20.
Table 1 Yields of extractions of Rosmarinus officinalis leaves using.
| Solvent | Treatment | Time (h) | % Performance |
|---|---|---|---|
| Ethanol | Ultrasound (USE) | 4 | 14.67 |
| Magnetic stirring (TAE) | 24 | 13.20 | |
| Ethyl acetate | Ultrasound (USA) | 4 | 14.48 |
| Magnetic stirring (TAA) | 24 | 12.99 |
It has been reported that this method offers advantages in terms of productivity, yield and selectivity, since it uses less extraction time, reduces chemical risks and is an environmentally friendly methodology, since less time, water and energy are used 21. Robles et al 22 characterized and identified bioactive compounds from tejocote (Crataegus mexicana). The extracts were obtained using mixtures of ethanol-water (70:30), acetone-water (70:30) and ultrasound for 30 minutes, where they found yield percentages of 21.1 %. While Flores et al 23 made extracts of dried leaves of Rosmarinus officinalis, using ethanol as an extracting solvent and ultrasound for 2 h, from which they obtained a yield percentage of 10.42 %.
Characterization of Rosmarinus officinalis extracts FTIR-ATR Infrared Spectroscopy
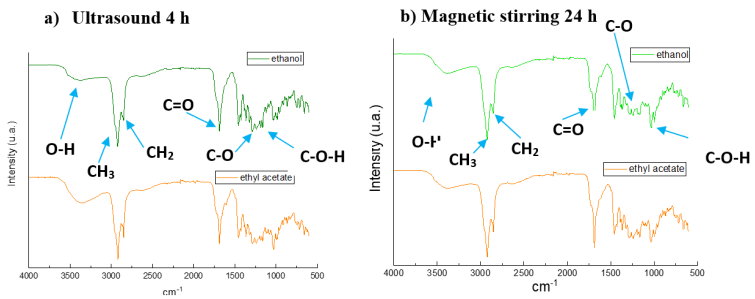
In Fig. 1 FTIR-ATR spectra are presented: a) ultrasound 4 h and b) magnetic stirring 24 h, both using ethyl acetate and ethanol, respectively, as extracting solvents. It should be noted that very similar results were obtained in the four different treatments. At 3345 cm-1 a broad band corresponding to the O-H stretching of alcohols and/or carboxylic acids attached to aromatic rings is observed; at 2930 cm-1 vibration of -CH3 asymmetric stretching is found, and 2850 cm-1, a band characteristic of the vibrations of the methylene group - CH2 symmetric stretching is present, while at 1670 cm-1 the band corresponding to the stretching of the carbonyl bond C=O carboxylic acids and their derivatives such as esters is shown and between 1400 to 1450 cm-1 the bands of the C- H bond torsions are found, as well as at 1000 cm-1 the bands of the C-O-H bonds out of the plane are found. The main groups to be highlighted from the secondary metabolites of Rosmarinus officinalis extract are hydroxyl, carbonyl, and carboxylic acids 24,25. Similar behavior is evidenced in both treatments, which leads to the deduction that the ultrasound treatment is the most recommended, as higher yield percentages are obtained in less time.
High performance liquid chromatography HPLC-MS
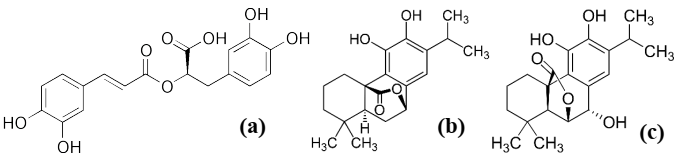
The analysis of the components present in each of the extracts was carried out by HPLC-MS chromatography, where the identification of the different compounds is shown in Table 2, in which the retention times, mass, compound and family are presented. Fig. 3 shows the HPLC-MS chromatograms of the different extracts obtained. As can be seen, the presence of phenolic terpenes, methoxyflavones, hydroxycinnamic acids and alkylphenols was confirmed. The most abundant compound was rosmarinic acid, which presented retention times of 41.31, 43.51 and a mass of 359 1, this compound could only be extracted using ethanol as extraction solvent, because the polarity index of ethanol is higher than that of ethyl acetate, which agrees with Bendif et al 26, who reported the identification of rosmarinic acid with a retention time of 11. 8 min and mass of 278.5, in ethanolic extracts of Rosmarinus eriocalyx obtained by the conventional method. Fig. 2 shows some of the compounds obtained in the extracts of Rosmarinus officinalis.
Table 2 Compounds identified by HPLC-MS of Rosmarinus officinalis extracts.
| Treatmen | Number | Retention time (min) | Mass (m/z) | Compound | Family |
|---|---|---|---|---|---|
| Magnetic stirring 24 h with ethyl acetate | 1 | 52.18 | 345.1 | Rosmanol | Phenolic terpenes |
| 2 | 54.67 | 330.3 | Jaceosidin | Metoxyflavones | |
| 3 | 56.13 | 345.1 | Rosmanol | Phenolic terpenes | |
| 4 | 55.47 | 325.1 | Ferulyl tartaric acid | Methoxycinnamic acids | |
| Magnetic stirring 24 h with ethanol | 5 | 41.31 | 359.1 | Rosmarinic acid | Hidroxycinnamic acids |
| 6 | 45.10 | 328.8 | Carnosol | Phenolic terpenes | |
| 7 | 50.39 | 345.1 | Rosmanol | Phenolic terpenes | |
| Ultrasound 4 h with ethyl acetate | 8 | 51.02 | 345.1 | Rosmanol | Phenolic terpenes |
| 9 | 52.62 | 325.1 | Ferulyl tartaric acid | Methoxycinnamic acids | |
| 10 | 53.7 | 347.1 | 5-Heptadecylresorcinol | Alkylpehnols | |
| Ultrasound 4 h with etanol | 11 | 43.51 | 359.1 | Rosmarinic acid | Hidroxycinnamic acids |
| 12 | 44.13 | 344.8 | Rosmanol | Phenolic terpenes | |
| 13 | 48.35 | 328.9 | Carnosol | Phenolic terpenes | |
| 14 | 55.04 | 359.1 | Rosmarinic acid | Hidroxycinnamic acids |
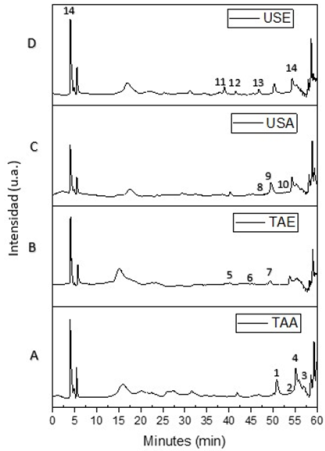
Fig. 3 HPLC- MS chromatograms of the extracts: (a) magnetic stirring for 24 h with ethyl acetate, (b) magnetic stirring for 24 h with ethanol, (c) ultrasound for 4 h with ethyl acetate and (d) ultrasound for 4 h with ethanol.
From this section, only the ethanol extracts of Rosmarinus officinalis obtained by ultrasound and magneto-magnetic agitation were used, since they were the ones that presented the highest yield percentages.
Thermogravimetric TGA analysis
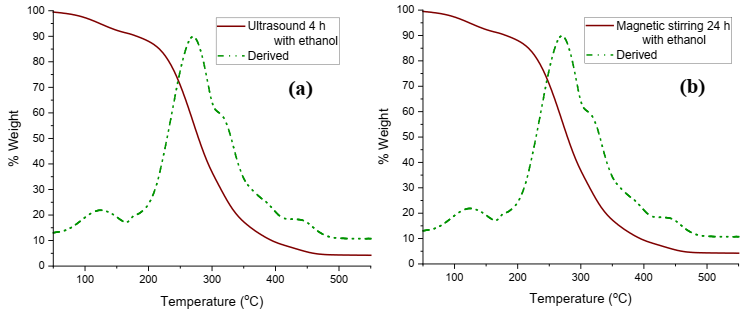
Fig. 4 shows the thermograms of the Rosmarinus officinalis extracts, where a first loss in weight of 8 % is found at 124.22 °C, which is attributed to the humidity of the extracts due to the solvents used. The second loss in weight is 80 % at 270.41ºC, which is where compounds such as rosmarinic acid and carnosic acid begin to degrade, since this weight loss is in the range of temperatures where the degradation of both compounds occurs and finally there is a third loss of 5 % at 417 °C, until reaching the total decomposition of the sample that occurs at 440.81 °C. From these thermograms it is assumed that the stability of the extracts is up to temperatures of 124.22 °C. Robinson et al 27 indicate that when there are 2 or 3 points of weight loss in natural extracts, it indicates the disappearance of certain families or secondary metabolites present in the extracts. This test is important because it indicates up to what temperature the organic compounds present in the extracts of Rosmarinus officinalis are stable 28.
Antioxidant capacity tests
Table 3 shows the results obtained for each of the techniques used, finding for DPPH results of percentage of antioxidant capacity of the radical of 56.03 - 69.82 %, several authors report similar inhibition percentages, among which are Briones et al 29 reported 73.85 % antioxidant capacity of the aqueous extract of Rosmarinus officinalis. While Perales et al 30 measured the antioxidant activity by the DPPH technique of methanol extracts of Rosmarinus officinalis obtained by ultrasound, finding a mean DPPH radical inhibition concentration of 31.65 Cl50 (μ/mL).
Table 3 Antioxidant capacity of Rosmarinus officinalis extracts with three different techniques.
| Extract | DPPH % antioxidant capacity | ABTS % antioxidant capacity | FRAP mg trolox/g capacity |
|---|---|---|---|
| Ultrasound 4 h with ethyl acetate | 69.82 | 84.13 | 0.4076 |
| Ultrasound 4 h with ethanol | 66.95 | 84.46 | 0.3363 |
| Magnetic stirring 24 h with ethanol | 63.50 | 82.44 | 0.3404 |
| Magnetic stirring 24 h with ethyl acetate | 56.03 | 81.93 | 0.3541 |
In the ABTS technique, antioxidant capacity percentages of 81.93 - 84.46 % were obtained, which is similar to that reported by Briones et al 29 who obtained an antioxidant capacity percentage of 75.3 % in aqueous extracts of Rosmarinus officinalis.
Finally, in the FRAP technique, results of 0.3363 - 0.4076 mg trolox/g sample were obtained, which is in agreement with that reported by Chen et al 31, where 0.226 mg trolox/g sample of antioxidant capacity of the aqueous extract of Rosmarinus officinalis was reported, as well as Briones et al 29, who obtained 0.349 mg trolox/g sample of antioxidant capacity of aqueous extracts of Rosmarinus officinalis. The antioxidant activity of Rosmarinus officinalis extracts is mainly attributed to terpenes, polyphenols and quinones, which act as free radical chain terminators, as chelators of reactive oxygen species and as chelators of metal ions (Fe2+), this is due not only to the high percentage of these components, but also to the presence of flavonoids and diterpenes found in small amounts or to the synergy between them 31,32.
In vitro antibiosis bioassay of the extracts of Rosmarinus officinalis
Extracts obtained by ultrasound showed mean inhibition halos of 23.25 mm against Escherichia coli and 23.5 mm against Staphylococcus aureus, while extracts obtained by magnetic stirring showed mean inhibition halos of 15 mm against Escherichia coli and 17. 25 mm against Staphylococcus aureus, as shown in Fig. 5. Martinez et al 33 obtained inhibition halos of 10.90 mm of the aqueous extract of Rosmarinus officinalis against Escherichia coli. On the other hand, Llanos et al 34, demonstrated the antibacterial activity of the mixture of essential oils of Origanum vulgare and Rosmarinus officinalis at 50 %, obtaining inhibition halos of 16 mm against Staphylococcus aureus, using the Kirby Bauer method. Rodenas et al 35 obtained inhibition halos of 15.81 mm of ethanolic extract of Rosmarinus officinalis obtained by maceration against Staphylococcus aureus. Sánchez et al 36, demonstrated the inhibitory effect of ethanolic extracts obtained by maceration of Rosmarinus officinalis against Staphylococcus aureus, finding inhibition halos of 20.88 mm.
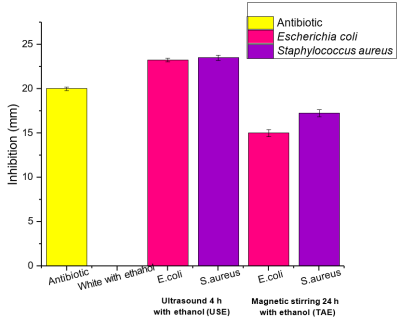
Fig. 5 Inhibition halos of Rosmarinus officinalis extracts against Staphylococcus aureus and Escherichia coli.
When comparing the inhibition halos of the extracts of Rosmarinus officinalis against the antibiotic, it can be observed that the halos of the extracts are greater, so that these extracts could be used in various applications due to the high demand for antibiotic resistance that is currently present.
The inhibitory activity of the ethanolic extract of Rosmarinus officinalis is attributed to rosmarinic acid, carnosol and carnosic acid. The mechanism of action of these compounds would be linked to inhibit the synthesis of the cell wall and membrane and also interfere in the synthesis of proteins and nucleic acids. Rosmarinic acid is the main compound to which the antimicrobial properties of Rosmarinus officinalis are attributed and which are enhanced by the flavonoids, since they increase the degree of liposolubility, which causes the separation of lipids from the cell membrane of the bacteria, disordering its structure and making it more permeable, thus allowing the filtration of ions and other cellular components by passive diffusion 37,38.
Obtaining the composite Characterization by infrared spectroscopy FTIR-ATR
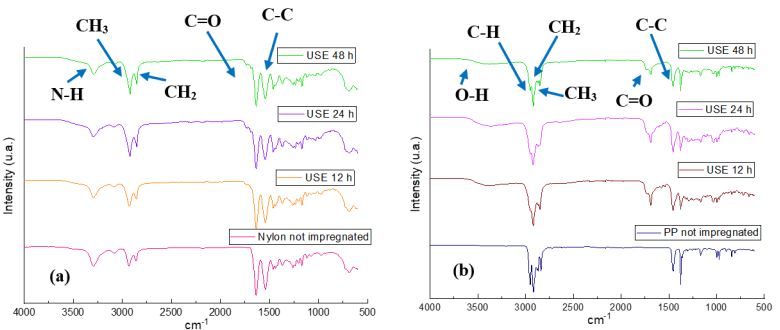
Fig. 6(a) shows the FTIR-ATR of Nylon 6 nonwoven fabrics impregnated for 12, 24 and 48 h with Rosmarinus officinalis extracts using a concentration of 10000 ppm of the extract. In which bands are shown at 3295 cm-1 corresponding to the stretching of the N-H bonds coming from amides present in the structure of the Nylon 6 nonwoven fabric, at 2931 and 2859 cm-1 two bands corresponding to the vibrations of -CH3 (asymmetric stretching) and of the methylene group -CH2 (symmetric stretching) are observed, at 1653 cm-1 the stretches of the carbonyl C=O bond coming from amides are shown, at 1539 cm-1 the bands corresponding to the bending of the N-H bonds are observed and at 1460 cm-1 a doublet corresponding to the C- N stretches coming from amides is shown. As can be observed independent of the impregnation time there are no changes in the chemical structure of nonwoven fabric after it is impregnated with the extracts of Rosmarinus officinalis, when compared with the infrared spectrum of the unimpregnated Nylon 6 nonwoven fabric. While in the incision (b) shows the infrared spectra of the PP fabrics impregnated with the extracts of Rosmarinus officinalis using a concentration of 10000 ppm of the extract, where at 2900 and 2950 cm-1 the bands corresponding to the C-H stretches coming from the polypropylene are shown, at 2850 cm-1 the bands corresponding to the vibrations - CH3 asymmetric stretching and at 2800 cm-1 the bands corresponding to the vibrations of the methylene group symmetric stretching are observed and at 1450 cm-1 the bands corresponding to the C-C17 bond tensions are found, In addition to these bands are shown others located at 3435 cm-1 which correspond to alcohols, phenols and at 1685 cm-1 is the band corresponding to the carbonyl groups present in the chemical structures of the secondary metabolites of the extract of Rosmarinus officinalis25.
Characterization by TGA thermogravimetric analysis
In Fig. 7, in section a) the thermograms of the Nylon 6 nonwoven fabrics impregnated with the extracts of Rosmarinus officinalis using a concentration of 10000 ppm are observed, where the unimpregnated fabric has only a loss in weight of 98 % at a temperature of 444.90 °C, while those impregnated with the ethanolic extracts of Rosmarinus officinalis present a very similar behavior, the first loss is between 120- 270 °C due to the loss of water, the second loss is between 250-400 °C due to the organic compounds present in the extract, as shown in the TGA of the extracts of Rosmarinus officinalis, which present the same behavior, finally the degradation of the fabrics begins around 450 °C.
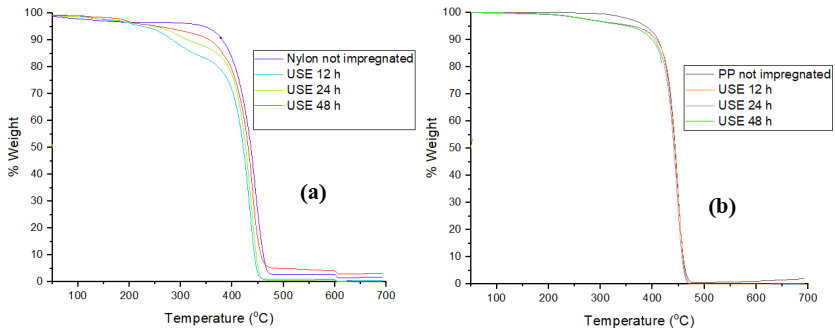
Fig. 7 Thermograms of nonwoven fabrics impregnated for 12, 24 and 48 h with Rosmarinus officinalis extracts, using a concentration of 5000 ppm: (a) Nylon 6 nonwoven fabric and (b) Polypropylene.
On the other hand, in section b) we have the PP nonwoven fabrics impregnated with the extracts of Rosmarinus officinalis, with a concentration of 5000 ppm, as can be observed, they have a very similar behavior, both the PP fabric without impregnation and those impregnated with the extract. In the unimpregnated fabric a loss in weight of 95 % is observed at a temperature of 449.11 °C, while in the impregnated fabrics a first loss is observed between 200 -270 ºC which is due to water, the second loss is mainly attributed to the loss of organic compounds and is found between 400-450 ºC and the total decomposition of the material occurs at a temperature of 450 ºC.
In conclusion, there is no improvement in the degradation temperature of the impregnated fabrics when compared to the unimpregnated fabric, however, these results show that the impregnated fabrics withstand temperatures above 400 ºC, which makes them excellent candidates for various applications.
In vitro antibiosis bioassay of Nylon 6 and polypropylene nonwoven fabrics impregnated with Rosmarinus officinalis extracts
The largest inhibition halos occurred on Nylon 6 and PP fabrics impregnated with a concentration of 10000 ppm of Rosmarinus officinalis extract. Inhibition halos of 18 mm were obtained for USE 12 h, 19 mm for USE 24 h and 20 mm for USE 48 h, while in the fabrics impregnated with the extracts obtained by magnetic stirring, halos of 8 mm were obtained for TAE 12 h, 6 mm for TAE 24 h and 9 mm for TAE 48 h, as shown in Fig. 8.
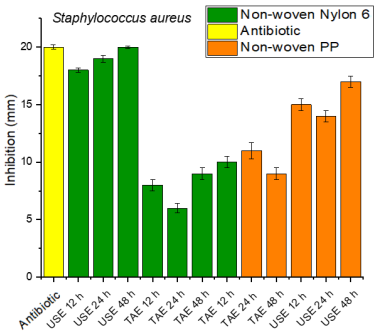
Fig. 8 Inhibition halos against Staphylococcus aureus of Nylon 6 and Polypropylene nonwoven fabrics impregnated with a concentration of 10000 ppm of the extract.
While in PP nonwoven fabrics impregnated with the extracts obtained by magnetic stirring, halos of, 10 mm TAE 12 h, 11 mm TAE 24 h and 9 mm for TAE 48 h were obtained, while in the fabrics impregnated with the extracts obtained by ultrasound, halos of 15 mm were obtained in USE 12 h, 14 mm USE 24 h and 17 mm USE 48 h, as shown in Fig. 7. Andrade et al 39 tested the antibacterial capacity of Nylon nonwoven fabrics impregnated with ZnO NPs, against Staphylococcus aureus, finding inhibition halos of 7 mm.
Nylon 6 fabrics impregnated with ethanolic extracts of Rosmarinus officinalis obtained by ultrasound showed halos very similar to the antibiotic, which leads us to conclude that they have excellent antimicrobial properties against Staphylococcus aureus, so this type of fabrics could be considered for the manufacture of drapes or surgical gowns and their subsequent use in hospitals.
Conclusions
Bioactive compounds such as rosmarinic acid, carnosol, rosmanol, among others, were obtained from the dried leaves of Rosmarinus officinalis, finding that the best extracting solvent was ethanol with 14.67 %. Likewise, it was possible to obtain a composite with antibacterial properties against Staphylococcus aureus, demonstrating that when Nylon 6 and PP fabrics are impregnated with Rosmarinus officinalis extracts, they do not suffer alterations in their chemical structures, making these composites excellent candidates for the manufacture of surgical clothing and their subsequent use in hospitals for the control of nosocomial infections.











 nueva página del texto (beta)
nueva página del texto (beta)