Introduction
Catalysis became crucial with the growth of the chemical industry through the last decades; 85 %- 90 % of these processes include at least one catalytic step to achieve them in a more sustainable way [1-3]. A catalyst is defined as a substance that increases the rate of a particular reaction under mild or soft reaction conditions without being part of the product. One of the most important aims for scientists who study this field is to design catalysts with highly active catalytic sites. These sites normally define whether the desired reaction will proceed with a higher rate and/or selectivity to avoid the formation of unwanted sub-products and under relatively mild conditions in comparison with a non-catalyzed reaction [3,4].
The catalyzed reactions can be carried out in a homogeneous manner where the reactants and the catalyst are in the same phase, usually in the liquid phase. Homogeneous catalysis has the advantage of allowing precise control of reaction conditions, which is very important in organic chemistry and the pharmaceutic industry [5]. Li and coworkers presented the synthesis of γ- Valerolactone using Sc(OTf)3 as an efficient homogeneous catalyst, obtaining yields over 90 % in this compound that is key in some industries related to green chemistry and biomass refinery [6]. However, this catalysis is less stable at high temperatures, and the catalyst is difficult to recover from the reaction mixture and, thereby, can elevate the cost of the process [5].
Heterogeneous catalysts are systems that present the property of being in a different phase as the reactants. In heterogeneous catalysis, the reactants (liquid, gas) adsorb onto the surface of a solid catalyst [5]. The advantages of these types of systems are the stability to high temperatures and easy removal from the reaction medium, reducing the overall cost, which is of great importance for industries such as oil and gas refining and wastewater, among others [7]. Thus, heterogeneous catalysis plays an integral role in producing over 80 % of chemical products worldwide. These catalysts can be synthesized with the control of important properties like particle size and morphology. However, a key feature in these systems is controlling the porosity because of the intimate relationship between the activity and product selectivity. The pore dimensions of solid catalysts can directly influence the diffusion properties of reactive species during a chemical process [8,9].
Metal nanoparticles have been used in catalysis since the 1940´s to the date, starting with precious metals like gold, silver, platinum or palladium and changing to the use of less expensive metals like nickel, cobalt or iron. Although these precious materials have been used for important reactions like the hydrogenation of polyvinyl alcohols [10], degradation of dyes in water like xylenol orange [11] or the oxidation of CO to CO2 [12]; and some of these nanoparticles are stabilized by weak linkers [13], an alternative to improve the activity of these materials and the use of less precious metals was presented when the catalyst was supported in other materials (solid matrix). The support can be chemically inert or active [14], resulting in interest when is catalytically active because it can take advantage of the inherent properties of the material, such as high surface area to interact and easy separation and reuse [13]. Traditional heterogeneous catalysts, such as metal oxides supported on a surface, are systems susceptible to dissolution and ion release, which can lead to environmental problems. 15 Furthermore, they possess lower turnover frequencies than homogeneous systems due to the non-homogeneous distribution of the catalytic sites [16]. Thus, a new wave of porous catalytic solids has been developed, such as silica-based materials, zeolites, and metal-organic frameworks (MOFs). Due to their predictable and designable long-range ordered structures, guest-accessible pores, and easily functionalized channels by pre- or post-modifications, decades of research on these materials have furnished numerous porous catalysts that are of substantial scientific and industrial interest [17,18]. In this review, we highlight some of these porous materials and their applications in heterogeneous catalysis. These materials have been studied for decades with promising results, presenting an alternative to conventional unsupported catalysts. Although there are some reviews related to this topic in the literature, we focus on the important contributions of research groups at the “Universidad Autónoma Metropolitana” (UAM) in this field over the years.
Silica-based materials in heterogeneous catalysis
In the 1990s, there was a great impulse in the investigation of mesoporous materials as heterogeneous catalysts, the synthesis of mesoporous silicate and aluminosilicate molecular sieves opened new possibilities for preparing catalysts with uniform pores 19, confinement of the catalyst within mesopores provided a means of introducing size and/or shape selectivity and greater specificity to reactions [20]. Later in the '90s, specifically in 1997, it was reported the possibility of generating Brønsted acid sites on the surface of the mesoporous structures, opening new possibilities for producing monofunctional acid as well as acid/metal oxide bifunctional catalysts. These molecular sieves presented very high surface areas, which were of great utility because they can act as support for catalytically active phases such as metal oxide complexes, among others [21].
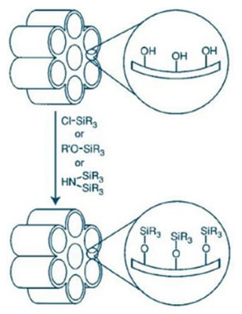
A widely employed material is the hexagonal MCM-41 (Mobil Composition of Matter), which became the focus for scientists due to its potential applications, both as a catalyst and as support [22]. In 2002, the encapsulation of copper complexes within MCM-41 and the partial oxidation of phenol was reported. The system demonstrated to be a good support and an excellent selective catalyst to produce catechol. Moreover, the absence of leaching copper through the process was observed [23]. A major limitation of this catalyst is its use in acidic conditions, as silica materials have low intrinsic stability in this environment. To overcome this problem, scientists studied the incorporation of strong acidic functional groups, like alkyl- or aryl-sulfonic acid, on the silica surface. It has been reported that these groups produce higher numbers of improved acid centres and enhance the activity in reactions catalyzed by the acid function. In 2005, Boveri and collaborators reported a method to prepare HSO3-functionalized MCM-41, as shown in Fig. 1; the materials were tested as catalysts for the reaction of benzene alkylation with 1-dodecane with moderate activity and good selectivity to 2-phenyl dodecane [24]. This methodology was also evaluated in the catalysis of stearic acid with glycerol, presenting interesting results like the selectivity to monoglyceride and the inhibition of di- and triglycerides, and the relation between the pore size and the increment of the activity in the reaction [25]. In 2010, the incorporation of Brønsted acid sites from HSO3 to Co-Mo/MCM-41 catalyst was presented in the treatment of heavy oil, performing sulfur removal and improving the properties of the expected products [26].
A further method to improve the acidity of the material is the incorporation of metals such as Al+3 or Ga+3 in the material MCM-41 tested with the isomerization of α-pinene reaction where the main products were camphene and limonene, and the selectivity to one product or the other could be varied depending on the content of Al or Ga ions [27]. Recently, these systems have been used in many different potential industrial applications, such as the production of biodiesel, where the basicity or acidity of these materials should play an important role in this process. These materials can also be used to immobilize enzymes or magnetic nanoparticles for the catalytic degradation of organic dyes in contaminated water. A novel application for silica-based materials is their use as support for anti-cancer drugs; for instance, some of these materials have been functionalized with different drugs or active proteins, and these systems showed good results in the elimination of the infected cells and presented null toxicity for the healthy ones [28,29].
Silica-based materials were one of the first porous materials studied in catalysis due to their large surface area reported in the 90´s decade, especially MCM-41, which was evaluated in several reactions presenting good results in the catalytic activity. Modifications were made in the material to overcome acidity problems; the addition of metal nanoparticles is another way to expand the activity of the catalyst. These materials were the pioneers in these applications and landed the foundations for other materials to be studied as heterogeneous catalysts, such as zeolites and Metal-organic Frameworks (MOFs) that we will discuss in the next topics.
Zeolites in heterogeneous catalysis
Zeolites are porous crystalline oxides consisting of TO4 tetrahedra, where T is generally Si, but it can be substituted for Al or other heteroatoms [17,30,31]. The tridimensional networks of well-defined micropores act as reactor vessels whose activity and selectivity can be enhanced by introducing catalytic active sites [32]. The success of these catalysts is related to their specific properties that rely on their surface area and adsorption capacity. Zeolites can be modified to control their adsorption properties, they can be varied from hydrophobic to hydrophilic type materials and active sites [21,32].
The control of acid strength, as well as the density of acid sites, has also led these materials to be successful catalysts; doping these materials with other atoms like aluminum can create protons that have Brønsted acidity and demonstrate unique properties for adsorption and catalysis [33]. Furthermore, in catalysis, it is of special importance for catalyzing reactions involving strong bases such as NH3 or pyridines [32]. The use of Zn has been reported in the methanol conversion to aromatics, showing great selectivity to aromatic compounds like benzene, toluene and xylene. The introduction of Zn particles in zeolites like ZSM-5 (Zeolite Socony Mobil) [34,35], MCM-22, TNU- 9 (Taejon National University) and MCM-68 [36], has shown the generation of strong acid sites while maintaining stability at high temperatures.
It is also possible to generate basic sites within the pores of zeolites to use them as catalytic active sites. The basic sites are of the Lewis type and correspond to framework oxygens, or they can also be exchanged with alkali metals. These systems have been reported to efficiently catalyze a variety of reactions, including Knoevenagel condensation, amide hydroxylation, one- pot multicomponent reactions (MCRs) and cycloaddition reactions [37]. An interesting feature of basic zeolites is that they are useful catalysts for some reactions that require acid-base pairs. In this situation, the Lewis acidity of the cation and the basicity of the oxygen can be balanced [32]. The use of Ni particles in zeolite ZSM-5 has been applied in CO2 methanation (conversion of carbon monoxide and carbon dioxide to methane through hydrogenation), presenting 99 % conversion related to weak and medium basic sites within the material [38].
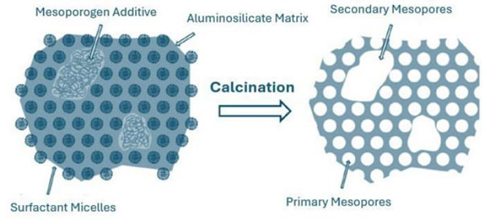
Controlling the size of the pores is a property that can be useful when the molecules cannot access or leave the pores of the zeolite. A reported strategy is to produce mesopores within the channels of these systems with the addition of agents that can generate mesopores in the zeolites (mesoporogenic agents), as shown in Fig. 2 [40]. In 2014, a study presented the synthesis of zeolite ZSM-5 with the addition of carbon nanoparticles and chitosan (biopolymer derived from crustacean shells); these additives changed the Si/Al ratio in the material and improved the pore sizes and volumes compared to the pure zeolite. The materials showed an interesting route for the catalytic cracking of low-density polyethylene (LDPE) to fuel [39].
Commonly, bulky molecules are of interest in catalysis; the reaction takes place only on the surface of the material, limiting the interaction with the reactants [41-43]. Another inconvenience is the irreversible adsorption of heavy secondary products or bulky molecules of products that may be trapped inside the cavities and lead to the deactivation of the catalyst [44]. New investigations are in progress to develop large pores zeolites; however, the synthesis of these materials is costly due to the use of expensive templates or multistep procedures [45]. More importantly, the chemical compositions for zeolites are limited to aluminosilicates, which can accommodate only marginal amounts of transition metals, primarily as defects [46]. Studies have shown us that zeolites can be competitive catalysts thanks to their properties. Nevertheless, more investigations and modifications must be performed on these materials so they can be applied to other industrial processes. The advances in technology can help them to play an interesting role in the transition to better technologies.
MOFs in heterogeneous catalysis
Metal-organic frameworks (MOFs) have attracted immense attention during the last two decades due to their potential to produce a wide variety of structures [47]. MOFs are constructed from metal cations (or clusters of metal cations) linked to one another by bridging organic linkers. The wide variety of these materials motivated research groups to seek a broad spectrum of applications such as gas storage, gas separation, and catalysis [48], among many others. There is continuous interest in seeking new MOF materials because these systems possess many outstanding features that commend them as catalysts, such as their large surface areas, adjustable pore sizes, and tunable properties [49,50].
MOFs have shown advantages over other porous materials, for instance, their crystalline morphology, unlike siliceous mesoporous materials which cannot be investigated by crystallographic techniques. MOFs can be modified using organic synthesis, and their pores may be decorated with catalytic sites, unlike some zeolites whose structures are fixed and unalterable. MOF materials show a balanced mix of crystallinity, porosity, and tunability, and there is significant potential for the commercial application of such materials in heterogeneous catalysis [51]. Some of the modifications can occur when the ligand is used to attach other active groups, such as Brønsted acid centres (sulfonic acid), basic centres (amino) or appropriate binding sites, which may engage in the development of metal complexes at lateral sites and implement some unique catalytic performance [52].
Another approach is to use the metal nodes of the MOF framework, as normally, these metal complexes possess solvent labile molecules coordinated, which can be removed to produce unsaturated metal sites. A common activation method is to expose the material to a vacuum at a certain temperature. Nevertheless, it has been reported that more methods, such as chemical activation, succeed in this task [53]. In several other instances, structural defects and imperfections might also insert certain coordinately unsaturated sites across the metal centres [52].
In 2023 several studies were made to demonstrate that these open metal sites could work in the alcoholysis of cyclohexene oxide using MOFs as catalysts. SU-101 (Stockholm University) presented conversion over 99 % with methanol, over 96 % with ethanol and over 24 % with propanol; the material presented retention of the catalytic activity over 5 cycles and a mechanism was proposed involving the Bi+3 open metal sites in the MOF [54], MOF-74 presented over 90 % conversion using methanol at 50 °C in less than 2 days, presenting no degradation, leaching or loss in the catalytic activity over 3 cycles [55] the mechanism is shown in Fig. 3. MOF-74 has also been used as a catalyst with different metal sites in the oxidation of styrene to benzaldehyde using peroxides as oxidants, where Cu-MOF-74 reported the highest yield, and Zn-MOF-74 acted as an inhibitor rather than as a real catalyst [56].
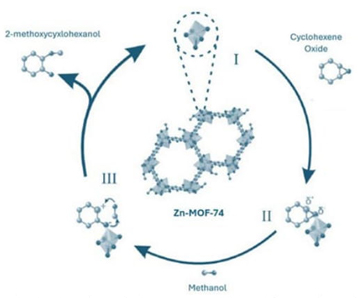
Fig. 3 Steps proposed for the alcoholysis of cyclohexene oxide with methanol catalyzed by Zn-MOF-74 55.
One of the most challenging processes in the industry is the production of vanillin; only 1 % corresponds to natural extraction, and the rest of the production is obtained by chemical synthesis. In the effort to facilitate the production of this molecule, the use of catalysts such as MOFs to produce vanillin by the transformation of trans-ferulic acid has been reported in this process. HKUST-1 (Hong Kong University of Science and Technology) was one of the first materials to be tested in this synthesis; the results showed that this material can be used for the effective conversion to vanillin with 95 % yield using hydrogen peroxide 57; later, a Cu(II) metal-organic polyhedron was tested, presenting a 60 % yield, but the material lost its crystallinity; however, no copper leaching in the reaction was presented [58]. Finally, Cu-MOF-74 presented yields between 71 and 97 % for vanillin production though the reaction presented copper leaching and the disruption of the framework structure [59]. This demonstrates that vanillin can be successfully synthesized using MOFs for this reaction; however, the reaction conditions and solvents need to be established before scaling up the reaction.
On the other hand, there can be semi-open metal sites where the linker offers the ability to switch on or switch off the active sites on the MOF. A good example is the use of MOF MFM-300(Sc) (Manchester Framework Material) as a catalyst for the Strecker reaction between N- Benzylideneaniline and trimethylsilyl cyanide (TMSCN) to form 2-phenyl-2-(phenylamino)acetonitrile, reported in 2022 by Peralta and collaborators. It was demonstrated that the material showed retention of the crystalline structure and catalytic activity over 5 cycles [60]. This same strategy was used by López-Olvera and collaborators with the MFM-300(Sc) material as a catalyst to transform H2S into polysulfides, compounds that can be used in other fields like battery electrodes [61].
One thing to take into consideration is the thermal stability of the material; under certain conditions, the structural integrity of MOFs can be compromised; such processes include amorphization or framework collapse. That changes the physical and chemical properties of a material, making it inappropriate for catalytic applications [62,63]. The election of the organic linker and metal plays a key role in the stability of the framework [62]; therefore it is crucial for the catalytic process. The design of new MOFs that can tolerate high temperatures should be considered an area of opportunity for many applications, not only catalysis.
Overall, research on MOFs in catalysis has continuously increased in the last decade thanks to their inherent properties. A salient feature of MOFs is their large surface area and their relationship with the catalytic activity, along with the robustness of the systems. For instance, gas adsorption and separation are classic MOF applications due to the pore size and stability. This last property depends on the strength of the metal−linker bond; the research of new MOFs with more robust frameworks could present a solution. Nevertheless, the structural and compositional complexity is a challenge that must be considered.
On the other hand, the challenge for heterogeneous catalysts is the loss of reactivity or selectivity; MOFs can present a solution with their open-metal sites that can act as acid or basic sites. Encapsulation of enzymes could be another solution to this problem. However, these systems have been barely studied, and considerations must be realized, such as the enzyme leaching within the framework due to a large pore of the MOF. It is worth mentioning that encapsulation could also lead to a decrease in porosity and thereby generate mass transport problems [46]. Porous materials could be a solution to the difficulties of the industry in the coming years, and the present review intends to show their importance and potential applications.
Conclusions
Porous materials have shown interesting advances in catalytic applications. Pre- and post-synthetic modifications can improve the selectivity in some reactions; these materials can also work as supports for other catalysts, improving their activity, which makes them perfect candidates for this type of application.
Silica-based materials were one of the first porous materials used in catalysts. Their activity was tested in important reactions, and modifications were made in their structure to overcome important problems in their use. Zeolites presented an improvement in porous materials with the presence of acid or basic sites, leading to interesting catalytic applications. Finally, MOFs have been one of the most studied porous materials in the last two decades thanks to their high crystallinity, large surface area, tuneable properties and adjustable pores. The catalytic process can occur within the pores of the MOFs thanks to the open metal sites or functional groups that act as acids or basic centres.
These materials have many possible applications, like gas adsorption, which is one of the most common since many industrial processes produce gases such as CO2 and H2S, which are harmful to the environment. Porous materials can also be applied to catalytically degrade contaminants, organic dyes, or other emergent pollutants like plastic and drug residues. Some of the other applications investigated for these materials are energy storage, drug delivery, and the sensing or detection of contaminants.
It is important to mention that there is still a lot of work to do before this can be applied in the industrial field; reaction conditions and solvent issues need to be tested so the materials can retain their structure and the catalyst can be easily recovered to be used again. Another alternative is the synthesis of new and more robust materials that can endure the harsh conditions that the industrial field presents. Understanding these materials is important to gain perspective on their significance in possible real-world applications.











 nueva página del texto (beta)
nueva página del texto (beta)