Introduction
The first theory of metallic sodium and a method to calculate the binding properties of metals was put forward by Wigner and Seitz since 1933.[1,2] Their work gave rise to the celebrated Wigner-Seitz cell model, whereby each atom in the crystal lattice is surrounded by a polyhedron formed by bisecting planes between the atom and its neighbors. Without further details, we deem this was the first confined system studied, which led to the first serious band structure calculations of metallic sodium. A few years later, Michels, De Boer and Bijl,[3] followed by Sommerfeld and Welker,[4] studied how the polarizability of the hydrogen atom varies when subjected to very high external pressures. Their work proposed that the hydrogen atom is enclosed in a spherical cavity of radius r 0 with impenetrable walls, which simulate the effects of the rest of the negative charges surrounding the atom. They solved approximately the Schrödinger equation for a set of states, and with an approximate wave function they calculated the polarizability through Kirkwood’s method.[5] We can say that these fundamental studies constitute a milestone for the quest for properties of confined quantum systems, which has become an active field of research in physics and chemistry worldwide.[6-9]
Noteworthy in this aspect, the significant contribution from professors and students of the Universidad Autónoma Metropolitana (UAM) - Iztapalapa is hereby acknowledged. Since the establishment of UAM 50 years ago, many research groups in this institution have made important contributions to science in different fields of knowledge. Particularly, in the case of the study of electronic properties of atomic and molecular systems submitted to non-conventional conditions, such as quantum confinement, the chemistry department and the physics department have combined efforts to generate new ideas and techniques to tackle various related problems. It is worth mentioning here that this research activity has promoted appropriate local and international meetings around this topic. This article appraises the efforts developed by our research groups at UAM-Iztapalapa to study confinement effects on the electronic properties of atomic and molecular systems. Of course, this summary is not meant to be exhaustive but to provide an insight into our main contributions to the knowledge and applications of confined quantum systems.
The starting point is the time-independent Schrödinger equation within the Born-Oppenheimer approximation
With
where N and M represent the number of electrons and nuclei, respectively, and positions of nuclei with atomic charge Z A are represented by R A . The position of electrons are represented by r i . In the context of this article, vc is quite important since this term represents the confinement imposed over the system. For molecules, there is an additional term to the total energy within the Born-Oppenheimer approximation that takes into account the interaction among nuclei,
Atomic units (a. u.) will be considered in this article.
The confinement models to be discussed here consist of closed or open geometric boundaries defining vc, whose barrier height may be infinite (hard wall confinement) or finite (soft wall confinement). For clarity of presentation, in the following sections, we shall refer separately to our achievements for hard wall and soft wall confinement in all cases.
Confinement imposed by hard walls
In this Section, we present an account of our relevant contributions concerning hard wall confinement for various atomic and molecular systems spatially limited by closed and open geometric boundaries of different shapes. We begin with the hydrogenic atom, which is the simplest yet fundamental system in the Periodic Table. Then, we proceed to the two-electron helium-like system, many-electron atoms, and molecular systems.
Hydrogen atom
Historically, the confinement imposed over a hydrogen atom was based on a model in which the hydrogen nucleus is clamped at the center of an impenetrable sphere, and the electron moves in the sphere under the coulombic attraction of the nucleus. The important ingredient in this model is an infinite potential on the sphere’s surface such that the wave function or electron density is canceled on the surface. In mathematical terms, the wave function must satisfy the Dirichlet boundary conditions. The confined hydrogen atom has spherical symmetry, so as in any central potential, the angular solution of the Schrödinger equation is given by the spherical harmonics Y l,m (θ,ϕ). Therefore, it is only necessary to solve the radial Schrödinger equation subject to the boundary conditions.
Researchers who worked on this problem before 1979, approximating the solution of the Schrödinger equation, could not obtain the energies accurately. They obtained 3-4 decimal places approximately in the energies calculations and a smaller number in the physical properties. Since the late 1970s, significant efforts have been reported to solve the Schrödinger equation of the hydrogen atom enclosed by hard walls.[10-12] Precisely, within these efforts, the UAM-Iztapalapa started to work with contributions in this field.
On one hand, the variational method was used with an elegant wave function, constructed as the product of a wave function of the free system multiplied by a cut-off function to ensure that the boundary condition is satisfied to obtain a good approximation to the energy of the confined system.[11,13] On the other hand, a series method gave a high-accuracy numerical approximation; the energies obtained by this method reported energies with 10-11 accuracy figures, giving energies below those reported previously.[12,14] One element to note in the used series method is that the wave function, in addition to depending on the coordinates, also depends explicitly on the energy.[12] In a later paper,[14,15] the power series method and a solution based on the confluent hypergeometric function were used to obtain energy with an error of less than 1 × 10-100. Although the hydrogen atom has one electron, this system exhibits many characteristics shown by many-electron atoms, which will be discussed below. For example, the orbital energy appears to cross between different states when the atom is under extreme confinement. If the hydrogen atom is confined at r 0 = 1.0 a. u. the orbital energy ordering is ϵ 1s < ϵ 2p < ϵ 3d < ϵ 2s ..., where the energy degeneracy disappears. The common electron configuration ordering no longer exists16 as can be appreciated from Fig. 1.
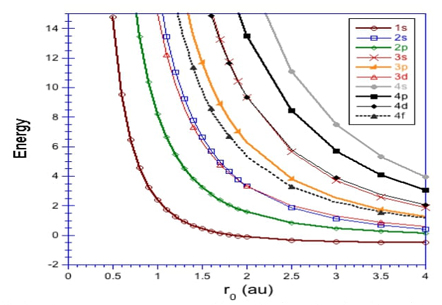
Fig. 1 Orbital energies for the hydrogen atom confined by spherical hard walls with several confinement radii r 0.
The confined quantum systems, particularly the spherically confined hydrogen atom, show abrupt changes in their physical properties when the size of the cavity containing them is reduced, i.e., as the pressure grows. The magnetic screening constant, Fermi contact term, polarizability, and the pressure as a function of the cavity radius r 0 are depicted in Fig. 2.
From Fig. 2, as the radius r 0 decreases the magnetic screening constant, the Fermi contact term and pressure increase rapidly, whereas the polarizability decreases quickly, indicating that the confined hydrogen atom is harder than the corresponding free atom. For these reasons, the confined hydrogen atom limited by spherical hard walls is a system of continuing interest.[14,15,17-23]
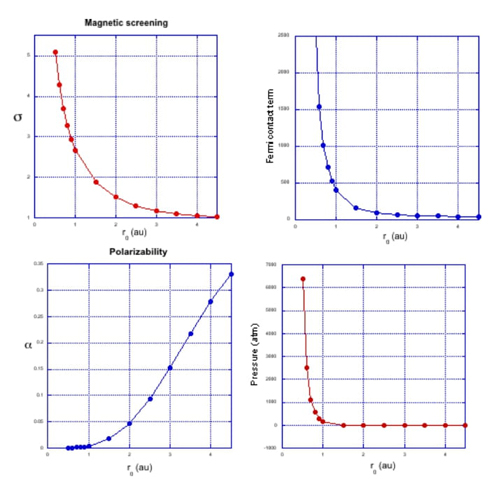
Fig. 2 Magnetic screening constant σ, Fermi contact term, polarizability and pressure for the ground state of the hydrogen atom confined by spherical hard walls with several confinement radii r 0.
The hydrogen atom has also been studied under spatial limitation due to closed and open hard walls with different shapes.[24-28] We must mention that the case of single atoms localized at arbitrary positions along the major axis of ellipsoids is implicit within the treatment of diatomic molecules. For this reason, the hydrogen atom is also considered when the molecular systems H2 or H2 + are under study.[29] The Fermi contact term, which has a direct relationship with the electron density at the nucleus, is one crucial result since this quantity is reduced for extreme confinements.[24] Thus, not for all confinement shapes is the electron density accumulated at the nuclei when the system is confined under small regions. Additional confinement shapes have been considered for the hydrogen atom.[30-36]
Many-electron atoms
The study of the electronic structure of many-electron atoms is a challenge since there is no analytical solution for the corresponding Schrödinger equation because the electron-electron interaction is present in this equation. Researchers have developed computational codes to obtain the wave function or electron density of atoms and molecules, and the number of these codes has increased with the arrival of new hardware. However, several of the mentioned computational codes have been designed to obtain wave function or electron density without spatial restrictions. Thus, confined many-electron systems pose additional difficulties in obtaining their electronic structure under these circumstances. Naturally, the helium atom is a good candidate for this class for elucidating changes in its electronic structure under hard-wall confinement. Using a simple variational ansatz for the wave function, the total ground state energy has been estimated for this atom confined by a sphere with hard walls.[11,37] With more sophisticated methods, such as those based on correlated Hylleraas functions, the total energy has been reported with high accuracy for the ground state and the first triplet state of this confined atom.[38-39] Also the correlation energy of this system has been estimated by several techniques.[14,40-46] The direct variational method or the use of correlated Hylleraas functions is limited to atoms with small number of electrons. Thus, methods designed to study many-electron systems, like Density Functional Theory (DFT) or Hartree-Fock (HF) method, have been applied to these systems by implementing new numerical codes. Along with DFT, there are several proposals, Thomas-Fermi approaches, [47-50] Kohn-Sham model, [51,52] or wave function methods.[40,43,44,52-55]
Estimations of confinement radii where atoms are ionized [54,56] and how the shell structure [57] is affected by this confinement are examples where the design of new computational codes exhibits their value. The s−d transition observed experimentally in some alkali-metals has also been observed as electronic configuration transitions [58-60] by using this confinement model. Chemical reactivity indices defined in the context of the DFT have been analyzed and show that for extreme confinement, an atom is not infinitely hard since the electronic transitions are such that the atom goes to states where it is soft.[61] Thus, this simple confinement model gives insight into how the electronic structure of atoms reacts under spatial restrictions.
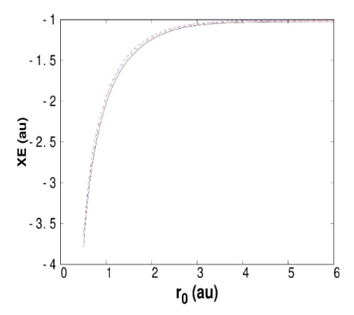
Fig. 3 Exchange energy, XE, for helium atom confined by hard walls obtained by Hartree- Fock (solid line), PBE (dashed line) and hybrid exchange functional with 50 % of Hartree- Fock (dot-dashed line).
It is well known that within the DFT, the exact exchange-correlation functional is unknown, and consequently, many approximations are used to account for the electron-electron interaction. For the confinement imposed by hard walls, the exchange energy delivered by some functionals is similar to that provided by the HF method. The behavior of the exchange energy obtained by the Perdew-Burke-Ernzerhob (PBE) exchange functional is similar to that observed by the HF method, as can be seen from Fig. 3. In the same plot, the half-and-half (0.5 of HF and 0.5 of PBE9 method shows behavior similar to that provided by PBE and HF. For this confinement, the HF exchange energy is the lower limit, and any of the approximations based on PBE are above HF results.[62] Thus, PBE exchange functional and hybrid functionals built with these approximations are appropriate for studying atoms confined by hard walls.
Several applications have been reported for many-electron atoms under the confinement imposed by hard walls defined either by closed or open boundaries. In one case, the pressure dependence of the mean excitation energy of bulk atoms in a given material has been estimated to account for the rate of energy loss of heavy ions penetrating the material.[63,64] In the case of hard open boundaries, the surface scattering potential of helium atoms close to a planar boundary is related to the ground-state energy shift imposed by the spatial restriction due to the plane. This approach has also been extended to study the scattering and penetration of heavy ions in nanolayered materials.[65,66]
Molecules
Some molecular systems have been studied under spatial restrictions. In this connection, the simplest molecular system under study is the one-electron diatomic molecular ion H2 + and its isomers like HeH++, confined by a hard spheroidal cavity. For convenience, the Schrödinger equation is written in terms of prolate spheroidal coordinates that incorporate the intrinsic symmetry involved in diatomic molecules.[24] This set of coordinates consists of families of mutually orthogonal confocal spheroids, ξ, and hyperboloids, η, such that fixed values of the variable ξ ∈ [1, ∞) define ellipsoids of revolution around the internuclear axis with eccentricity 1/ξ.
In this approach, the Dirichlet boundary conditions are satisfied when the wave function is canceled at an arbitrary ξ 0, which defines the confining cavity. When the nuclear positions coincide with the foci, the Schrödinger equation becomes separable, and analytical methods deliver exact results for the total energy and the corresponding wave function.[24] However, when the nuclear positions are set arbitrarily along the major axis, the Schrödinger equation becomes non-separable, and approximate methods such as the variational one have been used to obtain appropriate results.[26] The total energy of this system exhibits important changes when ξ 0 is reduced since it acquires large values when ξ 0 → 1.[24]
Using the exact electron density, the chemical bond of the H2 + molecule has been studied under extreme conditions.[67,68] The electron density along the internuclear axis is depicted in Fig. 4. In this case, the hydrogen atoms are localized at z = ±1 a.u. The electron density exhibits a cusp at the hydrogen nuclei positions for large values of ξ 0, corresponding to moderate confinements.
However, for strong confinements, ξ 0 close to 1, such a cusp disappears, and the electron density accumulates at the middle of the nuclei; the kinetic energy is very large, and the nuclei are incapable of binding the electron involved in this molecule.
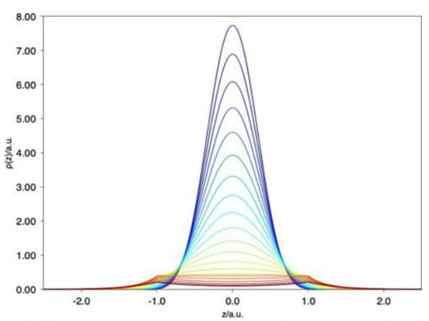
Fig. 4 Electron density of the H2 + molecule under several confinements defined by ξ 0. Hydrogen atoms are localized at z = ±1 a.u.
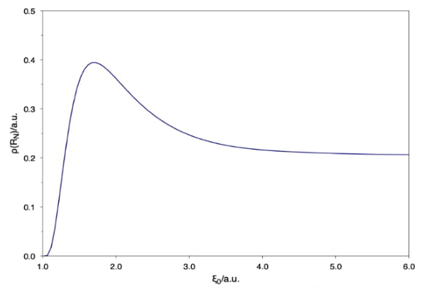
The electron density evaluated at a nucleus position ρ(R N ) as a function of ξ 0 is presented in Fig. 5. This figure shows that there is confinement where ρ(R N ) is a maximum, and it goes to zero when ξ 0 → 1, corroborating that the nuclei do not bind an electron for extreme confinements.
The H2 molecule involves the electron-electron interaction, so there is no exact solution, and consequently, the wave function must be approximated.[28,69] Estimating the correlation energy for the confined H2 molecule exhibits a behavior similar to that of the helium atom. It seems that two electrons confined under different circumstances respond similarly. Although the H2 + molecule contains only one electron, the behavior of the chemical bond is similar to that observed for the H2 molecule, which contains two electrons.[28] This result enhances the importance of the study of confined one-electron systems.
Concerning other studies of molecular confinement by hard spherical cavities, the use of the floating spherical Gaussian orbital (FSGO) representation for molecular orbitals allowed the calculation of pressure effects on the ground-state energy and molecular conformation of methane, ammonia and water as well as pressure-induced changes in the dipole moment of water and ammonia.[70,71] Finally, concerning molecular confinement by hard open boundaries, exact solutions of the Schrödinger equation for the H2 +and HeH2+ molecular ions, confined in dihedral angles and in the Born-Oppenheimer approximation, have been constructed, whereby the potential energy curves for the ground, first, and second excited states of the homonuclear H2 + molecule and for the metastable state of HeH2+ are reported illustrating their variations with the confining angle.[72]
Confinement imposed by soft walls
Hard wall confinement models constitute the first simple approach to exploring changes in the electronic properties of atoms and molecules due to spatial limitation. However, the confinement imposed by hard walls is known to overestimate, in general, the change in these properties compared to more realistic situations. Therefore, confining potentials with finite barrier height have been proposed as more appropriate models. This confinement is called finite potential, penetrable walls, soft walls or padded walls. This type of potential takes into account, in some way, the attractive forces between particles, such as the van der Waals force, among others. The potential v c = ∞ used for the hard wall confinement is replaced by one where v c =v o and the treatment of the Schrödinger equation is radically different from those used for hard wall. For this case, the logarithmic derivative of the wave function must be the same on the boundaries that define the confinement instead of the Dirichlet boundary conditions.
Hydrogen atom
Again, the hydrogen atom has been a hallmark study system for this kind of confinement. Consider a hydrogenic system with nuclear position of charge Z, clamped at the center of a spherical soft wall cavity of radius r 0 and barrier height v 0. In this case, the most commonly used confining potential is the step potential, which is defined as follows: Let r 0 be the radius of the spherical cavity, then, v c = 0 for 0 < r < r 0 and v c = v 0 for r > r 0, where v 0 is a constant. The corresponding Schrödinger equation has spherical symmetry, hence the angular solutions are the spherical harmonics Y l,m (θ, ϕ) and only the radial solutions have to be found. This step potential divides the space into two spherical regions, in the first one 0 < r < r 0, the radial solution is denoted by R in (r), while in the second, r > r 0, the radial solution is called R out (r). The interior and exterior solutions of the radial Schrödinger must be continuous with continuous derivative in the whole space, particularly at r = r 0. Then, the boundary conditions are: R in (r 0) = R out (r 0) and dR in /dr| r0 = dR out /dr| r0 .
There are several reports where this system is analyzed from different points of view. [10,14,73-76] One of the most successful approximate methods is the nonlinear variational method.[77] In that approach the authors use two different ansatz functions for each of the two regions of space, the energy is obtained by requiring that these two functions and their derivatives at r = r 0 are continuous, this implies the minimization of the energy functional in two variational parameters. The energies obtained by this procedure have 4 − 5 decimal places of accuracy for different values of r 0.
In another method, the authors [14] used a power series in the variable r, described in a previous section, to represent the wave function R in in the region 0 < r < r 0, and the exact solution for R out for the other region.[10] The energies obtained by this method have an accuracy of 10 digits. In another paper, the authors [15] used the exact wave functions for the internal and external solutions. Like most non-perturbative methods, the energies are obtained numerically by solving a transcendental equation. Using this approach, the authors obtained accurate energies for the ground state and some excited states. This method was also used to study the energy correction of a hydrogen atom confined in a penetrable spherical box by considering an atomic nucleus of finite size, [78] and to study the ground state energy of confined muonic atoms.[79]
For the hydrogen atom confined in a penetrable spherical cavity, a few physical properties such as magnetic screening constant, Fermi contact term, polarizability and pressure were calculated. Fig. 6 shows the behavior of these physical quantities as a function of the confinement radius and the barrier height v 0. In this figure, we can see the difference in the physical properties of the atom confined inside a penetrable barrier of different heights and an impenetrable one. The electron of an atom confined in a penetrable barrier escapes from the potential well as the confinement increases, i.e. as r 0 decreases. The consequence is that the magnetic shielding constant, the Fermi contact term, and the pressure do not grow indefinitely, whereas the polarizability grows quickly in this limit.
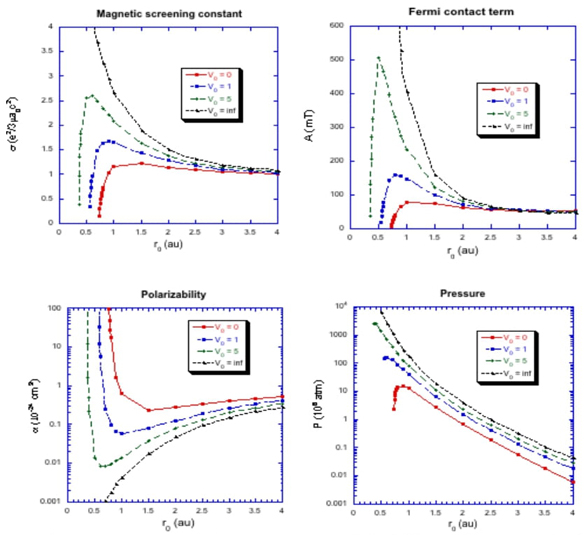
Fig. 6 Magnetic screening constant σ, Fermi contact term, polarizability and pressure for the ground state of the hydrogen atom confined by hard and soft walls as a function of the confinement radii r 0 .
Results from these studies show appreciable differences between hard and soft walls, where soft walls give a better idea about the confinement. We can mention how a finite potential allows the electron’s escape when the confinement is increased and how the information theory is useful to describe the delocalization of the electron density.[74,76] Similarly, for a given confinement radius and barrier height, the energy spectrum for all available bound states and several continuum states (pseudo-continuum) was obtained by solving the Schrödinger equation for the hydrogen atom within spherical soft and hard walls. The results indicate important differences in the behavior of the mean excitation energy of the confined atom as the confining barrier height changes.[80] Also, concerning other confining geometries with soft and hard walls, the bound-state energy spectrum and its evolution for a hydrogen atom located along the axis of a standard cylindrical confining cavity with either impenetrable or penetrable confining boundaries were obtained by solving the stationary Schrödinger equation using a finite differences approach. It was found that in general the energy levels evolve with an increasing value as the nuclear position is shifted from the central position up to a cylinder cap.[36,81]
Many-electron atoms
For many-electron atoms, numerical approaches are more sophisticated in this case than those used for the confinement imposed by hard walls.[46,52,62,77,82-86] Naturally, the richness delivered by many-electron atoms immersed within soft walls is impressive. For example, the shell structure of the beryllium atom is affected drastically when this atom is squeezed by a finite potential.[84,87] By contrasting the results obtained by DFT between hard and soft walls, approximations to DFT describe the exchange energy similarly to that presented by HF theory for confinement imposed by hard walls, since DFT exchange functionals and HF predict this quantity deeper when the confinement is increased. However, for confinement imposed by soft walls, there are appreciable differences between approximations of DFT and HF method.[62,85] In Fig. 7, there is a comparison between three exchange functionals (LDA, Becke88 and PBE) and HF method. From here, it is evident that the three exchange functionals predict an exchange energy above that predicted by HF for moderate confinement radii. However, this behavior is reversed for strong confinements where HF exchange energy exceeds the predictions of these three exchange functionals. This result suggests that people must carefully use computational codes that simulate systems under high pressure since not all DFT approximations work appropriately. It is worth noting that there are different ways to impose the confinement by soft walls, which are not limited to using model potentials.[86,88-90]
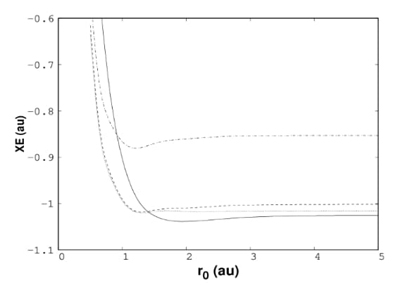
Fig. 7 Exchange energy, XE, for helium atom confined by soft walls obtained by Hartree-Fock (solid line), PBE (dashed line), Becke88 (dotted line) and LDA (dot-dashed line) exchange functionals.
Molecules
For molecular confinement imposed by soft walls only a few efforts have been put forward within the Born-Oppenheimer approximation, mainly to find the electronic structure of simple diatomic molecules under such conditions. A preliminary approach to model dense molecular hydrogen’s electronic properties and molecular conformation was proposed, where the H2 molecule is viewed as a caged-in system within a spherical boundary with finite potential barrier height v 0. A self-consistent treatment was employed using the FSGO representation of molecular orbitals. This molecular confinement model was put forward to calculate density effects on the rate of energy loss of energetic protons traversing dense hydrogen.[91] On the other hand, exact solutions to the corresponding Schrödinger equation have been reported for the hydrogen molecular ion H2 + enclosed by a penetrable spheroidal cavity with nuclear positions located at the focii.[92] When the nuclear positions are located arbitrarily along the major axis in this system, the Schrödinger equation is no longer separable. Hence, this non-separable problem for the confined H2 + and H2 molecules in their ground states was treated variationally.[93] In this way, given a finite height for the confining barrier potential, the independent variation of the nuclear positions from the cavity size and shape is allowed. In both cases, an important dependence of the equilibrium bond length and total energy on the confining barrier height is observed for fixed cavity sizes and shapes. As the cavity size is reduced, the limit of stability of the confined molecule is attained for a critical size. The advent of more powerful computing facilities makes the super-molecular approach for endohedral confinement studies a good alternative to obtain an idea of how the electronic structure can be changed in molecules enclosed by soft walls.[29,94]
Informational entropies
In the quantum theoretic information there are several measures and uncertainty relations, through which it is possible to find a deeper explanation of the physical properties of systems such as localization-delocalization, electronic correlation, quantum entanglement, avoided crossings, etc. The theoretic informational measures are functionals of the probability density, in configuration or momentum spaces. Some of those entropic measures are the Shannon entropy, Fisher information, Kulback-Leibler, Renyi and Tsallis entropies, disequilibrium, and, some entropic complexities. The Shannon entropy is a global measure, whereas the Fisher information is local and sensitive to local variations in probability density.
In confined systems, Shannon entropy and Fisher information are used as a measure of particle localization-delocalization in an atom or molecule.
Confinement imposed by hard walls
Studies about localization-delocalization have been made on the hydrogen atom, in two [32,33] and three dimensions,[73] the helium atom [95] and many-electron atoms.[87] In a spherical box with impenetrable walls, the atom cannot ionize as the pressure increases, decreasing the confining radius. So, as the external pressure increases, the electron density inside the box also increases, and the Shannon entropy in the configuration space decreases (localization). It was found that at the same box size closed-shell atoms have a lower Shannon entropy than the open-shell atoms. This indicates that closed-shell atoms have a more compact density than open-shell atoms.
Confinement imposed by soft walls
With this type of confinement, the Shannon entropy for the hydrogen atom [73,96] and many- electron [86] atoms have been mainly studied. For a finite barrier height v 0, as the pressure on the atom increases, the Shannon entropy decreases (localization) until it reaches a minimum value. As the pressure continues to increase, the Shannon entropy begins to increase rapidly (delocalization), and if the pressure continues to increase, there comes a point at which the electron escapes from the well (autoionization).
Challenges and perspectives
The study of atoms or molecules under confinement imposed by model potentials presents an intrinsic problem; there is no general computational code for obtaining the corresponding electronic structure. In our opinion, computational techniques like finite difference approx- imation or the finite element method (FEM) must be considered in large projects such as NWChem or Gaussian, to mention some examples, since these techniques are convenient for imposing different boundary conditions. Currently, in UAM-Iztapalapa there is an effort to solve HF or DFT using FEM for atoms and diatomic molecules confined by several potentials.[52] This is the first step in building a computational code to study the electronic structure of atoms and molecules confined by different potentials. This code opens many possibilities for studying the chemical bond of many-electron diatomic molecules under extreme conditions.
Concluding remarks
The present account of research efforts developed within the chemistry and physics departments at UAM-Iztapalapa in the past three decades toward understanding the behavior of confined quantum systems indicates a substantial degree of relevant contributions to knowledge in this field. This is a good theme to acknowledge and celebrate the 50th anniversary of UAM for the continuing support of our research activities along this time span. Of course, the growing interest of different research groups worldwide in this field makes it imperative to continue our efforts to contribute with timely new ideas.
The ideas and developments related to confined quantum systems by Professor Eugenio Ley-Koo have been crucial to our group. We dedicate this article to Eugenio in recognition of his contributions. We are deeply saddened by his passing and will greatly miss his lessons, seminars, and discussions on theoretical physics.











 nueva página del texto (beta)
nueva página del texto (beta)