Introduction
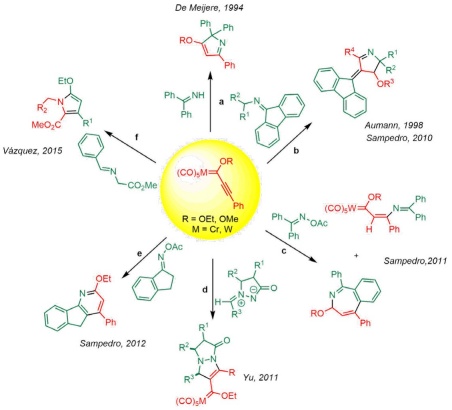
Transition metal Fischer carbene complexes undergo cycloaddition reactions with a variety of substrates, and the growing body of research on their use is creating a broad new field in chemistry [1-8]. Even the simplest carbene complexes have exhibited great versatility[9]. The scope of the reaction increases in the event of the formation of an unsaturated system and is controlled by the type of substituents[10-14]. As a result, these organometallic complexes are capable of reacting with imines and many other molecules. Imines are important building blocks because they can contain not only nucleophilic or electrophilic sites but also a combination of the two, thus allowing for different reaction pathways in some cases [15-17]. Usually, this reactivity favours the synthesis of cyclic compounds in a few steps[15,16,18-23].
The reactivity of imines and carbene complexes has been reported by various authors. Imines were reacted with simple Fischer-type carbenes by Hegedus via thermal and photochemical routes, leading to new carbenic complexes and lactam nuclei, respectively [23]. On the other hand, de Meijere found that chromium alkynyl carbene complexes react with imines to produce the Michael adduct, which undergoes cyclization in thermal conditions to generate 2H-pyrroles (Scheme 1(a)) [24]. Meanwhile, Aumman [15] obtained Michael adducts and mesoionic pyrrolium carbonyltungstates by the reaction of imines with alkynyl chromium(0) carbene complexes (Scheme 1(b)). Subsequently, Sampedro´s group [16,25] conducted a complete computational mechanistic study of the same reaction to afford potential molecular switches and motors. In a later publication, this group documented the reaction of oxime derivatives and alkynyl Fischer carbene complexes to furnish four distinct types of nitrogenated heterocycles [26,27] (Scheme 1(c) and (e)). Yu achieved [28] the regioselective [3+2] annulation of azomethine imines with 1-alkynyl Fischer carbene complexes to synthesize versatile functionalized N,N-bicyclic pyrazolidin-3-ones (Scheme 1(d)). Along the same lines, our group has described the reaction of alkynyl Fischer carbene complexes with α-imino glycine methyl esters to provide 1,2,3,5-tetrasubstituted pyrroles (Scheme 1(f)) [11].
A search of the literature revealed few reports on energy sources other than conventional heating to test the reactivity of Fischer-type carbenes, except for the extensive investigation on the photochemistry of these complexes[29-33]. In 2002, Magennis et al. [34] established an advantage for a Dötz reaction when assisted by microwave energy versus the traditional thermal heating method. This reaction was also carried out by utilizing resin-bound Fischer carbenes subjected to microwave irradiation, followed by an oxidative reaction to generate 1,4-naphthoquinones [35]. Uracil-containing Fischer carbenes [36,37] and 1,3-dienol esters [38] have been prepared with microwave irradiation as well.
Therefore, investigating the behavior of Fischer carbenes in chemical reactions promoted by microwave energy is a relevant approach. In the methodologies developed by our group for the reaction of alkynyl and vinyl(alkoxy)carbene complexes under thermal conditions, substituents proved to play an important role in reactivity and selectivity during the synthesis of a variety of compounds, such as ortho- and para-quinones [39], phenols [12,40,41], furans, pyran-2-ones [42], 4-amino-1-azadienes [14], and pyrroles. The aim of the current contribution was to explore the reactivity of alkynyl(ethoxy)carbene complex 1 with benzylidene anilines 2a-p under microwave irradiation.
Experimental
Chemistry
All solvents and reagents were purchased from Sigma-Aldrich and used without further purification. The products were purified by column chromatography with silica gel (MN Kieselgel 60, 230-400 mesh), employing ethyl acetate and n-hexane as eluents in different proportions. To identify the compounds, the mixture was submitted to thin-layer chromatography (TLC, utilizing aluminum sheets and silica gel 60 F/UV254) visualized with UV light. Melting points were measured on a digital Electrothermal 90100 melting point apparatus. 1H and 13C nuclear magnetic resonance (NMR) spectra were recorded on a Bruker Ascend 500 MHz or a Bruker Ultrashield 600 MHz spectrometer with CDCl3 or DMSO-d6 as the solvent. Chemical shifts are expressed in ppm, relative to tetramethylsilane as the internal reference. Infrared (IR) spectra were captured on potassium bromide plates with a Perkin-Elmer Spectrum 100 FT-IR spectrophotometer. Spectra from high-resolution mass spectrometry (HRMS) were acquired by carrying out electrospray ionization on a Bruker micrOTOF-Q II device, electron nebulization ionization on a Bruker QTOF mass spectrometer, and electron ionization techniques (70 eV) on a Jeol JSM-GC Mate II. X-ray data were collected on an Oxford Diffraction Gemini ‘A’ diffractometer with a CCD area detector. Microwave irradiation was performed in a Discover SP CEM microwave apparatus. The alkynyl(ethoxy)carbene complex of chromium 1 and the benzylidene anilines 2a-p were prepared by the reported methods [43].
General method
Alkynyl(ethoxy)carbene complex 1 and one of the benzylidene anilines 2a-p (1.5:1 mol equiv) were added to a reaction vessel containing anhydrous tetrahydrofuran (THF) (5 mL) under nitrogen atmosphere. Subsequently, the mixture was heated at 66 ºC with microwave irradiation (150 W) for 5 h. The reaction was monitored by TLC until completion. Purification of the crude mixtures by column chromatography over silica gel (n-hexane/EtOAc, 99:1) gave the corresponding products 3a-n. Compounds 5 and 6 were identified by NMR, and the results were compared to information in the literature [14,44,45].
Pentacarbonyl-(Z)-2-phenyl-2-{[(E)-(phenyl)(phenylimino)methyl]} (ethoxy)carbene chromium(0) (3a). According to the general method, the reaction between 1 (0.30 g, 0.85 mmol) and imine 2a (0.10 g, 0.57 mmol) provided 3a (0.18 g, 60 %) as a red oil. FT-IR (CH2Cl2) νmax 2061, 1988, 1936, 1712, 1593 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.74 (t, J = 6.6 Hz, 3H, H-18), 4.68 (bs, 1H, H-17), 4.86 (bs, 1H, H-17), 6.28 (s, 1H, H-3), 6.62 (d, J = 7.8 Hz, 2H, H-6), 6.87-6.92 (m, 1H, H-8), 7.06-7.11 (m, 2H, H-7), 7.14-7.17 (m, 2H, H-14), 7.22-7.26 (m, 2H, H-10), 7.30-7.38 (m, 6H, H-11, H-12, H-15, H-16). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 75.5 (C-17), 120.9 (C-6), 123.8 (C-8), 128.3 (C-11), 128.3 (C-7), 128.7 (C-15), 129.0 (C-9), 129.2 (C-10), 129.5 (C-16), 130.2 (C-12, C-14), 130.3 (C-3), 134.1 (C-13), 149.5 (C-2), 149.7 (C-5) 166.0 (C-4), 216.6 (COcis), 224.3 (COtrans), 353.4 (Cr = Ccarb). HRMS (EI) calcd for C29H21NO6Cr [M]+ 531.0774, found 531.0766.
Pentacarbonyl-(Z)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]-3-phenyl}(ethoxy)carbene chromium(0) (3b). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2b (0.12 g, 0.571 mmol) delivered 3b (0.20 g, 65 %) as a red solid (mp 92-94 °C). FT-IR (CH2Cl2) νmax 2061, 1988, 1932, 1605, 1566 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.72 (t, J = 7.2 Hz, 3H, H-18), 3.70 (s, 3H, OMe) 4.65 (bs, 1H, H-17), 4.82 (bs, 1H, H-17), 6.22 (s, 1H, H-3), 6.60 (d, J = 9.0 Hz, 2H, H-7), 6.64 (d, J = 9.0 Hz, 2H, H-6), 7.15 (d, J = 6.6 Hz, 2H, H-14), 7.24-7.29 (m, 2H, H-10), 7.30-7.35 (m, 3H, H-15, H-16), 7.36-7.41 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 55.3 (OMe), 75.5 (C-17), 113.6 (C-6), 123.2 (C-7), 128.5 (C-11), 128.7 (C-12, C-15), 129.0 (C-16), 129.2 (C-3), 129.3 (C-10), 130.1 (C-14), 133.7 (C-13), 134.6 (C-9), 142.5 (C-5), 150.0 (C-2), 156.6 (C-8), 164.6 (C-4) 216.6 (COcis), 224.4 (COtrans), 353.5 (Cr = Ccarb). HRMS (EI) calcd for C30H23CrNO7 [M]+ 561.0880, found 561.0873.
Pentacarbonyl-(Z)-2-{[(E)-(4-chlorophenylimino)(phenyl)methyl]-3-phenyl}(ethoxy)carbene chromium(0) (3c). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2c (0.12 g, 0.571 mmol) afforded 3c (0.14 g, 45 %) as a red oil. FT-IR (CH2Cl2) νmax 2061, 1989, 1933, 1583, 1567 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.73 (t, J = 7.2 Hz, 3H, H-18), 4.66 (bs, 1H, H-17), 4.81 (bs, 1H, H-17), 6.29 (s, 1H, H-3), 6.55 (d, J = 8.4 Hz, 2H, H-6), 7.05 (d, J = 8.4 Hz, 2H, H-7), 7.14-7.19 (m, 2H, H-14), 7.21-7.26 (m, 2H, H-10), 7.30-7.39 (m, 6H, H-11, H-12, H-15, H-16). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 75.5 (C-17), 122.3 (C-6), 128.5 (C-11), 128.5 (C-7), 128.7 (C-15), 129.1 (C-10), 129.2 (C-12), 129.7 (C-16), 130.2 (C-14), 130.7 (C-3), 133.4 (C-13), 133.8 (C-9), 148.2 (C-5), 149.2 (C-2), 166.6 (C-8), 166.6 (C-4), 216.6 (COcis), 224.1 (COtrans), 353.2 (Cr = Ccarb). HRMS (EI) calcd for C29H20ClCrNO6 [M]+ 565.0384, found 565.0377.
Pentacarbonyl-(Z)-3-(fluorophenyl)-2-{[(E)-(phenylimino)(phenyl)methy]} (ethoxy)carbene chromium(0) (3d). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2d (0.113 g, 0.571 mmol) furnished 3d (0.20 g, 66 %) as a red oil. FT-IR (CH2Cl2) νmax 2061, 1988, 1940, 1704, 1599 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.73 (t, J = 7.0 Hz, 3H, H-18), 4.65 (bs, 1H, H-17), 4.86 (bs, 1H, H-17), 6.24 (s, 1H, H-3), 6.62 (d, J = 8.5 Hz, 2H, H-6), 6.87-6.92 (m, 1H, H-8), 7.00-7.05 (m, 2H, H-15), 7.06-7.11 (m, 2H, H-7), 7.12-7.16 (m, 2H, H-14) 7.21- 7.25 (m, 2H, H-10), 7.30-7.35 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 75.6 (C-17), 115.9 (C-15, J = 25.8 Hz), 120.8 (C-6), 123.8 (C-8), 128.2 (C-7), 128.3 (C-11), 128.7 (C-13, J = 1.2 Hz) 128.8 (C-3), 129.0 (C-12), 129.1 (C-10), 132.0 (C-14, J = 10.0 Hz), 134.0 (C-9), 149.4 (C-2), 149.6 (C-5), 164.3 (C-16, J = 300.7 Hz), 165.9 (C-4), 216.5 (COcis), 224.0 (COtrans), 353.3 (Cr = Ccarb). HRMS (EI) calcd for C29H20FNO6Cr [M]+ 549.0680, found 549.0686.
Pentacarbonyl-(Z)-3-(4-chlorophenyl)-2-{[(E)-(phenyl)-(phenylimino)methyl}](ethoxy)carbene chromium(0) (3e). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2e (0.12g, 0.571 mmol) produced 3e (0.17 g, 53 %) as a red solid (mp 101-103 °C). FT-IR (CH2Cl2) νmax 2061, 1989, 1940, 1716, 1582 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.73 (t, J = 7.2 Hz, 3H, H-18), 4.64 (bs, 1H, H-17), 4.85 (bs, 1H, H-17), 6.21 (s, 1H, H-3), 6.61 (d, J = 7.8 Hz, 2H, H-6), 6.88-6.93 (m, 1H, H-8), 7.07-7.12 (m, 4H, H-7, H-14), 7.21-7.25 (m, 2H, H-10), 7.30-7.37 (m, 5H, H-11, H-12, H-15). 13C NMR (150 MHz, CDCl3): δ 14.8 (C-18), 75.6 (C-17), 120.9 (C-6), 124.0 (C-8), 127.7 (C-12), 128.3 (C-7), 128.5 (C-3), 129.0 (C-11), 129.1 (C-10), 131.2 (C-15), 131.2 (C-15), 132.1 (C-13), 133.9 (C-9), 135.7 (C-16), 149.6 (C-5), 165.9 (C-2), 167.4 (C-4), 216.4 (COcis), 224.0 (COtrans), 352.9 (Cr = Ccarb). HRMS (EI) calcd for C29H20ClNO6Cr [M]+ 565.0384, found 565.0370.
Pentacarbonyl-(Z)-3-(4-methoxyphenyl)-2-{[(E)-(phenyl)(phenylimino)methyl]}(ethoxy)carbene chromium(0) (3f). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2f (0.12 g, 0.571 mmol) provided 3f (0.20 g, 64 %) as a red oil. FT-IR (CH2Cl2) νmax 2060, 1938, 1601 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.72 (t, J = 6.6 Hz, 3H, H-18), 3.81 (s, 3H, OMe) 4.64 (bs, 1H, H-17), 4.84 (bs, 1H, H-17), 6.22 (s, 1H, H-3), 6.61 (d, J = 7.2 Hz, 2H, H-6), 6.82-6.93 (m, 3H, H-8, H-14), 7.05-7.13 (m, 4H, H-7, H-15), 7.20-7.25 (m, 2H, H-10), 7.28-7.34 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 55.3 (OMe), 75.3 (C-17), 114.2 (C-14), 120.9 (C-6), 123.6 (C-8), 126.3 (C-13), 128.2 (C-11), 128.3 (C-7), 128.9 (C-12), 129.2 (C-10), 129.9 (C-3), 131.9 (C-15), 134.2 (C-9), 147.7 (C-2), 149.9 (C-5), 160.8 (C-16), 165.9 (C-4), 216 (COcis), 224.4 (COtrans), 354.4 (Cr = Ccarb). HRMS (EI) calcd for C30H23CrNO7 [M]+ 561.0880, found 561.0883.
Pentacarbonyl-(Z)-3-(4-methylphenyl)-2-{[(E)-(phenyl)(phenylimino)methyl]}(ethoxy)carbene chromium(0) (3g). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2g (0.11 g, 0.571 mmol) generated 3g (0.16 g, 52 %) as a red oil. FT-IR (CH2Cl2) νmax 2060, 1986, 1939, 1715, 1605 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.72 (t, J = 7.2 Hz, 3H, H-18), 2.34 (s, 3H, Me), 4.65 (bs, 1H, H-17), 4.84 (bs, 1H, H-17), 6.24 (s, 1H, H-3), 6.61 (d, J = 7.8 Hz, 2H, H-6), 6.86-6.92 (m, 1H, H-8), 7.02-7.11 (m, 4H, H-7, H-14), 7.15 (d, J = 7.8 Hz, 2H, H-15), 7.22-7.26 (m, 2H, H-10) 7.29- 7.35 (m, 3H, H-11, H-12), 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 21.3 (Me), 75.4 (C-17), 120.9 (C-6), 123.7 (C-8), 128.2 (C-11), 128.3 (C-7), 128.9 (C-12), 129.2 (C-10), 129.5 (C-15), 130.2 (C-3), 130.3 (C-14) 130.8 (C-13), 134.2 (C-9), 140.0 (C-16), 148.7 (C-2), 149.8 (C-5), 165.9 (C-4), 216.7 (COcis), 224.4 (COtrans), 353.9 (Cr = Ccarb). HRMS (EI) calcd for C30H23CrNO6 [M]+ 545.0930, found 545.0925.
Pentacarbonyl-(Z)-3-(furan-2-yl)-2-{[(E)-(phenyl)(phenylimino)methyl]}(ethoxy)carbene chromium(0) (3h). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2h (0.09 g, 0.571 mmol) promoted the formation of 3h (0.14 g, 49 %) as a red solid (mp 95-97 °C). FT-IR (CH2Cl2) νmax 2059, 1986, 1929, 1580 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.66 (t, J = 6.0 Hz, 3H, H-18), 4.60 (bs, 1H, H-17), 4.71 (bs, 1H, H-17), 6.01 (s, 1H, H-3), 6.43 (s, 1H, H-16), 6.45 (s, 1H, H-15), 6.61 (d, J = 7.8 Hz, 2H, H-6), 6.87-6.92 (m, 1H, H-8), 7.04-7.11 (m, 2H, H-7), 7.20-7.26 (m, 2H, H-10), 7.28-7.35 (m, 3H, H-11, H-12), 7.53 (s, 1H, H-14). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 75.1 (C-17), 112.8 (C-15), 115.0 (C-3), 115.3 (C-16), 121.0 (C-6), 123.8 (C-8), 128.3 (C-11), 128.3 (C-7), 129.0 (C-12), 129.2 (C-10), 133.8 (C-9), 146.1 (C-16), 146.2 (C-2), 149.7 (C-5), 149.8 (C-13), 165.0 (C-4), 216.8 (COcis), 224.6 (COtrans), 350.0 (Cr = Ccarb). HRMS (EI) calcd for C27H19CrNO7 [M]+ 521.0567, found 521.0557.
Pentacarbonyl-(Z)-3-(furan-2-yl)-2-{[(E)-(4-methoxyphenylimino)(phenyl) methyl]}(ethoxy)carbene chromium(0) (3i). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2i (0.11 g, 0.571 mmol) led to 3i (0.21 g, 69%) as a red oil. FT-IR (CH2Cl2) νmax 2059, 1986, 1931, 1603, 1578 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.57 (t, J = 6.0 Hz, 3H, H-18), 3.61 (s, 3H, OMe), 4.50 (bs, 1H, H-17), 4.61 (bs, 1H, H-17), 5.88 (s, 1H, H-3), 6.32 (s, 1H, H-16), 6.37 (s, 1H, H-15), 6.51 (d, J = 9.0 Hz, 2H, H-7), 6.55 (d, J = 9.0 Hz, 2H, H-6), 7.14-7.22 (m, 2H, H-10), 7.25-7.33 (m, 3H, H-11, H-12), 7.43 (s, 1H, H-14). 13C NMR (150 MHz, CDCl3): δ 14.6 (C-18), 55.2 (OMe), 75.0 (C-17), 112.7 (C-15), 113.6 (C-6), 114.2 (C-3), 114.9 (C-16), 123.3 (C-7), 128.5 (C-11), 128.9 (C-12), 129.1 (C-10), 134.3 (C-9), 142.5 (C-5), 145.8 (C-14), 146.6 (C-2), 149.9 (C-13), 156.6 (C-8), 163.6 (C-4), 216.9 (COcis), 224.7 (COtrans), 350.0 (Cr = Ccarb). HRMS (EI) calcd for C28H21CrNO8 [M]+ 551.0672, found 551.0660.
Pentacarbonyl-(Z)-3-(4-bromophenyl)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]}(ethoxy)carbene chromium(0) (3j). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2j (0.16 g, 0.571 mmol) resulted in 3j (0.19 g, 53 %) as a red oil. FT-IR (CH2Cl2) νmax 2061, 1989, 1934, 1605, 1584 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.72 (t, J = 6.6 Hz, 3H, H-18), 3.70 (s, 3H, OMe), 4.60 (bs, 1H, H-17), 4.81 (bs, 1H, H-17), 6.13 (s, 1H, H-3), 6.58 (d, J = 7.0 Hz, 2H, H-7), 6.64 (d, J = 7.0 Hz, 2H, H-6), 7.02 (d, J = 6.5 Hz, 2H, H-14), 7.22- 7.28 (m, 2H, H-10), 7.36-7.42 (m, 3H, H-11, H-12), 7.46 (d, J = 6.5 Hz, 2H, H-15). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 55.2 (OMe), 75.6 (C-17), 113.6 (C-6), 123.3 (C-7), 123.9 (C-16), 127.6 (C-3), 128.6 (C-11), 129.1 (C-12), 129.1 (C-10), 131.3 (C-14), 131.9 (C-15), 132.6 (C-13), 134.5 (C-9), 142.3 (C-5), 150.4 (C-2), 156.7 (C-8) 164.5 (C-4), 216.6 (COcis), 224.2 (COtrans), 353.0 (Cr = Ccarb). HRMS (EI) calcd for C30H22BrNO7Cr [M]+ 638.9985, found 638.9980.
Pentacarbonyl-(Z)-3-(4-chlorophenyl)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]}(ethoxy)carbene chromium(0) (3k). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2k (0.139 g, 0.571 mmol) gave 3k (0.17 g, 50 %) as a red oil. FT-IR (CH2Cl2) νmax 2061, 1989, 1935, 1605 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.72 (t, J = 6.6 Hz, 3H, H-18), 3.70 (s, 3H, OMe) 4.61 (bs, 1H, H-17), 4.81 (bs, 1H, H-17), 6.15 (s, 1H, H-3), 6.58 (d, J = 8.4 Hz, 2H, H-7), 6.64 (d, J = 9.0 Hz, 2H, H-6), 7.08 (d, J = 8.4 Hz, 2H, H-14), 7.23-7.27 (m, 2H, H-10), 7.29-7.34 (m, 2H, H-15), 7.36-7.40 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 55.3 (OMe), 75.6 (C-17), 113.6 (C-6), 123.3 (C-7), 127.6 (C-3), 128.6 (C-11), 128.9 (C-15), 129.0 (C-12), 129.1 (C-10), 131.1 (C-14), 132.2 (C-16), 134.5 (C-9), 135.5 (C-13), 142.3 (C-5), 150.3 (C-2), 156.7 (C-8), 164.4 (C-4) 216.6 (COcis), 224.2 (COtrans), 353.1 (Cr = Ccarb). HRMS (EI) calcd for C30H22ClCrNO7 [M]+ 595.0490, found 595.0489.
Pentacarbonyl-(Z)-3-(4-methoxyphenyl)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]}(ethoxy)carbene chromium(0) (3l). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2l (0.13 g, 0.571 mmol) delivered 3l (0.23 g, 70 %) as a red solid (mp 46-48 °C). FT-IR (CH2Cl2) νmax 2060, 1932, 1604 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.70 (t, J = 7.2 Hz, 3H, H-18), 3.69 (s, 3H, OMe), 3.80 (s, 3H, OMe), 4.61 (bs, 1H, H-17), 4.81 (bs, 1H, H-17), 6.16 (s, 1H, H-3), 6.57 (d, J = 9.0 Hz, 2H, H-7), 6.63 (d, J = 9.0 Hz, 2H, H-6), 6.85 (d, J = 8.4 Hz, 2H, H-15), 7.09 (d, J = 8.4 Hz, 2H, H-14), 7.23-7.27 (m, 2H, H-10), 7.34-7.38 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.8 (C-18), 55.3 (OMe, OMe), 75.3 (C-17), 113.6 (C-6), 114.2 (C-15), 123.1 (C-7), 126.5 (C-13), 128.4 (C-11), 128.8 (C-3), 129.0 (C-12), 129.2 (C-10), 131.8 (C-14), 134.7 (C-9), 142.7 (C-5), 148.2 (C-4), 156.4 (C-8), 160.7 (C-16), 164.6 (C-2), 216.8 (COcis), 224.4 (COtrans), 354.4 (Cr = Ccarb). HRMS (EI) calcd for C31H25NO8Cr [M]+ 591.0985, found 591.0972.
Pentacarbonyl-(Z)-3-(4-methylphenyl)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]}(ethoxy)carbene chromium(0) (3m). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2m (0.12 g, 0.571 mmol) afforded 3m (0.20 g, 62 %) as a red oil. FT-IR (CH2Cl2) νmax 2060, 1987, 1932, 1606 cm-1. 1H NMR (CDCl3, 500 MHz,): δ 1.71 (t, J = 7.0 Hz, 3H, H-18), 2.34 (s, 3H, Me), 3.69 (s, 3H, OMe), 4.62 (bs, 1H, H-17), 4.80 (bs, 1H, H-17), 6.19 (s, 1H, H-3), 6.58 (d, J = 8.5 Hz, 2H, H-7), 6.63 (d, J = 9.0 Hz, 2H, H-6), 7.05 (d, J = 8.0 Hz, 2H, H-14), 7.14 (d, J = 8.0 Hz, 2H, H-15), 7.24-7.28 (m, 2H, H-10), 7.34-7.39 (m, 3H, H-11, H-12). 13C NMR (125 MHz, CDCl3): δ 14.7 (C-18), 21.3 (Me), 55.3 (OMe), 75.3 (C-17), 113.6 (C-6), 123.2 (C-7), 128.4 (C-11), 128.9 (C-12), 129.2 (C-10), 129.3 (C-3), 129.4 (C-15), 130.1 (C-14), 130.9 (C-13), 134.7 (C-9), 139.8 (C-16), 142.5 (C-5), 149.1 (C-2), 156.5 (C-8) 164.6 (C-4), 216.7 (COcis), 224.4 (COtrans), 353.9 (Cr = Ccarb). HRMS (EI) calcd for C31H25NO7Cr [M]+ 575.1036, found 575.1045.
Pentacarbonyl-(Z)-2-{[(E)-(4-chlorophenylimino)(phenyl)methyl]-3-(4-methoxyphenyl)}(ethoxy)carbene chromium(0) (3n). According to the general method, the reaction between 1 (0.30 g, 0.856 mmol) and imine 2n (0.13 g, 0.571 mmol) furnished 3n (0.170 g, 50 %) as a red oil. FT-IR (CH2Cl2) νmax 2060, 1931, 1604, 1580 cm-1. 1H NMR (CDCl3, 600 MHz,): δ 1.71 (t, J = 6.6 Hz, 3H, H-18), 3.82 (s, 3H, OMe) 4.62 (bs, 1H, H-17), 4.80 (bs, 1H, H-17), 6.22 (s, 1H, H-3), 6.54 (d, J = 8.4 Hz, 2H, H-6), 6.86 (d, J = 9 Hz, 2H, H-15), 7.04 (d, J = 8.4 Hz, 2H, H-7), 7.10 (d, J = 8.4 Hz, 2H, H-14), 7.20-7.24 (m, 2H, H-10), 7.32-7.37 (m, 3H, H-11, H-12). 13C NMR (150 MHz, CDCl3): δ 14.7 (C-18), 55.4 (OMe), 75.3 (C-17), 114.3 (C-15), 122.4 (C-6), 128.4 (C-7), 128.4 (C-11), 129.0 (C-8), 129.1 (C-12), 129.2 (C-10), 130.4 (C-3), 132.0 (C-14), 133.9 (C-9), 144.9 (C-13), 147.4 (C-2), 148.3 (C-5), 160.9 (C-16), 166.6 (C-4), 216.7 (COcis), 224.1 (COtrans), 354.1 (Cr = Ccarb). HRMS (EI) calcd for C30H22ClCrNO7 [M]+ 595.0490, found 595.0490.
Ethyl (E)-3-(4-methoxyphenyl)-2-{[(E)-(4-methoxyphenylimino)(phenyl)methyl]}acrylate (10b). Compound 3l (0.25g, 0.422 mmol) in THF (5 mL) was irradiated with visible light (fluorescent lamp, 22 watts, Osram) at room temperature for 48 h. The solvent was then removed under vacuum and the residue was separated by flash column chromatography (n-hexane/EtOAc, 99:1), obtaining 10b (0.1 g, 59 %) as a pale-yellow solid. FT-IR (CH2Cl2) νmax 2934, 1731, 1689, 1238, 1168 cm-1. 1H NMR (500 MHz, CDCl3) δ 1.06 (t, J = 7.1 Hz, 3H, H-18), 3.66 (s, 3H, OMe), 3.71 (s, 3H, OMe), 4.06 (m, J = 7.1 Hz, 2H, H-17), 6.75-6.56 (m, 6H, Ar), 7.45-7.26 (m, 5H, Ar), 7.69 (s, 1H, H-3), 7.92 (d, J = 7.5 Hz, 2H, Ar). 13C NMR (126 MHz, CDCl3) δ 14.1 (C-18), 16.0 (C-17), 55.5 (OMe), 55.6 (OMe), 76.8, 77.0, 77.3, 114.3, 114.4, 114.8, 115.1, 125.5, 125.6, 128.5, 128.8, 129.7, 131.7, 132.0, 132.9, 133.7, 190.8 (C-1). HRMS (ESI) calcd for C26H26NO4 [M+H]+ 416.1862 found 416.1865.
X-ray structure determination
Crystal data and refinement details for 10b are included in supplementary information. Single crystals were generated by the slow diffusion of n-hexane in a CHCl3 solution with 10b at 4 ºC. The pale-yellow crystal of compound 10b was mounted on glass fibers. Data was collected at 20 ºC on an Agilent SuperNova (single source at offset, Eos) S2 diffractometer using Mo Kα radiation (graphite crystal monochromator, λ 0.71073 Å). Hydrogen atoms were placed in idealized positions, and their atomic coordinates refined. An empirical absorption correction was applied based on spherical harmonics (implemented in SCALE3 ABSPACK scaling algorithm). The structure was solved with the SHELXT program package [46-48] running under the OLEX2 1.5 environment [49]. CCDC 2192850 contains the supplementary crystallographic data for this paper, which can be acquired free of charge from the Cambridge Crystallographic Data Centre via https://www.ccdc.cam.ac.uk/structures.
Computational details
All the stationary points along the reaction mechanisms were established by the density functional theory (DFT) at the M06-L level of theory [50] on the Gaussian 09 suite of programs [51], with the 6-311++G** basis set [52] for non-heavy atoms and the LANL2TZ effective core potential [53] for Cr atoms. The level of theory herein employed provides a good description of the thermochemistry, chemical kinetics (transition state energy), non-covalent interactions, and dissociation energy for organometallic compounds [51,54]. All the stationary points and transition state points were re-optimized with a non-explicit solvent model by means of the polarizable continuum model (PCM) [55,56]. The solvent considered for all the calculations was THF. The minima and transition states along the potential energy surface were optimized by performing a frequency calculation for each compound. The intrinsic reaction coordinate (IRC) was computed with Berny optimization to confirm the connection along a minimum energy path of reactants, intermediates, adducts, and products. Free energies at 363.15 K were calculated by including the corresponding thermal corrections to Gibbs free energies (TCGE).
Results and discussion
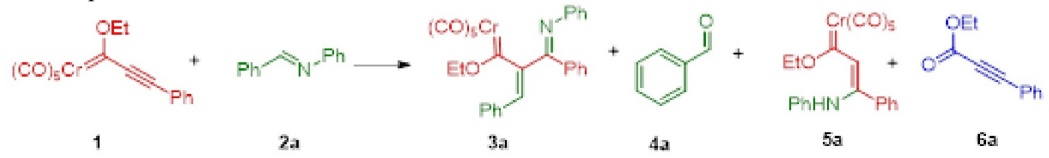
To initiate the reactivity study, benzylidene aniline 2a [43] and Fischer carbene complex 1 were utilized as model reagents [57]. Four anhydrous solvents (toluene, benzene, methyl t-butyl ether (MTBE), and THF) were tested to avoid the hydrolysis of 2a (Table 1, entries 1-4). Under thermal conditions, various products were detected by 1H NMR. For example, ester 6a resulted from the oxidation of 1 [44,45], benzaldehyde 4a from the hydrolysis of 2a, and enamine 5a from the 1,4-addition of aniline to 1 [14].
Table 1 Optimization of the reaction conditions of Fischer carbene 1 and imine 2a.a
a Complex 1 (1.5 equiv) and 2a (1 equiv), N2, 5 mL of solvent. b No solvent.
The spectroscopy data (HPLC and NMR) of the isolated product 3a were different than the expected result [15,25]. The 1H NMR spectrum of 3a showed signals for three aromatic rings located between 7.38 and 6.62 ppm, as well as a singlet at 6.28 ppm attributed to a vinyl proton. In the 13C NMR spectrum, a signal at 353.4 ppm indicated the existence of a carbene carbon (Cr = Ccarb), and the signals in 216.6 and 224.3 ppm evidenced the presence of COcis and COtrans groups. Hence, the structure of 3a corresponds to a new vinyl chromium carbene complex α-substituted by an (E)-phenyl(phenylimino)methyl group (i.e, a cross-conjugated metallatriene system, Table 1). Furthermore, the HRMS analysis is in agreement with the proposed structure of 3a.
Other experiments were carried out to improve the yield of 3a. With thermal heating and THF (entry 4), the low yield at 1 h did not improve when the reaction was monitored over the next few hours. Under neat conditions, infrared irradiation as the source of heat afforded a mixture of products comparable to those found with the thermal procedure, while microwave irradiation promoted the formation of only traces of the same (Table 1, entries 5-7). With microwave energy as the source of heat, the reactions performed in a sealed vessel furnished 3a in yields of 60 % and 30 % when using polar solvents (THF and CH3CN, respectively), and only a trace amount with toluene (Table 1, entries 8-10). This outcome may be due to the main properties of microwave energy, being dipolar polarization and ionic conduction [58]. Accordingly, the polar solvents probably provoke an efficient dipolar polarization effect over the generation and stabilization of polar species during the proposed reaction mechanism (Scheme 4).
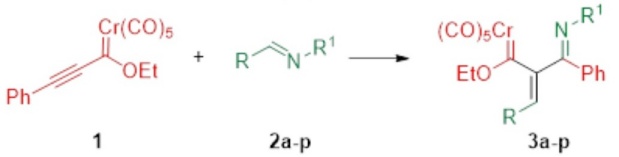
Under the optimized conditions, the scope of the methodology was explored by modifying the aromatic substituents of the benzylidene aniline (2a-p) while keeping 1 as the phenyl alkyne carbene complex (Table 2). The new complexes 3a-n were achieved in 45-70 % yields. Prolonging the irradiation time led to the decomposition of the carbene complex and only traces of the target compound.
Table 2 Scope of the synthesis of the new cross-conjugated metallahexatrienes 3a-p a
aComplex 1 (1.5 equiv) and 2a-p (1 equiv) in THF (5 mL) under N2, MW (150 W), 66 ºC, 5h.
The products were separated by column chromatography over silica gel, and their structures were established by 1H, 13C, 2D, and nuclear Overhauser effect (NOE) NMR experiments as well as IR and HRMS. In all cases, complexes 3a-n were obtained as single Z (double bond)/E (imino group) isomers, along with the corresponding by-products indicated in Table 1.
The impact of the electronic effect of the different aromatic groups of the imines on the efficiency of the reaction is illustrated in Table 2, although a quantitative structure-reactivity relationship was not established. A relatively low yield of complex 3c resulted from the presence of the chlorine atom (a weak electron donating group) in imine 2c (Table 2, entry 3) compared to a greater yield of complex 3b derived from the methoxy group (a stronger electron donating group) in the imine of 2b (Table 2, entry 2). The yield of complexes 3a and 3b were 60 % and 65 %, respectively (Table 2, entries 1 and 2), despite the lack of substituents in the phenyl rings of 2a (vide infra). On the other hand, electron donor groups in the imines 2l and 2m (R and R1) increased the yield of complexes 3l and 3m (Table 2, entries 12 and 13). The combination of electron donating and electron withdrawing groups in the imines 2j, 2k, and 2n afforded slightly lower yields of 3j, 3k, and 3n (53, 50, and 50%, respectively) compared to 3a (60 %), the latter furnished by the reaction with the unsubstituted imine 2a. In contrast, when the imine bore a strong electron withdrawing group, such as 2o and 2p (R = p-NO2C6H4, R1 = p-NO2C6H4), the reaction did not occur. Due to these differences in reactivity, the electronic effects of the substituents in imines 2 were analyzed by theoretical studies (vide infra).
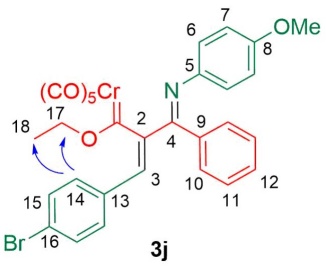
Even though it was not possible to obtain the crystalline structures of 3a-n by single crystal X-ray diffraction, NOE spectroscopy (NOESY and ROESY) experiments provided sufficient evidence to determine the conformation. Accordingly, complex 3j exhibited a NOE effect of the vinyl proton H-3 and the CH3CH2O group (protons H-17 and H-18) by irradiating the aromatic proton H-14 (7.02 ppm) (Fig. 1), indicating a Z configuration of the double bond C-C. Neither proton H-17 nor H-18 showed a NOE effect with H-6 or H-7, which suggests that the N- anisyl ring of the imine moiety remains distant from the metal center.
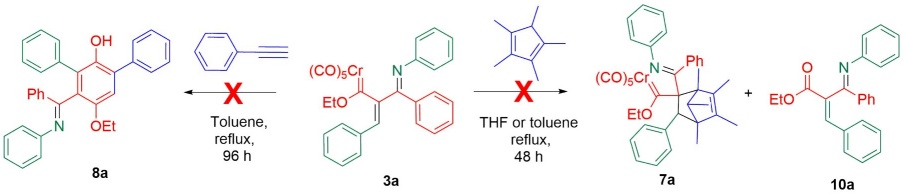
It is reported that metallatrienes are precursors of cyclopentadienes [58], indenes [59], naphthalenes [60], homopyrroles [61], and other carbo- and heterocycles [22]. Hence, some experiments were conducted to evaluate the reactivity of the new metallatrienes 3a-n. The reaction of 3a and pentamethylcyclopentadiene was attempted under the [4+2] cycloaddition conditions previously described. The reaction did not proceed in THF or toluene at reflux for 48 h, and a consequence, the starting materials were recovered. The results of the Dötz reaction of phenylacetylene with 3a in toluene was also examined after refluxing for 96 h. The 1H NMR analysis of the crude reaction mixture did not show the expected transformation, but the oxidation product 10a was detected (Scheme 2).
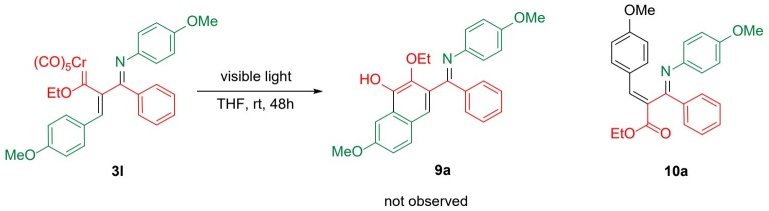
With the objective of forming a six-membered cycle in complex 3l, further experiments were performed with UV and visible light irradiation to attain the insertion of CO and the subsequent cyclization to phenol 9a. Nevertheless, the desired product was not found and only the oxidized compound 10b was isolated, afforded in a moderate yield of 59 % (Scheme 3). Despite there being an α,β-unsaturated system in complexes 3a-n, indicating susceptibility to Dötz benzannelation, such a reaction did not take place for 3l, probably because of the steric effect of the bulky substituents.
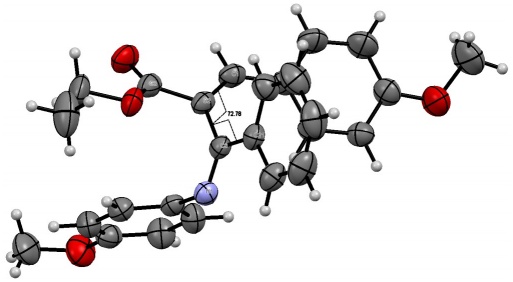
A crystal sample was obtained by the slow diffusion of n-hexane in a CHCl3 solution with 10b at 4 ºC. The crystallized compound fits in the monocyclic crystal system. The space group P21/c and unit cell parameters are a = 17.0269(12) Å, b = 9.6109(5) Å, and c = 14.1952(8) Å, as well as ( = 90°, ( = 104.895(7)°, ( = 90°, and a volume of 2244.9(2) Å3. According to the crystalline structure of 10b, oxidation of 3l and removal of the steric hindrance of the bulky Cr(CO)5 group leads to isomerization of the double bond of this compound to the E configuration [torsion angle: C1-C2-C6-C21 = 72.78°]. Moreover, the ester group adopts the s-cis conformation and the imino moiety is orthogonal to the double bond (Fig. 2).
Computational analysis
Reaction between the Fischer alkynyl carbene complex and benzylidene anilines
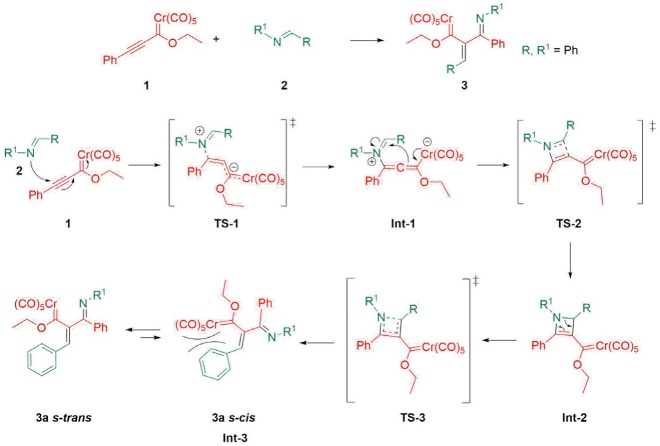
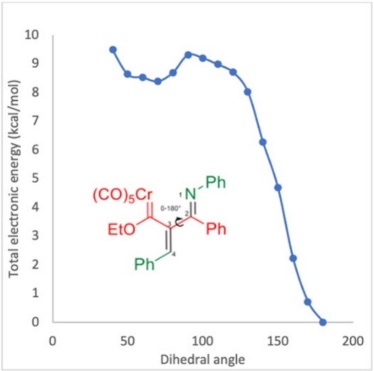
Considering the experimental findings, a computational study was performed to account for the mechanism of formation of the new compounds 3a-n. The potential energy surface was calculated for the proposed four-step mechanism (Scheme 4), involving three intermediates and three transition states (Scheme 5). The first step consists of the 1,4-addition of the nitrogen atom of the imine to the alkynyl Fischer carbene, leading to transition state TS-1 with an N-Cβ bond distance of 1.95 Å and an activation energy ((G≠) of 37.42 kcal/mol. Afterwards, intermediate 1 (Int-1) is generated, with a reactant energy ((Gº) of 13.76 kcal/mol. Int-1 undergoes an intramolecular attack by the carbenic anion and is converted into the iminium moiety by passing through the cyclic TS-2, which is the rate-determining step ((G≠ = 43.18 kcal/mol). The distance of the C-Cβ bond formed is 2.24 Å for Int-1 but only 1.40 Å for intermediate 2 (Int-2). The latter exhibits much greater stability than Int-1 ((Gº = 16.10 kcal/mol). Subsequently, the thermally allowed electrocyclic opening of the aza-cyclobutene of Int-2 brings about transition state 3 (TS-3) with an activation barrier ((G≠) of 29.92 kcal/mol, which in turn evolves into 3a (s-cis) with a dihedral angle of 68.83° (Figures 3 and 4). Interestingly, greater stability (-5.38 kcal/mol) was found for the s-trans than the s-cis conformation of 3a. To find the transition state barrier associated with the rotation, a SCAN of the dihedral rotation was performed (Scheme 6), using 18 steps spanning an interval of 0-180 degrees (consisting of 10 degrees for each step), starting from the s-cis and arriving to the s-trans conformation. The energetic profile shows two minima: the 3a s-cis structure at 68.83º and the more stable 3a s-trans conformer with an activation energy ((G≠) of 1.30 kcal/mol between both rotamers. The stability of the s-trans conformer is in agreement with the experimental results for compound 3a.
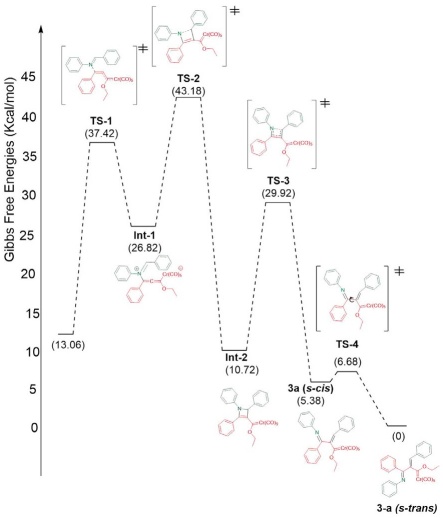
Scheme 5 Energetic profile of the Gibbs free energies (kcal/mol) relative to the energy of product at the M06L/6311++G**/Cr=LANL2TZ/SCRF=THF level of theory.
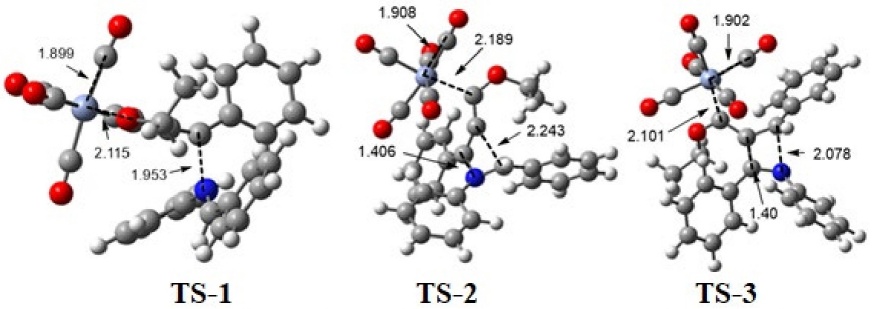
Fig. 3 Structures corresponding to the transition states TS-1, TS-2, and TS-3 at the M06-L/6-311++G**/Cr=LANL2TZ/SCRF=THF level of theory. Bond distances are given in Å. Color code of the spheres pictured: hydrogen atoms in white, carbon atoms in grey, nitrogen atoms in blue, oxygen atoms in red, and chromium atoms in light blue.
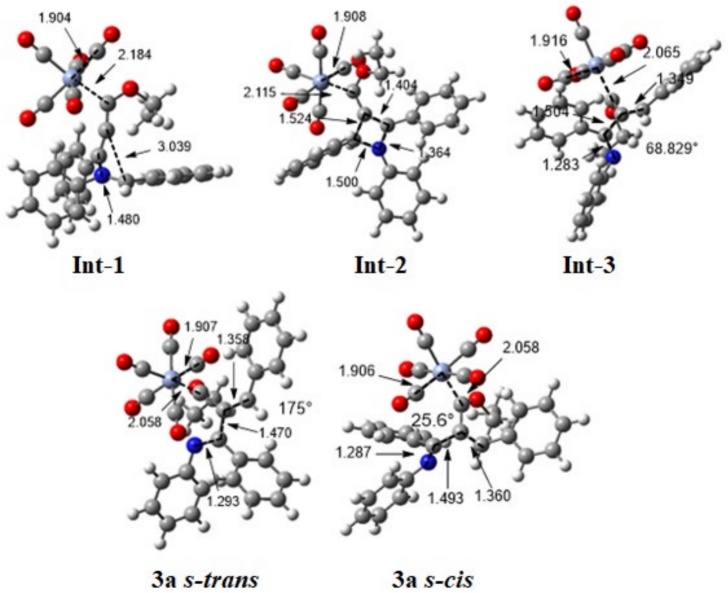
Fig. 4 Optimized structures of intermediates Int-1, Int-2, and Int-3, as well as products 3a (s-cis) and 3a (s-trans) at the M06-L/6-311++G**/Cr=LANL2TZ/SCRF=THF level of theory. Bond distances are given in Å. Color code of the spheres depicted: hydrogen atoms in white, carbon atoms in grey, nitrogen atoms in blue, oxygen atoms in red, and chromium atoms in light blue.
A comparative study was carried out to explore the impact of the R and R1 substituents on the aromatic system of benzylidene anilines 2a-p. Apparently, the R1 substituent did not have any relevant effect on the 1,4-addition, and the electronic effect of the R substituent was not relevant in the reactivity of the entire process (see supplementary data).
Conclusions
Microwave irradiation promoted the reaction between the Fischer carbene complex of Cr(0) (1) and benzylidene anilines 2a-n, obtaining moderate yields of new, stable, and polysubstituted Fischer carbenes with a cross-conjugated system. In all cases, complexes 3a-n were afforded as single Z (double bond)/E (imino group) isomers, while the oxidation product 10a changed to the E (double bond)/E (imino group) configuration. The computational study of the reaction mechanism revealed that the formation of the dihydroazete intermediate (Int-2) is the rate-determining step of the reaction. The proposed mechanism involves a 1,4-addition/ring closure/electrocyclic opening cascade reaction. The reaction did not proceed in any of the compounds 2 that had an electron withdrawing group in the aromatic ring of the imine moiety. Even though complexes 3a-n did not evolve into the Diels-Alder or the Dötz products (probably because of the great steric effects exhibited by the organometallic compounds), these kinds of structures are interesting from a synthetic and theoretical point of view. Further research on Fischer carbene complexes of W(0) and Mo(0) with different substituents is currently under way, and the results will be reported in due course.











 nueva página del texto (beta)
nueva página del texto (beta)