Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista odontológica mexicana
versión impresa ISSN 1870-199X
Rev. Odont. Mex vol.26 no.2 Ciudad de México abr./jun. 2022 Epub 08-Ago-2025
https://doi.org/10.22201/fo.1870199xp.2022.26.2.80761
Investigación original
Efficacy of Commercial Disinfectants in Reducing Microorganisms on Dental Chair Surfaces in Pediatric Clinics
*Especialidad en Odontología estética, cosmética, restauradora e Implantología. Facultad de Estomatología. Universidad Autónoma de San Luis Potosí, San Luis Potosí, México.
**Facultad de Odontología. Unidad Torreón, Universidad Autónoma de Coahuila, Coahuila, México.
***Facultad de Odontología, Río Blanco. Universidad Veracruzana, Veracruz, México.
****Facultad de Odontología. Universidad Autónoma del Estado de México, México.
*****Laboratorio de Bioquímica y Microbiología. Facultad de Estomatología, Universidad Autónoma de San Luis Potosí, San Luis Potosí, México.
******Especialidad de Periodoncia, Facultad de Estomatología, Universidad Autónoma de San Luis Potosí, San Luis Potosí, México.
Introduction:
reducing surface contamination in dental clinics with disinfectants is key to infection control.
Objective:
to evaluate bacterial growth on the dental spitoon/cuspidor, three-way syringe tip and saliva ejector converter of the dental chair in paediatric dental clinics after three working days.
Material and methods:
Microorganisms were isolated and Colony Forming Unit (CFU) were quantified from the surfaces evaluated during the 1st, 2nd and 3rd day of the working day. The disinfectants tested were Quaternary Ammonium Compounds (QACs) 5th generation 3% (FD 300® and Septalkan®) and 3rd generation (Bactogem EQ-110®); sodium hypochlorite 0.05% (NaClO) and chlorine dioxide (ClO2, TwinOxide®).
Results:
the microorganisms isolated prior to the use of disinfectants were: Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia faecalis, E. coli and Klebsiella-Enterobacter-Serratia-Citrobacter complex, KESC. Following the use of the disinfectants, P. aeruginosa and E. coli were completely eliminated; a significant reduction of E. faecalis and KESC was also obtained (p <0.05), and S. aureus showed no bacterial reduction. FD 300® obtained the highest reduction >5 Log10 on all three surfaces, followed by Septalkan® with a reduction between 3 and 4 Log10. NaClO 0.5% and TwinOxide® (ClO2) obtained a reduction of more than 3 Log10, Bactogem EQ-110® obtained smaller reductions between 2 and 3 Log10.
Conclusions:
The 5th generation 3% QACs FD 300® showed the highest CFU reduction on the three-way syringe tip and saliva ejector converter, and a total reduction on the cuspidor, followed by Septalkan®; NaClO and ClO2 obtained a similar CFU reduction; the 3rd generation QACs showed the lowest disinfectant efficacy.
Keywords: disinfectant solutions; surfaces; dental chair; infection control
Introducción:
la reducción de la contaminación de superficies en clínicas dentales con desinfectantes es clave para el control de infecciones.
Objetivo:
evaluar el crecimiento bacteriano en la escupidera, punta de la jeringa triple y conector del eyector de succión del sillón dental en clínicas dentales pediátricas después de tres jornadas de trabajo.
Material y métodos:
se aislaron los microorganismos y se cuantificaron las Unidades Formadores de Colonias (UFC) de las superficies evaluadas durante el 1.°, 2.° y 3.° día de la jornada de trabajo, los desinfectantes evaluados fueron compuestos de amonio cuaternario (CAQ) de 5.ª generación al 3% (FD 300® y Septalkan®) y de 3.ª generación (Bactogem EQ-110®), hipoclorito de sodio al 0.05% (NaClO) y dióxido de cloro (ClO2, TwinOxide®).
Resultados:
los microorganismos aislados previo al uso de los desinfectantes fueron: Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia faecalis, E. coli y complejos Klebsiella-Enterobacter-Serratia-Citrobacter, KESC. Tras el uso de los desinfectantes se eliminó totalmente P. aeruginosa y E. coli; asimismo se obtuvo una reducción significativa de E. faecalis y KESC (p <0.05), y S. aureus no mostró una reducción bacteriana. El FD 300® obtuvo la mayor reducción >5 Log10 en las tres superficies, seguido del Septalkan® con una reducción de entre 3 y 4 Log10. El NaClO al 0.5% y el TwinOxide® (ClO2) obtuvieron una reducción superior a los 3 Log10, Bactogem EQ-110® obtuvo menores reducciones entre 2 y 3 Log10.
Conclusiones:
el CAQ al 3% de 5.ª generación FD 300® mostró la mayor reducción de UFC en la punta de la jeringa triple y conector del eyector de succión y una reducción total en la escupidera, seguido del Septalkan®; el NaClO y ClO2 obtuvieron una reducción de UFC similar; el CAQ de 3.ª generación mostró la menor eficacia desinfectante.
Palabras clave: soluciones desinfectantes; superficies; sillón dental; control de infecciones
Introduction
Since the SARS-CoV-2 pandemic, there has been an increased awareness of the relationship of contaminated surfaces to the transmission of pathogens associated with dental care, which has highlighted the need for effective methods for cleaning and disinfection of inert surfaces1. Dental procedures involve contact of dental equipment or instruments with the patient’s sterile tissue or mucous membranes2, with these procedures being a major risk in the pathway of entry of pathogenic microbes; failure to adequately disinfect or sterilise surfaces of dental devices and equipment can lead to the transmission of infections3.
In odontology, infection control is a relevant issue to prevent the spread of diseases, so surface disinfection is a method to reduce the risk of contact with viruses and interrupt their spread4; therefore, it is necessary to have the knowledge of when the disinfection or sterilisation process should be performed; although both eliminate pathogens, only sterilisation eliminates endospores5. Disinfection and sterilisation of attachments and devices in dentistry for patient care is divided into three levels, depending on the intended use of each object: critical (contact with sterile tissue or bone: sterilisation), semi-critical (contact with mucosal membranes or non-intact skin: medium-level sterilisation or disinfection) and non-critical (contact with intact skin: medium or low-level disinfection)6, 7.
Microorganisms contaminating the environmental surroundings of dental clinics represent an important source of transmission of infections associated with dental procedures. It has been reported that transmission of infections in a clinical environment is caused by endogenous microbiota residues or pathogenic bacteria from the patient8. When these endogenous microbiota encounter inert surfaces of dental attachments and equipment that are not adequately disinfected, they become contaminated by viruses and bacteria9. In dental practices, infections can be acquired through microorganisms from aerosols, water lines, blood, saliva and respiratory secretions by microorganisms from the environment, oral cavity or respiratory tract, so cleaning of surfaces in dental practices should be performed before and after each patient care10,11.
There are desirable characteristics for a disinfectant solution: it should kill a wide variety of pathogenic microorganisms without causing the emergence of resistant forms; its action should be effective and efficient, even in the presence of organic matter. Furthermore, exposure to the disinfectant must not harm humans or the environment, be affordable, have a pleasant odour and a long shelf life6. The most commonly used disinfectant formulations today are quaternary ammonium compounds, which have amphoteric surfactant properties and possess positively charged polyatomic ion compounds12. Among the most popular disinfection chemicals are hydrogen peroxide, sodium hypochlorite, quaternary ammonium compounds and glutaraldehyde7, although the latter gives off highly toxic vapours when used at 2%13.
In a dental clinic there are surfaces that are more susceptible to continuous contamination, because they are more exposed to microorganisms or involve more contact of dental staff (dental light switch, X-rays, three-way syringe tip, saliva ejector), allowing for heterogeneous microbial populations, with some opportunistic pathogens for humans; when these surfaces are touched by the hands of staff, once contaminated they are a vehicle for surface contamination14, making surface disinfection one of the mandatory infection control procedures15. Due attention to proper cleaning, disinfection and sterilisation must be ensured in routine dental practice to avoid cross-infection13,14. Therefore, strict and proper implementation of infection control measures is essential for the safety of patients and dentists15. The aim of this study was to evaluate the antibacterial efficacy of different commercial disinfectant solutions used for three days during clinical days on different critical surfaces and dental chair attachments in a paediatric dental clinic. Total microorganisms recovered from each surface evaluated were quantified and microorganisms with pathogenic potential were identified.
Material and methods
This study was approved by the ethics committee of the Faculty of Stomatology, Autonomous University of San Luis Potosí (in Spanish: Universidad Autónoma de San Luis Potosí UASLP;) (CEI-FE-005-016). The site of this study was the Pediatric Dentistry postgraduate clinic of the Faculty of Stomatology of the UASLP. Inclusion criteria for the dental chairs studied included that they had been used by the same professional during each work shift, and that they attended between five and ten patients per day. Sampling was conducted over three consecutive days, so only chairs that met the inclusion criteria during this period were included. According to the schedule of patients seen by the student during his working day, the selection of the dental chair that met the inclusion criteria was made. The exclusion criteria for the dental chairs were that they had not met the number of patients seen during the shifts or that one of the three surface samples would not be taken.
Surface disinfection
Prior to the start of the study, a rigorous cleaning and disinfection protocol was applied to all dental chairs in the clinic to be studied; 0.5% sodium hypochlorite (NaClO) was used as the gold standard on all surfaces. The solutions tested were: 0.5% NaClO, chlorine dioxide (ClO2 at an application concentration of 50 ppm), two 5th generation QACs solutions and one 3rd generation QACs solution (Table 1). After randomisation of the dental units, the disinfectant to be used in each of them was assigned; the disinfectants were prepared following the instructions of each of the manufacturers in the Microbiology laboratory of the Faculty of Stomatology of the UASLP; the disinfectant solutions were placed in equal bottles with atomiser, labelled with a number that identified the product; the principal investigator was the only one who knew the product assigned to each number. The disinfection protocol was carried out between each patient during the work shift, which was performed by two participants in this study who were unaware of the product they were applying. Surface disinfection was carried out via aerosol, spraying the surface to be disinfected until visibly wet with an atomiser (0.6 ml/cm2), mechanical cleaning of the surfaces with a disposable multipurpose towel rubbing in several directions for one minute, leaving the surface wet for the following five minutes, and then collecting samples. These procedures were performed over three days, before the first patient of the day and between each patient seen at each dental chair.
Table 1 Disinfectant solutions evaluated in this study
| Solution | Trade name | Composition as described on the label or data sheet | Manufacture |
|---|---|---|---|
| Chlorine dioxide | TwinOxide® | Chlorine dioxide 0.3%. | TwinOxide International B. V. Netherlands. |
| 5th generation quaternary ammonium | FD 300® | Combination of synergistic effect of alkylamines and quaternary ammonium compounds. 100 g of FD 300 contains 12 g 3-aminopropyl-dodecyl-1,3-propandiamine, 7 g didecyl-methyl-poly(oxyethyl) ammonium propionate, as well as 5-15 % nonionic surfactants, < 5% complexing agents, < 5% alkaline detergent components, adjuvants and water. | Dürr Dental Orochemie GmbH + Co. KG, Germany. |
| 5th generation quaternary ammonium | Septalkan® | Didecyldimethylammonium chloride. Alkylbenzyldimethylammonium chloride (fourth and fifth generation quaternary ammoniums). | Alkapharm, France. |
| 3rd generation quaternary ammonium | Bactogem EQ-110® | The package describes that it contains: broad spectrum quaternary ammonium salts. No technical data sheet was found. | Mexico. |
Sample collection
Sampling was performed on three different surfaces of the dental chair: three-way syringe tip, ejector nozzle or saliva ejector converter and, surface of the spitoon. The initial disinfection protocol was carried out on day one, before any intervention and before starting the first clinical day. Subsequently, the baseline sample was taken (day one), the second sample was taken for follow-up (day two) and the third and final sample was taken after the last patient seen during the last day (day three). Sampling was performed with a sterile cotton-tipped swab previously moistened with sterile distilled water and rubbed ten times over each surface. The swabs were placed in a tube containing 5 ml of Tryptic Soy Broth transport medium.
Quantification of CFU
Samples were shaken vigorously for 10 seconds on a vortex shaker; serial dilutions were made, taking 500 µL with a pipette with a sterile tip from the basal samples, which were transferred to a test tube with 4.5 ml of sterile PBS; shaken for 30 seconds, and dilutions of 10-1, 10-2, 10-3, 10-4, 10-5, 10-6 and 10-7 carried out. After serial dilutions were made, 100 µL of each dilution was taken and seeded onto trypticase soy agar plates. The culture plates were incubated at 36 °C±1.5 °C for 24 to 48 hours. From the cultures grown, the colony forming unit CFU/ml of microbial growth were counted with the aid of a colony counter. Staphylococcus aureus, Enterococcus spp, Pseudomonas aeruginosa and Candida spp were determined from the primary culture of both the initial and final baseline samples. For species identification, selective, differential chromogenic media CHROMagarTM Staph aureus, CHROMagarTM Orientator, CHROMagarTM Pseudomonas and CHROMagarTM Candida were used. Species identification according to colony characteristics and colour was conducted according to the manufacturer’s instructions and the principal investigator’s experience. These selective plates were incubated at 36 °C±1.5 °C for 24 to 48 hours. Colony morphology examination was assessed on commercial chromogenic differential media. In addition, for confirmation, a smear and Gram stain were performed as well as subculturing on differential media using Salt Mannitol Agar, Enterocococcel Agar, Cetrimide Agar and MacConkey Agar.
For the statistical analysis, the data were captured in the SPSS version 20 program (IBM, Chicago, United States). In the descriptive statistics, medians, means, standard deviations and 95% confidence intervals were obtained for bacterial growth counts with the different disinfectants at the three times and on the different surfaces evaluated. The frequencies of the presence of pathogenic bacteria at basal and final times were obtained. For inferential analysis, Shapiro-Wilk test for Normality and Mauchly’s Test of Sphericity were used. To identify differences in bacterial growth considering the different times, disinfectants and surfaces, a two-way repeated measures ANOVA was used. To determine differences in the presence of bacteria between the basal and final times, McNemar tests were performed. For both two-way ANOVA and McNemar’s test, values of p =0.05 were considered statistically significant.
Results
The results of the effect of the disinfectants evaluated on the microbial growth (evaluated in Log10) per surface studied are shown in Table 2, the CFU count of the day one sample was always higher compared to the second day sample and the final sample on the third day. The efficacy of the disinfectant solutions was considered on the basis of a ≥3 Log10 reduction in bacterial growth in relation to the growth of sample 1, in addition to the application time of the disinfectant solutions and the surface studied. The highest efficacy in reducing bacterial growth was observed with the application of 3% 5th generation QACs (FD 300®) with >5 Log10 reduction at the three-way syringe tip, saliva ejector converter and zero growth on the dental spitoon surface. The 5th generation QACs (Septalkan®) evaluated was the second most effective disinfectant solution in reducing bacterial growth with a 3 to 4 Log10 reduction, being particularly effective in reducing bacterial growth of the three-way syringe tip. The 0.5% NaClO had a reduction of more than 3 Log10 and thus proved to be highly effective in reducing bacterial growth on cuspidor surfaces, being less effective in disinfecting the saliva ejector converter and even less effective on three-way syringe tip surfaces. ClO2 (TwinOxide®) showed similar efficacy to 0.5% NaClO with bacterial growth reducing effects of 3 to 4 Log10 on all three surfaces tested. The 3rd generation QACs (Bactogem EQ-110®) had the lowest reduction, with reductions between 2 and 3 Log10.
Table 2 Effect of the disinfectants evaluated on bacterial growth (Log10) on the surfaces studied.
| Disinfectant | Statistics | Three-way syringe tip Log10 | Ejector nozzle Log10 | Dental spitoon Log10 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline (1st day) | 2nd day | Final (3rd day) | Baseline (1st day) | 2nd day | Final (3rd day) | Baseline (1st day) | 2nd day | Final (3rd day) | |||
| Septalkan® | mean | 7.00 | 5.25 | 3.25 | 7.25 | 4.75 | 4.50 | 9.25 | 2.00 | 2.25 | |
| CI 95% | Min. | 5.70 | 3.73 | -.28 | 5.73 | 3.23 | 3.58 | 7.73 | -1.90 | -1.93 | |
| Max. | 8.30 | 6.77 | 6.78 | 8.77 | 6.27 | 5.42 | 10.77 | 5.90 | 6.43 | ||
| Median | 7.00 | 5.50 | 4.00 | 7.50 | 4.50 | 4.50 | 9.50 | 1.50 | 2.00 | ||
| standard deviation | .816 | .957 | 2.217 | .957 | .957 | .577 | .957 | 2.449 | 2.630 | ||
| NaClO 0.5% | mean | 7.00 | 5.75 | 3.75 | 8.00 | 6.00 | 3.25 | 8.25 | 5.25 | 2.00 | |
| CI 95% | Min. | 3.10 | 3.03 | -.43 | 6.70 | 2.56 | -.51 | 4.97 | -.76 | -1.67 | |
| Max. | 10.90 | 8.47 | 7.93 | 9.30 | 9.44 | 7.01 | 11.53 | 11.26 | 5.67 | ||
| Median | 7.00 | 5.50 | 4.50 | 8.00 | 6.50 | 4.00 | 8.50 | 6.00 | 2.00 | ||
| standard deviation | 2.449 | 1.708 | 2.630 | .816 | 2.160 | 2.363 | 2.062 | 3.775 | 2.309 | ||
| TwinOxide® | mean | 8.00 | 6.25 | 4.50 | 8.50 | 7.75 | 4.75 | 9.50 | 7.50 | 3.50 | |
| CI 95% | Min. | 6.70 | 2.72 | 2.91 | 6.45 | 6.95 | 2.75 | 8.58 | 5.45 | -.29 | |
| Max. | 9.30 | 9.78 | 6.09 | 10.55 | 8.55 | 6.75 | 10.42 | 9.55 | 7.29 | ||
| Median | 8.00 | 6.00 | 4.00 | 8.50 | 8.00 | 5.00 | 9.50 | 7.50 | 4.50 | ||
| standard deviation | .816 | 2.217 | 1.000 | 1.291 | .500 | 1.258 | .577 | 1.291 | 2.380 | ||
| FD 300® | mean | 7.25 | 4.75 | .50 | 7.50 | 3.75 | 2.75 | 9.25 | 4.75 | - | |
| CI 95% | Min. | 5.25 | 3.95 | -1.09 | 3.71 | -.82 | -.26 | 6.86 | 2.36 | - | |
| Max. | 9.25 | 5.55 | 2.09 | 11.29 | 8.32 | 5.76 | 11.64 | 7.14 | - | ||
| Median | 7.00 | 5.00 | .00 | 8.50 | 4.50 | 3.50 | 10.00 | 5.00 | - | ||
| standard deviation | 1.258 | .500 | 1.000 | 2.380 | 2.872 | 1.893 | 1.500 | 1.500 | - | ||
| Bactogem EQ-110® | mean | 7.50 | 6.00 | 5.75 | 8.50 | 6.50 | 6.00 | 7.75 | 5.25 | 5.75 | |
| CI 95% | Min. | 4.74 | 4.70 | 4.95 | 7.58 | 5.58 | 4.70 | 3.57 | 2.86 | 4.95 | |
| Max. | 10.26 | 7.30 | 6.55 | 9.42 | 7.42 | 7.30 | 11.93 | 7.64 | 6.55 | ||
| Median | 7.00 | 6.00 | 6.00 | 8.50 | 6.50 | 6.00 | 8.50 | 5.00 | 6.00 | ||
| standard deviation | 1.732 | .816 | .500 | .577 | .577 | .816 | 2.630 | 1.500 | .500 | ||
CI: 95% confidence interval, Min: Minimum value, Max: Maximum Value
The frequency of pathogenic microorganisms before and after the use of disinfectants according to the selective isolates from the surfaces evaluated is shown in Table 3. It was observed that all the solutions evaluated were effective in reducing the presence of the microorganisms found (E. faecalis, KESC, P. aeruginosa, E. coli), except for the case of S. aureus (Table 3). McNemar’s tests showed that the differences were statistically significant in the reduction of E. faecalis and KESC; for S. aureus the difference represented lack of efficacy; for P. aeruginosa and E. coli a reduction was obtained in its entirety and therefore the calculation did not apply.
Table 3 Frequencies of pathogenic bacteria by species before and after the use of the disinfectants on the three surfaces evaluated.
| Bacterial growth | Frequency of bacterial species | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| P. aeruginosa | S. aureus | E. faecalis | E. coli | KESC | ||||||
| Baseline | Final growth | Baseline | Final growth | Baseline | Final growth | Baseline | Final growth | Baseline | Final growth | |
| Negative | 32 | 60 | 47 | 35 | 39 | 56 | 54 | 60 | 34 | 54 |
| Positive | 28 | 0 | 13 | 25 | 21 | 4 | 6 | 0 | 26 | 6 |
| p* | - | 0.029 | <0.001 | - | <0.001 | |||||
p* = McNemar's test
KESC= Kliebsiella, Enterobacter, Serratia, Citrobacter complex
To compare the efficacy of the different disinfectants, a two-way repeated measures ANOVA was performed, not all groups passed the Shapiro-Wilk for normality test and Levene’s test for homoscedasticity. However, the data did pass Mauchly’s test of Sphericity (Mauchly’s W 0.977, d.f. 2, p =0.603) and were therefore considered homogeneous. For the interpretation of the multivariate contrasts, the Pillai’s trace was used, which showed significant statistical differences in the different contrasts evaluated: a) bacterial growth versus disinfectant tested (F =9.769, d.f. 4, p<0.001), b) bacterial growth versus time of sample collection from surfaces (F =116.808, p<0.001), c) the interaction of sample collection times versus disinfectant (F =4.182, p<0.001) and d) the interaction of sample collection times versus surfaces tested (F =3.769, p =0.007) (Figure 1 A-C ). Pairwise comparisons of the disinfectants showed that FD 300® 3% and Septalkan® were the most efficient compared to the other disinfectants evaluated. Comparison of the effect of bacterial growth reduction on the three critical surfaces showed significant difference in all groups except Septalkan® vs. NaClO 0.5%; Septalkan® vs. FD 300®, and TwinOxide® vs. Bactogem EQ-110® (Table 4).
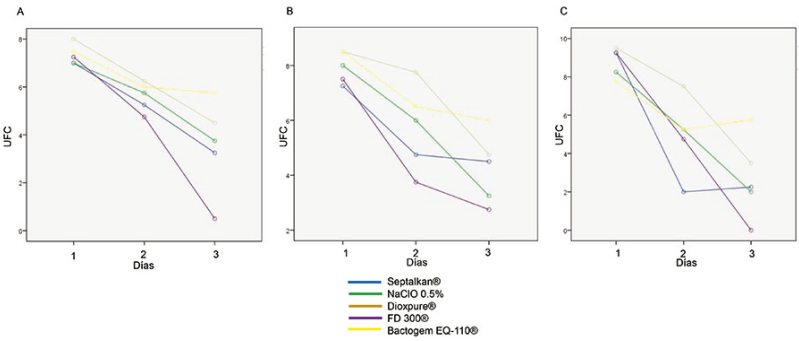
Figure 1 Relationship of bacterial growth versus days of sample collection (day one as baseline sample, day two and day three final sample) on the surfaces evaluated: three-way syringe tip (A) saliva ejector converter(B) dental spitoon (C).
Table 4 Comparative differences in the bacterial growth reduction effect of the disinfectants on the three surfaces and between the three time periods.
| Disinfectant | Disinfectant | p | 95% confidence interval for the statistical difference | |
|---|---|---|---|---|
| Lower value | Upper Value | |||
| NaClO 0.5% | Septalkan® | .338 | -.450 | 1.283 |
| FD 300® | .029 | .105 | 1.839 | |
| TwinOxide | Septalkan® | .000 | .772 | 2.506 |
| NaClO 0.5% | .007 | .355 | 2.089 | |
| FD 300® | .000 | 1.328 | 3.061 | |
| Bactogem EQ-110® | .748 | -.728 | 1.006 | |
| FD 300® | Septalkan® | .203 | -1.422 | .311 |
| Bactogem EQ-110® | Septalkan® | .001 | .633 | 2.367 |
| NaClO 0.5% | .015 | .217 | 1.950 | |
| FD 300® | .000 | 1.189 | 2.922 | |
| Time | Time | p | 95% confidence interval for the statistical differenceb | |
| Lower value | Upper Value | |||
| 1st day (basal) | 2nd day | .000 | 2.042 | 3.158 |
| 3rd day (final) | .000 | 3.926 | 5.141 | |
| 2nd day | 3rd day (final) | .000 | 1.296 | 2.570 |
b the two-way repeated measures ANOVA, p: p value
Discussion
The correct use of disinfectants after thorough and meticulous cleaning will prevent the risks of cross-infection in dental clinics; the proper and effective choice of disinfectant solution for dental chair surfaces and dental equipment is a key factor, as they have been reported to be a source of cross-contamination between patients, so proper disinfection is necessary7,16. The present study compared the effect of reducing bacterial growth with five commercial disinfectants on three critical surfaces of dental chairs in a paediatric dental clinic. The first result obtained in this study showed that 5th generation 3% QACs (FD 300®) achieved the greatest reduction of CFU at three-way syringe tip and at the ejector nozzle, and inhibited bacterial growth on the third day of evaluation in the dental spitoon. The 5th generation QACs (Septalkan®) was the disinfectant with the second highest CFU reduction, NaClO 0.5% and ClO2 obtained similar results, while the 3rd generation QACs (Bactogem EQ-110®) obtained the lowest CFU reduction.
Research has shown that in dental clinics there are surfaces and equipment with a higher risk of contamination than others, as they are more frequently used, which means that their surfaces are in greater contact with the dentist and assistant, including the patient, and therefore have a higher degree of exposure to microorganisms17. In this study, we chose the dental chair cuspidor which is a source of bacteria and is in direct contact with fluids such as saliva and blood, the ejector nozzle which contributes to the reduction of aerosols generated by rotary instruments and which is normally in use during most dental treatments and the three-way syringe tip which is exposed to the operative field. Another reason why these surfaces were chosen was because the aerosols generated by the rotating equipment reach a degree of dispersion of up to 2.5 metres, so that the concentrations of microorganisms are higher when they are closer to the patient’s mouth18.
The 5th generation QACs have a broad bactericidal, fungicidal and virucidal spectrum, with excellent germicidal performance under difficult conditions19; in this study the 5th generation QACs (FD 300®, Septalkan®) demonstrated the highest microbial reduction values. QACs are composed of polyatomic positive ions20; whereas, studies have shown that QACs are generally ineffective against polioviruses, rhinoviruses and hepatitis A21. Christensen et al. in their study demonstrated antibacterial activity of QACs against Pseudomonas, Salmonella and Staphylococcus but no effect against mycobacteria and polioviruses22; although, generation, concentration and time of action will have to be considered.
The 0.5% NaClO has demonstrated sufficient effectiveness for cleaning dental clinic workstation surfaces, and is considered one of the most widely used disinfectants worldwide23,24. The results of this study demonstrated lower efficacy of 0.5% NaClO than the QACs disinfectants; nevertheless, it demonstrated better efficacy in reducing microorganisms from the dental spitoon than from the three-way syringe tip and ejector nozzle. ClO2 had a very similar effect to 0.05% NaClO, both are used to disinfect inanimate surfaces and one of their main advantages is that ClO2is non-corrosive25,26. Biocides containing chlorine are used for surface decontamination, in the form of hypochlorite, are cheap and easy to produce. ClO2 is a highly oxidative biocide manufactured for water disinfection; with a disinfectant activity less influenced by pH resulting in trace by-products that are not very harmful, unlike conventional treatments with other chlorinated compounds27,28. Patel et al. demonstrated that ClO2 is effective against S. aureus, P. aeruginosa, S. mutans, Candida albicans, Bacillus subtilis, Mycobacterium tuberculosis, Mycobacterium avium subsp. avium and Hepatitis B virus29. In this study, the disinfectant evaluated TwinOxide® consists of 0.3% stabilised ClO2 with a ClO2 purity of more than 99% and a stability of more than thirty days according to its instructions and without by-products or residues such as free chlorine or chlorite, it kills bacteria, fungi, yeasts, viruses and spores, making it an alternative for contact disinfection of surfaces and equipment30.
Finally, the disinfectant with the lowest action was 3rd generation QACs; despite its increased biocidal activity and higher detergency versus 1st and 2nd generation quaternary ammoniums, in this study it was the disinfectant with the lowest efficacy in bacterial reduction in the three-way syringe tip, cuspidor and ejector nozzle.
The second result of this study showed the basal presence of P. aeruginosa, S. aureus, E. faecalis, E. coli and Klebsiella-Enterobacter-Serratia-Citrobacter complex, KESC, and after the disinfection protocol applying the evaluated solutions, P. aeruginosa and E. coli were completely eliminated. A statistically significant reduction of E. faecalis and KESC was obtained, though, in the case of S. aureus, the results obtained did not show a statistically significant reduction. Various studies have reported that some bacteria that can cause risks of infections linked to dental practice are S. pneumoniae, Mycobacterium tuberculosis, Klebsiella pneumoniae, E. coli, Legionella pneumophila and P. aeruginosa31, in our study we identified P. aeruginosa and the KESC complex of these bacteria. Other studies have reported that one of the most contaminated surfaces is the dental spitoon with a high number of CFU, which coincides with the result obtained in this study, while other studies have identified other microorganisms such as Klebsiella pneumoniae, Shigella sonnei y Staphylococcus aureus18.
The third result of this study demonstrated the efficacy of the disinfectants, as significant statistical differences were determined in bacterial growth versus the disinfectants evaluated, showing that 5th generation QACs significantly reduces bacterial growth versus NaClO, ClO2 and 3rd generation QACs. A difference was determined between the microbial growth obtained between the initial sample and the final sample, which means that the longer and more consistent the application, the greater the efficacy. Finally, a significant difference was demonstrated in the interaction of the times (basal and final) versus the disinfectant and the surfaces, which means that as the working days of the paediatric dental clinic went by and the greater the number of applications of the disinfectants on the surfaces evaluated in the dental units, there was a greater reduction in bacterial growth.
Future long-term studies of other critical surfaces in dental clinics should be conducted and other types of disinfectants available for medical use should be evaluated. It is recommended that dental clinic staff be trained to perform disinfection procedures, and follow previously established standardised disinfection protocols, as well as the proper use of barriers in clinical procedures32,33.
Conclusions
The implementation of a cleaning and disinfection protocol applied between each patient during three days of working days in a paediatric dental clinic proved to be effective in reducing CFU isolated from three critical surfaces in the dental units: three-way syringe tip, saliva ejector converter and cuspidor. The 5th generation QACs Septalkan® and FD 300® were the most effective, followed by ClO2 (TwinOxide®) and 0.5% NaClO. The 3rd generation QACs was insufficient to reduce the isolated microbial count. Bacterial growth decreased with all disinfectants evaluated as the evaluation periods elapsed on the three critical surfaces evaluated in the dental units.
REFERENCES
1. Weber DJ, Anderson D, Rutala WA. The Role of the Surface Environment in Healthcare-Associated Infections. Curr Opin Infect Dis. 2013; 26: 338-44. DOI: 10.1097/QCO.0b013e3283630f04 [ Links ]
2. Rutala WA, Weber DJ. Disinfection and Sterilization: An Overview. Am J Infect Control. 2013; 41(Suppl): S2-5. DOI: 10.1016/j.ajic.2012.11.005 [ Links ]
3. Rutala WA, Weber DJ. Sterilization, High-Level Disinfection and Environmental Cleaning. Infect Dis Clin North Am. 2011; 25(1): 45-76. DOI: 10.1016/j.idc.2010.11.009 [ Links ]
4. Dettenkofer M, Spencer RC. Importance of Environmental Decontamination. A Critical View. J Hosp Infect. 2007; 65(Suppl2): 55-7. DOI: 10.1016/S0195-6701(07)60016-4 [ Links ]
5. Yoo JH. Review of Disinfection and Sterilization - Back to the Basics. Infect Chemother. 2018; 50(2): 101-9. DOI: 10.3947/ic.2018.50.2.101 [ Links ]
6. Véliz E, Vergara T, Pearcy M, Dabanch J. Importancia del proceso de limpieza y desinfección de superficies críticas en un servicio dental. Impacto de un programa de intervención [Importance of Cleaning and Disinfection of Critical Surfaces in Dental Health Services. Impact of an Intervention Program]. Rev Chilena Infectol. 2018; 35(1): 88-90. DOI: 10.4067/s0716-10182018000100088. [ Links ]
7. Mupparapu M, Kothari KRM. Review of Surface Disinfection Protocols in Dentistry: A 2019 Update. Quintessence Int. 2019; 50(1): 58-65. DOI: 10.3290/j.qi.a41337 [ Links ]
8. Sánchez Vargas LO. El Control de Infecciones en el Ámbito Odontológico. Ciudad de México, México: Editorial Médica Panamericana; 2017. [ Links ]
9. Maki DG, Alvarado CJ, Hassemer CA, Zilz MA. Relation of the Inanimate Hospital Environment to Endemic Nosocomial Infection. N Engl J Med. 1982; 307(25): 1562-6. DOI: 10.1056/NEJM198212163072507 [ Links ]
10. Ibrahim NK, Alwafi HA, Sangoof SO, Turkistani AK, Alattas BM. Cross-Infection and Infection Control in Dentistry: Knowledge, Attitude and Practice of Patients Attended Dental Clinics in King Abdulaziz University Hospital, Jeddah, Saudi Arabia. J Infect Public Health. 2017; 10(4): 438-5. DOI: 10.1016/j.jiph.2016.06.002 [ Links ]
11. Quinn MM, Henneberger PK; National Institute for Occupational Safety and Health (NIOSH), Nati1onal Occupational Research Agenda (NORA), Cleaning and Disinfecting in Healthcare Working Group, Braun B, et al. Cleaning and Disinfecting Environmental Surfaces in Health Care: Toward an Integrated Framework for Infection and Occupational Illness Prevention. Am J Infect Control. 2015; 4385: 424-34. DOI: 10.1016/j.ajic.2015.01.029 [ Links ]
12. Ling ML, Ching P, Widitaputra A, Stewart A, Sirijindadirat N, Thu LTA. APSIC Guidelines for Disinfection and Sterilization of Instruments in Health Care Facilities. Antimicrob Resist Infect Control. 2018; 7: 25. DOI: 10.1186/s13756-018-0308-2 [ Links ]
13. Occupational Safety and Health Administration. Best Practices for the Safe Use of Glutaraldehyde in Health Care. OSHA 3258-08N 2006. [Internet]. [Fecha de consulta: 22 de julio de 2021]. Disponible en: Disponible en: https://www.osha.gov/sites/default/files/publications/glutaraldehyde.pdf [ Links ]
14. Engelmann AI, Dal AA, Miura CSN, Bremm LL, Boleta-Cerantio DC. Evaluation of Procedures Performed by Suregen-Dentists from Cascavel State of Parana and Surroundings for Biossecurity Control. Odontol Clin Cient. 2010; 9(2): 161-5. [ Links ]
15. American Academy of Oral and Maxillofacial. Radiology Infection Control Guidelines for Dental Radiographic Procedures. Oral Surg Oral Med Oral Pathol. 1992; 7382: 248-9. DOI: 10.1016/0030-4220(92)90202-2 [ Links ]
16. Condrin AK. Disinfection and Sterilization in Dentistry. Tex Dent J. 2014; 131(8): 604-8. [ Links ]
17. Weightman NC, Lines LD. Problems with the Decontamination of Dental Handpieces and Other Intra-Oral Dental Equipment in Hospitals. J Hosp Infect. 2004; 56(1): 1-5. DOI: 10.1016/j.jhin.2003.09.006 [ Links ]
18. Pasquarella C, Veronesi L, Castiglia P, Liguori G, Montagna MT, Napoli C, et al. Italian Multicentre Study on Microbial Environmental Contamination in Dental Clinics: A Pilot Study. Sci Total Environ. 2010; 408(19): 4045-51. DOI: 10.1016/j.scitotenv.2010.05.010 [ Links ]
19. Bureš F. Quaternary Ammonium Compounds: Simple in Structure, Complex in Application. Top Curr Chem (Cham). 2019; 377(3): 14. DOI: 10.1007/s41061-019-0239-2 [ Links ]
20. Smith GW, Smith AJ, Creanor S, Hurrell D, Bagg J, Lappin DF. Survey of the Decontamination and Maintenance of Dental Handpieces in General Dental Practice. Br Dent J. 2009; 207(4): E7; Discussion 160-1. DOI: 10.1038/sj.bdj.2009.761 [ Links ]
21. Petrocci AN. Surface Active Agents: Quaternary Ammonium Compounds. En: Block SS (Ed.). Disinfection, Sterilization, and Preservation. Philadelphia: Lea and Febiger; 1983. pp. 309-329. [ Links ]
22. Christensen RP, Robison RA, Robinson DF, Ploeger BJ, Leavitt RW, Bodily HL. Antimicrobial Activity of Environmental Surface Disinfectants in the Absence and Presence of Bioburden. J Am Dent Assoc. 1989; 119(4): 493-505. DOI: 10.1016/s0002-8177(89)94006-3 [ Links ]
23. Salvia AC, Dos Santos Matilde F, Silva FC, Kimpara ET, Cardoso AO, Balducci I, et al. Disinfection Protocols to Prevent Cross-Contamination between Dental Offices and Prosthetic Laboratories. J Infect Public Health. 2013; 6(5): 377-82. DOI: 10.1016/j.jiph.2013.04.011 [ Links ]
24. Karpay RI, Plamondon TJ, Mills SE, Dove SB. Combining Periodic and Continuous Sodium Hypochlorite Treatment to Control Biofilms in Dental Unit Water Systems. J Am Dent Assoc. 1999; 130(7): 957-65. DOI: 10.14219/jada.archive.1999.0336 [ Links ]
25. Rabenau HF, Kampf G, Cinatl J, Doerr HW. Efficacy of Various Disinfectants Against SARS Coronavirus. J Hosp Infect. 2005; 61(2): 107-11. DOI: 10.1016/j.jhin.2004.12.023 [ Links ]
26. Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of Coronaviruses on Inanimate Surfaces and their Inactivation with Biocidal Agents. J Hosp Infect . 2020; 104(3): 246-51. DOI: 10.1016/j.jhin.2020.01.022 [ Links ]
27. Russell AD, Hugo WB, Ayliffe GAJ. Principles and Practice of Disinfection, Preservation and Sterilization. 3a. ed. Londres: Blackwell; 2014. [ Links ]
28. Tzanavaras PD, Themelis DG, Kika FS. Review of Analytical Methods for the Determination of Chlorine Dioxide. Open Chemistry. 2007; 5(1): 1-12. DOI: 10.2478/s11532-006-0054-9 [ Links ]
29. Patel M, Ebonwu J, Cutler E. Comparison of Chlorine Dioxide and Dichloroisocyanurate Disinfectants for Use in the Dental Setting. SADJ. 2012; 67(7): 366-9. [ Links ]
30. Twin Oxide. [Internet]. [Fecha de consulta: 29 de junio de 2021]. Disponible en: Disponible en: https://www.twinoxide.com/es/sobre-twinoxide/ [ Links ]
31. Castiglia P, Liguori G, Montagna MT, Napoli C, Pasquarella C, Bergomi M, et al. Italian Multicenter Study on Infection Hazards during Dental Practice: Control of Environmental Microbial Contamination in Public Dental Surgeries. BMC Public Health. 2008; 8: 187. DOI: 10.1186/1471-2458-8-187 [ Links ]
32. Cardoso CT, Pinto Júnior JR, Pereira EA, Barros LM, Freitas ABDA. Contaminação de Tubos de Resina Composta Manipulados sem Barreira de Proteção [Contamination of Composite Resin Tubes Handled without a Protective Barrier]. Robrac. 2010; 18(48): 71-5. DOI: 10.36065/robrac.v19i48.421 [ Links ]
33. Zadoks RN, Van Leeuwen WB, Kreft D, Fox LK, Barkema HW, Schukken YH, Van Belkum A. Comparison of Staphylococcus Aureus Isolates from Bovine and Human Skin, Milking Equipment, and Bovine Milk by Phage Typing, Pulsed-Field Gel Electrophoresis, and Binary Typing. J Clin Microbiol. 2002; 40811: 3894-9302. DOI: 10.1128/JCM.40.11.3894-3902.2002 [ Links ]
Received: September 01, 2021; Accepted: September 01, 2022











 texto en
texto en 


