Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista odontológica mexicana
versión impresa ISSN 1870-199X
Rev. Odont. Mex vol.24 no.1 Ciudad de México ene./mar. 2020 Epub 31-Mar-2025
https://doi.org/10.22201/fo.1870199xp.2020.24.1.77547
Literature review
Fundamental principles for diagnosis, management, and treatment of odontogenic infections. A literature review
*Master’s in public health, Faculty of Nursing and Nutrition. Autonomous University of San Luis Potosí, San Luis Potosí, SLP, México.
§Department of Microbiology, Faculty of Stomatology. Autonomous University of San Luis Potosí, San Luis Potosí, SLP, México.
¶Specialty in Maxillofacial Surgery, Faculty of Stomatology. Autonomous University of San Luis Potosí, San Luis Potosí, SLP, México.
Odontogenic infections (OI) are the main cause of dental visits. They represent a significant number of hospital admissions in the emergency room and intensive care units. Therefore, the review of the literature is necessary to have greater scientific evidence that helps in the analysis and proposal of actions or guidelines to define, diagnose, and resolve odontogenic infections more effectively. In addition, it will contribute for planning more precise treatments and for preventing the inappropriate or indiscriminate use of antibiotics. This review comprised the years 2006 to 2018. It included review articles, consensus documents, and different clinical guidelines. This report aims to give the professionals the tools to timely identify the presence of odontogenic infections and associated factors (clinical and/or systemic) that may influence their approach on odontogenic infections management. It also provides the guidelines for the control and treatment of these infections.
Keywords: Odontogenic infection; antibiotics; clinical guidelines; Dentistry
Las infecciones odontogénicas (IO) son la principal causa de atención en la consulta dental, representan un número importante de ingresos hospitalarios en urgencias y unidades de cuidados intensivos, por lo que es pertinente la revisión de la literatura para sustentar la mayor evidencia científica, analizar y proponer acciones o principios para definir, diagnosticar y resolver efectivamente las infecciones odontogénicas, otorgando atención precisa que devuelva las funciones al sistema estomatólogico y que evite el uso inapropiado o indiscriminado de antibióticos. Está revisión se realizó entre el año 2006 y el 2018, incluye artículos de revisión, documentos de consenso y distintas guías clínicas, con base en ello, se proponen condiciones que identifican oportunamente la presencia de infecciones odontogénicas, factores asociados (clínicos y/o sistémicos) que pueden influir en el juicio del profesional para su manejo y las pautas de control y tratamiento de las infecciones odontogénicas.
Palabras clave: Infección odontogénica; antibióticos; guías clínicas; Odontología
Background
The odontogenic infection (OI) is the invasion and/ or multiplication of pathogenic microorganisms into dental structures or surrounding tissues. The most common OI are cavities, gingivitis, periodontitis, and pericoronitis.1 They are polymicrobial, mixed,2 and by their primary location they produce intense local symptomatology, limitation of oral functions dental loss, systemic compromise, and even death3.
OI are often a cause for emergency dental care,4,5 and the main cause of consultation at the first level of care.4-6 They account for 10% of the total antibiotic prescription in medical emergencies,7 with 6.3 days of average stay in intensive care units and mortality ranging from 0.128 to 20%.9 In Mexico, the Epidemiological Surveillance System of Oral Pathologies reported a prevalence of 93.2% for cavities10 and of 56.8% for periodontopathic bacteria,11 this report being similar to those in other countries.5,12 OI treated in emergencies are more common in men (55%) with a mean age of 37.5 ± 17.0 years. The most frequent OI are apical periodontitis (45%), abscesses (20.8%), marginal periodontitis (17.3%), pulpitis (16.3%), and pericoronitis (5.8%). 64.5% of patients receive surgical treatment and 31.7% antibiotic therapy, mostly with amoxicillin (54.5%).5
The objective of this review is to propose concrete actions for the control of OI on the basis of the highest level of evidence supported by the surveyed articles,1,4,6,13-16 consensus documents,17,18 and national/international clinical guidelines.19,20
Management and/or control
The fundamental principles for the proper diagnosis, management, and treatment of OI should be reflected in a complete medical record.6,21,22
Three steps are proposed for the control of OI as follows:
Stage I: Identification of associated factors and diagnosis
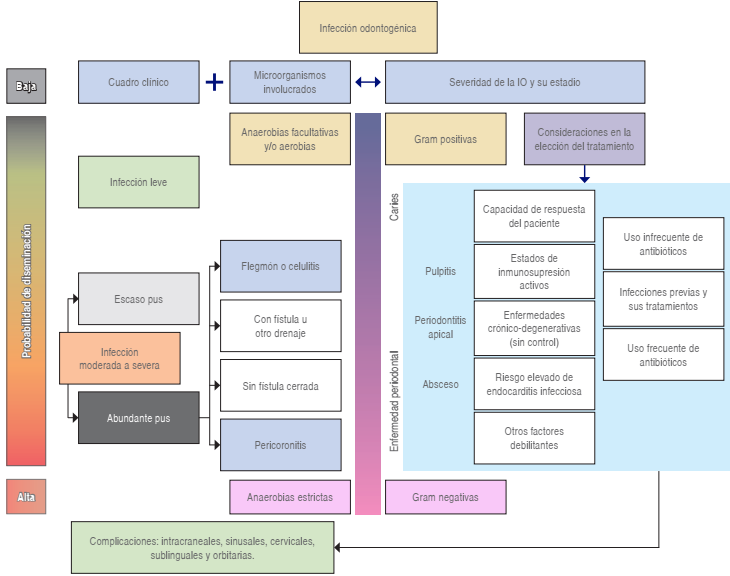
The OI must be characterized according to their nature, etiology, pathogenesis, patient’s current health status (oral and general), and health risks associated with the onset/evolution of the infection process.6,21,23,24
a) Clinical profile and microorganisms involved
A comprehensive anamnesis contributes to a presumptive or differential diagnosis.14 The symptomatology referred to by the patient, the triggering preconditions, general predictors such as inflammation,25,26 body temperature increase (> 38 °C),27 general discomfort, lymphadenopathy, pain, systemic involvement, odynophagia, dysphagia, and oral opening limitation (< 40 mm)4,28 are immediate considerations of an active infection process.4,6,25
The microbiota associated with the various OI is varied, depending on the origin of the infection and its time of evolution. Strict anaerobic gram-negative bacteria are more associated with a periodontal infection,2,24,29,30 while optional anaerobic gram-positive bacteria are highly associated with an endodontic infection.2,24,31
b) Severity and stage of infection
The ascertainment of the evolution time including days/ hours from the beginning of infection to the current status will help to establish the degree of severity of the OI. That, in addition to the signs and symptoms referred to by the patient will define the current stage of infection and the alternatives of treatment.
The OI stages are inoculation, abscess formation (location and origin of lesion), formation of phlegmon or cellulite, and resolution. Figure 1 outlines the main characteristics for identifying these stages.6,26,28,32
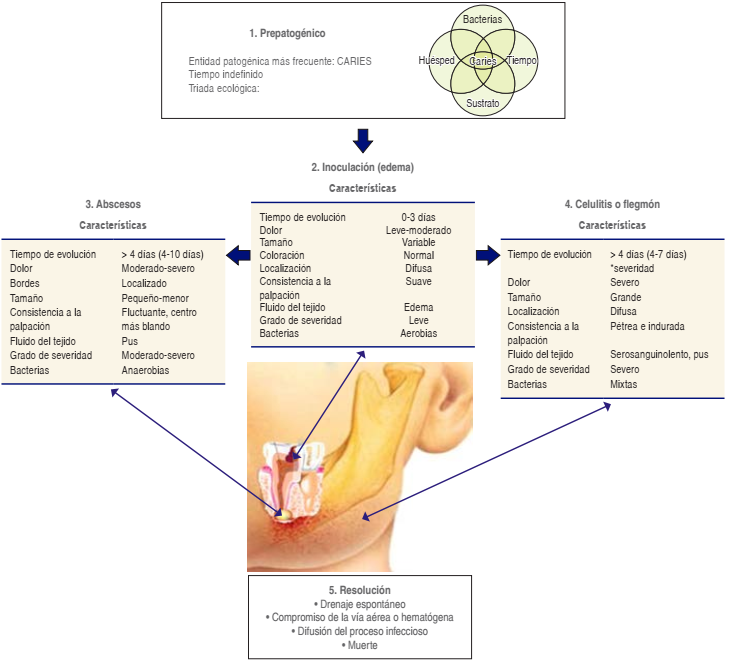
Figure 1 Odontogenic infections stages according to the natural history of the disease and its main characteristics.
c) Probability of infection spread
If the infectious process continues untreated, the risk of spread increases considerably.33 OI can extend to spaces that offer less resistance,1,2,28 causing the following:
Severe infections, such as odontogenic sinusitis, periorbital infections, or cavernous sinus thrombosis.33
Airway obstruction (main cause of mortality).6
Intracranial extension (cervical necrotizing fasciitis,34 brain abscess, meningitis,26 or mediastinitis),3 which requires immediate referral to a hospital environment.8,28
In severe cases, an odontogenic abscess can develop into peri-orbital cellulitis, a cause of intracranial and/or orbital complications.35 The thrombosis of the cavernous sinus is due to the spread of the OI into infraorbital space18 by intracranial extension direct to that sinus. Ludwig’s angina originates from second and third molar abscesses28 and can obstruct the upper airway.36
d) Patient’s response to infection
The presence of diabetes mellitus, nephropathies, heart disease, rheumatoid arthritis, malnutrition, hemophilia, and pregnancy, among others,1,6,16,17 demands adaptations in the management of OI.4,15 Most of these conditions are inflammatory as are oral diseases, which facilitates the establishment, rapid evolution, and aggravation of OI.
The adequate metabolic control and referral for consultations with other specialists are essential actions to ensure the health of patients by reducing the risk of the spread or OI complication.9
e) Patient’s history of previous infections and treatments
Alternate antimicrobial treatments should be considered,7 given the effects of synergy, competition, antagonism, or the occurrence of emerging or opportunistic infections1,22,37 due to the imbalance of the oral bacterial microbiota.30,38,39 Overuse of antibiotics, toxicity, intolerance, or development of hypersensitivity and selectivity must be documented.1,22
In Mexico, as of 2010 and after the analysis of economic factors and poor public policies,40 the government decided to regulate and control the prescription of antibiotics. However, self- medication, empirical treatment, postponement of local management,27 and ineffective posology caused by lack of follow-up and specificity are still common in the treatment of OI.16,41,42
Inadequate diagnoses and treatments, such as prescription of antibiotic medication for viral infections, increase the development of antimicrobial resistance,39,41,43 resulting in worsening or relapse processes that compromise OI control and increase recovery time and costs.44
Stage II: patient management
For the proper control of OI we propose the following actions (alone or combined):
a) Local management
OI management should focus on infection containment and dental treatment.26 In some situations it is necessary to implement supportive measures,6 including I) the prescription of NSAIDs to eliminate pain,36 II) the maintenance of metabolic balance with actions such as hydration,4 III) the control of body temperature, IV) the maintenance of glycemic balance, and V) the start of antibiotic treatment13-14 to prevent the spread of infection, thus reducing the bacterial inoculum in a qualitative and quantitative manner.1
Stage III: Types of treatment for control and/or resolution
The steps taken to control infections occurring between dental procedures prevent the risk of infection spread and/or cross-infection.1,41 Among these local antibacterial45 control measures are the use of chlorhexidine gluconate (0.12% mouthwash or gingival rinse at 2 or 3%), iodine povidone or thymol to disinfect cavities or wounds, 0.05% cetylpyridinium chloride as oral antiseptic, calcium hydroxide as intracanal medication, hydrogen peroxide hydrogen peroxide as irrigation solution, and sodium hypochlorite for irrigation in endodontic treatments.46
Surgical drains in early and self-limiting infections should be performed with simple incisions and proper tissue manipulation.26,27,47 Most OI of pulp and periodontal origin respond effectively to these procedures.22
Periodontal or surgical treatment includes mechanical debridement of necrotic tissues, removal of dental biofilm, abscess drainage, root scaling/planing, and even dental extractions, depending on the needs of the treatment to eradicate the infectious focus.1,22,30
With regard to the actions recommended for the management of OI, several authors1,13,14,16,18,48 indicate that the first line of action is the eradication of infection with local means and measures according to the origin of the infection.
b) Management of pharmacological antibiotic therapy
Management includes the use of antibiotics as adjuvants to prevent both the spread of OI and their complications due to hematogenous dissemination.14 For some authors, periodontal abscess with evidence of spread, necrotizing gingivitis, pericoronitis, cellulitis,45 as well as infections affecting cellulo-adipose spaces of head and neck region or rapidly evolving infections and fractures36 are indicative of antibiotic prescription. In the case of periapical infections, their use will depend on evolution and dissemination. Bascones et al.18 include periapical abscess and aggressive periodontitis45 if there are signs such as general discomfort, fever, trismus, dysphagia and/or dyspnea.47 Moreno Villagrana1 mentions that some conditions of pulpitis and periapical abscess may require antibiotic administration.
Table 1 summarizes the recommendations made by some authors for the antibiotic treatment of OI.1,13,16,18,48 Recommended antibiotics are beta-lactams,22 including natural penicillin,6,18,26 amoxicillin,16,18 and amoxicillin/clavulanic acid.14,49 In cases of penicillin allergy, it is suggested to use clindamycin1,15,28,49 and macrolides (such as erythromycin and azithromycin), as they have the advantage of reduced dosage.15
Table 1 Recommendations from some authors for dosage and duration of antibiotic treatment against odontogenic infections.
| Autores | Antibiótico | Dosis | Duración de tratamiento (días) |
|---|---|---|---|
| Bascones et al. (2004)18 | Amoxicilina | 1,000 mg/8-12 horas | 5 a 7 |
| Amoxicilina + ácido clavulánico | 875 mg/8 horas | ||
| 2,000 mg/12 horas | |||
| Clindamicina | 150-450 mg/6 horas | ||
| Claritromicina | 500 mg/12 horas | ||
| Doxiciclina | 100 mg/12 horas | ||
| Azitromicina | 500 mg/24 horas | 3 días consecutivos | |
| Eritromicina | 500-1,000 mg/6 horas | 5 a 7 | |
| Metronidazol | 500-750 mg/8 horas | ||
| López-Píriz et al (2007)48 | Amoxicilina + ácido clavulánico | 875/125 mg/8 horas (pulpitis o abscesos) | * |
| 2,000/125 mg/12 horas (pulpitis o abscesos) | * | ||
| Clindamicina | 600 mg/8 horas (infecciones periodontales) | * | |
| Dar-Odeh et al (2010)13 | Amoxicilina | 250-500 mg/8 horas | 3 a 7 |
| Amoxicilina + ácido clavulánico | 375-625 mg/8 horas | ||
| Clindamicina | 150-450 mg/6 horas | ||
| Metronidazol | 200 mg/8 horas | ||
| Claritromicina | 500 mg/12 horas | ||
| Doxiciclina | 100 mg 12 horas | ||
| Eritromicina | 500-750 mg/6-12 horas | ||
| Azitromicina | 500 mg/día | ||
| Moreno Villagrana et al (2012)1 | Amoxicilina | 500 mg/8 horas | * |
| 1,000 mg/12 horas | * | ||
| 250 a 500 mg/8 horas | * | ||
| 1,000 mg/8 a 12 horas | * | ||
| *500 mg/12-24 horas en ERC | * | ||
| Amoxicilina + ácido clavulánico | 500 q 875 mg + 125 mg/8 horas | * | |
| 2000 mg + 125 mg/12 horas | * | ||
| 375 a 625 + 125 mg/8 horas | * | ||
| 875 mg + 125 mg/12 horas | * | ||
| *500 mg/12-24 horas en ERC | * | ||
| Penicilina V | 500 mg/6 horas | * | |
| * | |||
| Azitromicina | 500 mg/24 horas | 3 a 7 | |
| Clindamicina | 300 mg/8 horas | 5 a 6 | |
| +600 mg/8 horas en ERC | * | ||
| 100 mg/12 horas | * | ||
| Ciprofloxacina | 500 mg/12 horas | * |
ERC = Enfermedad renal crónica.
*El autor no señala una recomendación de la duración de tratamiento antibiótico.
The American Dental Association indicates amoxicillin as the first choice. The Sanford Guide to Antimicrobial Therapy recommends broad-spectrum antibiotics such as clindamycin (300 to 450 mg/6 hours) and amoxicillin/clavulanic acid (2,000/125 mg/12 hours)20 and considers erythromycin as the best choice in case of allergy. The British National Formulary (BNF) indicates amoxicillin (250 to 500 mg/8 hours) or amoxicillin/clavulanic acid (375 to 625 mg/8 hours).18 In cases of allergy to beta-lactams, the BNF recommends clindamycin (150 to 450 mg/6 hours) or metronidazole (200 mg/8 hours).13
Recommendations on the duration of treatment are shortly mentioned.14,30 Duration will depend on the professional’s criterion and the stage of the infectious process. Herrera et al.30 argue that the duration of antibiotic therapy should be equivalent to the duration of acute lesion (two to three days); however, evidence shows that it can range from five to 10 days14,50 or up to three to four days after the eradication of signs and manifestations.14 The BNF recommends two to three days of antibiotic therapy for acute dentoalveolar OI.
c) Antibiotic prophylaxis (AP)
Prophylactic antibiotic administration for the dental procedure is recommended to decrease the possibility of complications by using high concentrations of antibiotics before, during, and after the procedures. There are some determinants for its indication; however, we recommend a thorough review of the issue for correct decision-making and choice of a proper scheme.15,20,51,52 Also, the following should be considered before use:
Trans-operative time should be reduced; there is a relative risk of bacteremia of 0.53 (95% CI, 0.49-0.57, p < 0.01).53
Clean incisions should be made with proper flap handling and gingival management.1,14,26
In case of using an antibiotic scheme, the plasma peak must be taken into account.1 Antibiotic intake is advisable two hours before or even after the intervention.15,2
Pharmacokinetic/pharmacodynamic antibiotic parameters.1
Discussion
Several drugs are used in Dentistry,6,13,14 so the proper mastery of clinical, scientific, and professional knowledge improves the consideration of basic principles related to Epidemiology, Public Health, and Evidence-Based Dentistry (EBD).52,53
Antibiotics are useful and effective in treating dental conditions, but overprescription or lack of treatment adherence triggers their excessive use or misuse.54-56 OI management often regards antibiotics as the first line of action omitting local or surgical management. Microbial dysbiosis and bacterial resistance are current alarming public health issues manifested by alarming public health issues manifested by WHO and PAHO.
Today, nonclinical factors for the issuance of prescriptions with antibiotic indication are still considered, including uncertainty in diagnosis, the patient’s influence, and even the prevention of complications.13,58
Conclusions
The omission or exclusion of diagnostic factors (OI clinical and physical manifestations, physiological or systemic conditions, and/or association between them) may have an impact on the success of treatment or damage to the patient’s health. It is key to consider all factors that contribute to the spread and severity of the dental infection (Figure 1).
The effective way to resolve OI is to first perform local mechanical therapeutic actions. The use of combined therapeutics will depend not only on the dental condition, but also on the clinical manifestations or the involvement of the systemic status.
On the basis of the clinical diagnosis of an OI, the recommendations summarized in this document aim to optimize the actions of the professional by gathering and standardizing care criteria (Figure 2).
REFERENCES
1. Moreno VA, Gómez CJ. Terapia antibiótica en odontología de práctica general. Antibiot Ther Gen Pract Dent. 2012; 69: 168-175. [ Links ]
2. Singh M, Kambalimath DH, Gupta KC. Management of odontogenic space infection with microbiology study. J Maxillofac Oral Surg. 2014; 13: 133-139. [ Links ]
3. Cariati P, Monsalve-Iglesias F, Cabello-Serrano A, Valencia- Laseca A, Garcia-Medina B. Cervical necrotizing fasciitis and acute mediastinitis of odontogenic origin: a case series. J Clin Exp Dent. 2017; 9: 150-152. [ Links ]
4. Sánchez R, Mirada E, Arias J, Paño JR, Burgueño M. Severe odontogenic infections: epidemiological, microbiological and therapeutic factors. Med Oral Patol Oral Cir Bucal. 2011; 16: 670-676. [ Links ]
5. Mahmoodi B, Weusmann J, Azaripour A, Braun B, Walter C, Willershausen B. Odontogenic infections: a 1-year retrospective study. J Contemp Dent Pract. 2015; 16: 253-258. [ Links ]
6. Velasco M I, Soto N R. Principios para el tratamiento de infecciones odontogénicas con distintos niveles de complejidad. Rev Chil Cir. 2012; 64: 586-598. [ Links ]
7. Oteri G, Panzarella V, Marcianò A, Di Fede O, Maniscalco L, Peditto M, Campisi G. Appropriateness in dentistry: a survey discovers improper proceduresin oral medicine and surgery. Int J Dent. 2018; 2018: 1-22. [ Links ]
8. Opitz D, Camerer C, Camerer D-M, Raguse J-D, Menneking H, Hoffmeister B et al. Incidence and management of severe odontogenic infections-a retrospective analysis from 2004 to 2011. J CranioMaxillofac Surg. 2015; 43: 285-289. [ Links ]
9. Rodríguez-Frausto M, Murillo-Bravo LD, Solorio S, Hernández- González MA, Villa-Barajas R. Frecuencia de infección cervicofacial odontógena que requiere hospitalización. Rev Médica Inst Mex Seguro Soc. 2011; 49: 137-140. [ Links ]
10. Grewal H, Verma M, Kumar A. Prevalence of dental caries and treatment needs in the rural child population of Nainital District, Uttaranchal. J Indian Soc Pedod Prev Dent. 2009; 27: 224-226. [ Links ]
11. Dirección General de Epidemiología. Resultados del Sistema de Vigilancia Epidemiológica de Patologías Bucales SIVEPAB 2015. [Internet]. Ciudad de México: Secretaría de Salud. Subsecretaría de Prevención y Promoción de la Salud, Centro Nacional de Programas Preventivos y Control de Enfermedades.2015. Disponible en: http://www.gob.mx/salud/documentos/informes-sivepab-2015. [ Links ]
12. Gonçalves L, Lauriti L, Yamamoto MK, Luz JGC. Characteristics and management of patients requiring hospitalization for treatment of odontogenic infections. J Craniofac Surg. 2013; 24: 458-462. [ Links ]
13. Dar-Odeh NS, Abu-Hammad OA, Al-Omiri MK, Khraisat AS, Shehabi AA. Antibiotic prescribing practices by dentists: a review. Ther Clin Risk Manag. 2010;6: 301-306. [ Links ]
14. Robles RP, de Frutos EE, Moreno MN, Mas CA, Sánchez CA, Morató AM. Me voy al dentista: ¿antibiótico como prevención o como tratamiento? Aten Prim. 2013; 45: 216-221. [ Links ]
15. Ramu C, Padmanabhan T. Indications of antibiotic prophylaxis in dental practice. Review. Asian Pac J Trop Biomed. 2012; 2: 749-754. [ Links ]
16. Teoh L, Stewart K, Marino R, McCullough M. Antibiotic resistance and relevance to general dental practice in Australia. Aust Dent J. 2018; 63: 414-421. [ Links ]
17. Merchant VA, American Academy of Orthopaedic Surgeons, American Dental Association. The new AAOS/ADA clinical practice guidelines for management of patients with prosthetic joint replacements. J Mich Dent Assoc. 2013; 95: 16-74. [ Links ]
18. Bascones-Martínez A, Aguirre-Urízar JM, Bermejo-Fenoll A, Blanco-Carrión A, Gay-Escoda C, González-Moles MA et al. Consensus statement on antimicrobial treatment of odontogenic bacterial infections. Med Oral Patol Oral Cir Bucal. 2004; 9: 369-376. [ Links ]
19. Prevención, Diagnóstico y Tratamiento de las Infecciones Odontogénicas en Adultos en el Primer y Segundo Nivel de Atención. México: Secretaría de Salud, 2014. [Internet]. Disponible en: http://www.cenetec.salud.gob.mx/descargas/gpc/CatalogoMaestro/517_GPC_Infeccionesodontogenicas/GPC_RR_PREV_DX_Y_TX_DE_INFECCIONES_ODONTOGENICAS.pdf [ Links ]
20. Agence Française de Sécurité Sanitaire des Produits de Santé (AFSSAPS). Prescription of antibiotics for oral and dental care. Médecine Mal Infect. 2012; 42: 193-202. [ Links ]
21. Cachovan G, Phark JH, Schön G, Pohlenz P, Platzer U. Odontogenic infections: an 8-year epidemiologic analysis in a dental emergency outpatient care unit. Acta Odontol Scand. 2013; 71: 518-524. [ Links ]
22. Patait M, Urvashi N, Rajderkar M, Kedar S, Shah K, Patait R. Antibiotic prescription: An oral physician's point of view. J Pharm Bioallied Sci. 2015; 7: 116-120. [ Links ]
23. Robles RP, Javierre MA, Moreno MN, Mas CA, de Frutos EE, Morató AM. Management of odontogenic infections in primary care: antibiotic? Aten Prim. 2017; 49: 611-618. [ Links ]
24. Matesanz-Pérez P, García-Gargallo M, Figuero E, Bascones- Martínez A, Sanz M, Herrera D. A systematic review on the effects of local antimicrobials as adjuncts to subgingival debridement, compared with subgingival debridement alone, in the treatment of chronic periodontitis. J Clin Periodontol. 2013; 40: 227-241. [ Links ]
25. Troeltzsch M, Lohse N, Moser N, Kauffmann P, Cordesmeyer R, Thiha A et al. A review of pathogenesis, diagnosis, treatment options, and differential diagnosis of odontogenic infections: A rather mundane pathology? Quintessence Int. 2015; 46: 351-361. [ Links ]
26. Bahl R, Sandhu S, Singh K, Sahai N, Gupta M. Odontogenic infections: microbiology and management. Contemp Clin Dent. 2014; 5: 307-311. [ Links ]
27. Igoumenakis D, Gkinis G, Kostakis G, Mezitis M, Rallis G. Severe odontogenic infections: causes of spread and their management. Surg Infect. 2014; 15: 64-68. [ Links ]
28. Brook I. Anaerobic bacteria in upper respiratory tract and head and neck infections: microbiology and treatment. Anaerobe. 2012; 18: 214-220. [ Links ]
29. Bhusari BM, Sanadi RM, Ambulgekar JR, Doshi MM, Khambatta XD. Abscesses of the periodontium: review with case series. Indian J Dent Sci. 2013; 5: 50-53. [ Links ]
30. Herrera D, Alonso B, Arriba L, Santa Cruz I, Serrano C, Sanz M. Acute periodontal lesions. Periodontol 2000. 2014; 65: 149-177. [ Links ]
31. Siqueira JF, Rôças IN. Microbiology and treatment of acute apical abscesses. Clin Microbiol Rev. 2013; 26: 255-273. [ Links ]
32. Gregoire C. How are odontogenic infections best managed? J Can Dent Assoc. 2010; 76: 114-116. [ Links ]
33. Yan W, Chakrabarti R, Choong J, Hardy T. Orbital cellulitis of odontogenic origin. Orbit Amst Neth. 2015; 34: 183-185. [ Links ]
34. Chueng K, Clinkard DJ, Enepekides D, Peerbaye Y, Lin VYW. An unusual presentation of Ludwig's angina complicated by cervical necrotizing fasciitis: a case report and review of the literature. Case Rep Otolaryngol. 2012; 2012: 1-4. [ Links ]
35. De Assis-Costa MDM, Santos GS, Maciel J, Sonoda CK, de Melo WM. Odontogenic infection causing orbital cellulitis in a pediatric patient. J Craniofac Surg. 2013; 24: 526-529. [ Links ]
36. DeAngelis AF, Barrowman RA, Harrod R, Nastri AL. Review article: Maxillofacial emergencies: oral pain and odontogenic infections. Emerg Med Australas. 2014; 26: 336-342. [ Links ]
37. Hoiby N. A short history of microbial biofilms and biofilm infections. APMIS. 2017; 125: 272-275. [ Links ]
38. Thompson JA, Oliveira RA, Xavier KB. Chemical conversations in the gut microbiota. Gut Microbes. 2016; 7: 163-170. [ Links ]
39. Aragon-Martinez OH, Isiordia-Espinoza MA, Tejeda-Nava FJ, Aranda-Romo S. Dental care professionals should avoid the administration of amoxicillin in healthy patients during third molar surgery: is antibiotic resistence the only problem? J Oral Maxillofac Surg. 2016; 74: 1512-1514. [ Links ]
40. Dreser A, Wirtz VJ, Corbett KK, Echániz G. Uso de antibióticos en México: revisión de problemas y políticas. Salud Pública México. 2008; 50: S480-487. [ Links ]
41. Oberoi SS, Dhingra C, Sharma G, Sardana D. Antibiotics in dental practice: how justified are we. Int Dent J. 2015; 65: 4-10. [ Links ]
42. Cope G, Cope A. Antibiotic resistance and how to act on it. Dent Nurs. 2013; 9: 706-709. [ Links ]
43. Cope AL, Chestnutt IG. Inappropriate prescribing of antibiotics in primary dental care: reasons and resolutions. Prim Dent J. 2014; 3: 33-37. [ Links ]
44. Jundt JS, Gutta R. Characteristics and cost impact of severe odontogenic infections. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012; 114: 558-566. [ Links ]
45. Herrera D, Matesanz P, Bascones-Martínez A, Sanz M. Local and systemic antimicrobial therapy in periodontics. J Evid- Based Dent Pract. 2012; 12 (3 Suppl): 50-60. [ Links ]
46. Bascones A, Morante S. Antisépticos orales: Revisión de la literatura y perspectiva actual. Av En Periodoncia E Implantol Oral. 2006; 18: 21-29. [ Links ]
47. Gutiérrez JL, Bagán JV, Bascones A, Llamas R, Llena J, Morales A et al. Consensus document on the use of antibiotic prophylaxis in dental surgery and procedures. Med Oral Patol Oral Cir Bucal. 2006; 11: E188-205. [ Links ]
48. López-Píriz R, Aguilar L, Giménez MJ. Management of odontogenic infection of pulpal and periodontal origin. Med Oral Patol Oral Cir Bucal. 2007; 12: E154-159. [ Links ]
49. Tancawan AL, Pato MN, Abidin KZ, Asari ASM, Thong TX, Kochhar P et al. Amoxicillin/clavulanic acid for the treatment of odontogenic infections: a randomised study comparing efficacy and tolerability versus clindamycin. Int J Dent. 2015; 2015: 1-9. [ Links ]
50. Leszczynska A, Buczko P, Buczko W, Pietruska M. Periodontal pharmacotherapy- an updated review. Adv Med Sci. 2011; 56: 123-131. [ Links ]
51. Bascones-Martínez A, Muñoz-Corcuera M, Bascones-Ilundain J. Relationship between odontogenic infections and infective endocarditis. Med Clínic. 2012; 138: 312-317. [ Links ]
52. Chrcanovic BR, Albrektsson T, Wennerberg A. Prophylactic antibiotic regimen and dental implant failure: a meta-analysis. J Oral Rehabil. 2014; 41: 941-956. [ Links ]
53. Cahill TJ, Harrison JL, Jewell P, Onakpoya I, Chambers JB, Dayer M et al. Antibiotic prophylaxis for infective endocarditis: a systematic review and meta-analysis. Heart Br Card Soc. 2017; 103: 937-944. [ Links ]
54. Skucaite N, Peciuliene V, Maneliene R, Maciulskiene V. Antibiotic prescription for the treatment of endodontic pathology: a survey among Lithuanian dentists. Med Kaunas Lith. 2010; 46: 806-813. [ Links ]
55. Tanwir F, Marrone G, Lundborg CS. Knowledge and reported practice of antibiotic prescription by dentists for common oral problems. J Coll Physicians Surg. 2013; 23: 276-281. [ Links ]
56. Wali A, Siddiqui TM, Shafiq K, Siddiqui S, Siddiqui A, Jabbar AA. Assessment of antibiotic prescription patterns amongst dental practitioners: a cross-sectional study. J Int Oral Health. 2015; 7: 5-10. [ Links ]
57. Araghi S, Sharifi R, Ahmadi G, Esfehani M, Rezaei F. The study of prescribing errors among general dentists. Glob J Health Sci. 2016; 8: 32-43. [ Links ]
58. Jayadev M, Karunakar P, Vishwanath B, Chinmayi SS, Siddhartha P, Chaitanya B. Knowledge and pattern of antibiotic and non narcotic analgesic prescription for pulpal and periapical pathologies- a survey among dentists. J Clin Diagn Res. 2014; 8: 10-14. [ Links ]
Received: August 01, 2019; Accepted: September 01, 2019











 texto en
texto en