Introduction
Diabetes is a chronic disease characterized by persistent hyperglycemia resulting from impaired insulin action or secretion (Antar et al., 2023). Type 2 diabetes (T2D) is the most prevalent form, accounting for over 90 % of cases (Ong et al., 2023). According to the International Diabetes Federation (IDF), 589 million adults are currently living with diabetes, and this figure is projected to rise to 853 million by 2050. In 2024, the disease caused approximately 3.4 million deaths (International Diabetes Federation, 2025).
Chronic hyperglycemia induces vascular damage, leading to complications that affect the kidneys, eyes, nerves, and heart, among other organs, ultimately deteriorating the quality of life and life expectancy of affected individuals (Lu et al., 2024). Glycemic control is essential for the prevention and management of diabetes-related complications. Various hypoglycemic therapies with different routes of administration (oral, subcutaneous, intranasal, etc.) have been developed to slow disease progression and improve quality of life. The current pharmacological management of diabetes includes the administration of insulin, metformin, sodium-glucose co-transporter-2 (SGLT2) inhibitor, glucagon-like peptide-1 (GLP-1) analogs, dipeptidyl peptidase-4 (DDP4) inhibitor, thiazolidinediones, and/or sulfonylureas (American Diabetes Association, 2024). Despite the wide range of treatments, the prevalence and associated morbidity and mortality of diabetes continue to rise (Zhou, B. et al., 2024), highlighting the urgent need for scientific research to develop therapies that optimize glycemic control, such as novel drug delivery systems.
Despite over a century since the discovery of insulin and over half a century since its introduction as a therapy for diabetes in humans, the development of new formulations, specifically for oral delivery, remains a critical area of research. This review discusses recent advances in oral insulin formulation and the strategies employed in preclinical trials using rats and mouse models of diabetes.
A literature search was conducted for experimental studies on oral insulin in murine models published in the last five years. Relevant publications were collected using the PubMed, ScienceDirect, and Google Scholar databases, employing a combination of keywords and MeSH terms, including “diabetes mellitus”, “insulin”, “oral insulin”, “oral administration”, “murinae”, “mice”, “rats”, “diabetes mellitus experimental”, and “drug therapy”. Priority was given to studies exploring the most recent and relevant strategies for oral insulin administration in diabetic rats and mice. All included studies declared compliance with ethical guidelines for animal experimentation.
Insulin
Insulin is a peptide hormone secreted by the β-cells of the pancreatic islets. It has a molecular weight of 5,800 Da and comprises 51 amino acids arranged in two polypeptide chains connected by two disulfide bonds. It is synthesized from the precursor pre-proinsulin and proinsulin, releasing C-peptide. Its receptor is a transmembrane tyrosine kinase protein composed of α and β subunits (Dağaşan and Erbaş, 2021). Insulin is an anabolic hormone that primarily regulates blood glucose levels. It acts on the liver, adipose, and muscle tissue, stimulating glucose storage, lipogenesis, and protein synthesis (Rahman et al., 2021). Insulin secretion is largely mediated by glucose levels but is also influenced by hormonal mechanisms (incretins, glucagon), metabolic substrates (ketones, lactate, fatty acids), and sympathetic activity (Bolli et al., 2021).
Insulin is released in cyclic pulses, with a surge in concentration occurring between 30 and 60 min postprandial, accounting for roughly one-third of the total prandial insulin release. The remaining two-thirds are secreted in response to incretin stimulation. In contrast, during fasting, insulin secretion is maintained at low concentrations and rates (Polinsky et al., 1988, cited in Bolli et al., 2021).
The discovery of insulin, a milestone in medical history, resulted from decades of research on pancreatic extracts. In 1921, Banting, Best, and MacLeod obtained a purified canine pancreatic extract and administered it in 1922 to a 14-year-old boy with type 1 diabetes (T1D). Early insulins, derived from porcine and bovine pancreases, had significant limitations, including adverse reactions because of their low purity and short duration of action. Therefore, researchers introduced purification methods like electrical precipitation and zinc crystallization to overcome these limitations (Karamanou et al., 2016). In 1950, NPH insulin (Neutral Protamine Hagedorn) was launched, becoming the first long-acting basal insulin (Bolli et al., 2021). In 1955, Sanger sequenced the primary structure of bovine insulin, enabling the synthesis of the first animal insulin in the 1960s. Finally, recombinant human insulin was effectively synthesized in 1974 (Karamanou et al., 2016). Introducing biosynthetic insulins via recombinant DNA technology in the 1980s marked a breakthrough, allowing for the development of insulin analogs with improved action profiles (Brange et al., 1988, cited in Bolli et al., 2022).
Insulin analogs are synthetic formulations designed with specific modifications in the amino acid sequence to achieve defined pharmacokinetic profiles. These include rapid-acting, short-acting, intermediate-acting, and long-acting analogs, as well as premixed combinations of rapid- and long-acting insulin for more convenient dosing based on individual requirements (Sharma et al., 2019).
Rapid-acting analogs, such as lispro and aspart, enhance absorption and minimize postprandial glucose fluctuations, making them ideal for bolus administration before meals. Long-acting analogs, like detemir and degludec, serve as basal doses, providing a flatter action profile and reducing the risk of hypoglycemia (Kramer et al., 2021). Although these analogs have improved glycemic control and patient quality of life, ongoing research focuses on developing faster and smarter insulin that automatically releases in response to plasma glucose levels (Rodbard and Rodbard, 2020).
Oral insulin
Insulin is traditionally administered subcutaneously (SC) at sites such as the abdomen, upper arms, thighs, and buttocks (American Diabetes Association, 2024). Although attempts to develop oral administration (PO) began shortly after its discovery, no commercial product is available because all tested formulations do not successfully pass clinical trials. This is mainly because gastrointestinal (GI) barriers hinder its bioavailability (Zhang et al., 2024).
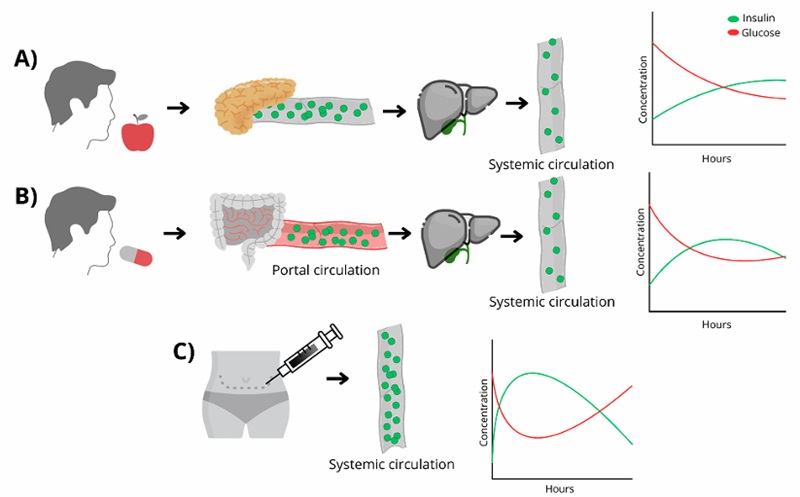
Oral insulin research holds great promise in biomedicine, as it aims to provide a non-invasive administration method that mimics the physiological secretion of the pancreas. Unlike the SC route, where insulin is absorbed into the systemic circulation, oral insulin is directed to the liver through the portal vein, thus replicating natural release patterns (Nabi-Afjadi et al., 2024) (Figure 1). This approach could optimize hepatic insulinization, reduce peripheral hyperinsulinemia, and minimize the risk of complications such as hypoglycemia, neuropathy, retinopathy, immunogenicity, and weight gain (Wong et al., 2016). Oral insulin would eliminate the need for injections, a frequent concern among patients due to fear of needles (Spain et al., 2016). Injectable insulin can also cause adverse reactions such as lipohypertrophy, hypersensitivity, infections, and pain (Demir et al., 2022). Other significant limitations of injectable therapy include affordability and the complexity of self-administration in older adults, both of which impact treatment adherence (Daniell et al., 2023).
The oral administration of peptides and proteins, such as insulin, faces multiple barriers related to their physicochemical properties and the GI environment, which limit their bioavailability and therapeutic efficacy (Verma et al., 2021). The acidic environment of the stomach compromises insulin integrity, as its peptide bonds are susceptible to enzymatic degradation by pepsin. Degradation continues in the intestine because of the action of trypsin and chymotrypsin (Peng et al., 2023). In addition, intestinal membranes present another obstacle because of the brush border enzymes and mucin secretion, a physical barrier hindering insulin absorption. Moreover, interactions with the gut microbiome influence the stability and absorption efficiency of peptides and proteins (Wong et al., 2016).
Bioavailability of oral insulin
Strategies to enhance the bioavailability of oral insulin focus on protecting its structure, overcoming physical and biological barriers, and promoting its absorption (Peng et al., 2023). Among the developed techniques, controlled-release systems stand out as they protect the hormone during its GI transit. Other approaches include using protease inhibitors to prevent intestinal degradation and applying absorption enhancers facilitating transport across epithelial cells (Su et al., 2012) (Table 1). Formulation with mucoadhesive polymers and micronization has shown potential for improving solubility and structural stability (Mumuni et al., 2020). Other innovations include chemical modification of the insulin molecule (Nabi-Afjadi et al., 2024) and administration via nanoparticles or encapsulation (Daniell et al., 2023).
Tabla 1. Clasificación de los sistemas de administración de péptidos y proteínas utilizados en la investigación de insulina oral.
| Peptide and protein drug delivery systems | ||
| According to release type | • Controlled release | |
| • Pulsatile release | ||
| According to release mechanism | • Targeted release | - Antibodies |
| - Receptors | ||
| • Stimuli-sensitive release | - Glucose-sensitive | |
| - pH sensitive | ||
| According to the delivery system used | • Nanoparticles | - Polymeric |
| - Solid lipid | ||
| - Inorganic (metal-based, silica-based) | ||
| - Liposomes | ||
| - Mucoadhesive | ||
| • Microparticles | - Polymeric | |
| - Lipid-based | ||
| - Inorganic | ||
| • Hydrogels | According to origin: | |
| - Natural | ||
| - Synthetic | ||
| In particles: | ||
| -Nanogels | ||
| -Microgels | ||
| • Self-emulsification systems | ||
| • CPP-based systems | ||
| • Mucoadhesive systems | ||
| Other mechanisms to improve bioavailability | • Enzyme inhibitors | |
| • Permeation enhancers | ||
| • Absorption enhancers | ||
| • Chemical modifications | ||
CPP: cell penetrating peptide
Protease inhibitors slow down protein degradation by binding to target enzymes, increasing the amount of protein available for absorption (Chouhan et al., 2017). Some examples that inhibit trypsin and chymotrypsin include chicken and duck ovomucoid, aprotinin, and the Bowman-Birk inhibitor (Muheem et al., 2016; Dan et al., 2020). However, these inhibitors may interact with other proteins, cause damage to the intestinal mucosa, and alter metabolic status (Verma et al., 2021).
Conversely, bile salts, surfactants, and certain fatty acids facilitate protein passage through the intestinal epithelium by enhancing absorption. These compounds can change tight junctions, increasing permeability (Alqahtani et al., 2021). Nevertheless, prolonged use may damage the intestinal mucosa and promote the translocation of pathogens and toxins (Muheem et al., 2016).
The chemical modification of peptides and proteins (whether through ligand conjugation, cyclization, or amino acid modification) can enhance resistance to enzymatic degradation, increase structural stability and cellular penetration, and reduce immunogenicity (Dan et al., 2020; Nicze et al., 2024). This approach offers a significant advantage over traditional absorption enhancers, as it does not compromise the integrity of cell membranes (Wong et al., 2016).
Mucoadhesive polymers encapsulate insulin, facilitating adhesion to the GI mucosa and creating a concentration gradient that enhances absorption. This strategy protects the drug and promotes a longer residence time at the absorption site (Dan et al., 2020). In contrast, cell-penetrating peptides (CPPs) are short peptides (fewer than 30 amino acids), either amphipathic or cationic, capable of interacting with the cell membrane and internalizing biomolecules (Kristensen and Nielsen, 2016). Endocytosis and direct translocation are the primary mechanisms by which CPP-biomolecule complexes enter cells (Korivi et al., 2021), with the advantage of not inducing significant damage to the cell membranes (Bashyal et al., 2016).
With the advancement of biotechnology, new transport systems have emerged, such as nanoparticles and microparticles, designed using natural or synthetic materials. These systems encapsulate insulin, protecting it from degradation and improving its absorption. Some examples of material used in this method include lecithin, chitosan, and poly(lactic-co-glycolic acid) (PLGA) (Wong et al., 2018; Cao et al., 2019).
Liposomes, vesicles formed by concentric lipid bilayers, can encapsulate hydrophilic and hydrophobic drugs, thus enhancing their bioavailability (He et al., 2019) (Table 1). Micro and nanoemulsions, thermodynamically stable isotropic solutions, comparably incorporate hydrophilic and hydrophobic molecules, improving drug distribution and penetration (Homayun et al., 2019; Nicze et al., 2024). Further innovative strategies include bilosomes, gas-enhanced delivery systems, and endogenous cellular transport systems (Xu et al., 2020).
Murine models for diabetes research
Using animals in research has been crucial for understanding the etiology and pathophysiology processes of diabetes, as well as for developing new drugs to treat it and its complications (Kottaisamy et al., 2021). Despite scientific and technological progress in the biomedical field, animal models continue to play an irreplaceable role in preclinical studies (Athmuri and Shiekh, 2023). Since the 17th century, diabetes research has relied on experimental animals, with murine models predominating since the 1940s (Martín-Carro et al., 2023; Pandey et al., 2023).
Laboratory rats and mice share biological similarities with humans, with close to 95 % of their genes in common. Their small size, short gestation period, rapid development, and lower cost compared to other animals are key factors that make them ideal for experimental studies (Bryda, 2013).
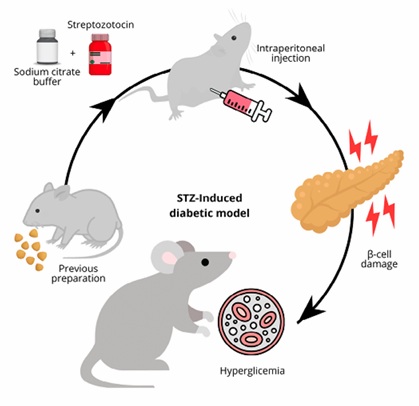
Experimental models classify according to the type of diabetes they represent (type 1 or type 2) and the method by which the disease develops (spontaneous, induced, or genetically manipulated) (López-Soto et al., 2024). Chemical induction models are the most used for studying the antidiabetic activity of new drugs and insulin formulations (Janapati and Junapudi, 2024). The induction protocols vary in dosage, age, sex, and weight of the specimens, as well as the route and timing of administration (Pandey et al., 2023). The most common chemical induction agents are streptozotocin (STZ) and alloxan, which progressively destroy pancreatic β-cells, resulting in insulin deficiency and sustained hyperglycemia (Athmuri and Shiekh, 2023) (Figure 2). Depending on the aims, high-fat diets sometimes accompany this process (Furman, 2021).
Some rodents spontaneously develop autoimmune diabetes, considering genetic predisposition. These models facilitate the evaluation of the activity of active compounds without the interference of adverse effects induced by agents like STZ (Sharma et al., 2016); for example, the Biobreeding (BB) rats, LEW.1AR1-iddm rats, and the NOD mouse (López-Soto et al., 2024). As shown in Table 2, the most frequently used rat strains in recent years include Sprague-Dawley and Wistar, along with various mouse strains such as ICR, C57BL/6J, KM, Akita, Kunming, and db/db.
Tabla 2. Ensayos preclínicos de insulina oral utilizando modelos murinos.
| Author, year | Diabetic model | Strategy for insulin delivery | Mechanism of action | Formulation | Results |
| Alfa et al. (2024) | Alloxan-induced model Wistar rats | Polymeric microparticles | Sustained release, protection from enzymatic degradation, improved stability. | Gelatinized PEG microparticles. | Formulations with a higher proportion of gelatin significantly reduced blood glucose levels—no signs of liver toxicity. |
| Li et al. (2024) | STZ-induced model ICR mice | pH- and hydrogen peroxide-sensitive nanoparticles | pH-responsive release, glucose-responsive release, and protection from enzymatic degradation. | Amphiphilic polymer incorporated with PBE-conjugated PHEMA and PCB to encapsulate glucose oxidase and insulin. | A sustained hypoglycemic effect, with an oral insulin bioavailability of 20.24 %. |
| Pang et al. (2024) | STZ-induced model C57BL/6 J mice | Polysaccharide-coated nanoparticles | Sustained release, protection from enzymatic degradation, enhanced cellular uptake. | Layered double hydroxide nanocomposites coated with chitosan and alginate. | Significant reduction in plasma glucose levels. Slower and more sustained release, compared to SC insulin. |
| Sharmah et al. (2024) | STZ-induced model Wistar rats | Glucose-responsive nanoparticles | Glucose-responsive release, enhanced cellular uptake and antioxidant activity. | Nanoceria nanoparticles are functionalized with phenylboronic acid and coated with dextran. | Excellent insulin loading capacity and controlled release, Increased bioavailability, maintenance of normoglycemia, and antioxidant activity. |
| Zhou et al. (2024) | STZ-induced model KM mice | Polysaccharide-based nanoparticles | Sustained release, anti-inflammatory effects, enhanced cellular uptake. | The nanoparticle system comprises Fucoidan, Concanavalin A, and Vitamin B12. | Prolonged and stable reduction in glucose levels with a lower risk of hypoglycemia. Anti-inflammatory benefits activity. |
| Chamsai et al. (2023) | STZ-induced model ICR mice | Polysaccharide-based nanoparticles | Mucoadhesion, sustained release, protection from enzymatic degradation. | Tablets and films were developed with thiolated chitosan-based nanoparticles, encapsulated with Cyclodextrin complexes. | Rapid disintegration, high insulin release, good mucoadhesive properties, and reduction in blood glucose levels with a prolonged hypoglycemic effect. |
| Chu et al. (2023) | STZ-induced model Sprague-Dawley rats | Lipid nanoparticles | Sustained release, protection from enzymatic degradation, enhanced cellular uptake. | Reverse micelles of insulin-containing sodium deoxycholate, sulfobetaine 12, and chitosan. | Significantly higher bioavailability compared to free oral insulin and a greater hypoglycemic response than SC insulin. |
| Rehmani et al. (2023) | STZ-induced model HsdOla:TO mice | CPP | Enhanced cellular uptake, sustained release, and protection from enzymatic degradation. | Glycosaminoglycan-binding enhanced transduction uses a peptide designed to bind to heparan sulfate glycans. | Enhance insulin absorption and facilitate its intracellular transit and progressive release within cells, thus achieving sustained normoglycemia. |
| Ren et al. (2023) | STZ-induced model C57BL/6J mice | pH-sensitive hydrogel | pH-responsive release, sustained release, improved insulin sensitivity. | Microalgae with Chlorella vulgaris, combined with sodium alginate. | More lasting and effective hypoglycemic effects compared to SC insulin, improved insulin sensitivity and favorably regulated the gut microbiota. |
| Shapira‐Furman and Domb (2023) | Spontaneous diabetic model Akita mice | Polymeric nanoparticles | Sustained release, protection from enzymatic degradation. | Biodegradable copolymer of polylactic acid (D-PLA) and PEG. | A significant reduction in blood glucose levels and a normal increase in body weight. |
| Asal et al., (2022) | STZ-induced model Swiss albino rats | Polymeric and nanoparticle composites | Protection from enzymatic degradation, sustained release, and enhanced cellular uptake. | Chitosan-based nanoparticles, gold nanoparticles coated with chitosan, and gold nanoparticles functionalized with PLGA. | Significant reduction in blood glucose and decrease in Hb1Ac (greater in ChAuNps/PLGA), with a gradual increase in insulin retention in acidic pH. |
| Goo et al. (2022) | STZ-induced model Sprague-Dawley rats | Self-micro emulsifying drug delivery system (SMEDDS) | Enhanced absorption, protection from enzymatic degradation, and improved stability. | Insulin complexed with anionic counterions (sodium octadecyl sulfate, sodium oleate, and sodium deoxycholate), surfactants (Tween 20, Brij L4, Cremophor EL), cosurfactants (Labrasol and Tetraglycol), and oils (Capmul MCM and Capryol 90). | Significant improvement in insulin stability against digestive enzymes and a greater reduction in glucose levels compared to free insulin. |
| Xu et al. (2022) | STZ-induced model Male BALB/c mice | Polysaccharide-based nanoparticles | Glucose-responsive release, sustained release, enhanced cellular uptake. | A combination of konjac glucomannan and concanavalin A is used as an oral delivery system. | Reduced blood glucose levels for up to 6 h, prolonged insulin release, and biocompatibility. |
| Zhang, F. et al. (2022) | STZ-induced model Sprague-Dawley rats | Polysaccharide-based nanoparticles | Sustained release, protection from enzymatic degradation, targeted delivery. | Pectin nanoparticles were modified with folic acid, using a double-crosslinking method with calcium and dihydrazide adipate. | High encapsulation efficiency and stability against degradation Significant reduction in glucose levels and improved bioavailability. |
| Zhang, H. et al. (2022) | STZ-induced model Wistar rats | Polysaccharide-based nano hydrogel | pH-responsive release, sustained release, and protection from enzymatic degradation. | Insulin-loaded nanohydrogel based on O-carboxymethyl chitosan and sodium alginate. | High insulin loading capacity and a controlled, sustained release of insulin for over 12 h, promoting its release in the small intestine and effective regulation of blood glucose levels. |
| Ito et al. (2021) | STZ-induced model ICR mice | CPP | Enhanced cellular uptake, sustained release, and protection from enzymatic degradation. | A small intestine-permeable cyclic peptide (cyclic L-DNP peptide) and Zn-insulin. | Enhance insulin absorption and show a rapid onset of blood glucose reduction. |
| Morales-Burgos et al. (2021) | STZ-induced model Wistar rats | Polysaccharide-based microspheres | Targeted delivery to the colon, protection from enzymatic degradation. | Arabinoxylan microspheres with a focus on colon-targeted release. | 39 % reduction in glucose levels (maximum effect after 18 h) and probably probiotic effect. |
| Muntoni et al. (2021) | STZ-induced model Wistar rats | Lipid nanoparticles | Sustained release and protection from enzymatic degradation. | Insulin glargine loaded in nanostructured lipid carriers. | Insulin release exhibited a biphasic pattern. However, the capsules were the only ones that showed efficacy compared to tablets and liquid formulations. |
| Yang et al. (2021) | STZ-induced model KM mice | Polysaccharide-based hydrogel microparticles | Sustained release, improvement in insulin sensitivity, protection from enzymatic degradation. | Polysaccharide-based hydrogel microparticles with chitosan and β-cyclodextrin. | Improve the clinical manifestations of diabetes and insulin resistance, increase in insulin sensitivity, reduction in fasting glucose levels, regulation of lipid metabolism, and antioxidant capacity. |
| Raguraman et al. (2020) | STZ-induced model Wistar rats | Magnetosomes microparticles | Targeted delivery, enhanced cellular uptake, and protection from enzymatic degradation. | Microscopic particles composed of magnetite crystals conjugated with PEG. | Significant reduction in fasting blood glucose levels and a notable improvement in triglyceride, total cholesterol, and liver enzyme levels. |
| Zhou et al. (2020) | STZ-induced model Wistar rats | Polyeric microparticles and iron-based nanoparticles | Sustained release, targeted delivery, and protection from enzymatic degradation. | mPEG-b-PLLA microspheres incorporated with an iron-based nanoparticle modified with sodium dodecyl sulfate. | Greater intestinal absorption compared to free insulin, reduced blood glucose in a more prolonged manner than SC insulin. Insulin was efficiently distributed to the liver and kidneys. |
| Mudassir et al. (2019) | STZ-induced model Sprague Dawley rats | Polyelectrolyte complex nanogels (polymers) | Protection from enzymatic degradation, enhanced cellular uptake, and sustained release. | pH-sensitive polyelectrolyte nanogels, composed of methyl methacrylate and itaconic acid. | Reduced glucose levels by 51.10 % after 6 hours. The nanogels facilitated absorption through the inhibition of proteolytic enzymes and the opening of intestinal tight junctions. |
| Wang et al. (2019) | STZ-induced model Sprague-Dawley rats | Liposomes | Enhanced mucosal penetration, sustained release, and protection from enzymatic degradation. | Cationic liposomes with a 'protein corona' formed by BSA. | Higher ability to penetrate the mucus layer and undergo transepithelial transport, and achieved a slower, sustained insulin release compared to free insulin. |
BSA, bovine serum albumin; CPP, cell-penetrating peptide; KM, Kunming; PBE, phenylboronic ester; PCB, poly(carboxybetaine); PEG, polyethylene glycol; PHEMA, (PBE)-conjugated poly(2-hydroxyethyl methacrylate); PLGA, poly(lactic-co-glycolic) acid; PLLA, polylactic acid; SC, subcutaneous; STZ; streptozocin.
Recent advances in the study of oral insulin
Nanoparticles
A predominant approach in the past few years is the use of nanoparticles, a versatile platform for insulin delivery with sizes smaller than 100 nm. It is essential to highlight that various strategies can be combined to improve bioavailability in these animal models when creating a single oral insulin formulation, for example, a glucose-sensitive polymeric nanoparticle integrated with an enzyme inhibitor.
Stimuli-responsive release nanoparticles
Sharmah et al. (2024), developed nanoceria nanoparticles functionalized with phenylboronic acid coated with dextran and showed excellent insulin loading capacity and glucose-sensitive release. This formulation showed higher bioavailability, more effective glucose control than SC insulin, and antioxidant properties in Wistar rats with STZ-induced diabetes. Li et al. (2024), investigated pH- and H2O2-sensitive nanoparticles to encapsulate insulin and glucose oxidase, achieving significantly higher oral insulin bioavailability than SC administration in an STZ-induced diabetic ICR mouse model.
Extended-release nanoparticles
Shapira‐Furman and Domb (2023), formulated nanoparticles made from a biodegradable copolymer of polylactic acid (D-PLA) and polyethylene glycol (PEG) that achieved sustained insulin release for 14 weeks in Akita mice, resulting in significant glucose regulation. Other studies have focused on nanoparticles with hypoglycemic effects and additional properties. Pang et al. (2024), developed double-layered hydroxide nanoparticles coated with chitosan and alginate, demonstrating a slower release than SC insulin in STZ-induced C57BL/6J mice. Analogously, nano-carriers with Fucoidan, concanavalin A, and vitamin B12 exhibited long-lasting hypoglycemic effects and anti-inflammatory properties, as well as a lower risk of hypoglycemia compared to SC administration in STZ-induced KM mice (Zhou, J. et al., 2024).
Mucosal penetration enhancement nanoparticles
Other plausible nanoparticles include chitosan-functionalized PLGA nanoparticles (Asal et al., 2022), pectin-modified folic acid nanoparticles (Zhang, F. et al., 2022), and konjac glucomannan and Concanavalin nanoparticles (Xu et al., 2022), which have achieved better penetration of the intestinal mucosa and greater bioavailability.
Liposomal and Lipid-Based Systems
Wang et al. (2018), employed cationic liposomes with a bovine serum albumin protein corona to enhance mucosal penetration and transepithelial transport of insulin in Sprague-Dawley rats with STZ-induced diabetes. Muntoni et al. (2021), used nanostructured lipid systems to deliver insulin glargine, achieving a biphasic release pattern. In their study, only capsules demonstrated efficacy in Wistar rats with STZ-induced diabetes, while tablets and liquid formulations failed to show a significant hypoglycemic effect.
Microparticles
Like nanoparticles but larger, the microparticles have also been explored for controlled insulin release. Yang et al. (2021), developed polysaccharide microspheres with chitosan and β-cyclodextrin. These microspheres improved clinical diabetes symptoms and insulin resistance, showing a significant reversal of pancreatic damage in Kunming mice with STZ-induced diabetes. Additionally, arabinoxylan microspheres have been studied for colon-targeted insulin release, exhibiting probiotic effects in Wistar rats with STZ-induced diabetes (Morales-Burgos et al., 2021). Biodegradable mPEG-b-PLLA microspheres incorporating iron-based nanoparticles revealed higher intestinal absorption and greater glycemic reduction than SC insulin or free oral insulin in vivo in Wistar rats with STZ-induced diabetes (Zhou et al., 2020).
Hydrogels
Hydrogels, polymeric networks capable of retaining a large amount of water and providing a protective environment for insulin, represent a promising system for oral insulin delivery and can be administered as nano- or microparticles. For instance, nanogels of methyl methacrylate and itaconic acid have demonstrated high encapsulation efficiency in Sprague-Dawley rat models with STZ-induced diabetes (Mudassir et al., 2019).
Self-Microemulsifying Drug Delivery Systems (SMEDDS)
SMEDDS are mixtures of oils, surfactants, and co-surfactants that form emulsions upon dilution in aqueous media, enhancing drug solubilization. Goo et al. (2022), developed a formulation based on this system that protected against enzymatic degradation and improved glucose absorption in STZ-induced diabetic Sprague-Dawley rats. However, challenges remain in matching the efficacy of SC insulin.
Cell-Penetrating Peptide (CPP)-Based Systems
Various CPP-based strategies have been examined to enhance cellular uptake and transport of insulin. Rehmani et al. (2023), employed a peptide to bind heparan sulfate glycans on cell membranes, resulting in improved insulin absorption, enhanced intracellular transit, and progressive release within cells in STZ-induced diabetic HsdOla:TO mice. Similarly, Ito et al. (2021), reported that intestinally permeable peptides such as the cyclic L-DNP peptide C-DNPGNET-C, when combined with more stable insulin formulations like Zn-insulin, exhibited efficient absorption into the portal vein without toxic effects. These CPP-based systems led to a significant reduction in glucose levels in STZ-induced diabetic ICR mice.
Smart Drug Delivery Systems (SDDS)
SDDS have gained prominence in modern medicine, particularly for blood glucose regulation in patients with T2D. These systems enable controlled drug release in response to specific stimuli, such as pH changes or certain enzymes (Elema et al., 2020). Examples of SDDS for oral insulin formulations include those loaded with glucose oxidase, phenylboronic acid, and lectin (Bordbar-Khiabani and Gasik, 2022).
The primary release mechanism involves glucose diffusion, its conversion to glucuronic acid, and subsequent pH reduction, which triggers degradation, swelling, or binding disruption, leading to insulin release (Wang et al., 2020). Key advantages of SDDS include enhanced therapeutic efficacy, resulting in better glycemic control and fewer side effects. Limitations include the complexity of their design and a restricted drug-loading capacity (Boppana et al., 2024).
Translation to clinical trials and limitations
Despite promising results in murine models, translating oral insulin to clinical practice remains challenging due to multiple factors hindering success in clinical trials. First, various methodological and biological issues compromise the external validity of preclinical studies. The lack of publication of negative results at this stage represents a bias, overestimating the likelihood of treatment success. Moreover, the absence of standardized protocols for animal experimentation, the inability of models to replicate accurately the complexity of human diabetes, and the stress associated with laboratory conditions can substantially influence results (Mak et al., 2014). These factors, along with inadequate experimental design, method flaws, cost, commercial competition, and suboptimal statistical analysis, contribute to the poor reproducibility of preclinical findings in clinical settings (McGonigle and Ruggeri, 2014; Ioannidis et al., 2018).
Rigorous processes such as randomization, blinding, including animals of all sexes and different age groups, and the transparent publication of results are essential for enhancing the predictability of preclinical studies (Mak et al., 2014). If a clinical trial fails, reverse translation of clinical findings to animal models can provide valuable insights to refine and guide future research (Denayer et al., 2014).
The difficulty in translating oral insulin formulation is evident in the low proportion that progresses beyond phase 2 clinical trials (Table 3). For example, the formulation I338, a basal insulin analog that demonstrated similar glucose control to SC insulin glargine in its early stages, was eventually discontinued due to the requirement for high doses (Halberg et al., 2019). Similarly, Novo Nordisk® halted the development of NN1952, a modified prandial insulin, due to unacceptable interactions with food intake (Zijlstra et al., 2014). The Eligen® formulation, even with a faster onset of action than regular SC insulin, exhibited high variability in absorption among patients, negatively affecting its clinical efficacy in a phase 2 trial and preventing further development (Kapitza et al., 2010; Dan et al., 2020). These cases highlight the complexity of translating preclinical results into real clinical settings and underscore the need to overcome multiple obstacles before achieving a viable formulation.
Tabla 3. Ensayos clínicos de insulina oral.
| Trial registration number | Name | Release strategy | Formulation | Status | References |
| NCT06731075 | ORMD-0801 | Absorption enhancer / Enzyme inhibitor | Human insulin, soytrypsin inhibitor, disodium ethylenediaminetetraacetic acid, Aerosil 200, Tween 80, and enteric coating. | Phase 2 (phase 3 scheduled for 2025) | Eldor et al. (2023) |
| CTRI/2018/08/015519 | Capsulin | Absorption enhancer | Recombinant human insulin, antioxidant bile salt, and enteric coating. | Phase 2 | New et al. (2023) |
| NCT04975022 | N11005 | Solid self-emulsifying microemulsion | Recombinant human insulin and solid self-emulsification system Oralpas Pro® | Phase 1 | Pan et al. (2023) |
| NCT02470039 | I-338 | Absorption enhancer / Chemical modification | Long-acting basal insulin analogue and sodium caprate. | Phase 2 | Halberg et al. (2019) |
| NCT03430856 | Tregopil (IN-105) | Absorption enhancer / Chemical modification | Chemically modified human insulin, sodium caprate, and polyethylene glycol (PEG). | Phase 2 / Phase 3 | Khedkar et al. (2019) |
| NCT01028404 | NN1952 | Chemical modification | Chemically modified human insulin and enteric coating. | Phase 1 | Novo Nordisk A/S (2017) |
| NCT00982254 | Eligen | Permeation enhancer | Insulin and N-(4-chlorosalicyloil)-4-aminobutyrate monosodium (4-CNAB). | Phase 2 | Kapitza et al. (2010) |
However, oral insulin research remains active, focusing on formulations with potential approval. ORMD-0801, an enteric-coated capsule, demonstrated a significant reduction in HbA1c levels in a phase 2 study (Eldor et al., 2023), which justifies initiating a phase 3 trial scheduled to begin in 2025 (Oramed, 2025). Tregopil (IN-105), although yielding mixed results in glucose control compared to SC insulin aspart, has made progress by reaching phase 3 and is still under investigation (Lebovitz et al., 2022). Likewise, the formulation N11005, a solid self-emulsifying system, exhibited a faster onset of action in a phase 1 study, consistent with preclinical results in rats (Beijing Hospital, 2021; Pan et al., 2023).
Perspectives
Advanced technologies and smart systems
Advanced approaches in micro- and nanotechnology, such as metallic nanoparticles, polymers, liposomes, emulsions, nanocapsules, and DNA nanoparticles, offer advantages in drug targeting and toxicity reduction (Sun et al., 2020; Sahu et al., 2021). Besides conventional strategies, stimulus-sensitive systems made from smart biomaterials represent a significant step forward in personalized insulin therapy and optimal glycemic control (Sultana et al., 2022).
Correspondingly, micro-electromechanical systems, including implantable microchips and micropumps, are emerging as key tools for drug-controlled release (Chircov and Grumezescu, 2022). Additionally, shape-memory materials and 4D printing technology, which enable dynamic and responsive drug delivery systems, open new avenues for designing even more sophisticated devices that can adapt to the individual characteristics of each patient (Amukarimi et al., 2021).
Advanced preclinical models
Optimizing existing animal model management through improved experimental design and execution, as well as investing in more sophisticated models that closely resemble human diabetes, is crucial. Innovations such as humanized mouse models (Fujiwara, 2018) and “organs-on-chips” technology (Wysoczański et al., 2024), which offer greater physiological relevance and simulate human physiology, could facilitate the analysis of insulin interactions in different organs (Ma et al., 2021).
Incorporating characteristics from clinical trials into preclinical studies can further improve the predictability of animal models. These characteristics include the evaluation of predictive biomarkers and the reverse translation of clinical results to experimental models (Denayer et al., 2014).
Although these technologies have made significant progress, many are still in the early stages of development. Therefore, increased investment in research is essential to translate these technologies into clinical practice (Xiao et al., 2020).
Conclusions
The development of oral insulin remains an up-and-coming area in medicine, potentially transforming diabetes treatment. Murine models have proven essential in diabetes research and the development of new therapies, leading to significant advancements in the design of drug delivery systems that aim to improve insulin bioavailability and reduce the side effects of SC administration. However, translating these innovations to clinical practice remains challenging, underscoring the need to optimize experimental models and continue investing in new technologies. In this context, the prospects for more effective, personalized, and less invasive insulin therapy continue to strengthen.











 nueva página del texto (beta)
nueva página del texto (beta)