Introduction
At the commercial level, there has been a recent increase in consumer interest for the use of substances of natural origin, especially plant extracts with therapeutic properties, to treat diseases such as obesity, kidney disease, cardiovascular disease, cancer, etc., among which is the stevia plant (Saraiva et al., 2020).
Stevia rebaudiana Bertoni is a plant of commercial interest, known for its high content of steviol glycosides (SGs) (Cauich-Cauich et al., 2022; Javed et al., 2017a). In 2024, the global market for S. rebaudiana will be valued at $405.6 million and is projected to reach $739.4 million in 2034, reflecting a compound annual growth rate (CAGR) of 6.2 % (Stevia Market, 2024).
Stevioside (Stev), rebaudioside A (Reb A) and rebaudioside C (Reb C), are the most abundant SGs in the leaves, and commercially these molecules have been attributed the sweetening power. Other SGs, such as rebaudiosides D, E, and M, have greater sweetening capacity, however, they are found at very low concentrations (Gupta et al., 2013). In addition, other studies have shown that the leaves of S. rebaudiana contain antioxidant compounds such as ascorbic acid, gallic acid and other phenolic compounds, including flavonoids, alkaloids, chlorophylls, xanthophylls and hydroxycinnamic acids (Pandey et al., 2018) that confer additional functional properties to the sweetening power, so the implementation of agronomic techniques that induce the biosynthesis of these compounds is an area of interest.
Today, the pharmaceutical and food industries have a growing interest in improving the quantity and quality of the active principles of medicinal plants (Ramezani et al., 2020). Increasing the production of secondary metabolites by modifying plant metabolism, is an efficient approach that could be achieved through several means.
Elicitors are biotic or abiotic substances or factors that can induce a metabolic response in plants depending mainly on their concentration, chemical nature, size and growth stage of the plant (Wang et al., 2017). Some phytohormones, such as methyl jasmonate (MeJa), have been used as elicitors. This phytohormone plays a crucial role in signaling pathways leading to the accumulation of secondary metabolites, which can have diverse applications including pharmaceuticals, flavors, and fragrances. However, their cost can be a challenge, especially for large-scale applications (Lucho et al., 2018; Moharramnejad et al., 2019).
Recently, research in the field of nanotechnology for signaling the effects of environmental stress on plant physiology is advancing at a rapid pace (Abdel-Aziz et al., 2020). Nanotechnology has the potential to revolutionize agriculture and significantly impact food and crop production (Magdaleno-García et al., 2023). Scientists are increasingly interested in employing biotechnological techniques to enhance the production of secondary metabolites and explore various applications in agriculture such as the formulation of nanopesticides and nanofertilizers (Nongbet et al., 2022).
Recent studies have shown that nanoparticles can act as elicitors (Vijayakumar et al., 2022) as these particles can rapidly penetrate plant cells, elevate protein levels and stimulate gene expression (Masarovicová et al., 2013). Javed et al. (2017a) investigated the effect of different concentrations of zinc oxide chemical nanoparticles (ZnO NPs) on growth and steviol glycoside production in micropropagated shoots of S. rebaudiana. They found that 1 mg/L of ZnO nanoparticles produced the highest percentage of shoot formation (89.6 %), by doubling the content of stevioside and rebaudioside A compared to the control, suggesting a positive influence of nanoparticles on plant growth and glucoside production.
The green synthesis of nanoparticles is particularly attractive due to their biocompatibility, low toxicity, and eco-friendly nature (Bouttier-Figueroa et al., 2023). Key considerations for synthesizing and producing plant-based nanoparticles include the selection of environmentally friendly or non-harmful solvents for stabilization (Raliya et al., 2014). Traditional chemical methods used in various industries can be costly and environmentally hazardous, especially when they involve toxic and hazardous chemicals (Krishnani et al., 2022). This has driven increased interest in green chemistry and sustainable practices that aim to minimize the environmental and health impacts of chemical processes (Gowri et al., 2019).
The main objective of this research was to determine how the application of ZnO and MgO nanoparticles as elicitors affects the content of certain compounds in dried Stevia rebaudiana leaves. Specifically, the study aims to measure changes in the levels of rebaudiosides A, D, M and address the metabolic profile of phenolic compounds such as gallic acid, chlorogenic acid, caffeic acid and caffeine.
Material and methods
Synthesis and characterization of ZnO and MgO phytonanoparticles from Moringa oleifera
The plant extract used for the synthesis of PTNPs was obtained from previously pulverized dehydrated leaves of Moringa oleifera, as described by Rodríguez-Pérez et al. (2015). Briefly, 500 mg of plant material were mixed with 25 mL of distilled water and maintained at 25 °C for 1 hour under continuous magnetic stirring. The mixture was sonicated at 360 W (COLE-PARMER, Vernon Hills, IL, USA) for 15 minutes at 25 °C and centrifuged at 4500 rpm for 10 minutes (HERMLE, Germany) to remove solids. The extract was filtered through a 0.45 μm Millipore membrane.
The synthesis of zinc and magnesium PTNPs was carried out following the methodology described by Chaudhuri and Malodia (2017), with modifications as reported by Velázquez-Gamboa et al. (2021).
Characterization techniques were used to obtain a comprehensive understanding of the chemical composition, size distribution, and physical morphology of ZnO and MgO PTNPs, which were characterized by FT-IR (NICOLET, Wisconsin, USA), SEM (JEOL 6010LA), dynamic light scattering (DLS, Montgomeryville, PA, USA), and ζ potential (Microtrac, Montgomeryville PA, USA). USA) (Selvakesavan et al., 2023).
Plant cultivation (Stevia rebaudiana Bertoni) and elicitation with ZnO and MgO phytonanoparticles
Stevia rebaudiana (Var. Morita II) plants were grown in plastic seedbeds using a mixture of peat and agrolite (4:1 v/v). After the development of 10 true leaves, plants were transplanted into polyethylene bags (20 x 30 cm) containing the same inert substrate. The experiment was carried out in an anti-aphid house in Cintalapa de Figueroa, Chiapas, Mexico (16°45′N 93°51′W). The temperature in the anti-aphid house ranged between 29 - 32 °C and relative humidity below 70 %. Plant nutrition was provided with modified Hoagland nutrient solution without zinc and magnesium.
At 30 days after transplantation (15 and 20 true leaves), different experimental treatments with ZnO and MgO PTNPs were applied. Plants were sprayed individually from the roots twice a week (1 mL per plant) for four weeks. The treatments evaluated were: T1 (control without PTNPs), T2 (MeJa 22 mg/L), T3 (PTNPs ZnO 1 mg/L), T4 (20 mg/L ZnO PTNPs), T5 (30 mg/L ZnO PTNPs), T6 (1 mg/L MgO PTNPs), T7 (20 mg/L MgO PTNPs), and T8 (30 mg/L MgO PTNPs). At the end of the experiment, plants were harvested and dried at 60 °C for 48 h. They were ground and stored in the dark at 4 °C until use.
Stevia rebaudiana extract preparation
The method described by Gasmalla et al. (2017) was used for the extraction of the steviol glycosides.
Rebaudiosides A, D, E and M quantification
The quantification of SGs (rebaudioside A, D and M) present in the extract of S. rebaudiana Bertoni was carried out by HPLC UV-Visible (High-performance liquid chromatography with UV-Vis detector, Flexar PerkinElmer, USA), according to the methodology reported by Velázquez-Gamboa et al. (2021).
Gallic acid, chlorogenic acid, caffeic acid, and caffeine contents
The system used for the analysis of the various phenolic compounds consisted of a Perkin Elmer high-performance liquid chromatograph. A reversed-phase C18 column (150 x 4.4 mm) was used at a wavelength of 270 nm for gallic acid and caffeine, and 325 nm for chlorogenic acid and caffeic acid. The mobile phases used were 0.1 % formic acid (solvent A) and methanol (solvent B). Twenty μL of sample were injected, a solvent gradient elution program was used starting at a 15 % (A)/85 % (B), increasing to 80 % (A)/20 % (B) throughout 50 min, then decreasing to 15 % (A)/85 % (B) over 2 min, and finally maintaining the same ratios for 5 min. External standards of gallic acid, chlorogenic acid, caffeic acid, and caffeine HPLC grade (Sigma Aldrich, USA) were used, and the phenolic compounds were expressed in mg/g of dry leaf.
Results and discussion
ZnO and MgO phytonanoparticles characterization
The formation of PTNPs was evidenced by the color changes in the precursor solutions and the subsequently obtained colloidal solution. The characterization of ZnO and MgO PTNPs synthesized from Moringa oleifera was carried out by scanning electron microscopy (SEM), dynamic light scattering (DLS) and FT-IR spectroscopy.
FT-IR and SEM analysis
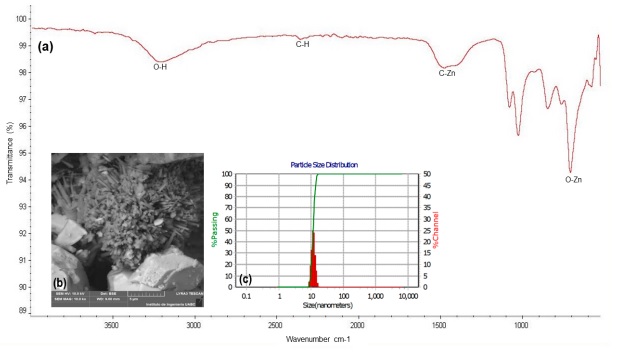
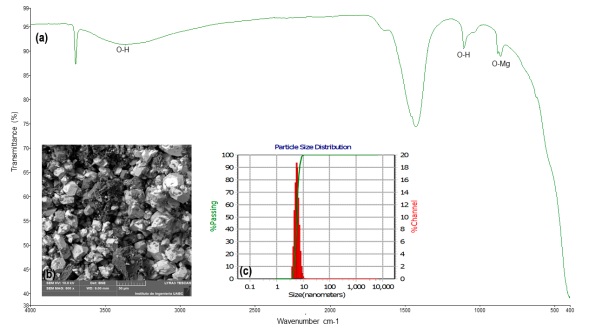
The FT-IR spectrum of the ZnO PTNPs is shown in Fig. 1a. Molecules such as alcohols, phenols, and carboxylic acids present in the plant extract can interact with the zinc surface and contribute to the stabilization of ZnO PTNPs (Shaikhaldein et al., 2021; Fakhari et al., 2019). Characteristic IR bands appeared at 3390 and 2825 cm-1 corresponding to the stretching vibrations of O-H and C-H bonds respectively (Senthilkumar et al., 2017). The band observed at 1500 cm-1 can be attributed to the asymmetric and symmetric stretching of carbonyls (C=O) (Moghaddam et al., 2017), while the band at 871 cm-1 could be related to the stretching vibrations of Zn-O bonds (Alamdari et al., 2020; Fakhari et al., 2019). On the other hand, the study of MgO PTNPs (Fig. 2a) shows the presence of a broad band of the hydroxyl group at 3381 cm-1 (Khajuria et al., 2021; Alamdari et al., 2020). Two distinct bands are observed in the wave number regions at 1429 and 1106 cm-1 which are attributed to the bending vibrations of the adsorbed water molecule and the surface hydroxyl group (-OH) (Boro et al., 2020). The band observed at 531 cm-1 indicates the formation of the Mg-O bond (Lucho et al., 2018).
SEM imaging of the ZnO PTNPs revealed agglomerated rod-like PTNPs with a diameter range of 3 - 8 nm (Fig. 1b). Velázquez-Gamboa et al. (2021) reported the synthesis of ZnO PTNPs from Moringa oleifera in the form of rod-shaped, elongated with an average size of 13 nm. The image corresponding to the MgO PTNPs shows that they are irregularly shaped, and highly agglomerated, with an average size of 45 nm (Fig. 2b). Similar results were reported by Amrulloh et al. (2021), who synthesized MgO PTNPs from M. oleifera with spherical shape and average size ranging from 20 to 80 nm. The study by Gupta et al. (2020) highlights the importance of various physicochemical factors in controlling the synthesis of green nanoparticles. In fact, the mentioned factors, including the optimal concentration of metal ions, pH, temperature, concentration of precursors and reducing agents, and reaction time, play fundamental roles in the synthesis of PTNPs, and significantly affect their physicochemical properties and functionality. However, to date, the synthesis and application of specific forms of PTNPs have not been widely reported or established in the scientific literature. The synthesis and application of nanoparticles, especially in the context of green or environmentally friendly methods, are active areas of research and new developments occur periodically.
DLS and ζ potential analysis
The DLS technique is widely used to determine the hydrodynamic diameter of suspended particles. The average hydrodynamic size of the ZnO PTNPs (Fig. 1c) synthesized in this study was in the range of 9 nm to 30 nm in a stabilized medium. The ζ potential defines the colloidal stability, it is a typical measure of the surface charge of a particle (Mehta et al., 2021). In this study, the ζ potential of ZnO PTNPs in distilled water was +200 mV, therefore, they can be considered as strongly cationic which favors their dispersion in solution. When the particle is partially dispersed in the medium, the ζ potential tends to be positive (Campbell et al., 2019). The exact ζ potential required for stability depends on the specific colloidal system, including the nature of the particles, their size, surface charge, and the surrounding medium. In addition, factors such as temperature and ionic strength can also affect the ζ potential and thus the stability of colloids.
The histogram in Fig. 2c, shows the size distribution of MgO PTNPs, which were observed in the range of 5 - 10 nm with a mean size of 7.5 nm. Moorthy et al. (2015) reported the synthesis of MgO PTNPs from Neem leaf with a size of 43 nm, while Ali et al. (2020) reported the size MgO PTNPs from Persimmon extract with a size of 23.88 nm. Regarding the surface charge of the biosynthesized MgO PTNPs MgO a ζ potential of +39.5 mV was determined. These results were similar to those reported by Suresh et al. (2014), who reported a ζ potential of +39 mV in MgO PTNPs from Calotropis gigantean, indicating the formation of stable nanoparticles in solutions at slightly acidic pH.
Rebaudioside A, D, E, and M content in elicited Stevia rebaudiana Bertoni plants
Recently, some researchers have reported that PTNPs influence the production of secondary metabolites in plants with medicinal properties (Selvakesavan et al., 2023). Rebaudioside A is the most abundant compound in Stevia rebaudiana extract; therefore, the commercial value of this product is mainly defined by its content. However, there are other rebaudiosides with similar sweetening power to Reb A, such as Reb D and Reb M, which, although found in low concentrations, could have a significant impact on the sensory properties of S. rebaudiana. Regarding the content of rebaudioside A, in Table 1 shows that there is a significant statistical difference (p < 0.05) between treatments, where the plants that were elicited with MeJa showed an increase of 18 % with respect to the control plants. These results were consistent with those described by Lucho et al. (2018), who reported that the application of this phytohormone induced an increase in rebaudioside A, after 24 and 72 hours of application.
Cuadro 1 Contenido de rebaudiósidos A, D y M en extracto acuoso de hojas secas de Stevia rebaudiana Bertoni estimulados con fitonanopartículas de óxido de zinc y magnesio.
Table 1. Rebaudiosides A, D and M content in aqueous extract of Stevia rebaudiana Bertoni dried leaves elicited with zinc and magnesium oxide phytonanoparticles.
| Treatments | PTNPs mg/ L | Rebaudioside A | Rebaudioside D | Rebaudioside M | |
| mg/ g DW | |||||
| T1 | Deionized water | 0 | 43.38 c | 15.64 a | 2.55 b |
| T2 | Metil jasmonate | 0 | 51.19 a | 15.82 a | 2.44 c |
| T3 | ZnO | 1 | 45.22 b | 13.12 b | 2.63 a |
| T4 | 20 | 38.8 de | 12.05 cd | 2.54 b | |
| T5 | 30 | 37.67 e | 11.71 d | 2.51 bc | |
| T6 | MgO | 1 | 45.86 b | 12.77 bc | 2.5 bc |
| T7 | 20 | 42.3 c | 12.32 cd | 2.52 bc | |
| T8 | 30 | 40.49 e | 12.4 bcd | 2.55 b | |
| LSD (0.05) | 1.73 | 0.72 | 0.085 | ||
LSD: Least significant difference. Values with the same letter in each column have no statistically significant difference between treatments. T2 (22.43 mg/L Metil jasmonate)
Regarding the treatments with added PTNPs, it was observed that at low concentrations (T3 and T6) there was a significant increase between 4 and 5 %, respectively, compared to the control, while at higher concentrations their content decreased. Javed et al. (2017a) reported similar results in the content of steviol glycosides using 1 mg/L ZnO PTNPs in the culture of micropropagated S. rebaudiana shoots. These results suggest that PTNPs of both metals at concentrations of 1 mg/L could generate a signaling cascade that probably induces a higher expression or activity of the enzyme UGT76G1. On the other hand, the increase in PTNPs concentrations could induce greater stress in plants by modifying the metabolism, towards the synthesis of molecules with antioxidant capacity to regulate cell damage (Table 2), thereby inhibiting the synthesis of steviol glycosides.
Table 2 Content of gallic acid, chlorogenic acid, caffeic acid and caffeine, in aqueous extract of Stevia rebaudiana Bertoni dried leaves elicited with zinc and magnesium oxide phytonanoparticles.
Cuadro 2. Contenido de ácido gálico, ácido clorogénico, ácido cafeico y cafeína en extracto acuoso de hojas secas de Stevia rebaudiana Bertoni estimulados con fitonanopartículas de óxido de zinc y magnesio.
| Treatments | PTNPs mg/ L | Gallig acid | Chlorogenic acid | Caffeic acid | Caffeine | |
| mg/ kg DW | ||||||
| T1 | Deionized water | 0 | 63.1 a | 749.2 de | 20.1cd | 7.8 b |
| T2 | Metil jasmonate | 0 | 62.3 ab | 818.0 d | 23 cd | 6.0 c |
| T3 | ZnO | 1 | 55.3 c | 746.7 e | 27.3 c | 7.4 bc |
| T4 | 20 | 55.2 c | 1777.0 a | 26.4 c | 10.4 a | |
| T5 | 30 | 58.7 bc | 1425.2 b | 41.6 b | 7.4 bc | |
| T6 | MgO | 1 | 62.2 ab | 142.0 g | 17.7 d | 5.9 c |
| T7 | 20 | 64.5 a | 451.9 f | 26.8 c | 4.2 d | |
| T8 | 30 | 59.2 b | 1011.9 c | 57.4 a | 6.5 bc | |
| LSD (0.05) | 4.82 | 69.3 | 1.54 | 1.54 | ||
LSD: Least significant difference. Values with the same letter in each column have no statistically significant difference between treatments. T2 (22.43 mg/L Metil jasmonate)
The results for Reb D and Reb E content were reported as Reb D equivalents, because both molecules are isomers and have the same retention time in the chromatographic analysis used. Table 1 shows that there is no significant statistical difference (p < 0.05) between the controls, although the same enzyme catalyzes the glycosylation for the synthesis of these molecules using Reb A as precursor. On the other hand, the application of ZnO and MgO PTNPs, at all the concentrations studied, significantly reduced their content up to 26 %.
As for Reb M, the plants treated with ZnO at 1 mg/mL increased its content by 3 % with respect to the control, while the other treatments did not show significant statistical differences with respect to the control plants.
Several authors have reported that the use of elicitors and phytohormones such as salicylic acid, paclobutrazol, MeJa, some auxins such as gibberellins and nanoparticles (Ag, CuO, ZnO-PEG, ZnO-PVP, CuO-PEG and CuO-PVP) (Gupta et al., 2015; Karimi et al., 2015; Javed et al., 2017a; Javed et al., 2017b; Saraiva et al., 2020; Wang et al., 2021) of chemical, biological and commercial synthesis, affect the synthesis of secondary metabolites, mainly in the production of SGs and compounds with antioxidant capacity. Therefore, ZnO and MgO PTNPs may exhibit similar catalytic properties in certain chemical reactions and could play a similar role in the synthesis of steviol glycosides, depending on the specific reaction conditions and mechanisms involved. However, it is important to note that the catalytic behavior of nanoparticles can be highly dependent on several factors, including size, morphology, surface properties, and specific reaction conditions. Lucho et al. (2018) reported that MeJa does not negatively affect the expression of genes involved in SGs synthesis. They demonstrated the positive influence of MeJa on the activation of the genes responsible for encoding enzymes in the SGs biosynthesis pathway, reporting that about 12 (80 %) genes catalogued by this elicitor as a strong candidate for use in SGs production optimization. A statistically significant difference was observed in Reb M content, and with these results, it could be assumed that PTNPs act similarly in gene activation; Therefore, PTNPs could induce a higher gene expression of enzymes responsible for stevioside glycosylation, these enzymes are known as UDP-glycosyltransferases.
Gallic acid, chlorogenic acid, caffeic acid, caffeine content
The production of non-enzymatic antioxidants (phenolic compounds) is the protective mechanism against reactive oxygen species (ROS) in plants and their accumulation varies in different environments. Several studies have reported that the use of metal PTNPs induces the synthesis of these metabolites (Gowri et al., 2019; Moharramnejad et al., 2019; Velázquez-Gamboa et al., 2021). Several researchers have reported that metal PTNPs influence the synthesis of secondary metabolites in plants. Stevia rebaudiana has an added value because it is a plant that has a high content of phenolic compounds. However, there are some of them with greater antioxidant capacity, such as flavonoids, which, although found in lower concentrations, have a significant impact on the commercial value of the plant.
Near the flavonoid content, those of gallic acid, chlorogenic acid, caffeic acid and caffeine present in the leaves of S. rebaudiana nanoelicited are shown in Table 2. It was observed that the control with MeJa did not modify the content of gallic acid, chlorogenic acid, or caffeic acid, only caffeine decreased significantly. However, the elicitation with PTNPs mainly with ZnO at 20 mg/L induced the production of molecules with antioxidant capacity. Chlorogenic acid increased up to 137 %, while caffeic acid increased by 185 %. Likewise, in the results of the caffeine studies, a significant statistical difference was observed between treatment T4 (20 mg/L ZnO PTNPs) and T1 (negative control) showing a 133 % increase. In this case, the results obtained indicate that the application of ZnO and MgO PTNPs induces an increase in antioxidant compounds content. In general, Chandra et al. (2017) suggests that PTNPs can stimulate a strong immune response in Camellia chinensis plants, leading to the upregulation of defense-related genes, increased activity of defense enzymes, and accumulation of phenolic compounds, all of which play a crucial role in the plant’s ability to defend itself against potential threats such as pathogens.
The results of the study indicate that an increase in PTNPs concentration leads to an increase in the production of reactive oxygen species (ROS), but does not reach toxic levels for the plant. Instead, the plant responds by increasing the production of antioxidant compounds. This response is consistent with the concept of homeostasis in plants. Hussain et al. (2021) indicate that oxidative stress was ameliorated in Persicaria hydropiper L. seedlings exposed to ZnO NPs through increased production of free proline, phenolics, flavonoids, and the activation of antioxidant enzymes.
Conclusions
The elicitation of S. rebaudiana with ZnO and MgO PTNPs induces the biosynthesis of rebaudioside A and M, which could improve the sweetening potential of its extracts, in addition to a significant increase in the content of chlorogenic acid and caffeic acid, molecules that contribute to the antioxidant activity. Consequently, the application of ZnO and MgO PTNPs to S. rebaudiana plants probably gives added commercial value to the crop. However, it is necessary to continue the studies to evaluate the phytotoxic effect of the extract obtained from plants treated with these PTNPs.
Funding
The authors thank CONAHCyT Mexico for providing financial support to Velázquez-Gamboa with Scholarship: 849710 to carry out this research.
Conflicts of interest
Consent for Publication: All authors agreed to publish this paper in this journal.
Ethical approval: This study does not contain any studies with human participants or animals performed.
Conflict of interest: The authors declare no conflict of interest.
Acknowledgments
The authors thank the Instituto Técnologico de Tuxtla Gutierrez for providing the facilities











 nueva página del texto (beta)
nueva página del texto (beta)