Introduction
The December holiday season is the festive period with the greatest impact on adult body weight (Zorbas et al., 2020; Díaz-Zavala et al., 2017). Observational studies have shown that more than 50 % of the annual weight is gained during this period (Yanovski et al., 2000; Sturm et al., 2016). Globally, few preventive interventions have been conducted during the December holidays and have been implemented in US and UK (Kaviani et al., 2019; Hirsh et al., 2019; Mason et al., 2018; Watras et al., 2006; Zorbas et al., 2020). Therefore, interventions are needed to prevent weight gain in this critical period, and outside these populations. This work sought to evaluate the effect of two online interventions on the prevention of body weight gain, compared to a control group in Mexican adults during the holiday season, in order to test the feasibility of the methodology and obtain data to calculate the sample size of a definitive study. The study coincided with the COVID-19 pandemic, which appears to have negatively affected diet, physical activity, and body weight of the population (Bennett et al., 2021).
Materials and methods
This exploratory pilot randomized controlled trial of parallel groups, with three arms, was approved by the Research Ethics Committee of the Department of Nursing at the University of Sonora (registration number: CEI-ENFERMERÍAEPD-002-2020). This report follows the recommendations of the Consolidated Standards of Reporting Trials guide, in its extension for pilot studies (Eldridge et al., 2016) and was registered in ClinicalTrials.gov platform (NCT05060978) prior to conduct of the trial.
Participants
Participants were adults with normal weight, overweight or obesity, recruited online (through flyers, posters, direct invitation, and social networks) at the city of Hermosillo, Sonora, México. Eligibility criteria were evaluated and their consent was obtained. Participants were eligible for inclusion if they were adults with a BMI classified as normal weight, overweight or obesity (BMI ≥ 23 kg/m2 and ≤ 40 kg/m2), aged between 20 and 65 years, and residents of the city of Hermosillo, Sonora. They also had to commit to not undertake any other intervention outside the one assigned in the study during the 8 weeks of the intervention. We excluded people with a previous diagnosis of a medical condition that constituted a contraindication for the intervention or that could affect body weight.
Randomization
Participants were randomized by blocks, stratified by sex and age, with a 1:1:1 allocation ratio, to the three different intervention groups: 1) Watch your Weight During the Holidays Program (WWDH), 2) Relative fasting 5:2 (RF5:2), or 3) control group. Assignment to groups was performed by a member of the research group not involved in the recruitment of participants, nor intervention implementation.
Interventions
The WWDH group comprised self-efficacy and habit formation through encouragement of participants to record their weight at least one time during each week (self-weighing), follow 10 tips for weight management during the holidays, graphical information about physical activity calorie equivalents (PACE) of Mexican foods and drinks, and three video calls for nutrition counseling (one group and two individual video calls). The RF5:2 group received a dietary recommendation to follow a 5:2 intermittent fasting approach, along with two video calls for nutrition counseling related to intermittent fasting (one group and one individual video call). They were given low-energy menus of 550 kcal/day and 660 kcal/day for women and men, respectively, to follow during two fasting days. During the remaining five days, there was no energy restriction. The control group received an explanation of a healthy eating leaflet, based on a Mexican Standard for health promotion: NOM-043-SSA-2012. To encourage retention, study participants obtained a weighing scale as a reward for completing the ten-week measurements. Detailed information about each intervention group can be seen in Supplement 1.
Outcomes
The primary outcome was change in body weight at 8-weeks. Measurements were originally planned to be at eight-weeks but high reports of COVID-19 infections by study participants during the holiday season resulted in a delay of two weeks (10-weeks). Secondary outcomes of body size and composition, biochemical variables and health-related quality of life were also measured at 10-weeks. Baseline measurements were assessed from November 1 to 12, 2021, and final measurements from January 24 to February 2, 2022. The outcome assessors were blinded to treatment allocation status (TJMC and ALMP). Techniques and details about how measures were taken for each outcome can be consulted in Supplement 1.
Statistical analysis
Statistical analysis was performed with one-way ANOVA tests for parametric samples or Kruskal-Wallis tests for nonparametric samples, using statistical software NCSS 10 (Number Cruncher Statistical System for Windows, Kaysville, UT, USA). All analyses were performed using the intention-to-treat approach and, secondarily, with those who completed all measurements.
Results
Thirty-two participants were recruited and randomized to the three different intervention groups: WWDH Program (n = 10), RF5:2 (n = 11) and control group (n = 11). Figure 1 shows the flow diagram of study participants. At the end of the study measurements, a retention of 78.1 % of the participants was achieved.
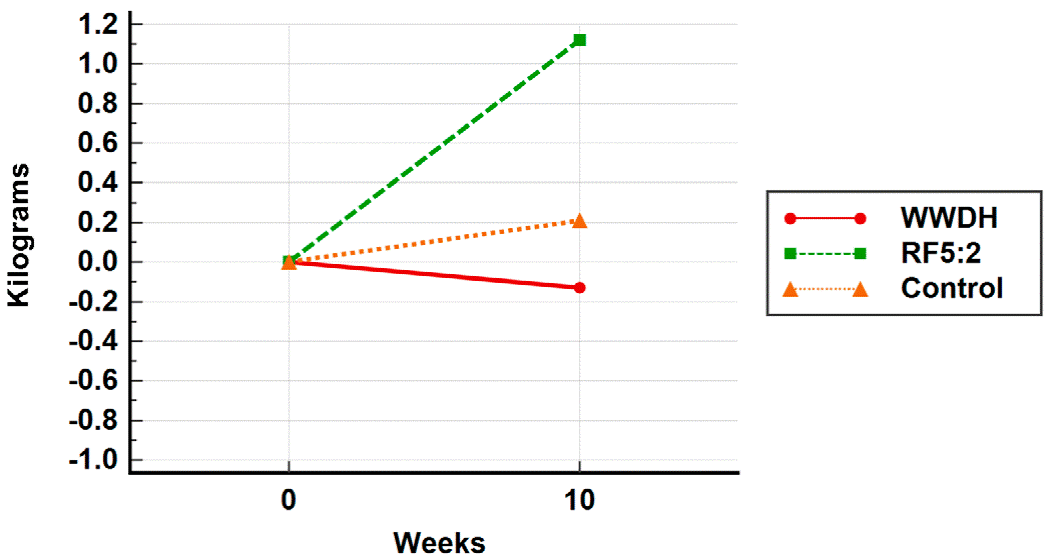
Participants were predominantly female (65.6 %), with a mean age ± standard deviation of 38.1 ± 11.3 years, and BMI of 30.4 ± 3.84 kg/m2. There were no significant differences between groups at baseline in any of the characteristics as was expected due to the randomization process (Table 1). Table 2 shows the results of the variables analyzed by the ITT approach. Results for weight change at 10 weeks showed no difference between groups (WWDH: -0.10 ± 1.81, 95 % CI -1.40 to 1.19; RF5:2: 0.92 ± 1.18, 95 % CI 0.12 to 1.71 and control group: 0.15 ± 1.68, 95 % CI from -0.98 to 1.28) (Figure 2), though a positive trend was observed for the WWDH group and a negative trend for the RF5:2 group, both compared to the control group. For secondary outcomes, no differences were observed between the groups.
Tabla 1 Características basales de los participantes.
| Outcome | WWDH group (n= 10) | RF5:2 group (n= 11) | Control group (n= 11) | P Value |
|---|---|---|---|---|
| Sex n (Femenine %) | 7 (70.0) | 7 (63.6) | 7 (63.6) | 0.94 |
| Age (years) | 37.5 (11.5) | 39.7 (12.5) | 38.1 (11.3) | 0.84 |
| Weight (kg)a | 77.1 (68.8, 97.4) | 77.1 (69.4, 109) | 87.0 (79.1, 99.5) | 0.58 |
| BMI (kg/m2) | 29.5 (3.60) | 30.8 (4.98) | 30.7 (2.87) | 0.69 |
| Waist circumference (cm) | 97.5 (11.8) | 100 (15.8) | 101 (9.37) | 0.71 |
| Fat mass (kg) | 31.3 (7.68) | 35.0 (9.49) | 35.1 (7.02) | 0.49 |
| Fat free mass (kg)a | 43.3 (40.8, 67.5) | 42.1 (39.9, 54.1) | 49.2 (44.5, 64.7) | 0.68 |
| Systolic blood pressure (mm/Hg) | 124 (12.3) | 118 (11.9) | 120 (14.5) | 0.62 |
| Diastolic blood pressure (mm/Hg) | 82.1 (10.1) | 78.2 (9.74) | 79.1 (11.1) | 0.68 |
| Biochemical outcomes | ||||
| Fasting glucose (mg/dL) | 89.6 (10.1) | 81.3 (10.2) | 84.4 (12.1) | 0.21 |
| Total cholesterol (mg/dL) | 186 (40.7) | 213 (50.6) | 186 (44.8) | 0.30 |
| LDL cholesterol (mg/dL) | 117 (32.0) | 134 (36.9) | 114 (38.8) | 0.39 |
| HDL cholesterol (mg/dL) | 42.9 (6.69) | 47.6 (9.61) | 43.0 (7.18) | 0.32 |
| Triglycerides (mg/dL) | 129 (64.5) | 157 (134) | 145 (66.6) | 0.84 |
| Physical functiona | ||||
| Health-related quality of life outa | 95.0 (85.0, 100) | 95.0 (83.7, 100) | 89.1 (85.0, 100) | 0.92 |
| Physical problemsa | 100 (68.7, 100) | 100 (93.7, 100) | 100 (50.0, 100) | 0.47 |
| Pain | 80.2 (15.5) | 89.5 (10.3) | 80.2 (16.8) | 0.27 |
| Social function | 83.7 (15.6) | 85.0 (17.4) | 79.5 (18.7) | 0.75 |
| Mental health | 72.0 (14.3) | 77.6 (17.6) | 70.1 (13.8) | 0.52 |
| Emotional problems | 56.6 (38.6) | 70.0 (39.9) | 60.6 (29.1) | 0.69 |
| Vitality | 62.5 (16.8) | 62.0 (23.7) | 64.0 (16.4) | 0.96 |
| Perception of general health | 69.5 (15.7) | 77.5 (14.9) | 65.4 (20.3) | 0.28 |
| Health changes over time | 65.0 (24.1) | 62.5 (21.2) | 61.3 (30.3) | 0.94 |
Baseline characteristics of participants, expressed as mean (percentage) or mean (standard deviation), one-way ANOVA test; and amedian (percentiles 25 and 75), Kruskal-Wallis test.
Abbreviations: WWDH: Watch your Weight During the Holidays, RF5:2: Relative Fasting 5:2.
Tabla 2 Cambios en las variables desde el inicio hasta las 10 semanas de seguimiento.
| Outcome | WWDH group (N= 10) | RF5:2 group (N= 11) | Control group (N= 11) | P value |
|---|---|---|---|---|
| Weight (kg) | -0.10 (-1.40, 1.19) | 0.92 (0.12, 1.71) | 0.15 (-0.98, 1.28) | 0.30 |
| BMI (kg/m2) | -0.04 (-0.51, 0.42) | 0.34 (0.05, 0.63) | 0.06 (-0.32, 0.45) | 0.27 |
| Waist circumference (cm) | -0.36 (-1.71, 0.99) | 0.14 (-0.92, 1.21) | -0.97 (-2.43, 0.49) | 0.39 |
| Fat mass (kg)a | 0.12 (-0.31, 1.80) | 0.56 (-0.02, 1.67) | 0.12 (0.00, 0.45) | 0.57 |
| Fat free mass (kg)a | -0.63 (-1.71, 0.14) | 0.05 (-0.33, 0.66) | 0.16 (0.00, 0.81) | 0.57 |
| Systolic blood pressure (mm/Hg) | 1.00 (-6.53, 8.53) | 2.00 (-2.29, 6.29) | 4.54 (1.41, 7.67) | 0.57 |
| Diastolic blood pressure (mm/Hg) | -3.40 (-8.87, 2.07) | -0.45 (-5.59, 4.68) | -1.18 (-5.75, 3.39) | 0.57 |
| Biochemical outcomes | ||||
| Biochemical outcomes Fasting glucose (mg/dL)a | 0.00 (-4.00, 2.00) | 0.00 (0.00, 12.0) | 0.00 (0.00, 10.0) | 0.16 |
| Total Cholesterol (mg/dL)a | 28.6 (5.41, 51.7) | 16.2 (-21.6, 54.1) | 8.45 (-12.4, 29.3) | 0.13 |
| LDL cholesterol (mg/dL)a | 20.5 (0.00, 35.7) | 0.00 (-1.00, 14.0) | 0.00 (-4.00, 27.0) | 0.11 |
| HDL cholesterol (mg/dL)a | 0.00 (0.00, 8.75) | 0.00 (0.00, 13.0) | 0.00 (0.00, 4.00) | 0.92 |
| Triglycerides (mg/dL)a | 0.00 (-29.0, 33.5) | 0.00 (-8.00, 8.00) | 0.00 (-45.0, 12.0) | 0.77 |
| Health-related quality of life outcomes | ||||
| Physical functiona | 0.00 (-1.25, 6.25) | 0.00 (0.00, 5.00) | 0.00 (0.00, 0.00) | 0.49 |
| Physical problemsa | 0.00 (-6.25, 6.25) | 0.00 (0.00, 0.00) | 0.00 (0.00, 0.00) | 0.93 |
| Pain | 5.44 (-10.0, 14.5) | -5.90 (-20.7, 8.94) | 5.00 (-1.14, 11.1) | 0.31 |
| Social functiona | 0.00 (-18.7, 15.6) | 0.00 (0.00, 0.00) | 0.00 (0.00, 12.5) | 0.56 |
| Mental health | -2.40 (-11.3, 6.56) | -2.18 (-8.46, 4.10) | 4.36 (-0.93, 9.66) | 0.22 |
| Emotional problems | -30.0 (-53.7, -6.28) | 6.05 (-21.9, 34.0) | 6.07 (-10.7, 22.8) | 0.03 b |
| Vitality | -1.00 (-10.6, 8.65) | 10.0 (-0.62, 20.6) | -1.36 (-6.58, 3.85) | 0.07 |
| Perception of general healtha | -2.50 (-10.0, 6.25) | 0.00 (-5.00, 10.0) | 0.00 (-5.00, 0.00) | 0.65 |
| Health changes over time | -7.50 (-26.4, 11.4) | 4.54 (-15.1, 24.1) | -9.09 (-20.4, 2.23) | 0.38 |
Changes in outcomes from baseline to 10-weeks of follow-up, expressed as mean (percentage) or mean (standard deviation), one-way ANOVA test; and amedian (percentiles 25 and 75), Kruskal-Wallis test. b Statistically significant differences with one-way ANOVA test, without differences with Bonferroni test. Abbreviations: WWDH: Watch your Weight During the Holidays, RF5:2: Relative Fasting 5:2.
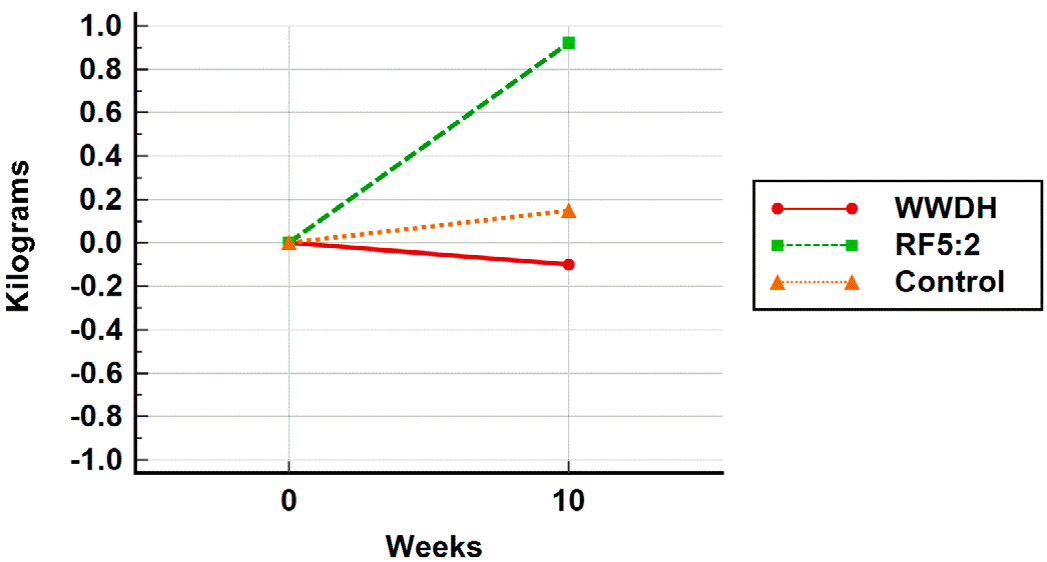
Analysis by those who completed all study measurements also showed no difference between groups in body weight change at 10 weeks or in any of the secondary outcomes evaluated (Figure 3). During the 10-week study period, no adverse effects were reported by study participants.
Discussion
This pilot study showed non-significant positive effects on weight gain prevention in favor of the WWDH group, compared to the RF5:2 and the control groups. The positive result of the WWDH group was a bit less than expected, compared to other studies on the prevention of weight gain during the holiday season. A systematic scoping review showed that intervention studies during this holiday period have achieved significant weight loss, mostly in favor of the intervention group (Zorbas et al., 2020). The review showed weight losses from 0.13 kg to 1.30 kg in the intervention groups and weight gains from 0.09 to 1.10 kg in the control groups (Zorbas et al., 2020). The effect of the RF5:2 group was opposite to what we expected and showed a similar behavior to the control groups of previous studies. We are not sure if this is due to this type of intervention not being culturally acceptable in Mexican participants, as it has not been tested previously outside of high-income countries (Harris et al., 2018; Cioff i et al., 2018), or whether it was due to the difficulty of maintaining normal eating habits on the five non-fasting days during the holiday season.
The data from this study will allow the implementation of this methodology in a definitive randomized controlled trial with a larger sample size. Given the results obtained by the RF5:2 intervention during this period, we have removed this intervention from the definitive study (NCT05580926). We also intend to use face-to-face rather than online sessions for the WWDH intervention, and remove the last individual session in the intervention group. Finally, considering the data from this pilot study (mean differences and SDs from WWDH and RF5:2, assuming that the RF5:2 group acted as a control group) and taking into account other published studies (Mason et al., 2018; Kaviani et al., 2019), it was possible to calculate a sample size of 64 participants for a definitive study (32 in each arm).
Among the strengths of our pilot study is that, to our knowledge, is the first randomized controlled trial seeking to prevent December holiday body weight gain remotely and during the COVID-19 pandemic. It is also the first in this period in a population outside of the United States of America and the United Kingdom. Among the limitations of our study was the presence of the uncertain pandemic outlook, which we believe strongly affected the retention of study participants which may have negatively affected the ability of the participants to adhere to the interventions, particularly the RF5:2 intervention. Likewise, the small sample size typical of a pilot or exploratory study could be taken as a limitation, but it fulfilled its purpose in proving that the intervention was feasible (while suggesting areas for improvement) and allowed the calculation of a sample size for a definitive study.
In conclusion, the results of this study showed that a trial aimed at preventing weight gain during the December holiday period is feasible in Mexican adults, having retained 78.1 % of the participants. It also showed that, while the WWDH intervention was promising, the RF5:2 intervention was not promising in Mexican adults during this period.











 nueva página del texto (beta)
nueva página del texto (beta)