Presented in part as abstracts at the “35th Annual Meeting of the European Society for Pediatric Infectious Diseases (ESPID 2017),” Madrid, Spain, May 23-27, 2017 and “European Academy of Pediatrics Congress and Mastercourse (EAP 2023),” Pavda, Italy, 18-21 May, 2023
Introduction
Human metapneumovirus (hMPV) is a leading cause of respiratory tract infection in the pediatric population, causing acute lower respiratory tract infections (LRTI) that frequently require hospital admission1-3. hMPV has been identified as responsible for 5-25% of pediatric hospitalizations for bronchiolitis and pneumonia, being more prevalent in children under 5 years of age, especially in patients from 6 to 11 months of age4-6. Reports of estimated cases in 2018 were of 14.2 million hMPV-associated acute LRTI cases, from which 643,000 needed hospitalization. Around 58% of the hospital admissions were in infants under 12 months, and 64% of in-hospital deaths resulted from infants > 6 months7. Clinical presentation is similar to other viral infections like respiratory syncytial virus (RSV) in children and includes upper and lower respiratory symptoms5,8.
Since its first identification in 2001 in the Netherlands, several studies have provided important information associated with hMPV infection worldwide9,10. Nevertheless, there are only a few studies in Costa Rican children about hMPV among the few from Central America11-13. The first pediatric reports in Latin American children came from South America (Brazil and Argentina), and then, other reports in this region have been published14-16. The aim of this study was to describe in a prospective cohort the epidemiological characteristics of patients with LRTI due to hMPV admitted to the only pediatric hospital of Costa Rica.
Methods
This was a prospective, cross-sectional study of patients admitted at the Hospital Nacional de Niños Dr. Carlos Sáenz Herrera, Centro de Ciencias Médicas in San José, Costa Rica. Our nation has 5,044,197 million (2023) inhabitants and is ranked with a per capita GDP of US$ 12,472 for 2021. Our hospital is the only tertiary referral and teaching pediatric center in the country, with ~320 beds, receives 95 000 annual emergency department admissions, and is the only hospital in the country with a pediatric intensive care unit (PICU).
Inclusion criteria were as follows: all children, male or female, of any ethnic group, aged 1 month to < 13 years presenting to our center with a discharge diagnosis of acute LRTI (predominantly bronchiolitis and pneumonia) requiring hospitalization from September 1, 2015, to September 30, 2017. Identification of acute LRTI cases was based on discharge diagnosis and daily results reported by the Immunology Division Laboratory and Epidemiological Surveillance Division to the principal investigator of the study (M.T.V-A) and available to the physicians in charge of hMPV patients, through the electronic laboratory results platform. Respiratory samples included nasopharynx, tracheal aspirates, and bronchoalveolar lavages. The detection of hMPV was made with direct immunofluorescence assay (Light Diagnostics, Chemicon), as no molecular testing for hMPV was available at that time. We excluded patients who had incomplete data on their clinical charts and nosocomial infections by hMPV.
A standard data collection form was used to obtain information from clinical charts. The data were extracted with a structured data collection tool. Data were abstracted from electronic ED records, admission, and progress notes as well as laboratory. Data collected included demographics, age, past medical history, risk factors, presenting symptoms and signs, imaging, investigations, interventions, and complications.
A statistical analysis was performed using Stata 16 (StataCorp, College Station, TX). Presenting symptoms and signs were analyzed descriptively. Analyses were performed on the total number of episodes and presented as percentages with corresponding 95% confidence intervals (CI). Continuous variables were presented as mean and standard deviation (SD) (normal distribution) or median and interquartile range (IQR) (non-normal distribution). We performed a multivariable logistic regression for categorical variables and a linear regression for continuous variables to identify the ones that were independently associated with age < 1 year old and the use of respiratory ventilatory support. Variables were chosen based on significance in the univariate analysis (p < 0.2), and on clinical discretion based on known predictors in the literature. We reported both odds ratios with their corresponding 95% CI and p values accordingly depending on the type of variable. The study was approved by the Institutional Review Board of our hospital (CLOBI-HNN-032-2015) and authorization to publish this manuscript was obtained from (Centro de Desarrollo Estratégico e Información en Salud y Seguridad Social, [CENDEISSS] San José, Costa Rica: CENDEISSS-AB-0161-2023). Due to the study nature, no informed consent was required to collect the information. All bioethical and safety information aspects were protected.
Results
During the study period, 188 children were included with a positive confirmatory laboratory test for hMPV, of which 62% (116) were male. When assessing age distribution, children under 15 months of age represented the majority (65%) of patients admitted for hMPV infections. The median age was 12 months (IQR 6-23).
Children had a median hospital stay of 5 days (IQR 3-8). Patients were admitted for symptomatic hMPV all year round, and distributed in the two seasons of the country: summer and rainy season (mid November to mid April). However, cases were likely to peak during October, November and December (32%), creating a tendency of greater number of hospitalizations in the second semester (Fig. 1). White race was predominant with 93.6% of patients, 3.7% were black and 2.7% indigenous.
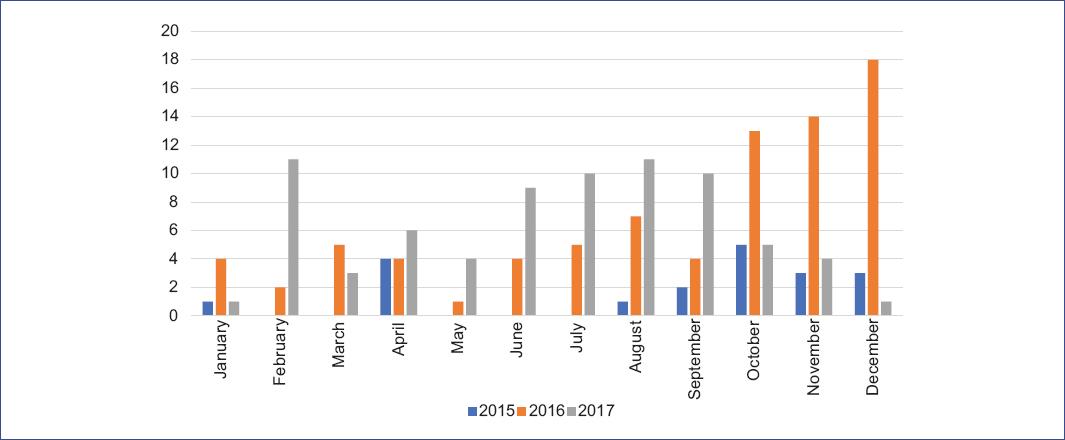
Figure 1 Seasonal distribution of number of cases of human metapneumovirus-associated hospitalizations, according to year and month of hospitalization.
Sixty-two (33%) of the patients were previously healthy. The most common underlying conditions were prematurity (21.8%; 4), recurrent wheeze-asthma (22,8%; 43), and low birth weight (17%; 32). Some risk factors for the severity of illness found were lack of breast milk in 67.0% (126) of the patients, followed by home overcrowding in 29.3% (55) and passive smoking in 15.4% (29). The most common documented symptoms were cough (86%;162), respiratory distress (83.5%;157), fever (62.2%;117), and rhinorrhea (60.6%;114). Physical examination findings included intercostal retractions 55.3% (104), wheezing 46.3% (87), and crackles in 44.1% (83) of the patients.
A total of 148 patients (78.7%) underwent a complete blood count testing on admission. Hemoglobin ranged from 6.6 g/dL to 18 g/dL with a median of 11g/dL (SD = 2.5). Regarding leukocyte count, the median value was 8,955/mm3 with a range of 520/mm3-25,660/mm3. Regarding CRP, it was determined in 68.6% (n: 129) of the patients, with an average of 39.5 mg/L, median of 15 mg/L, mode of 5 mg/L, minimum of 1 mg/L, and maximum of 326 mg/L.
Co-infection was detected in 10 (5%) patients, mainly by RSV (3), influenza A (2), parainfluenza 3 (2), adenovirus (1), and Mycoplasma pneumoniae (1). Ninety-four percent (177) of our patients had a chest radiograph performed on admission, in whom diffuse infiltrates were reported in 46.3% (82), followed by air trapping in 37.9% (67) and consolidations in 14.7% (26).
Oxygen therapy was the most frequent treatment in 93% (175) of patients, followed by bronchodilators in 59.5% (112), steroids in 42% (79), blood products in 13.2% (25), ipratropium bromide 12.7% (24), inotropic support 11% (21), and nebulized adrenaline in 4.7% (9). Regarding antibiotic treatment, 52.6% of hospitalized patients received one or more intravenous antibiotics during the hospitalization. The main reason for antibiotic administration was clinical worsening in 36% of cases, 25% due to abnormal chest X-rays findings, and 12% due to an abnormal physical examination on admission. Antibiotics were also used in those with positive blood cultures for one or more microorganisms (5) 2.6%, and with concomitant diagnosis of acute otitis media, neonatal sepsis, and urinary tract infection.
In relation to complications, 45 (23.9%) patients needed management with high-flow nasal cannula (HFNC) during hospitalization, most of these in average for 3.4 days (SD = 2.08). Fifty-four (28.7%) children required intensive care unit (ICU) admission. Twenty-eight (14.8%) of them were on assisted mechanical ventilation for a mean duration of 6.3 days (SD = 6.03). Among the documented complications, ventilatory failure occurred in 17% (32) of hospitalized patients, shock 4.2% (8), pleural effusion 1.6% (3), pneumothorax 1% (2), and 1 patient presented acute encephalopathy. No deaths occurred during the present study; however, ten patients were discharged requiring supplementary oxygen at home.
A univariate analysis was performed including variables of interest. As shown in tables 1 and 2, the outcome variables used in this analysis were age < 1 year old and use of mechanical ventilation or HFNC. In table 1, where age < 1 year old was the outcome variable, we found no statistical association between age and the variables of interest.
Table 1 Univariate analysis of patients with age < 1 year old with hMPV during the study period
| Variables of interest | Age < 1 year (n = 94) n (%) | Age > 1 year (n = 94) n (%) | OR (CI) | p |
|---|---|---|---|---|
| Male sex | 63 (67) | 53 (56) | 1.5 (0.8-2.9) | 0.13 |
| Length of hospital stay in days (mean, SD) | 7.7 (9.7) | 7.1 (5.9) | - | 0.60 |
| Known healthy | 28 (29.7) | 23 (24.4) | 1.3 (0.6-2.6) | 0.41 |
| Asthma/recurrent wheeze | 18 (19.1) | 25 (26.6) | 0.6 (0.3-1.3) | 0.22 |
| Prematurity | 24 (25.8) | 17 (18) | 1.5 (0.7-3.3) | 0.21 |
| Passive smoking | 15 (16) | 14 (14) | 1.0 (0.4-2.6) | 0.84 |
| Lack of breastfeeding | 61 (64) | 65 (69) | 1.2 (0.6-2.3) | 0.53 |
| Presence of fever | w58 (61) | 59 (62.7) | 0.9 (0.5-1.7) | 0.8 |
| Consolidations on chest X-ray | 7 (7.4) | 14 (14.8) | 0.4 (0.1-1.2) | 0.10 |
| Need of HFNC | 25 (26.5) | 20 (21.2) | 1.3 (0.6-2.7) | 0.39 |
| Need of mechanical ventilation | 11 (11.7) | 17 (18) | 0.6 (0.2-1.4) | 0.21 |
| ICU admission | 28 (29.7) | 26 (27.6) | 1.1 (0.5-2.1) | 0.74 |
| Days in mechanical ventilation (mean, SD) | 8.9 (8.3) | 4.7 (2.3) | - | 0.07 |
| Days with HFNC (mean, SD) | 3.4 (2.3) | 3.5 (1.7) | - | 0.81 |
HFNC: high-flow nasal cannula; hMPV: human metapneumovirus; CI: confidence interval; ICU: intensive care unit; IQR: interquartile range; OR: odds ratio; SD: standard deviation.
Table 2 Univariate analysis of patients with any respiratory support (mechanical ventilation and HFNC) with the diagnosis of infection by hMPV during the study period in the National Childrens Hospital
| Variables of interest | Any respiratory support device (MV or HCNF) (n = 59) n (%) | No respiratory support device (MV or HCNF) (n = 129) n (%) | OR (CI) | p |
|---|---|---|---|---|
| Male sex | 38 (64.4) | 78 (60.4) | 249. 7 (41-10079.1) | < 0.001 |
| Age in months (mean, SD) | 15.2 (15.8) | 25.1 (32.9) | - | 0.03 |
| Age < 1 year | 29 (49.1) | 65 (50.3) | 0.95 (0.5-1.8) | 0.87 |
| Length of hospital stay in days (mean, SD) | 10.7 (10) | 5.9 (6.4) | - | < 0.001 |
| Known healthy | 19 (32.2) | 32 (24.8) | 1.4 (0.6-2.9) | 0.28 |
| Asthma/recurrent wheeze | 16 (27.1) | 27 (21) | 1.4 (0.6-3.2) | 0.34 |
| Prematurity | 13 (22) | 28 (21.7) | 1.0 (0.4-2.2) | 0.95 |
| Passive smoking | 9 (15.2) | 20 (15.5) | 0.9 (0.3-2.4) | 0.96 |
| Breastfeeding | 26 (44) | 36 (27.9) | 2.0 (1.0-4.0) | 0.02 |
| Presence of fever | 39 (66.1) | 78 (60.4) | 1.2 (0.6-2.5) | 0.45 |
| Consolidations on chest X-ray | 13 (22.3) | 8 (6.2) | 4.2 (1.5-12.6) | 0.001 |
| ICU admission | 53 (89.8) | 1 (0.7) | 1130 (132-45759) | < 0.001 |
CI: confidence interval; HFNC: high-flow nasal cannula; hMPV: human metapneumovirus; ICU: intensive care unit; IQR: interquartile range; MV: mechanical ventilation; OR: odds ratio; SD: standard deviation.
In table 2, where the use of any ventilatory support was chosen as an outcome variable in the analysis, male sex, age < 1-year-old, a longer length of stay, and ICU admission were associated with this outcome. In the multivariate analysis, only ICU admission and a longer length of hospitalization was associated with patients that needed mechanical ventilation (MV) or HFNC (p = 0.05 and < 0.001, respectively).
Discussion
Since its discovery, findings suggest that hMPV infection in some children may be as severe as RSV infection and reports from low-middle income countries suggest that infants < 1 year of age have disproportionately high risks of severe infections across all income regions and all child mortality settings, similar to RSV and influenza virus7,17. In our study, we did not find this association between age and severity and surprisingly no deaths occurred.
Although hMPV seasonality was variable, cases tended to peak from August to December, possibly associated to the rainy season in our country. These findings seem to correlate with other studies conducted in tropical and subtropical areas where epidemic peaks tended to occur during periods of high rainfall. The seasonality of this agent suggests that climate features and meteorological conditions are important factors in hMPV prevalence worldwide8,12.
Studies have shown that hMPV is an important cause of acute respiratory infection in children, particularly in those under 1 year of age8,9. This is consistent with our study, where most of our symptomatic hMPV cases requiring hospitalization occurred in children under 15 months of age. We also found a male gender predominance, which correlates with previous international and local epidemiological studies11,18. Fortunately, most cases of acute LRTI due to hMPV are mild; nevertheless, there are well-studied risk factors that make patients vulnerable for severe hMPV illness, leading to hospitalization, PICU admission, respiratory support, and/or death9,19. In our study, we found that children under 12 months, history of prematurity, low birth weight, absence of breastfeeding, and asthma were more frequent in severe hMPV disease and had longer hospital stay. On the multivariate analysis, only longer hospital stay was shown to be associated with this outcome.
The primary treatment strategies recommended for acute bronchiolitis are supportive care as needed, including hydration, supplemental oxygen, and mechanical ventilation when needed. Little consensus exists about the best management strategies for bronchiolitis; therefore, care varies significantly across settings and countries20. In our study, most of our patients required oxygen therapy throughout hospitalization, either through nasocannula, high-flow cannula, or assisted mechanical ventilation; nevertheless, physicians commonly used bronchodilators and steroids4,21. During the rainy season in our country, when most hospitalizations due to ARTI to viral pathogens including hMPV occur, hospital beds plethora may prompt physicians to manage these patients with respiratory support in beds outside the PICU until there is a bed available. Therefore, in some of these cases, patients were extubated and did not require PICU transferal at the end.
One of the most important and concerning findings in our study was the high rate of antibiotics use. According to international guidelines, there is minimal evidence to support the use of empirical antibiotics for bronchiolitis. However, these are prescribed at rates of 34-99% in uncomplicated cases. Findings in our study were consistent, which given that an incidence of 52.6% of antibiotic therapy use was reported. Establishing protocols to initiate antibiotic coverage could reduce the unjustified use of antibiotic therapy in patients with uncomplicated bronchiolitis or viral pneumonia, minimizing prolonged hospitalizations, nosocomial infections, and costs.21 Due to the nature of the study and the initial objectives, we could not determine the specific reason and indication for antibiotics in these patients, nor compare those patients that were started on antibiotics on admission with those in whom these drugs were started during the course of their hospitalization.
In our study, 37.5% of our patients suffered complications during their hospitalization. Respiratory associated complications were found among the most frequent ones including ventilatory failure, atelectasis, shock, pleural effusion, and pneumothorax. Despite that, no deaths occurred during the present study, and only 5% of our patients were discharged requiring supplemental oxygen.
We acknowledge the limitations of our study, including the fact that patients were recruited from the only tertiary pediatric center, which could increase the number of patients with severe illnesses. Children with hMPV are also admitted in other Costa Rican hospitals with milder infections; therefore, we could not generalize the results of our findings to other patients from the country, nor determining the disease burden. Furthermore, the amount of patients in the sample size could be underestimated due to underreporting of viral infections, lack of screening to all patients with ARTI, and problems with samples aspiration and processing, among others. Finally, we need updated data as this situation may have changed in the past 8 years and we have now PCR and other sensitive diagnostic tests for hMPV and other viruses. Among the strengths of our study, this is the only prospective study of hMPV-associated hospitalizations in Costa Rican children and one of the few in Central America and the Caribbean. Second, although our study period was relatively small, analyzing two consecutive respiratory seasons of 24 months, allowed us to confirm previous observations that hMPV hospitalizations occur more commonly in the second semester of the year, when additionally RSV produces significant morbidity and high impact on health services care and hospitalizations. The role of hMPV and RSV co-infections in our population should be a future area of research and analysis.
Conclusions
hMPV is associated with significant morbidity but low mortality in hospitalized Costa Rican children with acute respiratory infection, particularly those under 1 year of age. We found no clinical characteristics that that could distinguish hMPV from other pathogen types at the time of presentation, as well as no association with age or risk factors to the severity of hMPV. However, several risk factors were associated with severe hMPV illness, leading to hospitalization, admission to ICU, respiratory support, and/or death. The most concerning finding from or study was the high rate of antibiotic use in these patients, either at admission or during hospitalization. Since then, this has been a priority among our antimicrobial stewardship program personnel.











 nueva página del texto (beta)
nueva página del texto (beta)


