Introduction
Type 1 diabetes mellitus (T1DM) is characterized by the destruction of pancreatic Beta cells, normally due to an autoimmune process resulting in a loss of endogenous insulin production1. Worldwide, at least 108,300 children < 15 years old were diagnosed with T1DM in 20212. In Chile, the incidence has almost tripled from 2006 to 2021 (8.0-23.1/100,000 inhabitants, respectively)3. The fluctuating levels of blood glucose related to T1DM increase the risk of ketoacidosis and hypoglycemic events, among other complications4. Moreover, the life expectancy of this population could be reduced by 15-20 years, while the risk of death could be increased 3-4 times compared to peers without T1DM4. Therefore, glycemic control becomes fundamental in the population with T1DM. In addition, the pediatric population with T1DM presents lower levels of muscle function5 and quality of life6 than their peers without T1DM. Importantly, physical exercise has evidenced a positive role in glycemic control (e.g., hypoglycemic events)7,8, muscle function9, and quality of life7,8 in the pediatric population with T1DM. Unfortunately, the pediatric population with T1DM presents lower levels of physical activity10. The above is associated principally with fear of hypoglycemic events during exercise11. An alternative to decrease the hypoglycemic events is continuous glucose monitoring (CGM)12. Thus, the combination of physical exercise and CGM could be relevant in T1DM control and reducing its negative consequences. Therefore, the aim of this pilot study was to describe the results of a pilot program conducted in a public health hospital facility to analyze the effect of concurrent exercise plus CGM on weekly hypoglycemic events, muscle function, aerobic capacity, and quality of life of the pediatric population with T1DM attended in a public health system.
Method
Participants
The inclusion criteria were a pediatric population between 7 and 14 years old diagnosed with T1DM and managed by the endocrinology specialist team of the HEGC. To be considered in the analysis, the participants had to attend at least 75% of the training sessions. The exclusion criteria were moderate-to-severe cognitive deficiency, traumatological condition that would prevent performing the exercise, and cardiorespiratory disease that contraindicated performing the exercise. The study followed the national and international ethics declarations and was approved by the scientific ethics committee of the Servicio de Salud Metropolitano Sur, Santiago, Chile (code: 04-19012022).
Study design
The participants underwent measurements over three consecutive days:
Day 1: pre-participative evaluation13, capacitation, installation of CGM, and anthropometric measurements (body weight, height, and body mass index [BMI]), Clark test (hypoglycemic events auto-recognition).
Day 2: glycated hemoglobin (HbA1c) after 8-12 h of fasting in the HEGC dependences.
Day 3: quality of life questionnaire, muscle function (standing broad jump, push-up, prone plank, squat, and chest press 10 maximum repetitions [10RM]), and aerobic capacity (20 m shuttle run test).
Training protocol
Before, during, and after the concurrent training protocol, we implemented a rigorous security protocol related to hypoglycemic or hyperglycemic events in the pediatric population with T1DM following the guidelines of Moser et al. 202014. The concurrent training protocol was developed and executed by a multidisciplinary team of experts in training15,16. The participants completed a concurrent training protocol throughout 12 weeks, 2 times/week. The endurance training protocol consisted of 25 min of endurance training at 50% of the aerobic maximum velocity (AMV), which was estimated following the proposal of García et al.17. The AMV increased by 5% every 2 weeks until it reached 75%. The strength training protocol consisted of different exercises for the upper extremities (push-ups and chest press), lower extremities (long jump and squats), and trunk (prone plank)15,16. The details of concurrent training protocol intensity and volume are presented in table 1.
Table 1 Concurrent training program
| Aerobic training planning | ||||||
|---|---|---|---|---|---|---|
| Weeks | 1-2 | 3-4 | 5-6 | 7-8 | 9-10 | 11-12 |
| Working time (min) | 25 | 25 | 25 | 25 | 25 | 25 |
| Maximal aerobic velocity (%) | 50 | 55 | 60 | 65 | 70 | 75 |
| Strength training planning | ||||||
| Weeks | 1-2 | 3-4 | 5-6 | 7-8 | 9-10 | 11-12 |
| Standing broad jump (set × repetitions) | 2 × 10 | 3 × 8 | 3 × 10 | 3 × 12 | ||
| Push-up (set × repetitions) | 2 × 10 | 3 × 8 | 3 × 10 | 3 × 12 | ||
| Prone plank (set × repetitions) [s]) | 2 × 30 | 2 × 45 | 2 × 60 | |||
| Squat (set × intensity [RM])) | 3 × 10 at 30% | 3 × 10 at 40% | 3 × 10 at 50% | 3 × 8 at 60% | 3 × 8 at 70% | |
| Chest press (set × intensity [RM]) | 3 × 10 at 30% | 3 × 10 at 40% | 3 × 10 at 50% | 3 × 8 at 60% | 3 × 8 at 70% | |
RM: repetition maximum.
Measurements
CGM
The Medtronic® CGM dispositive was used. The reports submitted 1 week before the start of the training protocol and throughout the 12-week period were analyzed. The weekly hypoglycemic (54-70 mg/dL [< 54 was considered a hypoglycemic event]), percentage of time in range (70-180 mg/dL), and mean weekly glycemic concentration (mg/dL) were used.
ANTHROPOMETRICS
Body weight and height were measured using a lever scale and stadiometer SECA®. Then, the BMI was calculated. BMI and height z-scores were calculated using height and BMI plus biological sex and age.
GLYCEMIC CONTROL
Blood venous samples were collected after 8-12 h of fasting. The HbA1c was used to analyze glycemic control.
QUALITY OF LIFE
Measured through the Kidscreen-27 questionnaire18. The five items (physical well-being, psychological well-being, autonomy and relationship with the environment, friends and social support, and school environment) plus the total score were used.
CLARKE TEST
Questionnaire to assess the sensitivity and perception of hypoglycemic events in the pediatric population with T1DM.
STANDING BROAD JUMP
The participant stood behind the jump line with feet shoulder-width apart. From that position, they jumped as far as possible, landing with both feet simultaneously19. Three attempts were made, and the best score in centimeters (cm) was considered.
PUSH-UP TEST
The participants positioned themselves on a mat, supported their feet and hands at shoulder width, and performed maximum repetitions of trunk lifts with a 90° arm flexion20. The maximum number of repetitions (reps) was considered.
PRONE PLANK
Only the forearms and toes were allowed to be in contact with the mat, maintaining the isometric position for as long as possible21. The total time recorded in seconds (s) was used.
10RM SQUAT AND CHEST PRESS
The subject performed a bench press and a squat press with a weight in kilograms (kg) that allowed them to perform up to 10 repetitions. The indirect RM was then calculated using the formula proposed by Martínez-Cava et al.22. The estimated 1RM was used.
20 M SHUTTLE RUN TEST
It involved running 20 m back and forth at an incremental speed starting at 8.5 km/h, which increased by 0.5 km/h/min. Maximum oxygen consumption (VO2 max) in ml/kg/min was estimated using the formula by Leger et al.23, and the distance covered in meters (m) was recorded.
Statistical analysis
Due to the small sample size, all statistical analyses were conducted using non-parametric tests. A Wilcoxon test was applied to evaluate changes in the dependent variables after the concurrent training protocol. Changes in the variables measured by the CGM were compared using a Friedman test at 4, 8, and 12 weeks of intervention. When significant differences were identified with the Friedman test, a post hoc Dunn test was performed. Statistical significance was set at p ≤ 0.05. Data are presented as the median with interquartile range (IQR). Statistical analyses were conducted using PRISM 9.0 (GraphPad, California, 2019).
Results
The participants (n = 7, 11 [10-13] years, 4 males and 3 females) were recruited from the Pediatric Endocrinology Unit to the Physical Activity and Sports Medicine Unit of the Hospital Dr. Exequiel González Cortés (HEGC). The participants had a period of 12 (8-28.5) months from the first T1DM diagnosis to the start of protocol training. Baseline insulin treatment before protocol training consisted of insulin glargine (Lantus, Sanofi, Paris, France), n = 3, at a dose of 11 (10-31) UI and insulin degludec (Tresiba, Novo Nordisk, Bagsværd, Denmark), n = 4, at a dose of 16 (5.7-31.5) UI. Of the 8 participants who started, one withdrew from the training protocol due to personal problems. The baseline characteristics of the participants are shown in table 2.
Table 2 Dependent variables before and after 12 weeks of the concurrent training protocol in the pediatric population with T1DM
| Variable | Before intervention (median plus IQR) | After intervention (median plus IQR) | p |
|---|---|---|---|
| Anthropometric | |||
| Weight (kg) | 47.7 (38.5-67.2) | 47.4 (39.4-70.6) | 0.50 |
| Height (cm) | 143.0 (138.7-168.0) | 146.0 (140.75-170.0) | 0.03 |
| Height-z (SD) | 0,5 (0.9-2.3) | 0.7 (0.8-2.3) | 0.23 |
| BMI (kg/m2) | 20.8 (19.7-24.8) | 20.9 (19.2-25.7) | 0.93 |
| BMI-z (SD) | 0.9 (0.7-2.3) | 0.82 (0.4-2.4) | 0.20 |
| Glycemic control | |||
| Hb1AC (%) | 6.9 (5.8-7.6) | 6.7 (6.3-9.0) | 0.04 |
| Muscle function | |||
| Standing broad jump (cm) | 129.0 (103.5-152.0) | 129.0 (107.5-180.0) | 0.03 |
| Push-up test (rep) | 4.0 (0.5-29.0) | 10.0 (5.0-39.0) | 0.06 |
| Prone plank (s) | 43.5 (26.0-99.2) | 81.5 (79.9-139.3) | 0.01 |
| 10RM squat (kg) | 31.3 (25.9-44.7) | 36.0 (30.6-51.7) | 0.03 |
| 10RM chest press (kg) | 17.9 (17.0-41.9) | 25.1 (22.6-48.5) | 0.01 |
| Quality of life | |||
| Physical well-being (points) | 88.8 (86.1-100) | 100 (100-100) | 0.03 |
| Psychological well-being (points) | 96.4 (92.8-100) | 96.4 (96.4-100) | 0.12 |
| Autonomy and relationship with the environment (points) | 85.7 (83.9-92.8) | 89.2 (85.7-92.8) | 0.50 |
| Friends and social support (points) | 87.5 (87.5-100) | 93.7 (87.5-100) | 0.50 |
| School environment (points) | 93.7 (84.3-100) | 93.7 (87-5-100) | 0.50 |
| Total score (points) | 90.4 (88.7-97.4) | 94.1 (93.1-97.8) | 0.01 |
| Aerobic capacity | |||
| VO2max (mL/kg−1/min−1) | 41.5 (39.6-46.3) | 41.5 (39.6-46.3) | 0.50 |
| Distance running in shuttle 20 m run test (m) | 200.0 (180.0-760.0) | 280.0 (240.0-960.0) | 0.01 |
BMI: body mass index; BMI-z: body mass index by z-score; Hb1AC: glycated hemoglobin; Height-z: height by z-score; RM: repetition maximum; rep: repetitions; VO2max: maximal oxygen consumption; IQR: interquartile range.
Time slots associated with hypoglycemic events
The hypoglycemic events were divided into time slots (00:01-06:00, 06:01-12:00, 12:01-18:00, and 18:01-00:00) and counted. The hypoglycemic episodes from 00:01 to 06:00 were 19.1% of the events, from 06:01 to 12:00 were 25.7% of the events, from 12:01 to 18:00 were 33.8% of the events and from 18:01 to 00:00 were 21.3% of the events. The time slot from 06:01 to 12:00 was divided into 06:01-09:00 or pre-training and 09:01-12:00 or immediately post-training, considering that the training sessions took place at 09:00. Thus, of the 25.7% of hypoglycemic episodes that occurred in this time slot, 65.7% occurred in the pre-training time slot and 34.2% in the post-training time slot. Therefore, the number of hypoglycemic episodes directly related to exercise was 8.7%. The hypoglycemic episodes that occurred during the training protocol were treated with 30g of fast-assimilating carbohydrate.
CGM
One week before the start of the training protocol, a median plus IQR of 0.30 (0.0-0.50) hypoglycemic episodes/week was recorded. A median plus IQR of 0.30 (0.19-0.40) hypoglycemic episodes/week was recorded during the 1st month of intervention, 0.20 (0.10-0.58) episodes/week during the 2nd month, and 0.15 (0.08-0.55) episodes/week during the 3rd month. One week prior to the start of the training protocol, the median plus IQR of the weekly glucose concentration was 149.00 (133.00-213.00) mg/dL. The median plus IQR of weekly glucose concentration was 158.25 (133.00-214.75) mg/dl during the 1st month of intervention, 160.75 (139.38-226.00) mg/dL during the 2nd month, and 153.00 (137.75-208.08) mg/dL during the 3rd month. One week before the start of the training protocol, the median plus IQR time in range was 80.00 (65.00-94.00)%. The median plus IQR time in range was 70.00 (51.63-93.75)% during the 1st month of intervention, 66.00 (52.63-94.00)% during the 2nd month and 66.50 (52.00-94.00)% during the 3rd month. No significant differences were found between the baseline and concurrent protocol training scores for the three variables.
Anthropometrics
Significant differences were evidenced in the pre-to-post-intervention for height, increasing from 143.0 (138.7-168.0) to 146.0 (140.75-170.0) cm (p = 0.03). No significant differences were observed for height z-score (p = 0.23), weight (p = 0.64), BMI (p = 0.82) and BMI z-score (p = 0.20).
Glycemic control
Significant differences were observed in the pre-to-post-intervention for HbA1c, increasing from 6.9 (5.8-7.6)% to 6.7 (6.3-9.0)% (p = 0.046).
Quality of life
Significant differences were observed in the pre-to-post-intervention in the physical well-being domain, increasing from 88.8 (86.1-100) to 100 (100-100) points (p = 0.03) and the total score from 90.4 (88.7-97.4) to 94.1 (93.1-97.8) points (p = 0.001). No significant differences were observed for the rest of the Kidscreen-27 domains (Table 2).
Muscle function
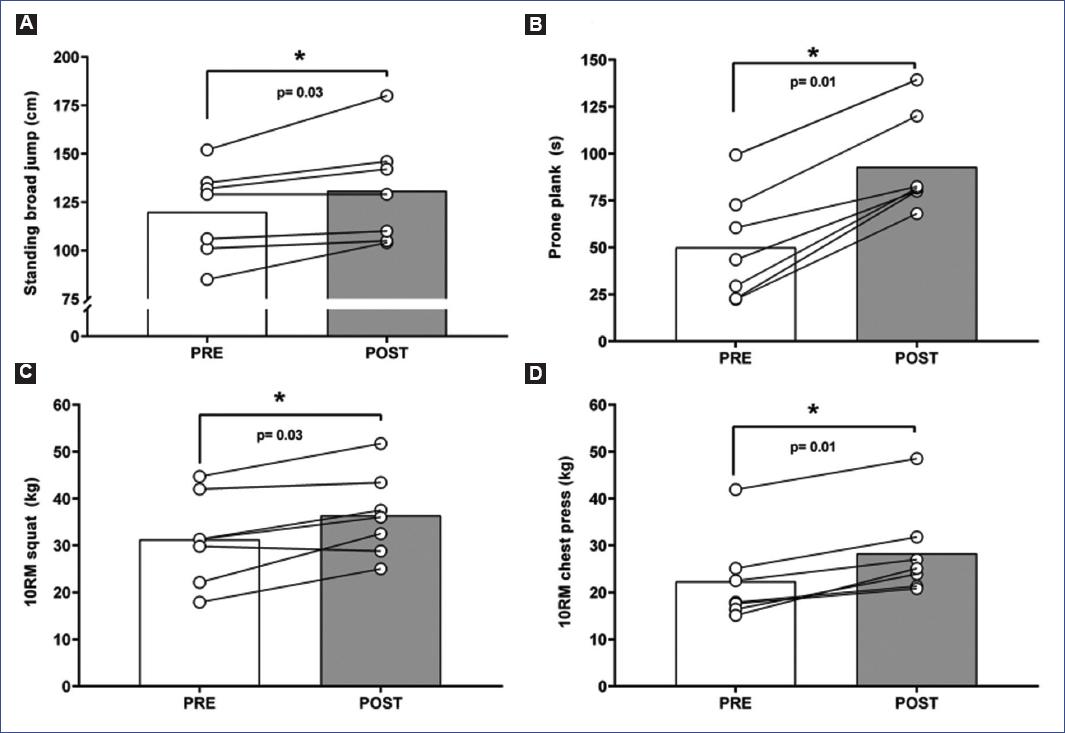
Significant differences were observed in the pre-to-post-intervention in the standing broad jump, increasing from 29.0 (103.5-152.0) to 129.0 (107.5-180.0) cm (p = 0.03) figure 1A. Similarly, the prone plank increased from 43.5 (26.0-99.2) to 81.5 (79.9-139.3) s (p = 0.01) figure 1B, the 10RM squat increased from 31.3 (25.9-44.7) to 36.0 (30.6-51.7) kg (p = 0.03) figure 1C, and the 10RM chest press increased from 17.9 (17.0-41.9) to 25.1 (22.6-48.5) kg (p = 0.01) figure 1D. No significant differences were observed for the push-up test (p = 0.06).
Discussion
Following the concurrent training protocol, the HbA1c, muscle function, quality of life, and aerobic capacity increased significantly in the pediatric population with T1DM. Throughout the intervention, no changes were evidenced in the weekly hypoglycemic episodes, weekly glycemic concentration, and time in range.
Throughout the three intervention months, the pediatric population with T1DM showed no changes in the weekly hypoglycemic episodes, and the mean of hypoglycemic episodes per day was 1.6-1.8. Similarly, Laffel et al.24 showed 1.4-1.5 daily hypoglycemic episodes. Therefore, our results could support that physical exercise programs in T1DM patients are safe and could be used as a strategy to include physical activity and sports habits throughout the 1st year of the appearance of the disease. Despite the lack of results related to weekly glycemic concentration and time in range, our results are below those reported in the literature24. Laffel et al.24 reported weekly glycemic concentrations and time in the range between 199-209 mg/dL and 37-43%, respectively. In our study, we reported weekly glycemic concentration and time in the range between 163-168 mg/dL and 44-66%, respectively, which could demonstrate better glycemic control in our study population. It is important to mention that the results of Laffel et al.24 incorporated only CGM without any exercise intervention. To the best of our knowledge, there is no evidence of the effects of concurrent protocol training plus CGM on the variables mentioned above in the pediatric population with T1DM.
At the end of the concurrent training protocol, the HbA1c decreased from 6.9 (5.8-7.6)% to 6.7 (6.3-9.0)%. Our results differ from those reported in the literature25,26, which showed that following diverse training protocols, the HbA1c decreases in the pediatric population with T1DM. We hypothesized that the increase in HbA1c was due to the planning of our concurrent training protocol. Thus, the meta-analysis of Garcia-Hermoso et al.25 reported that concurrent training was superior to endurance training alone, but concurrent training should have a duration of ≥ 24 weeks and be of high intensity (aerobic phase), characteristics that contrast with our protocol of 12 weeks and moderate intensity (aerobic phase). Therefore, in the future, the aerobic phase should include high-intensity components such as high-intensity interval training (HIIT). Importantly, the fear of exercise-associated hypoglycemic events is greater in the pediatric population who has completed exercise programs compared to their peers who have not completed exercise programs27. One strategy to reduce this fear of exercise-associated hypoglycemia is to consume carbohydrates28. Therefore, we hypothesized that this strategy could have increased HbA1c levels even though participants were under nutritional control. Thus, in a larger trial, we need to consider nutritional education sessions related to carbohydrate overconsumption before the start of the training protocol to avoid this HbA1c increase.
The muscle function improved after the concurrent training protocol, showing an improvement in the standing broad jump (p = 0.03), prone plank (p = 0.01), 10RM squat (p = 0.03), and 10RM chest press (p = 0.01). Our results are in line with those reported by other authors9,26, in which other concurrent training protocols improved the muscle function of the pediatric population with T1DM. Specifically, Dhooge et al.9 showed that after a concurrent training protocol of 20 weeks, 2 times/week, the strength of the upper and lower extremities (1RM) increased by 72.2% and 23.1%, respectively. The reported improvements in muscle function were relevant, considering the detrimental effects of T1DM on muscle function and mass5,29. Thus, Maratova et al.5 after a 9-year follow-up, found that the pediatric population with T1DM showed a decrease in muscle function and mass even following the pharmacological treatment for the disease. Similarly, Tan et al.29 compared muscle function and architecture between pediatric populations with and without T1DM, and showed that the pediatric population with T1DM presented lower muscle function expressed by 40.5% and 31.5% lower torque of the knee flexors and extensors and architectonic changes, such as 12.8% lower thickness of the rectus femoris. Therefore, the addition of a concurrent training protocol could maintain and prolong ideal values for muscle function and mass in the pediatric population with T1DM.
The quality of life improved after the concurrent training protocol in the physical function domain (p = 0.03) and the total score (p = 0.01) of the Kidscreen-27 questionnaire. Our results are relevant considering the lower quality of life of the pediatric population with T1DM compared with healthy peers6. The systematic review of Absil et al.26 showed that in only 1 of 7 studies, the quality of life improved after different training protocols in the pediatric population with T1DM. Therefore, our results are novel compared with the available evidence30. The quality of life of this population is reduced due to the fear of hypoglycemic episodes30. Thus, when the parents are in the highest quartile of fear of hypoglycemic episodes, the pediatric population with T1DM showed a 20% lower quality of life compared to those in the lowest quartile of fear of hypoglycemic episodes30. Interestingly, the use of CGM devices evidenced a decrease in the fear of hypoglycemic episodes among parents of the pediatric population with T1DM31. Therefore, we hypothesized that the use of CGM, the lower hypoglycemic episodes reported (fewer than 2 daily), and the improvement in muscle function could contribute to the increases in the quality of life in the pediatric population with T1DM.
The concurrent training protocol showed an increase in the distance covered in the 20m shuttle run test (p = 0.01) without change in the VO2 max estimated by the same test. We hypothesized that the improvement in the distance covered in the 20m shuttle run test (26.9%) is related to a neuromuscular improvement in response to the resistance training phase of the concurrent training protocol32. Thus, the improvement in intra and intermuscular coordination would be responsible for a better running capacity. Alternatively, we believe that no changes in VO2 max could be a result of the aerobic phase intensity proposed in the concurrent training protocol not being high enough to generate central and peripheral adaptations. Moreover, the estimation equation used23 could underestimate the changes due to considering only the age and level reached in the test. Similarly, other concurrent training protocols9 have also not produced improvements in VO2 max. However, when HIIT protocols are compared with moderate-intensity continuous training (MICT) in the pediatric population with T1DM33, the HIIT protocols produce significant improvements (6.1%) versus no changes (3.1%) in the MICT protocol for VO2 max33. Therefore, if we aim to improve the VO2 max of the pediatric population with T1DM, the recommendation should be centered on ensuring that the aerobic phase of the concurrent training protocol is based on the HIIT protocol.
The main strength of this study is providing safe planning of concurrent training protocol with positive results in glycemic control, muscle function, and quality of life, which could guide clinical practice in public health. In contrast, the study design (pilot study) presents certain limitations, such as the small number of participants and the absence of a control group. Therefore, this represents one of the first steps in exercise treatment for the pediatric population with T1DM in public health, and more studies are needed in this area.











 nueva página del texto (beta)
nueva página del texto (beta)