Introduction
Tuberous sclerosis complex (TSC) is an autosomal dominant systemic disease that can affect any human organ with lesions known as hamartomas. It is caused by pathogenic variants in the tumor suppressor genes TSC1 and TSC2, which encode hamartin and tuberin, respectively. These genes are located on chromosomes 9q34 and 16p13.3, respectively. The hamartin-tuberin complex downregulates mTORC11.
The presence of angiofibromas in tuberous sclerosis can have significant repercussions for patients, with both physical and psychological implications. Angiofibromas are commonly found in school-aged patients with TSC. Although they rarely pose a life-threatening risk, they often become a source of stigma for these patients, affecting their self-esteem, social interaction, and consequently, their quality of life. Due to their potential to become disfiguring, various therapeutic options have been considered, including radiosurgery, cryosurgery, electrofulguration, CO2 laser, and chemical peels, most of which are painful. Angiofibromas present as multiple erythematous or skin-colored papules in centrofacial areas, primarily on the nose and surrounding regions1.
The accidental observation of facial angiofibroma regression in a patient with TSC who received oral sirolimus after a renal transplant has led to the development and increasing use of this drug through topical application. Sirolimus is a macrolide consisting of a ring with lactonic and lactam-like groups derived from the fermentation products of Streptomyces hygroscopicus. The disappearance of angiofibromas has been observed with its use2.
The concentrations of topical sirolimus, the vehicle used, and the dosage vary among different studies. The use of topical sirolimus has been reported in active ingredient concentrations ranging from 0.003% to 1%, with the most common concentrations being 0.1% and 0.2%3,4.
At the Instituto Nacional de Pediatría, 105 patients were registered from 2005 to 2017, all of whom were evaluated in the dermatology service outpatient clinic.
There is no consensus on management, with most treatments being painful, and multiple sessions are often required to achieve satisfactory results. Topical sirolimus represents a therapeutic option to achieve a sustained response in preventing the appearance of new angiofibromas.
The objective of this study was to analyze the effectiveness and safety of topical sirolimus in the treatment of children with angiofibromas and TSC. The study was approved by the Institutional Project Review Committees of the Instituto Nacional de Pediatría in Mexico City, with registration number 2019/003.
Method
After obtaining approval from the Research Ethics Committee, Biosafety Committee, and Research Committee of the Instituto Nacional de Pediatría, parents of the patients completed the informed consent form, and children provided assent letters (for children aged 6-18 years). Ten patients diagnosed with TSC were recruited. Patients with potential pregnancy, autoimmune conditions, or those taking oral sirolimus were excluded.
A quasi-experimental, open-label before-and-after study was conducted. The study included patients with angiofibromas and TSC treated at the dermatology service of the Instituto Nacional de Pediatría from 2019 to 2020, aged 8-17 years, who agreed to participate and signed both informed consent and assent letters.
Patients who met the selection criteria underwent baseline photography, lesion counting, documentation of lesion locations, and measurement of the largest lesions' diameters. Subsequently, they received the compounded sirolimus preparation with instructions to apply it once daily on the right side of the face for 6 months, followed by discontinuation on that side and initiation on the left side for another 6 months. Patients attended monthly follow-up appointments for photography and evaluation of lesion number and size. In addition, lesion resolution and sirolimus-related adverse events were assessed. After the initial 6-month treatment period, lesion recurrence was also evaluated.
Sirolimus solution (1 mg/1 mL) and CeraVe moisturizing lotion were used. The 1% sirolimus compounded formulation was prepared by incorporating 0.1 g of sirolimus into 19.9 g of CeraVe moisturizing lotion, which was then provided to patients. They transported it under refrigeration, and follow-up appointments were scheduled monthly. Details of the sirolimus preparation are recorded in figure 1.
Results
Ten patients were included, with a median age of 15.5 years (range: 8-17 years). Six patients (60%) were male. All patients had numerous lesions of varying sizes. Table 1 shows the evolution of the lesions, demonstrating a decrease in both number and size from the 3rd month onward. Regarding recurrences, evaluation of the right side of the face 6 months after treatment discontinuation revealed new lesions appearing from the 4th-month post-treatment.
Table 1 Evolution of facial angiofibromas
| Evaluated features | n | Month of treatment | p* | |||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |||
| Number of lesions | 20 | 113 | 113 | 94 | 74 | 47 | 53 | < 0.001 |
| Average size (mm) | 20 | 39 | 39 | 34 | 30 | 24 | 20 | < 0.001 |
| Disappeared lesions | 20 | 0 | 0 | 18 | 37 | 48 | 59 | < 0.001 |
| Recurrences Average number of lesions | 10 | 0 | 0 | 0 | 0.3 | 2.7 | 6.5 | < 0.001 |
*Repeated measures analysis of variance. Within-subjects effects.
There was a positive correlation between age and the number of lesions, with older patients having more lesions (r = 0.466, p = 0.038). A positive correlation was also found between average lesion size and number of lesions, with larger lesions associated with greater lesion numbers (r = 0.401, p = 0.08). Conversely, a negative correlation was found regarding the percentage of resolved lesions, with older patients showing a lower percentage of resolved lesions (r = −0.501, p = 0.024).
The percentage of resolved lesions showed negative correlations with both the number of lesions (r = −0.857, p < 0.001) and lesion size (r = −0.67, p = 0.001). These findings suggest that early treatment initiation may be beneficial.
Regarding safety, two patients experienced local irritation during sun exposure, which resolved with sunscreen application, and treatment discontinuation was not required.
Discussion
TSC is a genetic, autosomal dominant disorder characterized by the formation of benign tumors in multiple systemic organs4. Facial angiofibromas are present as red or pink centrofacial papules, particularly in the nasofacial fold, cheeks, and chin. They develop in early childhood (6-8 years) and are present in over 80% of individuals with TSC (Figs. 2-5). These tumors, which contain vascular and connective tissue elements, constitute major criteria for TSC and commonly affect school-age children and adolescents, significantly impacting their self-esteem4.
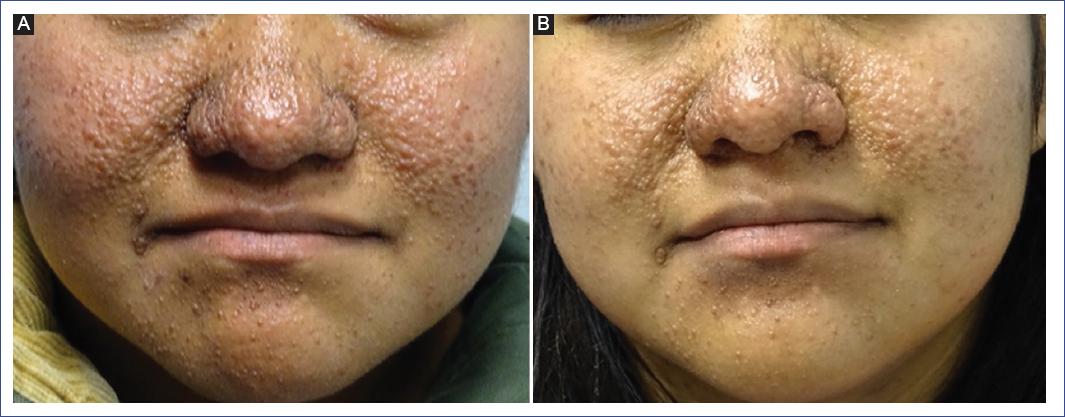
Figure 2 A and B: a 15-year-old adolescent with tuberous sclerosis and angiofibromas shows decreased lesion size and disappearance of small angiofibromas after 12 months of treatment with 1% sirolimus lotion. Note the absence of erythema.
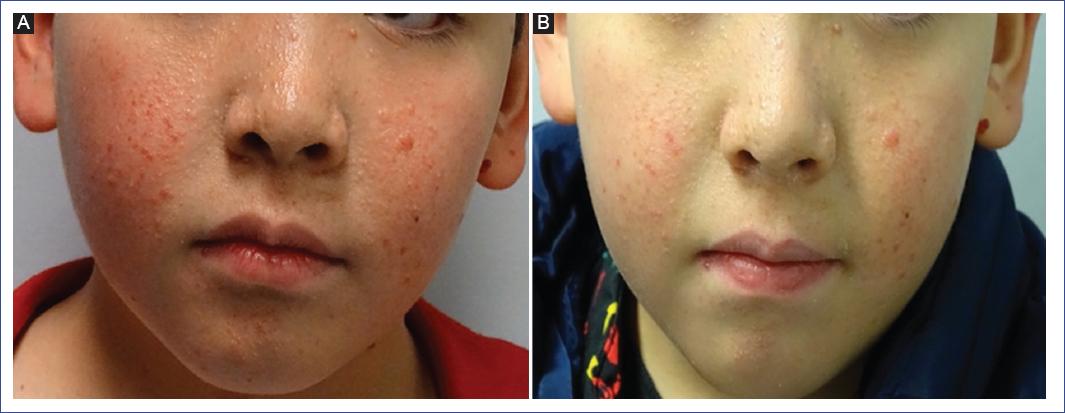
Figure 3 A and B: an 11-year-old boy with tuberous sclerosis and angiofibromas shows results after 1 year of treatment with topical sirolimus.
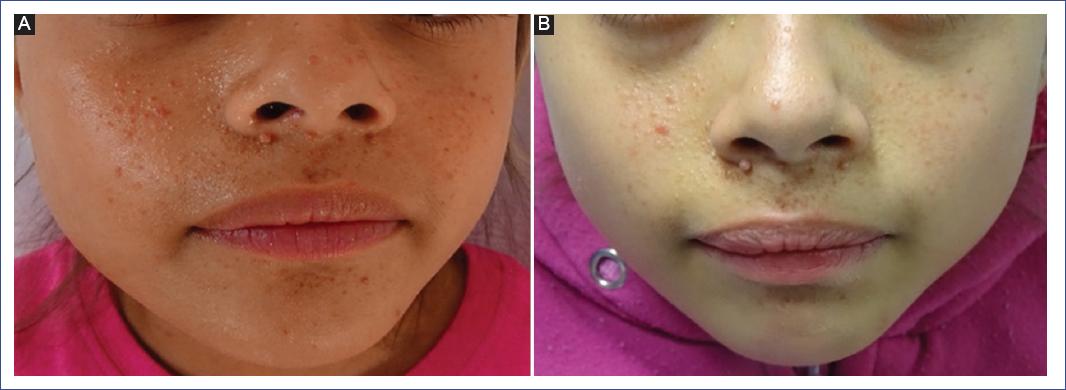
Figure 4 A and B: a 13-year-old girl with tuberous sclerosis and angiofibromas after 1 year of treatment with 1% sirolimus lotion.
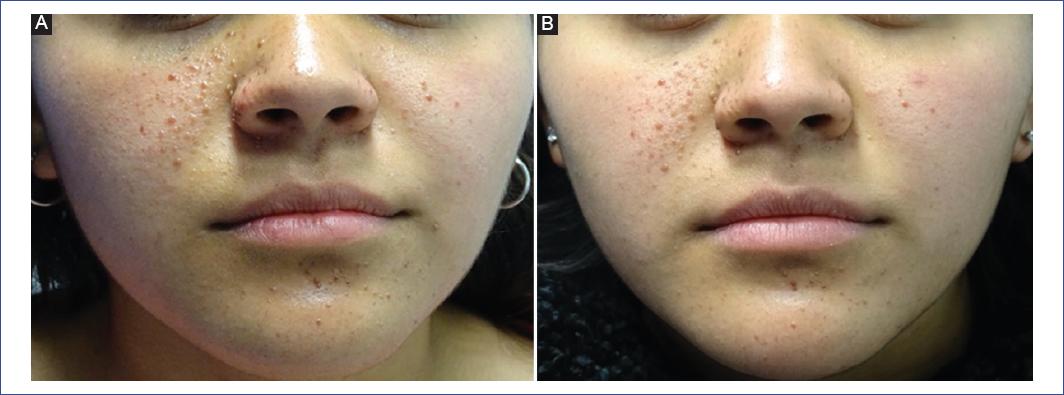
Figure 5 A and B: a 16-year-old adolescent, after 1 year of treatment with 1% sirolimus lotion. A decreased lesion size and disappearance of multiple angiofibromas can be observed.
An ideal treatment for TSC has not yet been established; however, various procedures such as radiosurgery, cryosurgery, electrofulguration, CO2 laser, and chemical peels are available. These treatments are often painful, invasive, and expensive1,4.
The literature reports promising outcomes with topical sirolimus, which has been compounded at concentrations ranging from 0.001% to 1%. However, traditional preparation processes involve crushing and sieving sirolimus tablets and incorporating them into a vehicle (such as cold cream, polyvinylidene fluoride [PVDF] in ointment, gel, or petroleum jelly), resulting in poor cosmetic outcomes. Direct application of sirolimus solution (1 mg/mL) on angiofibromas resulted in intense irritation, necessitating a reduction in application frequency from twice daily to once daily. However, a compounded formula with sirolimus solution (1 mg/mL) mixed with emollients has been reported to be better tolerated by patients in previous studies4,5.
Consequently, we used sirolimus oral solution (1 mg/mL) mixed with a moisturizing lotion, which significantly improved the organoleptic properties and cost of the formula. This formulation was prepared by the pharmacology department, and physicochemical stability tests were conducted on the 1% sirolimus formulation. The preparation requires refrigerated storage and has an optimal duration of 1 month.
In general, our patients showed improvement starting from the 3rd month of using 1% sirolimus in CeraVe moisturizing lotion, and this improvement persisted throughout both the initial 6-month application on the right cheek and the subsequent 6-month application on the left cheek. However, patients with large angiofibromas exhibited only a moderate response (40-80%). In addition, we found that children with smaller and fewer lesions showed a good response (80%) (Figs. 2-5). Therefore, while the efficacy and tolerability of 1% sirolimus in CeraVe moisturizing lotion have been demonstrated for small angiofibromas, this was not the case for larger lesions. This aligns with Foster's findings5, which indicated a decrease in size and disappearance of smaller lesions but no significant change in larger lesions.
Multiple reports have demonstrated a good safety profile, with only six cases of local irritation reported: four related to the direct application of sirolimus oral solution (1 mg/mL) and two associated with the PVDF vehicle. In our study, we observed two patients with local irritation that resolved with sunscreen application. In studies monitoring systemic effects, no adverse effects have been reported, and sirolimus plasma levels have remained below detection limits, well below both therapeutic values and the toxicity range2,5.
We consider 1% topical sirolimus to be a good treatment option for facial angiofibromas in children. While the ideal formulation has not been established, we found that 1% sirolimus in CeraVe lotion is highly effective in children, causing neither discomfort nor pain. Early initiation of treatment helps improve patients' self-esteem. However, in adolescents, ablative treatments with shaving and electrodesiccation can be used, followed by the application of 1% or 2% sirolimus. Long-term studies are needed to establish which treatment is most effective and have the lowest recurrence rate.
In our patients, the recurrence time after treatment cessation was 4 months. This information may guide treatment scheduling, allowing for planned treatment interruptions and resumption after this period.
It is important to emphasize that formulations, regardless of type, should be prepared by trained personnel under appropriate pharmacological laboratory conditions. Home preparation without proper controls should not be encouraged.
Conclusion
Topical sirolimus is an effective and safe treatment for facial angiofibromas in children with TSC, showing significant improvement, particularly in smaller lesions. The treatment has a good safety profile, with minimal side effects, mainly local irritation. While larger lesions showed a more moderate response, early treatment improved patients' self-esteem. Recurrence of lesions occurred about four months after discontinuation, suggesting the need for periodic treatment cycles. Further studies are needed to optimize dosage and treatment duration.











 nueva página del texto (beta)
nueva página del texto (beta)



