Introduction
Arthritis is a chronic degenerative disease characterized by joint inflammation that limits the range of joint motion with pain or joint tenderness. Juvenile idiopathic arthritis (JIA) is the most common rheumatic disease in the pediatric age group and is one of the leading causes of acquired disability in this age group1. The age of onset is typically between 1 and 16 years, with peak incidence around 5 years of age, and it frequently extends into adulthood. The disease is twice as common in females. The annual incidence of JIA worldwide is 8-23 cases/100,000 children under 16 years of age2. In Mexico, an annual incidence of 80-90 cases/100,000 children is reported3. The International League of Associations for Rheumatology proposes that JIA be classified into seven clinical subtypes: Oligoarticular, Rheumatoid-factor-positive polyarthritis and Rheumatoid-factor-negative polyarthritis systemic, enthesitis-related, psoriatic, and undifferentiated. These are mutually exclusive and have different clinical management approaches. In Mexico, it has been reported that the polyarticular course represents between 50% and 66% of patients with JIA3-6 and that females account for 63% of JIA cases4.
Treatment promotes better control of the disease’s inflammatory activity, prevents disease progression and complications such as chronic damage, functional disability, and negative impact on growth and development, and achieves disease remission with a good quality of life. However, the costs of treatment can limit its adequate application in the population.
Treatment intends to achieve greater control of the inflammatory activity of the disease, avoiding consequences such as chronic damage and functional disability. The first line of treatment is non-steroidal anti-inflammatory drugs; these medications are used as adjuvants for the disease until the administration of disease-modifying antirheumatic drugs (DMARDs), such as methotrexate (MTX). The objective of MTX is to inhibit cell replication and block the production of cytokines such as interleukin 1 and tumor necrosis factor-α, preventing the replication of synovial cells and the production of collagenases (cartilage-destroying enzymes). When MTX fails to achieve the objective (due to intolerance or refractoriness), the following can be administered: leflunomide, sulfasalazine, cyclosporine, or methylprednisolone. In cases where there is no improvement because the arthritis is refractory to MTX, treatment continues with biological agent therapies such as etanercept, abatacept, tocilizumab, and anakinra7,8.
Direct medical costs (DMC) are those incurred during the process of medical care. JIA’s mean annual DMC internationally ranges from 193 USD in India9 to 49,429 USD in the United Kingdom10. Costs have not been reported for other Latin American countries, specifically Mexico. Significant differences exist in the components of DMC across various studies. In some cases, the main cost corresponds to the pharmacological treatment component9-15; however, in other studies, the main cost component corresponds to health-care personnel11,16-18. The JIA subtype with the highest DMC is polyarthritis19, followed by polyarthritis and systemic arthritis, although the differences between the latter two are minimal19,20. The management costs of JIA show significant variations between countries due to the specific characteristics of the health-care system (human, technological, and economic resources) and treatment protocols.
There is a clear absence in the scientific literature of cost evaluation studies for JIA in Latin America, particularly in Mexico. This situation confers the need to obtain estimates of the DMC of JIA in Mexico, especially for the polyarthritis type. The study aimed to determine the DMC of polyarthritis21.
Methods
A partial economic evaluation of the cost-of-illness type was developed from the perspective of the Hospital Materno Infantil (HMI). The time horizon was 12 years, covering from the diagnosis of the disease to the last available measurement. All patients diagnosed with polyarticular JIA treated by the pediatric rheumatology service of the HMI who had an active clinical record and had not been referred to another Instituto de Seguridad Social del Estado de México y Municipios (ISSEMYM) medical unit for follow-up were included. Resource identification was carried out through the review of clinical records.
Medications were classified into seven groups according to the therapeutic objective established by the HMI’s pediatric rheumatology service: (a) biological disease-modifying drugs, (b) synthetic disease-modifying drugs, (c) concomitant medications, (d) prophylactics, (e) treatments for adverse effects of other medications, (f) inflammation and pain, and (g) those to address complications from the disease.
Each patient contributes information depending on their chronological follow-up time within the institution, from their diagnosis to record review in 2022. Patients were classified according to their management status as (a) patient at the time of record review (in treatment [TX]), (b) in clinical remission with medication (CRMM), and (c) in clinical remission without medication (CRWM). Information on the patient’s age, sex, and rheumatoid factor (+ or −) was also included.
Costs were estimated for each patient, including diagnostic, treatment, and follow-up costs. Cost components were divided into consultation costs, medications, hospitalization, imaging studies, and laboratory tests. The micro-costing technique (unit price × quantity) was used for medication and medical supply components. The average cost technique (institutional cost/recovery fees × quantity) was also used for imaging and laboratory studies, bed day/hospitalization costs, procedures, and consultations.
Costs primarily come from two sources of information: 1. The unit acquisition costs of medications and medical supplies from the HMI in 2021, and 2. The average costs/recovery fees reported by ISSEMyM, both published in the electronic version of the Official Gazette "Gaceta del Gobierno"21. In addition, costs from the recovery fee schedules of the Hospital Infantil de México Federico Gómez were used22.
Two estimates of DMC were made. The first DMC estimate corresponds to an administrative-type approach, which provides information on the total average cost and per component for each patient with JIA per year for the entire patient population and projects the institution’s budget according to its future service offerings. This estimate includes the report of central tendency measures, mean and standard deviation (SD), as well as the percentage contribution of each component to the total cost for each year. In addition, the average percentage contribution of each component over the 12 years of follow-up for this population is reported.
The second estimate includes a more analytical approach, with information corresponding to the specific DMCs for the population that did incur expenses in a given component in a specific year of the disease. This is considered valuable information for decision-making regarding its management. Since the costs obtained do not present a normal distribution, the distributions were evaluated using the Kernel density graphical method and the skewness/kurtosis statistics method. In this second analysis, non-parametric measures are reported (median and 25th-75th percentiles). Finally, a cost analysis of the resource structure of the medication component was performed according to the previously proposed classification.
All costs are reported in US dollars, using the exchange rate of December 31, 2021 (1 USD = 20.5157 MXN)23. This study was approved by the HMI research committee, registration 01-2022.
Results
Twenty-six medical records of patients with polyarticular arthritis were analyzed. Twenty-two records corresponded to female patients (84.6%), the frequency of positive rheumatoid factor was 43%, the mean age of patients was 16.1 years (SD = 1.8), the age of disease onset was 9.6 years (SD = 3.5) with an average disease treatment time of 6.5 years (SD = 2.9), with a minimum of 2 years and a maximum of 12 years. The results of the clinical evaluation indicate that 18 patients were in treatment (TX) (69.2%), six were in CRMM (23.1%), and two were in CRWM (7.7%).
Table 1 reports the total DMCs and by component for each of the 12 years, finding a mean of USD 4,555.2 (SD = 1,456.7) in the first 10 years, and the relative weight of the components of consultation, hospitalization, imaging, and laboratory studies, and finally medications of 1.8%, 8.0%, 8.4%, and 82.7%, respectively.
Table 1 Annual direct medical costs by component and percentage
| Year | Cost component | n | Mean | Standard deviation | % |
|---|---|---|---|---|---|
| 1 | Hospitalization | 26 | $305.3 | $1,019.1 | 17.8 |
| Consultation | 26 | $124.3 | $257.9 | 7.2 | |
| Diagnostic imaging and laboratory | 26 | $417.0 | $339.9 | 24.3 | |
| Medications | 26 | $869.7 | $1,729.9 | 50.7 | |
| Total cost | 26 | $1,716.4 | $2,246.4 | ||
| 2 | Hospitalization | 26 | $269.7 | $760.9 | 5.5 |
| Consultation | 26 | $81.8 | $48.6 | 1.7 | |
| Diagnostic imaging and laboratory | 26 | $409.8 | $252.6 | 8.4 | |
| Medications | 26 | $4,142.3 | $4,592.9 | 84.5 | |
| Total cost | 26 | $4,903.6 | $4,943.0 | ||
| 3 | Hospitalization | 24 | $435.4 | $1,478.8 | 6.4 |
| Consultation | 24 | $63.6 | $38.6 | 0.9 | |
| Diagnostic imaging and laboratory | 24 | $298.3 | $173.2 | 4.4 | |
| Medications | 24 | $6,037.6 | $6,169.9 | 88.3 | |
| Total cost | 24 | $6,834.8 | $6,577.6 | ||
| 4 | Hospitalization | 22 | $421.2 | $1,186.7 | 7.0 |
| Consultation | 22 | $65.4 | $36.8 | 1.1 | |
| Diagnostic imaging and laboratory | 22 | $295.6 | $164.7 | 4.9 | |
| Medications | 21 | $5,517.2 | $5,074.0 | 91.2 | |
| Total cost | 22 | $6,048.6 | $5,657.4 | ||
| 5 | Hospitalization | 20 | $347.1 | $849.0 | 7.1 |
| Consultation | 20 | $63.6 | $49.7 | 1.3 | |
| Diagnostic imaging and laboratory | 19 | $344.7 | $376.9 | 7.1 | |
| Medications | 19 | $4,348.6 | $3,694.5 | 89.3 | |
| Total cost | 20 | $4,869.3 | $3,953.4 | ||
| 6 | Hospitalization | 18 | $180.3 | $629.1 | 3.5 |
| Consultation | 18 | $46.9 | $29.0 | 0.9 | |
| Diagnostic imaging and laboratory | 18 | $258.6 | $184.6 | 5.0 | |
| Medications | 18 | $4,660.6 | $4,787.6 | 90.6 | |
| Total cost | 18 | $5,146.3 | $4,744.6 | ||
| 7 | Hospitalization | 14 | $489.9 | $1,321.8 | 10.7 |
| Consultation | 14 | $38.6 | $33.3 | 0.8 | |
| Diagnostic imaging and laboratory | 14 | $255.2 | $244.0 | 5.6 | |
| Medications | 14 | $3,783.0 | $4,146.8 | 82.8 | |
| Total cost | 14 | $4,566.7 | $4,181.8 | ||
| 8 | Hospitalization | 9 | $698.4 | $2,095.3 | 16.3 |
| Consultation | 9 | $91.6 | $182.7 | 2.1 | |
| Diagnostic imaging and laboratory | 9 | $306.0 | $251.0 | 7.2 | |
| Medications | 9 | $3,179.9 | $3,265.3 | 74.4 | |
| Total cost | 9 | $4,276.0 | $3,465.9 | ||
| 9 | Hospitalization | 7 | $255.2 | $675.2 | 5.9 |
| Consultation | 7 | $35.8 | $18.2 | 0.8 | |
| Diagnostic imaging and laboratory | 7 | $415.3 | $316.4 | 9.6 | |
| Medications | 7 | $3,638.0 | $2,217.9 | 83.7 | |
| Total cost | 7 | $4,344.3 | $2,180.0 | ||
| 10 | Hospitalization | 5 | $- | $- | 0.0 |
| Consultation | 5 | $29.0 | $19.5 | 1.0 | |
| Diagnostic imaging and laboratory | 5 | $212.6 | $118.8 | 7.5 | |
| Medications | 5 | $2,604.9 | $2,761.5 | 91.5 | |
| Total cost | 5 | $2,846.5 | $2,816.2 | ||
| 11 | Hospitalization | 3 | $- | $- | 0.0 |
| Consultation | 3 | $17.6 | $20.1 | 6.8 | |
| Diagnostic imaging and laboratory | 3 | $214.1 | $62.3 | 82.6 | |
| Medications | 3 | $27.6 | $47.8 | 10.7 | |
| Total cost | 3 | $259.2 | $103.8 | ||
| 12 | Hospitalization | 1 | $- | $- | 0.0 |
| Consultation | 1 | $13.2 | $- | 13.0 | |
| Diagnostic imaging and laboratory | 1 | $88.4 | $- | 87.0 | |
| Medications | 1 | $- | $- | 0.0 | |
| Total cost | 1 | $101.6 | $- | ||
| Promedio 10 años | Hospitalización | 8.0 | |||
| Consulta | 1.8 | ||||
| Gabinete and laboratorio | 8.4 | ||||
| Medicamentos | 82.7 |
The average of the first 10 years is reported because the information corresponding to years 11 and 12 primarily relates to patients in remission on conventional synthetic disease-modifying antirheumatic drugs, a situation that distorts the structure of cost components compared to the rest of the years.
Table 2 presents the median estimate and interquartile range (IQR) of patients who required medical attention from the institution for each component, total DMC, and the number of patients for each year (n). It is worth mentioning that because the population size is different for each component, it is not valid to attempt to sum the medians of each component to estimate the total DMC for each year; the latter is reported based on the median of the DMC variable. In the first 10 years of treatment, the total DMCs of patients with JIA have an average of USD 3,828 (SD = 1,492). For the different components, these presented the following costs: consultations USD 54.7 (SD = 17.3), imaging and laboratory studies USD 308.0 (SD = 52.0), hospitalization USD 2,348.5 (SD = 1,776.4), and finally medications USD 4,285.1 (SD = 2,008.4).
Table 2 Annual cost of treatment
| Year | Cost component | n | Median | P25 | P75 |
|---|---|---|---|---|---|
| 1 | Hospitalization | 4 | $1,519.8 | $301.6 | $4,132.3 |
| Consultation | 20 | $65.9 | $26.4 | $115.3 | |
| Diagnostic imaging and laboratory | 24 | $356.3 | $158.4 | $703.9 | |
| Medications | 18 | $148.8 | $24.2 | $2,132.8 | |
| Total cost | 26 | $509.5 | $182.6 | $2,909.9 | |
| 2 | Hospitalization | 5 | $1,196.9 | $221.7 | $2,686.0 |
| Consultation | 24 | $92.3 | $52.7 | $115.3 | |
| Diagnostic imaging and laboratory | 26 | $353.2 | $262.0 | $494.6 | |
| Medications | 24 | $3,468.6 | $207.7 | $7,598.7 | |
| Total cost | 26 | $3,824.1 | $782.7 | $7,676.7 | |
| 3 | Hospitalization | 3 | $4,373.9 | $100.2 | $5,975.3 |
| Consultation | 24 | $65.9 | $39.5 | $79.1 | |
| Diagnostic imaging and laboratory | 24 | $296.1 | $200.1 | $406.4 | |
| Medications | 23 | $3,971.0 | $99.5 | $12,410.3 | |
| Total cost | 24 | $4,535.4 | $380.9 | $12,032.0 | |
| 4 | Hospitalization | 3 | $2,658.1 | $1,792.5 | $4,816.3 |
| Consultation | 22 | $59.3 | $39.5 | $92.3 | |
| Diagnostic imaging and laboratory | 20 | $331.5 | $227.7 | $431.0 | |
| Medications | 20 | $5,144.2 | $542.1 | $9,852.6 | |
| Total cost | 22 | $5,673.8 | $48.0 | $8,747.7 | |
| 5 | Hospitalization | 3 | $2,259.0 | $2,215.5 | $2,468.3 |
| Consultation | 20 | $52.7 | $26.4 | $79.1 | |
| Diagnostic imaging and laboratory | 16 | $282.9 | $188.1 | $475.3 | |
| Medications | 19 | $3,087.6 | $2,266.8 | $6,029.1 | |
| Total cost | 20 | $4,174.8 | $9.9 | $6,161.2 | |
| 6 | Hospitalization | 2 | $1,622.5 | $610.7 | $2,634.3 |
| Consultation | 16 | $52.7 | $39.5 | $65.9 | |
| Diagnostic imaging and laboratory | 17 | $299.3 | $80.5 | $397.2 | |
| Medications | 13 | $7,496.5 | $1,632.8 | $8,935.8 | |
| Total cost | 18 | $4,589.0 | $- | $8,254.5 | |
| 7 | Hospitalization | 3 | $1,783.2 | $300.2 | $4,775.3 |
| Consultation | 12 | $46.1 | $13.2 | $79.1 | |
| Diagnostic imaging and laboratory | 12 | $251.2 | $91.1 | $441.3 | |
| Medications | 9 | $6,575.3 | $2,506.8 | $7,914.6 | |
| Total cost | 14 | $4,069.5 | $- | $4,923.4 | |
| 8 | Hospitalization | 1 | $6,285.9 | $6,285.9 | $6,285.9 |
| Consultation | 7 | $39.5 | $26.4 | $79.1 | |
| Diagnostic imaging and laboratory | 9 | $243.4 | $107.8 | $541.0 | |
| Medications | 6 | $4,716.5 | $2,961.2 | $7,462.4 | |
| Total cost | 9 | $4,277.5 | $169.0 | $208.6 | |
| 9 | Hospitalization | 1 | $1,786.3 | $1,786.3 | $1,786.3 |
| Consultation | 7 | $39.5 | $13.2 | $39.5 | |
| Diagnostic imaging and laboratory | 6 | $403.9 | $268.2 | $719.0 | |
| Medications | 6 | $4,600.0 | $2,511.3 | $5,833.7 | |
| Total cost | 7 | $4,666.7 | $3,186.1 | $6,088.1 | |
| 10 | Hospitalization | 0 | |||
| Consultation | 4 | $32.9 | $26.4 | $49.4 | |
| Diagnostic imaging and laboratory | 5 | $263.2 | $86.5 | $313.3 | |
| Medications | 4 | $3,642.3 | $562.1 | $5,564.2 | |
| Total cost | 5 | $1,966.1 | $297.9 | $5,835.3 | |
| 11 | Hospitalization | 0 | |||
| Consultation | 2 | $26.4 | $13.2 | $39.5 | |
| Diagnostic imaging and laboratory | 3 | $244.7 | $142.4 | $255.0 | |
| Medications | 1 | $82.9 | $82.9 | $82.9 | |
| Total cost | 3 | $294.6 | $142.4 | $340.8 | |
| 12 | Hospitalization | 0 | |||
| Consultation | 1 | $13.2 | $13.2 | $13.2 | |
| Diagnostic imaging and laboratory | 1 | $88.4 | $88.4 | $88.4 | |
| Medications | 0 | ||||
| Total cost | 1 | $101.6 | $101.6 | $101.6 |
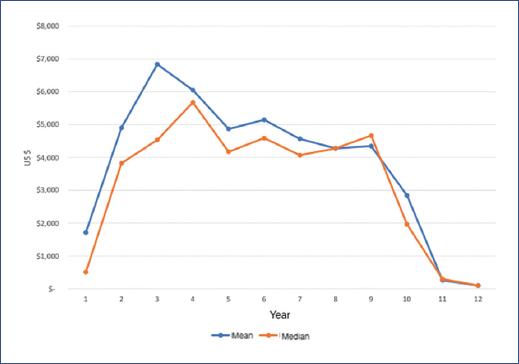
Figure 1 shows the evolution of total DMCs for each year of follow-up obtained with both estimates, which are relatively consistent in the behavior of DMCs. However, it can be seen that the first estimate (mean) tends to overestimate the cost results compared to the second estimate (median). From the 2nd year onwards, treatment DMCs increase, remaining relatively stable, only decreasing after the 9th year.
Analyzing each component over time, we observe that in the hospitalization component, during the 12 years of follow-up, only 10 of the 26 patients (38.46% of the total) used hospitalization services, with a total of 123 hospitalization events. Hospitalizations occurred only in the first 9 years, with a mean cost of USD 2,348.6 (SD = 1,776.4) and a median of USD 1,784.8 (1,510-3,087) for the first 10 years. Compared to the 1st year, an increasing cost trend was found, with some peaks in years 3 and 8 (Fig. 2A).
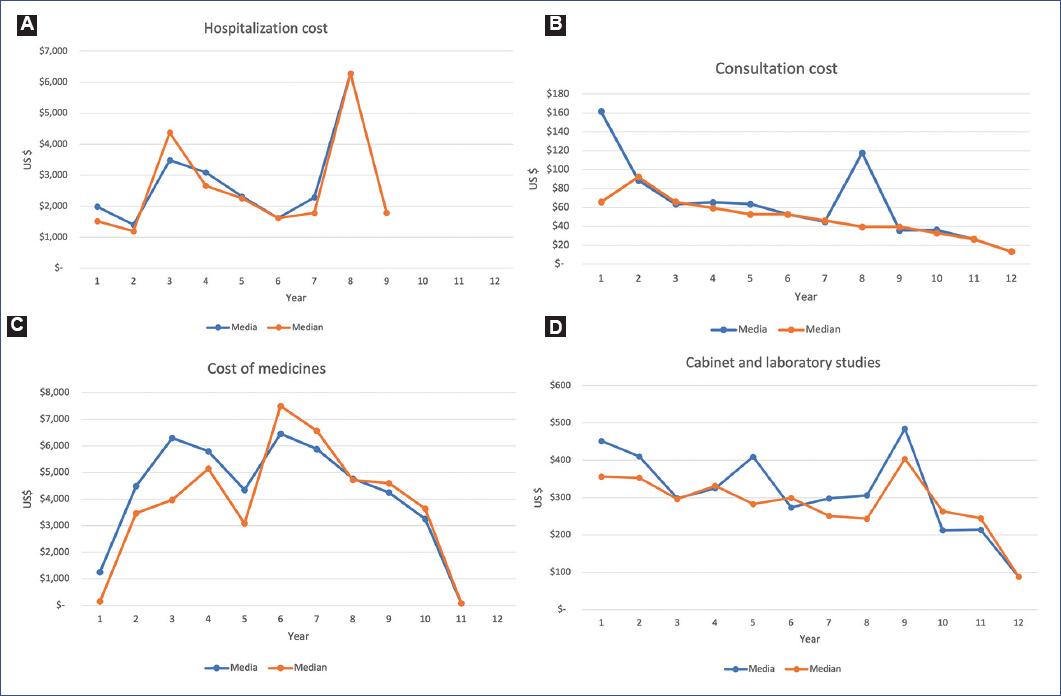
Figure 2 Evolution of the different cost components of polyarthritis. A: hospitalization. B: medications. C: consultations. D: cabinet and laboratory studies.
The medication component constitutes the highest cost within the total DMC. A total of 43 different medications were administered to the patients, with a median number of medications administered being 9 (IQR = 4). The mean cost was USD 4,285.1 (SD = 2,008.4), and the median was USD 4,285.5 (3,457.13-5,502.0) per patient per year during the first 10 years. We found that the cost of medications gradually increased during the first 6 years and subsequently decreased until reaching a minimum below the cost reported in year 1 (Fig. 2B).
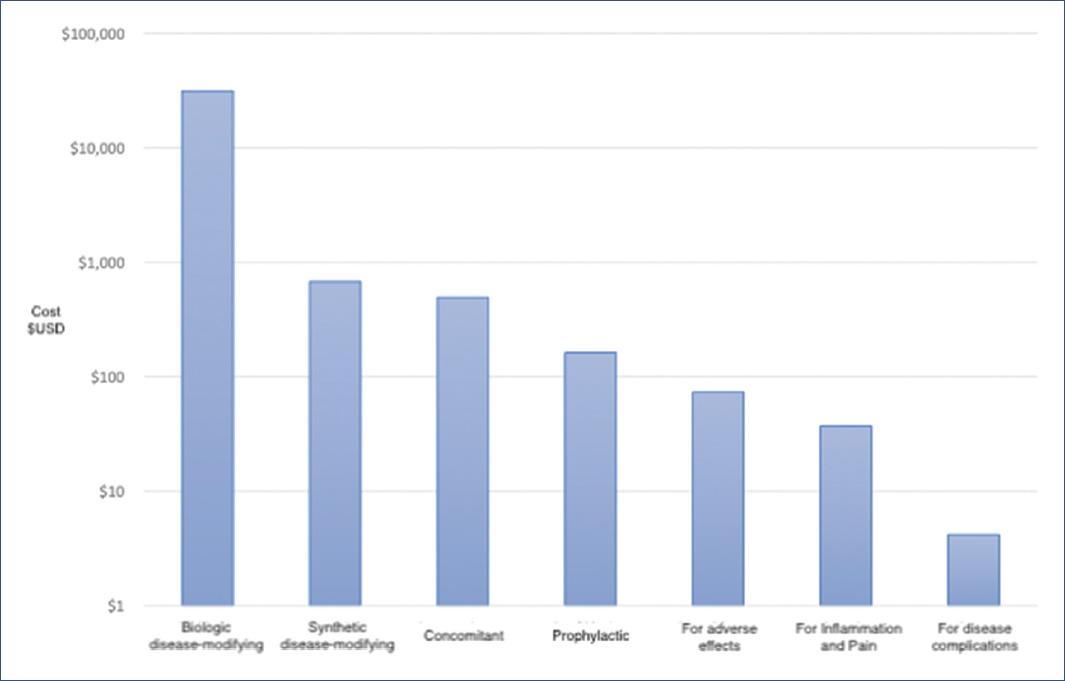
The medication groups from highest to lowest cost (percentage share) are biological disease-modifying drugs (abatacept, adalimumab, etanercept, secukinumab, and tocilizumab) representing 95.6%, followed by synthetic disease-modifying drugs (hydroxychloroquine, leflunomide, MTX, and sulfasalazine) representing 2.04%, concomitant medications (calcitriol, calcium carbonate/Vitamin D3, carboxymethylcellulose, ferrous fumarate, mycophenolate sodium, prednisone, polyethylene glycol/propylene glycol, and tobramycin) 1.7%, prophylactic treatments (folic acid, rifampicin, rifampicin/isoniazid, cholecalciferol, and Vitamin D) 0.5% of the total, pharmacotherapy for addressing adverse effects of other medications (esomeprazole and omeprazole), treatment for inflammation and pain (acetylsalicylic acid, diclofenac, etoricoxib, ibuprofen, naproxen, and acetaminophen) 0.1%, and medications for treating disease complications (alendronic acid and colchicine) (Fig. 3). According to clinical management, biological disease-modifying drugs are gradually incorporated from the 2nd year of treatment, which coincides with the pattern of cost increase for this component (Fig. 2B).
The consultation component represents the lowest DMC for the institution, with a mean of USD 54.7 (SD = 17.3) and a median of USD 52.7 (39.5-65.9) in the first 10 years of follow-up. However, this component shows an increase in the 2nd year (40%), a cost that subsequently decreases until reaching 20% of that reported in the initial year (Fig. 2C).
Finally, the diagnostic imaging and laboratory studies component presents a mean of USD 308.0 (SD = 52.0) and a median of USD 297 (262.8-353.9). In this component, we observe that costs decrease consistently over time; in the 12th year, the cost represented 25% of the initial year’s cost (Fig. 2D).
Discussion
There are no reports on the estimation of DMC for JIA of the polyarthritis subtype from the perspective of public health service providers in Latin America. This study represents one of the first efforts to estimate these costs and demonstrates that the medication component represents 82.7% of the total DMC for care in the first 10 years of these patients, with biological disease-modifying drugs accounting for 95.6% of the total cost.
In this study, medical records were retrospectively analyzed over 12 years; only the studies by Luca et al. 201624, Mars et al. 201925, and Kip et al. 202114 conducted cost follow-ups over long periods of 5, 9, and 10 years, respectively. The study by Kip et al. Only examined costs derived from the use of synthetic and biological disease-modifying drugs. In contrast, Luca et al. 201624 study included costs of consultations, laboratory tests, and hospitalizations. Other studies that analyzed the same cost components are those by Thornton et al. in 2008a, Thornton et al. in 2008b16,18, and Minden et al. in 200919, although the latter presents these components in a more disaggregated form. The duration of this study offers us a broader panorama of treatment throughout the disease and includes the estimation of the same components, so it can be considered to have a good design and allows comparability with these studies.
This study did not include estimates related to the rehabilitation process, as this stage of care is carried out in another hospital unit. Bernatsky et al. 200715 and Thornton et al. 2008a16 did include these types of costs without specifying the proportion relative to the total DMC. Only the study by Yucel et al. 201211 mentions that rehabilitation costs (physiotherapist) represent 0.5% of the DMC. While the lack of cost information for the rehabilitation component could be considered a weakness in the present study, as it represents a percentage of 0.5% of the cost reported in another study11, it should not affect the DMC estimation and distort the estimated relative weight of participation of the four components estimated here. Although the consultation component has the lowest participation within the DMC, it is a key component since the treatment of patients with JIA is mainly carried out on an outpatient basis, a period during which, in addition to clinical assessments of patients, the delivery and application of pharmacological treatments are performed.
Among the limitations of our study, it should be considered that in recent years, a smaller number of patients were observed compared to the initial years. Therefore, the results of the last 3 years could be considered less robust. When analyzing the information, it should be considered that the results are representative of the disease for the first 10 years of illness. It is worth noting that the two cases of patients with Remission on Conventional Synthetic DMARDs occur in years 11 and 12, which is why, in those years, there is a modification in the relative weight of resource use and DMC within the disease. Another limitation of the study is that the average costs/recovery fees reported by ISSEMyM mainly contemplate the variable costs of services or procedures offered by the institution, not the fixed costs. However, considering the conditions of most public institutions in Mexico, which generally, due to their age, already have depreciated equipment and infrastructure and considering the type of disease management, we believe that underestimating fixed costs in this case does not affect the estimated results.
For the present study, the median annual DMC per patient amounted to a mean of USD 4,555.2 (SD = 1,456.7) and a median of USD 3,828 (SD = 1,492) in the first 10 years of treatment. These costs are similar to those previously reported26. However, there are studies in which the reported costs are below those reported in the present work and preceding studies. One of these is the study by Ens et al. 2013, which reports costs well below those reported in the present study. Nevertheless, the work of this group was conducted from the family’s perspective, not from the service provider’s perspective. The study by Khatun et al.9 also reported lower costs, with annual DMCs of USD 193. In that study, few individuals used biological disease-modifying drugs; however, in cases where this medication was used, costs doubled, although they remained low despite this.
Another prospective study conducted in the United Kingdom in patients with polyarthritis, which compared the change in DMC during the first 6 months of treatment in 1989 and 10 years later, showed that the average DMC per patient was USD 1,003 in 1989 and almost doubled by 1999, with a cost of up to USD 2,578 (adjusted for exchange rate and inflation to 2021)27. Through the data from the present study, we identified that the median costs increase exponentially from the 1st year, with the 4th year being where the median cost is ten times higher than the 1st year. After that year, the median costs decrease gradually until reaching values similar to those obtained during the 1st year, between years 11 and 12. These data are similar to those reported by Nikiphorou et al. 201527, in which it is observed that as time passes, patients incorporate biological disease-modifying drugs into their treatment, thereby increasing the costs of the medication component. This coincides with what was observed in years 3 and 4 of our study and with other studies reporting that when this type of medication is incorporated, there is an increase in the cost of between 200%9 and 300%13 compared to the non-use of these biological drugs. In our study, an increase of 550% is observed between the 1st and 2nd years, and the total DMC increases by about 651%. However, the increase in the cost of the medication component is 2,327%, which is well above what was expected even when not compared to the value of the 7th year of treatment, where the maximum value is found (4,930%). This leads us to believe that this increase is due to the low prices of disease-modifying drugs (non-biological) during the 1st year or to the excessive increase observed in the cost of biological disease-modifying drugs from the 2nd year onward.
The pharmacological component represents a very high percentage (82.7%) of the total DMC, a figure that is very similar to that reported by Yucel et al. (85%)11. In other studies, this component represents a highly variable percentage, between 9 and 54%9,10,15,16,20,27. Considering that the total DMCs are relatively similar, we are inclined to think that this variability is due to an overpricing of medications in the Mexican health system.
Based on these results, we suggest replacing patented biological disease-modifying drugs with effective and safe generic medications that can generate savings for the health institution and expand current coverage. If this is not possible, public health institutions should conduct a detailed review of purchase prices from pharmaceutical companies.
According to other authors, the polyarticular clinical subtype of JIA is the most expensive among the different subtypes, followed by the polyarthritis and systemic arthritis subtypes19,20. These figures suggest that the estimated cost of polyarthritis could overestimate the average DMC of the disease if it were used for the rest of the JIA patient subtypes in Mexico, except the polyarthritis and systemic arthritis subtypes, according to the cost differences reported in other studies11,19,20,28.
We consider that future studies should make estimates for other JIA subtypes in the Mexican context. Likewise, it is suggested to make estimates of out-of-pocket expenses or indirect costs, which could be useful to estimate the cost of the disease from the user’s and/or society’s perspective. In addition, it would be interesting to include the costs and disease burden on caregivers. Finally, because the natural course of polyarthritis continues into adulthood, it would be important to estimate the costs over the lifetime of these patients.
Conclusion
Patients with JIA of the polyarthritis subtype at HMI presented an annual mean DMC of USD 4,555.2 (SD = 1,456.7) and a median of USD 3,828 (SD = 1,492) in the first 10 years of the disease.
The main component of DMC in the first 10 years of treatment was the medication component (82.7%), followed by diagnostic imaging and laboratory studies (8.4%), hospitalization (8.0%), and finally, the consultation component (1.8%).
The cost of biological disease-modifying drugs represented 95.6% of the DMC within the medication component and is reflected in the 2nd year of treatment. Therefore, it is important to include generic medications or review purchase prices by Mexican public health institutions from pharmaceutical companies.











 nueva página del texto (beta)
nueva página del texto (beta)