Introduction
Primary infection with the Epstein-Barr virus (EBV), also known as human herpesvirus 4, typically occurs in early childhood after exposure to the oral secretions of seropositive individuals following intimate contact. The virus spreads mainly through saliva and infects B cells and epithelial cells, persisting in the former for a lifetime. Infection activates both humoral and cellular immunity. CD8+ lymphocytes and NK cells play a crucial role in controlling infection1, as massive, atypical lymphocytosis can be seen in infectious mononucleosis2. Infectious mononucleosis has been considered the most common disease associated with EBV infection in adolescents and young adults. Mononucleosis is characterized by the production of heterophile antibodies, which are predominantly immunoglobulin (Ig) M. The detection of these antibodies in patients' serum is indicative of acute EBV infection3.
Although most EBV infected children are asymptomatic, some develop symptomatic disease. Clinical manifestations include typical and atypical presentations. Typical manifestations include infectious mononucleosis-like symptoms, such as pharyngitis, fever, fatigue, severe headache, general malaise, lymphadenopathy, and splenomegaly3-5. However, these manifestations can vary, ranging from mild to more severe cases with various complications. Atypical manifestations encompass a wide range of conditions, including acute dacryocystitis6, airway obstruction7, pneumonia8, acute myocarditis9, atherosclerosis10, thrombocytopenia, hemolytic anemia, agranulocytosis, hemophagocytic lymphohistiocytosis (HLH)11,12, neurological disorders13-18, hepatic involvement19, renal manifestations20, acute acalculous cholecystitis21, splenic rupture22, Kawasaki disease23, Henoch-Schönlein purpura24, velvety oral leukoplakia25, neoplasms26, psychiatric symptoms27, and more complex inflammatory and/or autoimmune diseases28. The prognosis varies depending on the clinical presentation, with most patients recovering within a few weeks. However, severe and chronic complications can occur, especially in cases of HLH, neurological involvement or neoplasia.
Reported laboratory findings in acute or chronic EBV infection often include leukocytosis, relative lymphocytosis, mild thrombocytopenia, and elevated levels of aspartate transaminase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH) and ferritin29. Diagnosis can be confirmed through the detection of heterophile antibodies, specific EBV antibodies (IgM and IgG), and viral load measurement. Treatment is primarily supportive, as EBV infections are usually self-limiting. However, antiviral treatment may be considered in cases with atypical symptoms or complications30-32, and steroids can be used to manage autoimmune and inflammatory symptoms. Rituximab, a monoclonal antibody targeting CD20, may be beneficial in lymphoproliferative disorders.
In the realm of pediatric virology in the hospital setting, symptomatic EBV infections are common; however, there is a notable gap in research regarding atypical manifestations. This study aims to describe the clinical manifestations and results of EBV infection in a tertiary pediatric hospital in Mexico. We identified the most common clinical manifestations delineating the differences between atypical manifestations of EBV infection and their typical counterparts in a hospitalized pediatric population. The results of this study will enrich our understanding of EBV infection in pediatric patients, especially within the Mexican population, thereby facilitating early recognition and competent management of atypical presentations.
Methods
Patients
An observational, transversal, retrospective, and descriptive study was conducted at the Hospital de Pediatría of Centro Médico Nacional Siglo XXI (CMNSXXI) of the Instituto Mexicano del Seguro Social. The study was approved by the local Health Research Committee (Institutional registration number R-2022-3603-048). Records of pediatric patients with positive EBV viral load results from January 2012 to December 2022 were collected. Patients without complete medical records were excluded from the study.
Quantification of EBV load
EBV loads were quantified by a reverse – polymerase chain reaction (RT-PCR) –based method using 400 μL of whole blood and using a commercial kit according to the hospital's routine protocol. Results are reported as the number of viral copies per mL.
Statistical analysis
Correlation analysis was performed using Spearman rank-based testing. In the correlation analysis between the hepatic enzymes and viral load, a p < 0.05 between a marker and any of the viral load measurements was the threshold to include that marker in the reported results. Logistic regression and other statistical analyses were performed using GraphPad Prism 6.
Results
A total of 68 patients exhibited positive viral load results indicative of EBV infection. Following strict inclusion criteria, 26 hospitalized patients were found to be eligible for our study, with the remainder excluded due to incomplete medical records. Age at diagnosis ranged from 1 to 14 years, with a notable concentration in the preschool and school-age demographic groups (Table 1). Males were predominant, representing 62% (n = 16) of the final cohort, with the majority from Mexico City. A diverse distribution across different regions of the country is also shown in table 1. Initial pediatric medical care accounted for 38.4% of patients, while the remainder sought care under different services such as hematology, oncology, immunology, surgery, and gastroenterology, consistent with the diverse symptomatic presentations observed at the time of diagnosis.
Table 1 General characteristics of patients with Epstein-Barr virus infection (n = 26)
| Variable | Medium | Min-Max |
|---|---|---|
| Age (years) | 5 | 1-14 |
| n | % | |
| Sex | ||
| Male | 14 | 53.8 |
| Female | 12 | 46.2 |
| Age group | ||
| Infants | 5 | 19.2 |
| Preschoolers | 8 | 30.7 |
| School-age | 8 | 30.7 |
| Adolescents | 5 | 19.2 |
| Place of origin | ||
| Mexico city | 16 | 61.5 |
| Chiapas | 3 | 11.5 |
| Guanajuato | 2 | 7.7 |
| Guerrero | 2 | 7.7 |
| Estado de México | 1 | 3.8 |
| Sinaloa | 1 | 3.8 |
| Tabasco | 1 | 3.8 |
| Comorbidities | ||
| Without comorbidities | 6 | 2 |
| With comorbidities* | ||
| Hepatic | 9 | 34.6 |
| Transplant | 7 | 26.9 |
| Aplastic anemia | 7 | 26.9 |
| Oncologic | 5 | 19.2 |
| Juvenile idiopathic arthritis | 2 | 7.7 |
| Chronic kidney disease | 2 | 7.7 |
| Severe combined immunodeficiency | 1 | 3.8 |
*Some patients had multiple comorbidities.
Among the patient cohort, 6 individuals (23%) were previously healthy, with no identified comorbidities before the EBV infection diagnosis. The remaining subjects presented with a spectrum of comorbidities, including oncological conditions such as four cases of leukemia and one of lymphoma, one of juvenile idiopathic arthritis, one severe combined immunodeficiency, and two cases of chronic kidney disease. Liver-related comorbidities were evident, with 8 individuals (31%) having acute hepatitis and 1 patient (4%) experiencing intrahepatic cholestasis. In addition, 6 patients (23%) had aplastic anemia. A history of transplantation was noted in 7 cases (27%). Regarding to other infections, 4 individuals (15%) were HIV-positive, 3 (11.5%) were diagnosed with cytomegalovirus (CMV) infection, and 1 patient (4%) had concurrent SARS-CoV-2 infection (Table 1 summarizes all these data).
Fever was the predominant symptom in 23 patients (85%), followed by lymphadenopathy in 9 cases (35%). Of note, only 2 patients (8%) presented with the classic triad of sore throat, lymphadenopathy, and fever, characteristic of infectious mononucleosis. Therefore, atypical symptoms were prevalent, including gastrointestinal abnormalities in 14 patients (54%), hepatic abnormalities in 11 patients (42%), splenomegaly in 6 patients (23%), HLH in 6 patients (23%), pneumonia in 3 patients (11.5%), and airway obstruction, renal abnormalities, and post-transplant lymphoproliferative disorder all in 1 patient each (4%). Leukopenia was observed in 5 patients (19%) and five patients had lymphocytosis (Table 1).
Analysis of viral load data revealed a range of 555 to 355,500 copies/mL, with a median of 2,826 copies/mL (Table 2 and Fig. 1). The most common biochemical abnormalities included liver function test abnormalities and elevated LDH levels. Specifically, 27% of individuals had elevated LDH, while 61.5% had elevated liver function test results. Six patients had missing LDH measurements and four had missing AST and/or ALT measurements (Table 2). We did not observe a significant correlation between the levels of LDH, AST and ALT and the EBV loads (Supplementary Fig. 1).
Table 2 Biochemical alterations in patients with Epstein-Barr virus infection
| Variable | Medium | Min-Max |
|---|---|---|
| DHL | 467.5 | 39.4-1083 |
| AST | 57.5 | 9.6-151.5 |
| ALT | 64.1 | 2.5-240.9 |
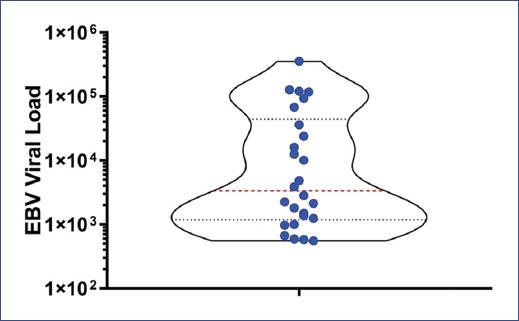
Figure 1 Distribution of Epstein-Barr virus viral load among the 26 patients included in the analysis. Viral load values are presented on a logarithmic scale to accentuate the variability observed between individuals. Each data point on the graph corresponds to the viral load of a specific patient.
The treatments given to patients are shown in table 3, the most common therapeutic approach was supportive management, given to 22 (85%) of the patients, 5 patients (19%) required specific antiviral treatment (acyclovir or a related drug), and 3 (11.5%) were treated with Rituximab. Regarding the total length of hospital stays for the studied patients, the median duration was 17.5 days, ranging from 7 to 32 days. Twenty-two (85%) patients were discharged home, while the remaining patients died during their course of illness due to the direct manifestations of EBV infection. Table 3 describes six patients who developed HLH secondary to EBV infection, with two of these unfortunate cases resulting in death. Additional details on the deceased patients, including comorbidities, atypical manifestations, and the direct cause of death, are summarized in table 4. While the EBV load in deceased patients ranged from a minimum of 500 copies/mL to a maximum of 120,000 copies/mL, we did not find a significant difference between the deceased patients and those who survived (Fig. 2). This lack of significance may be attributed to the small number of deceased patients in our sample.
Table 3 Characteristics of patients with HLH secondary to Epstein-Barr virus infection (n = 6)
| Patient | Year of diagnosis | Age (years) | Sex | Comorbidity | Viral load (copies/mL) | Therapy* | Outcome |
|---|---|---|---|---|---|---|---|
| 1 | 2019 | 12 | Male | Acute lymphoblastic leukemia | 36,270 | Antiviral | Discharge |
| 2 | 2020 | 1 | Male | Juvenile idiopathic arthritis/chronic kidney disease | 594 | Steroids | Deceased |
| 3 | 2019 | 8 | Male | None | 584 | None | Discharge |
| 4 | 2021 | 6 | Female | None | 973 | None | Discharge |
| 5 | 2020 | 1 | Female | SARS-CoV2 infection and aplastic anemia | 2,826 | Rituximab | Deceased |
| 6 | 2020 | 10 | Male | Severe combined immunodeficiency | 117,900 | Antiviral/Rituximab | Discharge |
*Refers to any treatment besides the supportive care.
Table 4 Characteristics of patients who died with complications secondary to EBV infection (n = 4)
| Patient | Age (years) | Sex | Comorbidity | Atypical Manifestations | Viral Load (copies/mL) | Treatment | Cause of death |
|---|---|---|---|---|---|---|---|
| 1 | 1 | Male | Juvenile idiopathic arthritis/chronic kidney disease | HLH | 594 | Steroids | Septic Shock |
| 2 | 1 | Female | Sars-Cov2 coinfection/aplastic anemia | HLH/Acute Renal Lesion | 2,826 | Rituximab | EBV |
| 3 | 13 | Male | Chronic hepatitis | Pneumonia | 12,510 | Steroids/Antiviral | Septic and hypovolemic Shock |
| 4 | 3 | Male | Limphoma/CMV coinfection/transplant | LPD | 121,000 | None | Septic and Hypovolemic Shock |
HLH: hemophagocytic lymphohistiocytosis; LPD: lymphoproliferative disease; CMV: cytomegalovirus; EBV: Epstein-Barr virus.
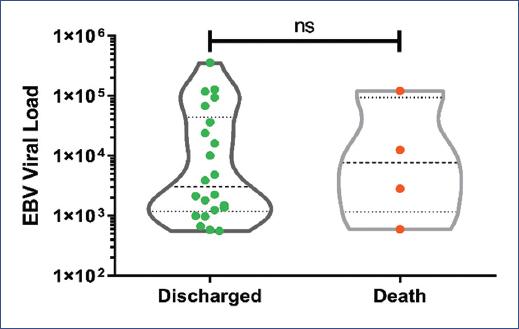
Figure 2 Comparison of Epstein-Barr virus load in patients who succumbed to the infection with those who were discharged. The viral load values were logarithmically scaled, with each data point representing an individual patient. Mann–Whitney U test for non-parametric data, without normal distribution, statistical significance was determined with a p = 0.05.
Discussion
While the majority of patients in this study presented with a self-limiting clinical course and were discharged home in good condition, we observed four fatalities (15%) directly linked to EBV infection. It is noteworthy that all four of these patients had severe comorbidities coexisting with the positive EBV load, such as HLH, lymphoma, pneumonia, and renal disease. This suggests that patients with compromised immunity have a more severe disease course. Three of the deceased patients had higher than 2000 copies/mL. We found eight patients coinfected with other agents at the time of EBV diagnosis, including four with HIV, three with CMV, and one with SARS-CoV-2. This latter patient was one of those who died due to this coinfection. Singh et al. found that among viral infections associated with SARS-CoV-2, EBV had the highest incidence, without knowing the exact cause of this association33. Moreover, SARS-CoV-2 has the ability to reactivate EBV34.
Our study revealed a diverse spectrum of both typical and atypical clinical manifestations in pediatric patients with EBV load detection. Notably, these manifestations frequently correlated with underlying comorbidities within our study cohort, underscoring the imperative role of contextual clinical assessment when faced with EBV infection in children. Surprisingly, the classic triad of symptoms conventionally associated with infectious mononucleosis -pharyngitis, adenomegaly, and fever- was present in only a minority of cases. This observation underscores the critical need to consider EBV infection in individuals with complex underlying pathologies or compromised immune status, even when the presentation deviates from the typical infectious mononucleosis pattern. In our study, a variety of atypical manifestations of EBV infection was observed, so diverse that they are difficult to distinguish from other pathologies35. The atypical manifestation ranges from renal abnormalities, airway obstruction, bicytopenias, pancytopenias, and the development of HLH. HLH is a syndrome that usually occurs in patients with severe systemic hyperinflammation, which includes persistent fever, cytopenias, hepatosplenomegaly, coagulopathies, and elevated ferritin36. A study conducted in Japan in 2007 reported 277 cases of HLH, of which 130 (46%) had EBV as the causative agent37. We can observe that out of the six patients who developed HLH in our study, two died, with one receiving treatment with steroids and the other with rituximab. Chellapandian et al., conducted a study to evaluate the treatment with rituximab in pediatric patients with EBV-positive HLH, in which an immediate improvement in the signs and symptoms of HLH was observed38. In our study, rituximab was used in only three patients, of whom two were diagnosed with EBV-induced HLH. Unfortunately, one of these patients died despite the treatment.
While the majority of patients received primarily supportive care, a variety of treatment modalities was implemented on a case-by-case basis. Supportive care was aimed at alleviating symptoms, while some patients underwent more targeted therapies, including the administration of rituximab. In addition, three patients were treated with steroids. Corticosteroids, known to alleviate symptoms such as fever, lymphadenopathy, and oropharyngeal manifestations, are beneficial in atypical presentations or when associated with severe atypical symptoms such as airway obstruction, acute hemolytic anemia, and significant cardiac or neurologic involvement30. However, the specific reasons for prescribing steroids and rituximab, as well as the discernible benefits of their use, were not explicitly documented in the patients' medical records.
A notable finding was the lack of a clear association between EBV viral load and patient outcomes. Although some patients had elevated viral loads, the majority experienced favorable clinical outcomes, while others with lower viral loads succumbed to more severe disease. This underscores the critical role of factors beyond viral load, such as a patient's immune status and underlying comorbidities. This finding is consistent with previous research highlighting the multifaceted nature of EBV and its direct capacity to cause damage or complicate other underlying conditions.
EBV infection in pediatric populations has been the subject of various studies worldwide, shedding light on its diverse clinical presentations and outcomes. García-Peris et al., conducted a study in Madrid, Spain, involving 103 children39. They reported that 63% of the children exhibited typical mononucleosis symptoms, while the remaining 37% presented with atypical manifestations, all with favorable outcomes. These findings also underscore the diverse clinical spectrum of EBV infection, although the frequency of typical mononucleosis symptoms was higher than in our population. Examined 61 pediatric patients in Shanghai and found typical symptoms in the majority, but a significant portion exhibited atypical symptoms, including respiratory (in 31% of cases), urinary (14.8%) and hematological (14.8%) manifestations5. Similarly, Jeon et al. found typical symptoms as the most frequent, reporting fever, lymphadenopathy, liver involvement and various cytopenias in their study of 38 EBV-infected children in Korea40. In the Mexican context there is a lack of comprehensive studies addressing the atypical manifestations of EBV infection in pediatric patients. Napoleón et al. conducted a study spanning four decades and reported lymphadenopathy as the most common symptom. Although they documented some atypical manifestations, no fatalities were reported41. Lozano et al. described clinical manifestations in seven patients with EBV infection, primarily presenting typical symptoms (faringitis, esplenomegalia, hepatomegalia, and linfoadenopatias y fiebre)42.
The present study underscores the need to recognize the diverse clinical presentations of EBV infection in pediatric cohorts. Our findings are consistent with the global literature and reinforce the concept that EBV infection should be considered in children presenting with a broad spectrum of symptoms. In particular, our research reveals an increased prevalence of atypical manifestations beyond the typical symptoms associated with infectious mononucleosis. Examples include juvenile idiopathic arthritis or aplastic anemia, each observed in 23% of patients. Thus, EBV should be considered as a potential contributor to a variety of diseases, particularly in patients with underlying comorbidities or immunosuppression. Furthermore, the lack of a discernible correlation between viral load and outcome underscores the multifaceted nature of EBV-related disease and emphasizes the need for thorough patient evaluation and a comprehensive management approach.
In conclusion, this study adds to the evolving body of knowledge regarding EBV infection in pediatric patients and highlights the ongoing need for research in this area. Future investigations should address novel treatment modalities, especially in complicated cases with comorbidities or severe multisystemic involvement. Our study, conducted in a tertiary hospital specializing in severe pediatric pathologies, provides valuable insights into the peculiar clinical characteristics of EBV infection in this specific population.
Study limitations
Our study, while providing insights into EBV infection in pediatric patients, has several limitations. Firstly, its observational nature precludes establishing causality between EBV infection and clinical outcomes, given the associative study design. Although viral load measurement is informative, it alone does not fully predict clinical outcomes, which are likely influenced by multiple factors such as underlying diseases or coinfections besides EBV activity. The heterogeneity of the patient population, drawn from various hospital services without standardized treatment guidelines, introduces variability in clinical presentation, interpretation, and management. Moreover, incomplete patient records may have biased our findings, potentially underestimating certain parameters. Despite these limitations, our study underscores the need for further research to better understand EBV infection in pediatric cohorts.











 nueva página del texto (beta)
nueva página del texto (beta)


