Introduction
Cardiovascular (CV) disease is the leading cause of morbidity and mortality worldwide. The current global health policy goals include a 25% reduction in premature mortality from non-communicable diseases by 20251.
There is a strong association between respiratory infections and acute CV events. All strains of influenza and Streptococcus pneumoniae infections can trigger a variety of CV alterations that may lead to hospitalization or death2-6.
Evidence has shown that influenza vaccination (IV) and pneumococcus vaccination (PV) are associated with a reduction in the rate of several CV outcomes, such as myocardial infarction (MI), heart failure (HF) hospitalization, and CV mortality7-17. However, several challenges have been identified in the implementation of these prevention strategies. These challenges include patient decisions (vaccine hesitancy, previous experience, and misinformation), healthcare providers' knowledge and attitudes toward vaccination, and healthcare system barriers. All these factors contribute to lower-than-expected immunization rates in Latin America and globally18-28.
Despite the efforts by the World Health Organization, government authorities, and health leaders in most countries to encourage compliance with vaccination recommendations, uptake remains low. At present, there is no accurate information on vaccination rates among patients undergoing primary or secondary prevention of cardiometabolic diseases in the Americas29.
The aim of this study is to determine the rates of vaccination against influenza and pneumococcal disease in the general population of the Americas, both healthy and sick, and to analyze the factors influencing these rates.
Methods and design
Study population
The registry for this study is observational. A total of 19 countries will be prospectively enrolled in the Inter-American Vaccination Registry of Influenza and Pneumococcus (CorVacc Study). Eligibility for the registry includes ambulatory patients over 18 years of age who provide their informed consent to participate in the survey. Patients under the age of 18 years and those unable to provide informed consent will be excluded from the study.
Informed consent
Patients will be informed about the survey's objective and the anonymity of their responses. No identifiable personal data will be collected. Ethics approval was obtained from the Inter-American Society of Cardiology (SIAC) Research Ethics Board.
Study design
A cross-sectional online survey comprising 34 questions will be developed using Google Forms (Mountain View, CA). The research team will invite patients to complete the questionnaire in person or through email, social media, telephone calls, and paper questionnaires.
The survey will be divided into three sections: (i) questions that examine a patients demographic profile; (ii) questions that examine their CV risk profile; and (iii) questions that examine their vaccination profile.
The questionnaire consists of a variety of question types, including dichotomous, Likert-type, multiple-option, and open-ended response choices. The respondents will not be obligated to provide a response and they may select multiple responses, in accordance with the content of the question.
Study organization
A call for cardiologists and other physicians from Latin American countries to join a team of collaborators for the CorVacc Study was conducted. A total of 19 countries were represented by cardiologists who registered to participate in the study. The countries will be divided according to a prespecified geographic distribution (Fig. 1), as follows:
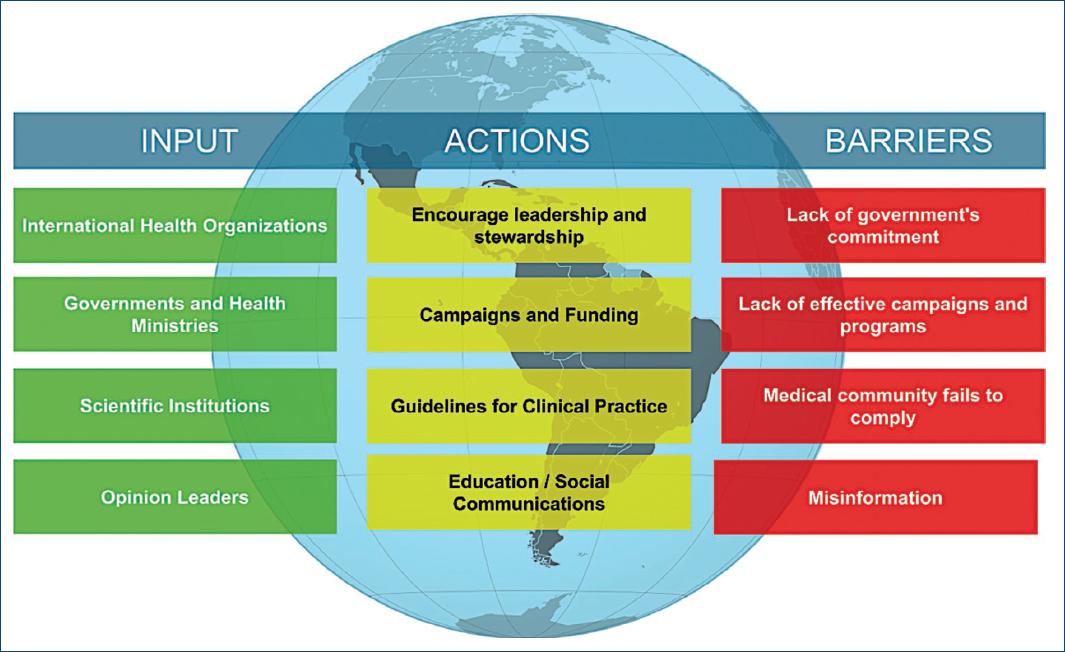
Figura 1 Key factors influencing immunization rates, actions for improvement and barriers encountered.
−North, Central, and Caribbean region: Mexico, Guatemala, El Salvador, Honduras, Nicaragua, Costa Rica, Panama, Cuba, Puerto Rico, and Dominican Republic
−Andean region (AR): Venezuela, Colombia, Ecuador, Bolivia, and Peru
−Southern Cone region: Paraguay, Chile, Uruguay, Brazil, and Argentina.
The total population within Latin American countries is estimated to be approximately 662 million. A sample size of 20,000 surveys was calculated to achieve a margin of error of 5%, a confidence level of 90%, and an error of distribution of 50%.
Survey
The survey platform will be made for enrolling patients. To enhance the response rate, periodic reminders will be sent through mail, text message, or social media. In addition, information about the progression of surveys by country will be made available. The survey is comprised of 34 questions (Table 1), which address the following topics:
Table 1 Description of the questions included in the survey
| Questions | Possible responses |
|---|---|
| Region | North, Central America, and the Caribbean |
| Andean | |
| Southern cone | |
| Age | |
| Gender | Female |
| Male | |
| Transgender | |
| Other | |
| What perception do you have of your state of health? | Scale of 1-10 (Unhealthy-very healthy) |
| Number of inhabitants city/town of residence | < 10,000 population |
| 10,000-100,000 population | |
| 100,000-500,000 population | |
| > 500,000 population | |
| Don't know | |
| Marital relationship | Married |
| Divorced | |
| Single | |
| United to | |
| Widower | |
| None of the above | |
| Work relationship | Dependent asset |
| Independent asset | |
| Retired/pensioned/retired | |
| Independent worker | |
| Irregular worker | |
| None of the above | |
| Education level | None |
| Primary | |
| Secondary | |
| University | |
| Master's degree | |
| If you are a professional, could you describe your occupation? | Lawyer |
| Business Administration | |
| Architect | |
| Engineer | |
| Entrepreneur | |
| Finance | |
| Doctor | |
| Marketing/Advertising | |
| Business | |
| Health area personnel | |
| Others | |
| Economic Income in US dollars per month | < 499 |
| 500-999 | |
| 1000-1499 | |
| 1500-1999 | |
| 2000-2499 | |
| 2500-2999 | |
| > 3000 | |
| Access to health services | Private with direct payment |
| Private with medical insurance | |
| Social security | |
| Public health service | |
| None | |
| What chronic diseases do you have? | Cancer |
| Mellitus diabetes | |
| Diseases of the immune system | |
| Psychiatric mental illness | |
| Psychological | |
| Chronic lung diseases | |
| Renal disease | |
| Fatty liver | |
| Arterial hypertension | |
| Hypothyroidism | |
| Polycystic ovary | |
| Cholesterol and/or triglyceride problems | |
| Overweight/Obesity | |
| Smoking | |
| None of the above | |
| Have you been diagnosed by a doctor with the following diseases? | Angina pectoris |
| Arrhythmias | |
| Disease of the arteries of the lower limbs and/or aorta | |
| Stroke | |
| Myocardial infarction | |
| Heart failure | |
| None of the above | |
| Have you had any of the following procedures? | Cardiac catheterization with stent placement in any coronary artery |
| Cardiac surgery for heart valve prostheses | |
| Bypass surgery or revascularization of the coronary arteries | |
| Lower limb vascular surgery | |
| Pacemaker | |
| None of the above | |
| For his chronic illnesses, I take my medications | Every day without fail, at the corresponding time |
| Every day without fail, but not at the corresponding time | |
| I remember from time to time | |
| I forget from time to time | |
| I don't take them | |
| I don't need medication | |
| How many medications do you take? | 1 |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| > 5 | |
| None | |
| I don't like taking medications | |
| Please select your medication scheme | Single-drug pills |
| One pill with two medications | |
| One pill with three medications | |
| They told me it's called Polypill | |
| None of the above | |
| Have you had the flu? | Yes |
| No | |
| Don't know | |
| Have you had pneumococcus? | Yes |
| No | |
| Don't know | |
| How confident are you in the influenza vaccination? | Scale 1-10 (Not at all confident-very confident) |
| How confident are you in the pneumococcus vaccination? | Scale 1-10 (Not at all confident-very confident) |
| How often do you get the influenza vaccine? | Annually in season |
| Every 2 years | |
| Every 3 years | |
| When I remember | |
| From time to time | |
| I don't put it on | |
| Have you had a pneumococcus vaccine? | Yes |
| No | |
| Who prescribes the vaccines? | General physician |
| Internal medicine | |
| Family medicine | |
| Cardiologist | |
| Endocrinologist | |
| Geriatrician | |
| Gynecologist | |
| Pulmonologist | |
| Neurologist | |
| Pediatrician for my children/grandchildren | |
| Others (Gastroenterologist, Rheumatologist, etc.) | |
| I prescribe myself | |
| None of the above | |
| What health service does vaccination provide you? | Public |
| Private | |
| Social Security | |
| Annual vaccination campaign in my country | |
| I buy it at the pharmacy without prescription | |
| None of the above | |
| How much do you know about the cardiovascular benefits of vaccination? | Scale 1-10 (I don't know them-I know them) |
| If you are not a doctor, do you suggest other vaccinations? | Yes |
| No | |
| Does not apply | |
| If you are a doctor, do you indicate vaccination for Influenza and Pneumococcus? | Yes |
| No | |
| Does not apply | |
| Do you know when the Influenza season is in your country? | Yes |
| No | |
| Are there vaccination campaigns for Influenza and Pneumococcus in your country? | Yes |
| No | |
| Don't know | |
| Have you been vaccinated for Covid-19? | Yes |
| No | |
| Number of doses given | 1 |
| 2 | |
| 3 | |
| 4 | |
| 5 | |
| None | |
| All necessary | |
| What type of vaccine do you use? | Abdala (Cuban) |
| Moderna | |
| J and J/Janssen | |
| Oxford/Astra Zeneca | |
| Pfizer/BioNTech | |
| Sinopharm (China) | |
| Sinovac/Biotech (China) | |
| Sputnik 5 (Russia) | |
| None | |
| How confident are you in the COVID-19 vaccination? | Scale 1-10 (Not at all confident-very confident) |
Population demographics
It is crucial to ascertain the factors that may influence behavior, such as age and gender. It is important to characterize the country in which patients reside, considering factors such as income, education, and health coverage. There are significant differences between the three regions with regard to environmental and cultural factors. Furthermore, some countries have experienced migratory flows, particularly from Europe, resulting in ethnic differences, in contrast to other countries with larger native and black populations.
Socioeconomic status and education level
The Prospective Urban and Rural Epidemiological study demonstrated that socioeconomic status is associated with differences in risk factors for CV disease incidence and outcomes, including mortality30. According to this study, individuals with a lower level of education in low-and middle-income countries, such as the Latin American community, experience higher incidence and mortality rates from CV diseases30. Factors such as access to health services, treatments, and vaccination against influenza and pneumonia are crucial for understanding the true situation of patients with cardiometabolic diseases.
Cardiometabolic profile
Patients will be characterized by their history of cardiometabolic diseases, and the time elapsed since their last admission to hospital due to CV events. It is well established that individuals who have survived a MI or ischemic stroke, particularly those with diabetes, are at higher risk of subsequent CV events. These patients would potentially benefit from intensified treatment and comorbidities management31.
Medical controls and treatments
Suboptimal adherence to medications prescribed chronically for secondary prevention of CV and metabolic diseases continue to burden the healthcare system, despite the well-established prevention benefits of adherence32. Potential barriers to optimal adherence include the number of pills per day, access to healthcare systems, and the availability of drugs and medical prescriptions33. Therefore, evaluating the percentage of the population receiving treatment according to medical recommendations is extremely valuable. Furthermore, assessing adherence to recommended immunizations against influenza and pneumococcus is crucial, as vaccines are particularly indicated in this group of patients33.
Discussion
In 2021, the SIAC published an article evaluating the status of cardiometabolic patients without COVID-19 infection during the pandemic, including their immunization profile against influenza and pneumococcus34. A total of 4216 subjects were included in the study, where the mean age was 60 (± 15) years, and 49% of patients were females. The study population comprised of 1764 patients (42%) aged 65 years or above, 899 patients (21.3%) with diabetes, and 606 patients (14.3%) who were current or former smokers. A total of 769 (18.2%) patients had a known history of computer-aided design, while 538 patients (12.7%) had a history of HF. The global IV rate was 46.5% (n = 1963), PV 24.6% (n = 1039), and double vaccination rate was 21% (n = 887)29.
The immunization rate was found to be lower than expected for this population. Furthermore, no significant sex differences in vaccination rates were identified by the authors35.
In the same cohort, the impact of different variables on immunization rates was analyzed, including geographic region. The vaccination rates of patients from the Southern cone (Argentina, Paraguay, and Chile) were approximately double those of patients from the tropical regions. The IV rate was 69% in the Southern cone, 34% in AR (Peru, Ecuador, Colombia, and Venezuela), and 35% in Central America and the Caribbean (Costa Rica, Cuba, El Salvador, Guatemala, Mexico and Dominican Republic) (χ = 452, df = 2, p < 0.001). The rates of PV were 43%, 20%, and 11%, respectively (χ = 406, df = 2, p < 0.001). The rates of double vaccination were 40%, 11%, and 9%, respectively (χ2 = 458, df = 2, p < 0.001)36.
In this context, several variables influence the decision to be vaccinated or not in the continent. These variables are presented in figure 2.
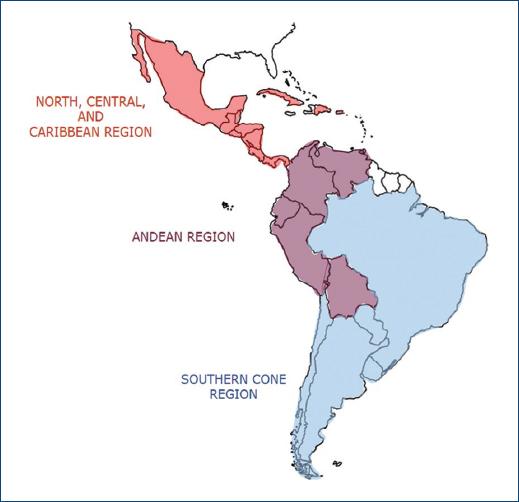
Figure 2 Distribution of participating countries by region. North, Central, and Caribbean region (NCC): Mexico, Guatemala, El Salvador, Honduras, Nicaragua, Costa Rica, Panama, Cuba, Puerto Rico and Dominican Republic. Andean region (AR): Venezuela, Colombia, Ecuador, Bolivia and Peru. Southern Cone region (SCR): Paraguay, Chile, Uruguay, and Argentina.
Despite the advantages associated with IV and PV, the recommendations for its prescription by scientific societies and health regulatory agencies, the vaccination rates globally, as well as in the Americas, appear lower than expected. This phenomenon can be attributed to the existence of implementation barriers affecting doctors, patients, and the broader health system. Recognizing these barriers is crucial for developing strategies to achieve vaccination targets37.
A thorough understanding of these implementation barriers, which involve doctors, patients, and their context, is essential when designing continuous improvement strategies. The current and unavoidable challenge for our scientific societies is to turn recommendations into action37.
The Pan-American Health Organization urges the use of scientific evidence to guide decision-making and program implementation to improve immunization rates. In this context, the present study will help advocate for Latin America and enhance vaccination coverage for influenza and pneumococcus, thereby improving the CV prognosis of patients.
Conclusion
This study will provide insight into the impact of influenza and pneumococcus vaccinations in Latin American populations and the barriers preventing the immunization targets from being actualized. Hopefully, this will help to facilitate the development of targeted and focused health prevention strategies.











 nueva página del texto (beta)
nueva página del texto (beta)


