Introduction
Acute myocardial infarction (AMI) stands as a prominent global cause of mortality. Despite advancements in prompt reperfusion and pharmaceutical treatments, patients continue to face considerable risks of morbidity and mortality post-discharge. While serial cardiac troponin tests are crucial for AMI differentiation, they come with various limitations1-3. Moreover, cardiac troponin levels may not accurately reflect the severity of coronary artery disease (CAD). Thus, identifying dependable biomarkers remains pivotal for early AMI detection and risk stratification4,5.
Osteoprotegerin (OPG), the receptor activator of nuclear factor-kβ (RANK-kβ) (RANK and its ligand [RANKL]), constitute a crucial system in bone remodeling and several bone disorders6. OPG is expressed in multiple tissues other than bone, including the vascular endothelium, and it appears to be stored within WeibelPalade bodies together with P-selectin and von Willebrand factor7. It is released with these mediators after stimulation with tumor necrosis factor α and interleukin-1 beta. OPG also contributes to vascular injury, inflammation, and atherosclerosis. In fact, atherosclerosis and vascular calcification are active processes that also involve this so-called bone-vessel axis6. Prior studies have confirmed OPG’s association so-called with acute coronary syndrome (ACS) severity and prognosis8.
Another essential molecule in this bone-vessel system is osteopontin (OPN). Although it does not form part of the OPG/RANKL/RANK axis, OPN is a highly versatile cytokine with complex biological functions, including roles in inflammation, immune responses, wound healing, cell adhesion, cell migration, and cell survival9-13. Some studies indicate that the overexpression of OPN favors atherosclerosis and other cardiovascular conditions12,13. For example, OPN is a strong predictor of outcomes in patients with calcified aortic valve disease and ischemic vascular pathologies, including stroke, myocardial infarction, and peripheral arterial disease11,13. Finally, recent studies of microRNA have found associations between OPN and NT-proBNP levels in ACS.
Percutaneous coronary intervention (PCI) with drug-eluting stents is currently considered as the standard treatment in patients with significant CAD14. The SYNergy between percutaneous study coronary intervention With TAXus and cardiac surgery (SYNTAX), published more than 10 years ago, changed the paradigm of modern cardiology15-17. The SYNTAX score serves as a commonly utilized risk stratification tool, although with the drawback of requiring an invasive procedure and being ascertainable only after angiography. The primary objective of this study is to ascertain the levels of OPG and OPN, alongside osteoprotegerin/RANKL ratio (ORR), and assess their association with the SYNTAX score and other pertinent clinical indicators. The aim is to ascertain the potential of these molecules as predictive markers for risk, aiding in risk stratification. Eventually, they could potentially be employed even before angiography to gauge the severity of coronary lesions.
Materials and methods
This was a cross-sectional, unicentric, and single-blinded study. We enrolled adult patients diagnosed with CAD who presented to the ER with acute chest pain and demonstrated clinical and electrocardiographic evidence of ischemia. In addition, these patients underwent coronary angiography confirming significant CAD necessitating either PCI or coronary artery bypass grafting. Exclusion criteria encompassed pathologies hindering revascularization treatment: hemorrhagic diatheses, active infectious or neoplastic conditions, and pregnancy. The institutional ethics committee board approved the study (protocol ID number 18-1067). The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The participants signed written informed consent forms.
The study comprised three primary groups: (1) patients with ACS who underwent PCI following documented ischemia as per inclusion criteria (ACS-PCI), (2) individuals without ACS who underwent coronary angiography for reasons other than ischemia but did not receive PCI (non-ACS without PCI), and (3) a control group consisting of asymptomatic individuals, specifically healthy blood donors, who did not undergo coronary angiography.
Collection and processing of samples (OPG and OPG)
A blood sample was collected by venipuncture from patients and controls; the plasma fraction was isolated in aliquots and stored at −70°C until required for biochemical analysis. In the case of ACS-PCI and non-ACS patients, blood sample collection was taken before the coronary angiography or PCI.
Detection of OPG, OPP, and ORR (OPG/RANKL)
Levels of OPP and RANKL were measured by ELISA (R&D Systems, Minneapolis, MN) according to the protocols suggested by the manufacturer. Furthermore, the levels of OPG were measured by Bio-Plex Multiplex Immunoassay (Bio-Rad Laboratories, California, USA) according to the manufacturer’s protocols. ORR is simply the arithmetic division of OPG by its ligand (RANKL) for value normalization purposes.
SYNTAX score calculation
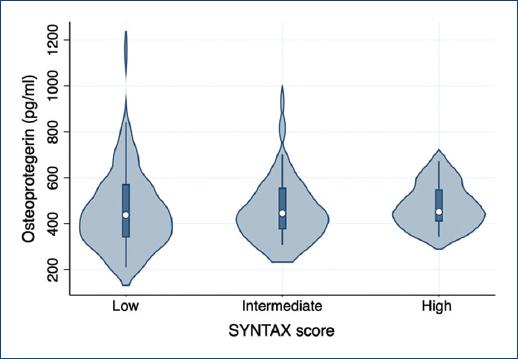
Following left heart catheterization, angiographic images were assessed by a cardiologist not affiliated with the study. The SYNTAX score was computed using the official website of the Syntax Score Task Force (http://www.syntaxscore.org/calculator/start.htm)18. Patients were categorized according to the classifications outlined in the original SYNTAX study: low (0-22), intermediate (23-32), and high scores (≥ 33).
Statistical analysis
Continuous variables were expressed as mean with standard deviation, and categorical variables were expressed as frequencies and percentages. The normality of the variables was evaluated using the ShapiroWilk test, and those variables with normal distribution were analyzed with parametric tests (Student’s t-test). In contrast, non-parametric tests were used to test variables without normal distribution. The association between OPG and OPN levels and the patients’ demographic variables was investigated with the χ2 test for categorical variables. According to their distribution, continuous variables were contrasted with the analysis of variance or the Kruskal-Wallis (H) test. Multivariate logistic regression analysis was performed, including variables associated with OPG levels; results are presented as relative risk (RR) with 95% confidence intervals (95% CI). Statistical analysis was performed with Stata version 16.0 software (StataCorp LLC).
Results
The study included 147 participants: 46 were women (31%) and 101 men (69%) with a mean age of 60 years. Thirty eight of the patients with ACS did not have dyslipidemia. However, a negligible number of patients (4) were taking statins before the study. The baseline characteristics of the three groups are summarized in table 1. Levels OPG, OPN, and ORR are displayed in table 2.
Table 1 Patient baseline characteristics
| Features | Total (n = 147) | ACS-PCI (n = 47) | Non-ACS without PCI (n = 50) | Control group (n = 50) | p* |
|---|---|---|---|---|---|
| Male, n (%) | 69 | 83 | 60 | 64 | 0.0001 |
| Age, years | 60 ± 13 | 64 ± 9 | 60 ± 15 | 58 ± 15 | 0.07 |
| BMI, kg/m2 | 27.25 ± 4.07 | 27.44 ± 4.19 | 26.76 ± 4.25 | 27.57 ± 3.82 | 0.5 |
| DM, n (%) | 38 (26) | 20 (43) | 16 (32) | 2 (4) | 0.0001 |
| HT, n (%) | 71 (48) | 28 (60) | 25 (50) | 2 (3) | 0.293 |
| TC, mg/dL | 154 ± 45 | 141 ± 40 | 140 ± 43 | 178 ± 41 | 0.0001 |
| LDL, mg/dL | 91 ± 34 | 85 ± 33 | 86 ± 36 | 102 ± 29 | 0.01 |
| HDL, mg/dL | 37 ± 18 | 34 ± 13 | 40 ± 13 | 38 ± 9 | 0.07 |
| TG, mg/dL | 145 ± 81 | 154 ± 93 | 117 ± 41 | 163 ± 92 | 0.1 |
| Glu, mg/dL | 119 ± 47 | 145 ± 56 | 116 ± 37 | 97 ± 33 | 0.0001 |
| Creatinine, mg/dL | 1.03 ± 0.6 | 1.10 ± 0.77 | 1.07 ± 0.7 | 0.91 ± 0.19 | 0.2 |
| eGFR, mL/m2/1.73‡ | 81.5 ± 30.9 | 84.4 ± 35.1 | 78.8 ± 34.9 | 81.5 ± 21.5 | 0.6 |
| CRP, mg/dL§ | 6.4 (0.26-246) | 30.9 (1.06-246) | 5.9 (0.36-169) | 1.45 (0.26-46.4) | 0.0001† |
| LVEF, n (%) | 51 ± 14 | 42 ± 12 | 50 ± 15 | 61 ± 5 | 0.0001 |
| TAPSE, mm | 20 ± 6 | 18 ± 4 | 21 ± 9 | 21 ± 2 | 0.02 |
| PASP, mmHg | 32 ± 13 | 32 ± 11 | 39 ± 18 | 27 ± 6 | 0.0001 |
Continues variables are reported as mean (standard deviation) and categorical variables as percentages.
*The p values were determined using the analysis of variance and KruskalWallis† test for non-normally distributed variables.
‡Obtained by the modification of diet in renal disease (MDRD) glomerular filtration rate equation.
§Reported as median (p25-75). ACS: acute coronary syndrome; PCI: percutaneous coronary intervention; DM: diabetes mellitus; HT: hypertension; TC: total cholesterol; TG: triglycerides; LDL: low-density lipoprotein cholesterol; HDL: low-density lipoprotein cholesterol high density; CRP: C-reactive protein; LVEF: left ventricular ejection fraction; TAPSE: tricuspid annular plane systolic excursion; PSAP: pulmonary artery systolic pressure.
Table 2 Serum concentrations of OPG, OPN, and ORR
| Biomarkers | Total (n = 130) | ACS-PCI (n = 47) | Non-ACS without PCI (n = 33) | Control group (n = 50) | p* |
|---|---|---|---|---|---|
| OPG, pg/mL | 589.5 (343-1034) | 445 (211-1157) | 757 (227-1537) | 828 (25.6-2288) | 0.001 |
| OPN, pg/mL | 747.7 (37.5-7369.5) | 361 (37.5-25.8) | 626 (114-1541) | 2259 (189.5-7369.5) | 0.001 |
| ORR | 13.2 (0.05-45.77) | 8.9 (4.2-23) | 19.9 (0.99-30.47) | 16.5 (0.05-45.7) | 0.001 |
Continues variables are reported as median and percentile 25 and 75.
*p values were determined using the KruskalWallis test. In 17 patients, appropriate determination of OPG, OPN, or ORR was not possible (missing completely at random, MCAR).OPN: osteopontin; OPG: osteoprotegerin; ORR: osteoprotegerin/RANKL ratio.
As presented in figure 1, patients admitted with ACS-PCI presented lower levels of OPG (p = 0.001) (Fig. 2). Most of the patients with ACS-PCI showed low-risk SYNTAX scores (52%), but we did not find significant differences between SYNTAX risk categories (p = 0.70). Regarding the type of ACS, we did not identify differences in OPG levels between patients with STEMI or non-STEMI (p = 0.57). Furthermore, no correlation between OPG and SYNTAX score (Spearman’s r of 0.01, p = 0.89) was found (Fig. 3). There was a positive correlation between OPG, OPN, ORR, and age in patients with ACS and there was an inverse correlation of OPG and age in control groups. Moreover, ORR was lower in patients admitted with ACS-PCI than in the non-ACS and control group (p = 0.001).
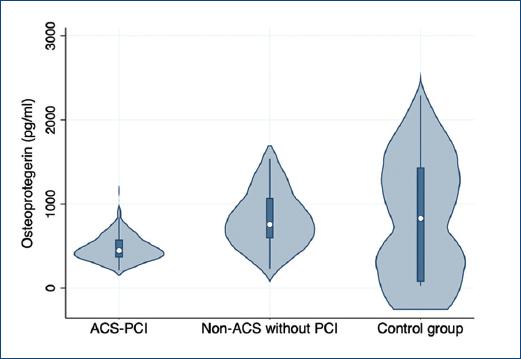
Figure 1 Differences of osteopontin between patients with acute coronary syndrome-percutaneous coronary intervention (ACS-PCI) versus non-ACS without PCI and control group.
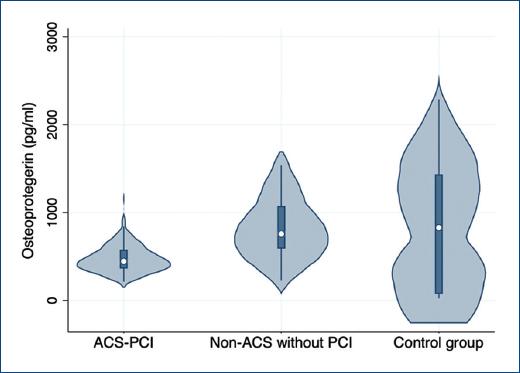
Figure 2 Osteoprotegerin levels are decreased in patients with acute coronary syndrome (ACS)-percutaneous coronary intervention compared with non-ACS and control groups (p = 0.001).
On the other hand, patients with ACS exhibited decreased OPN levels compared to the control group (Fig. 2). The pairwise post hoc Dunn test with Bonferroni adjustments revealed significance only between the ACS-PCI group and the control group and the Non-ACS without PCI group versus the control group (p = 0.000). However, no differences were observed in the levels of OPG among the various SYNTAX groups (p = 0.79).
A logistic regression model was constructed to assess established clinical risk factors for CAD alongside ORR. A decrease in ORR corresponds to a RR of 1.16 (95% CI 1.06-1.25) for having a high SYNTAX score (≥ 33). Furthermore, other clinically relevant variables demonstrated significance in the regression model: hyperglycemia is associated with a RR of 1.027 (95% CI 1.010-1.038) for high SYNTAX scores, while elevated CRP levels are associated with a RR of 1.024 (95% CI 1.010-1.038).
Discussion
Recent evidence underscores a close link between bone-related proteins and atherosclerosis, reflecting an important interplay that contributes to vascular injury, inflammation, and atherosclerosis7,12,13. The main objective of this study was to assess the usefulness of possible biomarkers within the bone-vessel axis in patients with ACS. Our findings confirm the previously known relation between OPG and ACS patients19. However, neither OPG nor OPN values correlated with specific SYNTAX scores and only ORR showed a slight increase in the RR for high scores. This suggests that the concentration of OPG or OPN is not useful as a marker of the severity of CAD, at least determined angiographically.
Patients with both STEMI and non-STEMI ACS showed lower levels of OPG than controls. In fact, in a post hoc analysis of our data (not shown), we found that in controls, OPG levels are elevated regardless of whether they are obese or have DM or hypertension. Regarding OPN levels, they were also decreased in patients with ACS and were lower in the presence of comorbidities such as DM (analysis post hoc, data not shown).
These findings contradict those observed in other similar studies where OPG levels are usually elevated in patients with ACS. In a systematic review, it was concluded that OPG is not a reliable marker of atherosclerosis. This was due to the enormous variability in the results since there was not homogeneity in the methodology used in each study, the selection of patients, of the sample obtained (serum or plasma) and the presence of possible confounders. We consider that the time in which the sample was obtained can influence the results since we obtained the sample before the intervention and in other studies, the sample was obtained at baseline. Other studies also evaluated the levels serially at 24-48 h, and the increase in OPG could be interpreted as a compensatory effect of OPG. Another factor that can influence the contradictory results of the levels of OPG with those in the literature is the use and application of the different methodologies to OPG determination.
Our results could support the proposal that OPG has a protective effect on atherosclerosis and that the finding of increased OPG levels in ACS could be an epiphenomenon. This has been suggested in the literature and should be evaluated specifically.
Our study is not the only one to document contrary data. Sandberg et al.20 studied OPG levels in 20 healthy subjects and in 40 patients with stable angina and 40 with unstable angina and found elevated levels. This author found no difference between groups and the levels reported are like our control groups. In this study, determinations were carried out with kits like ours. Another study supported that OPG levels are elevated in this condition and could indicate that OPG, OPN, and ORR have a protective role, by including two control groups, one with great utility because it was shown that they did not have arterial damage and with blood donors. Furthermore, Koshikawa et al.21 who studied patients with peripheral arterial disease found no difference in OPG levels compared to controls. Vik et al.22 studied OPG through arterial images by echography and control subjects had higher levels of OPG. Another study23 that evaluated the role of OPG in STEMI included 42 patients whose OPG levels were measured serially and concluded that high OPG levels are related to the extent of myocardial damage; however, this study did not include a control group.
We consider that to explain these findings, we must think about the atheromatous plaque at the molecular level. The decrease in OPG activity may indirectly imply an increased activity of matrix metalloproteinase (MMP)24, favoring the degradation of the extracellular matrix, reducing the thickness of the fibrous cap of the plaque, and eventually selecting its rupture and thrombosis in situ. Furthermore, other studies have shown that OPG can protect large blood vessels from arterial calcification based on observation of renal and aortic calcifications25,26.
Therefore, it is necessary to investigate further whether OPG can be a marker or perhaps a mediator of vascular pathology by modulating osteogenic, inflammatory, and apoptotic responses. Integrating the results of recent experimental research and animal models where OPG+/+ versus OPG−/− mice receiving a high phosphate diet and treatment with l,25-dihydroxy vitamin D3 were compared, and a correlation between the increased activity of aortic alkaline phosphatase (crucial initiator in mineralization) and the area of aortic calcification in OPG−/− mice was found27. This suggested an anti-calcification role of OPG through negative regulation of the phosphatase protein.
Finally, OPG also influences the production of other essential agents in plaque stability and MMP [37]. For example, in in vitro studies, incubation of VSMC and monocytic cells with OPGB stimulated the expression of MMP associated with matrix degradation and the production of IL6 and 1728. Therefore, OPG could contribute to endothelial dysfunction by blocking RANKL signaling, and activating protective intracellular endothelial pathways, such as the nitric oxide synthase pathway29-31.
Furthermore, we found that OPN levels were decreased in patients with ACS-PCI. OPN is one of the non-collagenous proteins present in the bone matrix of the atherosclerotic plaques (more strikingly in calcified plaques)9,32,33, and the role of OPN in cardiovascular alterations is well documented. Osteogenic proteins have been evaluated in key network connections between biological signaling pathways, and they are involved in multiple signaling pathways in cardiovascular diseases. The interconnection of these pathways provides insights into the potential treatment of CAD. In this study, we evaluated OPN to seek alternate participation with OPG and found it increased in the peripheral blood of patients without ACS. One possible explanation of this finding is that the soluble distribution of OPN may be lower than the local or tissular concentration, mainly in the calcified atherosclerotic plaques. OPN levels can be altered in individuals with CAD and the presence of calcification of the mitral annulus and aortic valve25,34.
The lack of correlation between OPG and OPN levels with the SYNTAX score may have different explanations. First, in our study be the significant, there is a disproportionate number (52%) of patients with low-risk lesions (SYNTAX < 22). Furthermore, the calculation of the score depends on other factors apart from the vascular calcification itself since it also considers the degree of angulation, the number of affected vessels, the presence of collateral circulation, the TIMI flow of a particular lesion, and the presence of thrombus, among others, which could make this tool an insensitive parameter for vascular calcification.
This is one of the first studies focused on evaluating the performance of this marker in individuals with ACS-PCI and in which evaluation by angiography and the SYNTAX score was also determined, although no association of OPG was found. Our results do not support OPG as a marker of the extent of arterial damage. A graphic summary of the work is shown in figure 4.
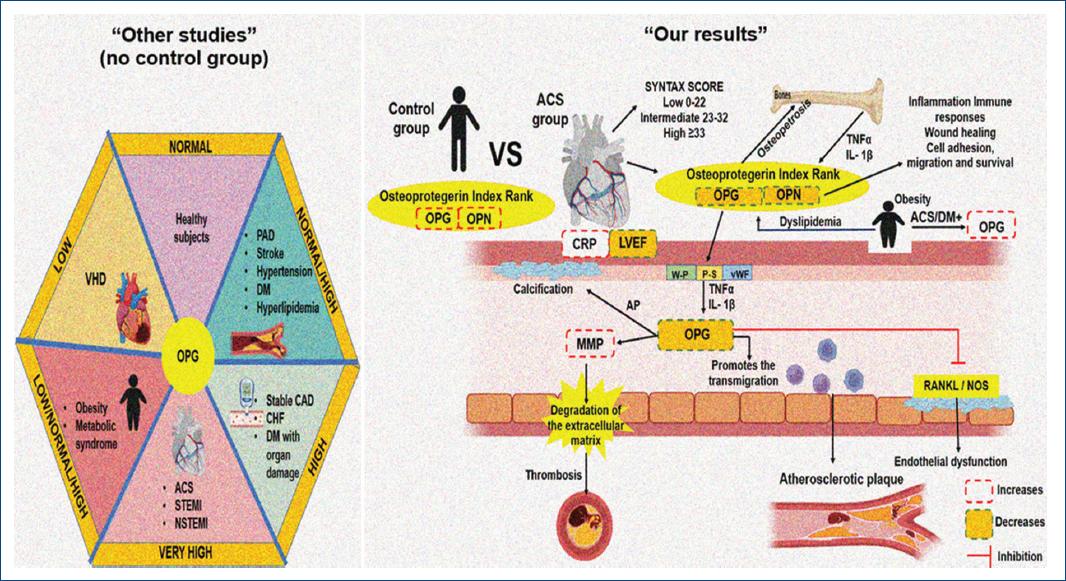
Figure 4 A: diseases that contribute to the increase in various degrees of OPG according to other studies. B: the results of our study show an increase in the OPG/OPN ratio compared to healthy patients, which could indirectly contribute to the increase in MMP and block the RANKL/NOS signaling pathway, contributing to endothelial dysfunction. The formation of atherosclerotic plaque, thrombosis, and other cardiovascular diseases. ACS: acute coronary syndrome; AP: alkaline phosphatase; CAD: coronary artery disease; CHF: chronic heart failure; CRP: C-reactive protein; DM: diabetes mellitus; LVEF: left ventricular ejection fraction; MMP: matrix metalloproteinase; NOS: nitric oxide synthase; NSTEMI: myocardial infarction without ST elevation; OPG: osteoprotegerin; OPN: osteopontin; PAD: peripheral artery disease; P-S: p-selectin; RANKL: nuclear factor kappa-B ligand; STEMI: myocardial infarction with ST elevation; VHD: valvular heart disease; vWF: von willebrand factor; W-P: Weibel-Palade bodies.
Limitations
First, this was a single-center study that could lead to a selection bias. The study included a disproportionate number of men. Furthermore, the SYNTAX may be a less precise tool for evaluating the complexity of CAD and heavy calcification than other computed tomography scan techniques. Finally, although more than 70% of the patients in our study had hypercholesterolemia, an insignificant number of patients were on statin treatment. This is undoubtedly a limitation of our study, but it is since we included patients who reflect the reality of the country’s public health services where a large number of individuals do not have access to lipid-lowering medications.
Conclusion
OPG and OPN (and ORR) levels are decreased in patients with ACS and show no correlation with the SYNTAX score. As an exploratory study our work suggests that increased OPG and OPN levels in non-ACS patients may have, in fact, a protective effect. The reproducibility and comparison of the various findings will require prospective studies where uniformity in the selection of patients and equivalence in the methodology allow a consensus of the participation of OPG and OPN as markers of atherosclerosis.
Author contributions
(I) Conception and design: J.A. Vega-Rosales, and M.E. Soto. (II) Administrative support: M.E. Soto, (III) Supply of study materials or patients: H. Saucedo-Orozco, D.F. Zazueta-Salido, G. Salinas-Arteagas, L. Pech-Manzano, (IV) Data collection and assembly R. Cruz-Soto, V. Guarner-Lans, I. Pérez-Torres and G. Salinas-Arteaga, (V) Drafting of manuscripts: All authors, (VI) Final approval of the manuscript: All authors.











 nueva página del texto (beta)
nueva página del texto (beta)