Case presentation
This is the case of a 68-year-old woman with a 4-month history of total thyroidectomy due to papillary thyroid cancer. Her treatment was levothyroxine, calcium carbonate, and calcitriol. She was transferred to our emergency department with a diagnosis of ST-elevation myocardial infarction. Urgent coronary angiography showed findings of non-obstructive Takotsubo syndrome (TTS) (Fig. 1A and B). Due to signs of cardiogenic shock, she was started on dobutamine and norepinephrine, and an intra-aortic balloon pump (IABP) was placed. Here, we present a rare presentation of TTS and dilated cardiomyopathy due to severe hypocalcemia.
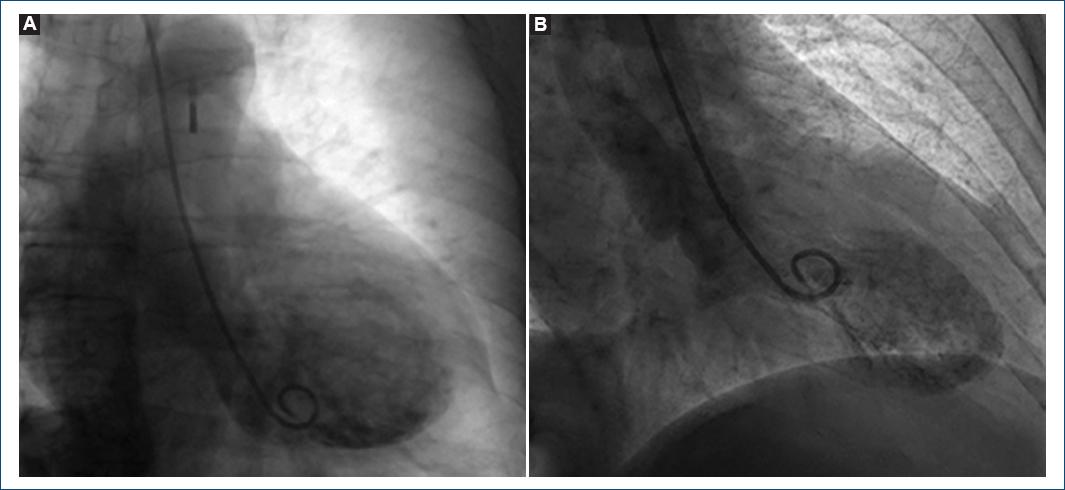
Figure 1 Left ventriculography A: during diastole and B: systole showing typical left ventricular apical ballooning.
She complained of dyspnea, paresthesias, numbness, and Trousseau´s sign were present; no chest pain or other angina equivalents were referred. The initial electrocardiogram (ECG) showed ST-segment elevation and a prolonged QT interval of 560 mseg (Fig. 2A). Laboratory tests showed positive cardiac enzymes NT-proBNP 932 pg/mL and hs-TnT 946 pg/mL (normal range 3-14); hyperlactatemia 2.9 mmol/L (normal range 0.6-1.9); and significant electrolyte abnormalities characterized by severe hypocalcemia with serum calcium of 4.4 mg/dL and 4.6 mg/dL with albumin correction (normal range 8.6-10.2); ionized calcium of 0.5 mmol/L (normal range 1.1-1.3); hyperphosphatemia 6.7 mg/dL (normal range 2.5-5.0); and hypomagnesemia 1.37 mg/dL (normal range 1.9-2.7). Transthoracic echocardiography (TTE) revealed a dilated left ventricle (LV), a low left ventricular ejection fraction of 31%, and signs of typical TTS.
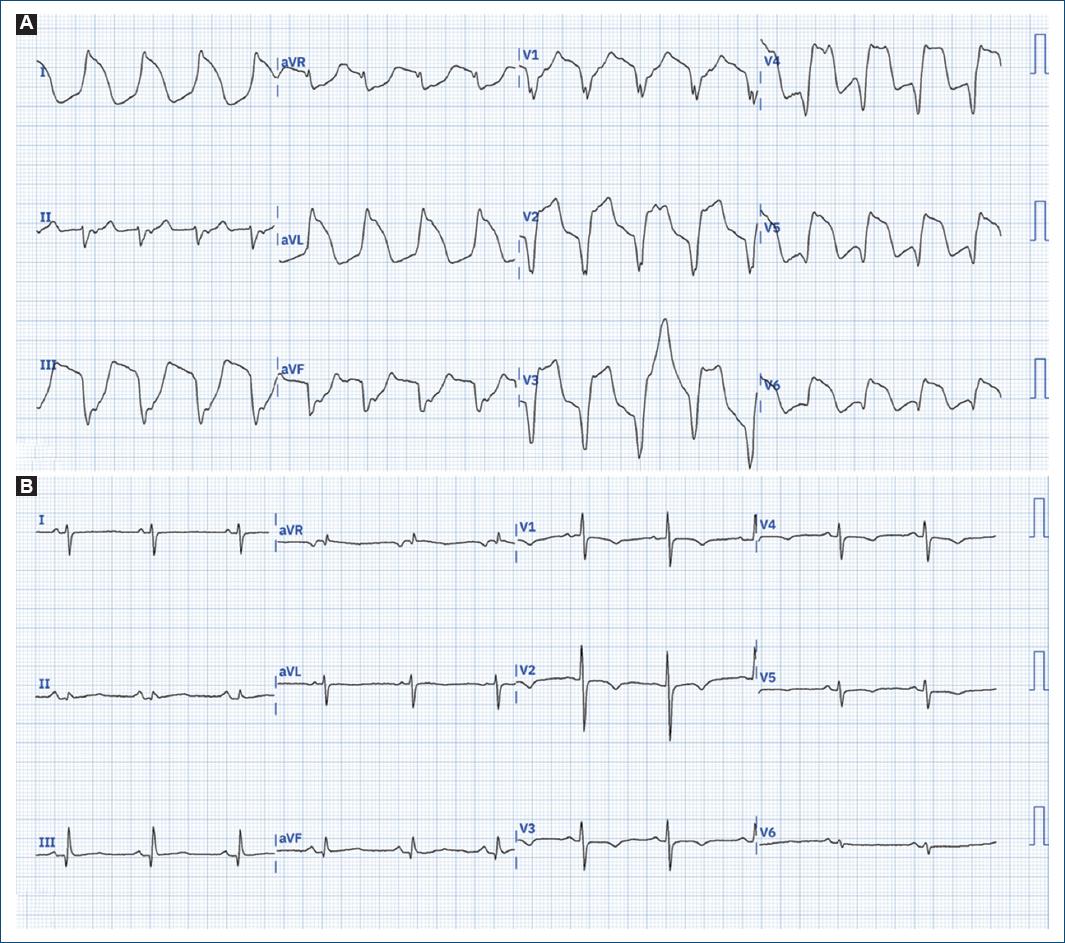
Figure 2 ECG. A: initial ECG on arrival to the enforcement directorate showing a wide QRS complex, diffuse ST-segment elevation, and a prolonged corrected (Bazett) QT interval of 560 mseg. B: ECG on day 3 which shows sinus rhythm with abrupt QRS and QT interval normalization corresponding with normalized calcium. ECG: electrocardiogram; QRS: QRS complex.
She was admitted to the coronary care unit with inotropic and mechanical support. Intravenous and oral calcium replacement and calcitriol were initiated. Further workup showed low vitamin D at 13 ng/mL (normal above 30) and undetectable levels of 1-84-PTH. Endocrinology was consulted and confirmed the diagnosis of post-surgical hypothyroidism and hypoparathyroidism. No other precipitant factor for TTS was identified.
After established therapy, she started showing signs of recovery: she was off of inotropes on day 2; calcium and ECG were normalized on day 3 (Fig. 2B); and IABP was removed on day 4. A second TTE showed recovered LVEF of 63% with normalized LV geometry and volumes without any signs of regional wall motion abnormalities (RWMA). She was discharged after 7 days of hospitalization.
Discussion
TTS is an acute cardiac syndrome characterized by RWMA of the left ventricle leading to transient acute heart failure in the absence of a culprit epicardial coronary lesion1. Commonly described risk factors are: female sex, anxiety, depression, obstructive lung disease, diabetes, substance abuse disorders, post-menopausal women, and endothelial dysfunction2. The specific pathophysiology of TTS remains unknown; however, catecholamine-mediated myocardial dysfunction has been proposed as one of the key elements1. Also, a long list of triggers has been described to play a role, and, specifically, electrolyte abnormalities are among that list. However, even though a relationship between global LV hypokinesia and hypocalcemia has been previously described, as far as we are concerned, a direct correlation between hypocalcemia and TTS has only been reported once3.
Calcium has a tight homeostatic regulation, and it is known to play a central role in the myocardial contractile apparatus. Severe hypocalcemia can result in both systolic dysfunction, and diastolic dysfunction leading to a reversible cause of dilated cardiomyopathy and heart failure4. Also, vitamin D deficiency and hypoparathyroidism, which individually alter calcium homeostasis, have been associated with reversible LV systolic dysfunction and myocardial fibrosis4. The precise mechanism of hypocalcemia-induced TTS is unclear. It has been previously suggested to be multifactorial due to microvascular dysfunction, multi-vessel spam, and catecholamine-mediated β1- and β2-adrenergic receptors (β1AR and β2AR)3. The activation of β1AR and β2AR in the heart increases intracellular cAMP, which phosphorylates L-type calcium channels, increasing calcium influx and enhancing cardiac contractility5. This mechanism is highly relevant because it has been previously described that the distribution of β1AR and β2AR in the left ventricle is not homogeneous but instead shows an apical-basal variation1. Thus, explaining a potential relationship between calcium and the catecholamine-mediated pathophysiology. Nevertheless, besides that, there are no studies showing that β1AR and β2AR activation in the presence of hypocalcemia can cause significant myocardial dysfunction characteristic of TTS. It can be considered that this unbalance in patients with the presence of TTS risk factors (i.e., pre-disposition) ultimately can lead to TTS.
Conclusion
We think that in this specific case, our patient had the presence of TTS risk factors (female sex and post-menopausal) and that hypocalcemia was the trigger leading to TTS. A thorough work-up and clinical evaluation performed did not find another potential trigger. The clear correlation between the correction of calcium levels with ECG normalization and the the fast improvement of hemodynamic and LV function parameters supports hypocalcemia as the etiology. Finally, the correct identification of triggers for TTS is crucial, as the treatment strategy to restore heart function can change dramatically, leading to a different prognosis. Based on this clinical case, we strongly suggest always checking for calcium levels in the initial workup of TTS.











 nueva página del texto (beta)
nueva página del texto (beta)


