Introduction
For many years, arrhythmogenic cardiomyopathy (ACM), known as arrhythmogenic right ventricular (RV) dysplasia, has a significant potential for sudden cardiac death. Its prevalence is 1 in 5,000 and represents an important cause of sudden cardiac death in people younger than 60 years old. ACM is characterized by replacing normal myocardium with fibro-adipose tissue, mainly at the anterior infundibulum, RV apex, and the diaphragmatic area of the right ventricle1.
Cardiac magnetic resonance imaging (CMR) is important in diagnosing and managing patients with ACM. The diagnostic criteria focus on RV volume and ejection fraction (EF), and only the Padua consensus considers late gadolinium enhancement2,3.
Simulations show abnormal deformation patterns of ACM not by changing the electrical properties of the system but only when altered mechanical characteristics are altered by reducing contractility and increasing stiffness4,5. Thus, the ventricular-arterial coupling (VAC) can be useful by being related to efficiency (RV and left ventricular [LV] EF) in RV echocardiography. TAPSE/pulmonary arterial systolic pressure (sPAP) index can reflect the length-force relationship is a useful RV function evaluation and is not affected by the grade of LV dysfunction6.
CMR is an important tool that allows stratification and prognosis of ACM. VAC might play an important role in ACM prognosis based on mechanical and structural abnormalities related to the structural information provided by CMR.
We aimed to describe and correlate mechanical uncoupling by echo with CMR findings and clinically relevant arrhythmias in patients with ACM.
Methods and study population
We retrospectively screened 97 patients with suspicion of ACM at Instituto Nacional de Cardiología, in Mexico City from January 2008 to November 2021 with a follow-up of 12 months. Our exclusion criteria were pregnancy/postpartum period, infectious, autoimmune, hepatic, or neoplastic diseases, previous dialysis, transplant, coronary syndromes, or not fulfilling the criteria mentioned earlier. Fifty-seven were excluded as they did not meet the clinical and imaging definition and 12 for other reasons. Therefore, 28 were diagnosed using the 2019 Modified Task Force Criteria for arrhythmogenic ventricular cardiomyopathy (ACM) and Padua criteria. Moreover, three patients were eliminated as they did not have CMR and echo at the time of diagnosis (Fig. 1).
The RV function variables were obtained from echocardiography reports conducted by trained personnel. The TAPSE was calculated through the systolic excursion of the tricuspid annulus assessed by M-mode and expressed in centimeters.
We explored two critical pathophysiological factors in ACM: right VAC as the ratio of TAPSE to echocardiographic derived sPAP (TAPSE/SPAP) and clinically relevant ventricular tachycardia (need for emergency electrical or pharmacological cardioversion) compared with clinical data, electrocardiographic findings and multi-imaging with transthoracic echocardiogram; plus CMR as the gold standard.
Patients were managed under the Declaration of Helsinki. They provided written informed consent, and the current registry was approved by the Research Committee and the Ethics in Research Committee of the Instituto Nacional de Cardiología.
Statistical analysis
Statistical data analysis of clinical and imaging parameters was expressed as median and interquartile ranges. A Spearman's rank test was used to obtain correlations. The Fisher's exact test was used to compare nominal characteristics, and exact Mann-Whitney U-tests for group comparisons were performed.
In preconceived regression models of left and RV function with VAC were conceived in the frame of low-rank regression splines, generalized additive models and fitted to penalized likelihood estimation (restricted by 5 knots) were performed (GAMPL in SAS) to produce flexible nonparametric regressions. A p < 0.05 was considered statistically significant. The analysis was done in SPSS version 22 and SAS-University-Edition©.
Results
The median age of our cohort was 34 years old, 72% were men, two patients had hypothyroidism (12%), 36% had a family history, and the median body mass index was 25.39 kg/m2.
Electrocardiographic abnormalities were present across the cohort, with 52% having incomplete right bundle branch block (iRBBB) and T wave inversion, 68.2% having Epsilon waves, and 52% having wide QRS (> 120 ms).
The echocardiography parameters had a median LVEF of 55% and RV shortening of 34%. The calculated VAC (TAPSE/sPAP) was 0.63. The CMR showed a median LVEF of 47% and right ventricular ejection fraction (RVEF) of 34.45%, with 19 patients (76%) with late gadolinium enhancement (Table 1).
Table 1 Clinical, electrocardiographic, and multi-imaging characteristics
| Cohort (n = 25) | Value |
|---|---|
| Age (years) | 34 (26-50) |
| Men (%) | 18 (72) |
| BMI (kg/m2) | 25.39 (20.2-28.7) |
| Weight (kg) | 70 (55-85) |
| Height (cm) | 167 (160-172) |
| Body surface area (m2) | 1.78 (1.59-1.96) |
| IRBBB (%) | 13 (52) |
| Inverted T wave (%) | 13 (52) |
| QTc Bazzet (ms) | 430 (420-480) |
| Epsilon wave (%) | 17 (68.2) |
| Wide QRS > 120 ms (%) | 13 (52) |
| Implantable cardiac defibrillator (%) | 17 (68) |
| Ventricular tachycardia or fibrillation | 16 (64) |
| Echocardiography | |
| LVEF (%) | 55 (40-56) |
| RV shortening (%) | 34 (20-40) |
| TAPSE mm | 18 (16-20) |
| sPAP mmHg | 31 (25-41) |
| VAC (TAPSE/sPAP) | 0.63 (0.55-0.8) |
| Cardiac magnetic resonance | |
| LVEF | 47 (40.7-55) |
| RVEF | 34.45 (30.35-39.6) |
| Mass (g/m2) | 44.89 (36.81-54.50) |
| LV final diastolic volume (mL/m2) | 64.88 (55.79-73.73) |
| LV final systolic volume (mL/m2) | 37.02 (28.1-41.45) |
| LV beat volume (mL/m2) | 29.05 (24.21-36.09) |
| RV final diastolic volume (mL/m2) | 86.85 (66.25-113.76) |
| RV final systolic volume (mL/m2) | 55.35 (34.21-68.81) |
| RV beat volume (mL/m2) | 29.05 (24.21-36.09) |
| Gadolinium enhancement | 19 (76) |
Values in interquartile ranges unless stated otherwise. IRBBB: incomplete right bundle branch block; LV: left ventricle; RV: right ventricle; EF: ejection fraction; sPAP: pulmonary arterial systolic pressure; VAC: ventriculo-arterial coupling.
VAC
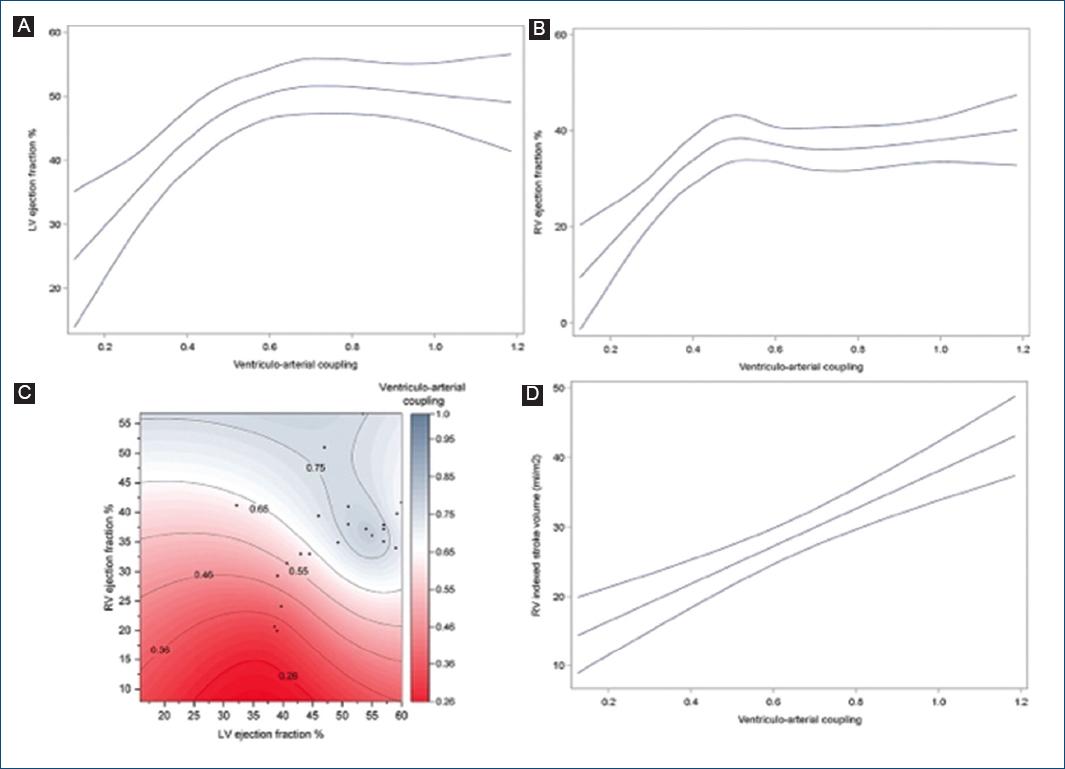
At the Spearman regression, we found a positive relation with LVEF by echo ρ = 0.461 (p = 0.027) and by CMR ρ = 0.472 (p = 0.023), RVEF by CMR ρ = 0.522 (p = 0.038); TAPSE ρ = 0.498 (p = 0.022); and the RV indexed stroke volume ρ = 0.79 (p < 0.001) and a negative with the tricuspid regurgitation gradient ρ = −0.489 (p = 0.033) and sPAP ρ = −0.772 (p < 0.001).
Low-rank regression splines showed a mainly positive association of LVEF (df = 3 F = 20.75 p < 0.001) and RVEF (df = 4 F = 24.34 p < 0.001) with VAC. Furthermore, a positive linear association was seen when VAC was compared against the RV-indexed stroke volume (df = 1 F = 31.67 p < 0.001).
When evaluating RVEF and LVEF against VAC (df = 3 F = 13.04 p < 0.001) in a contour model, it showed an overall positive correlation between EFs and coupling in the patients with ACM (Fig. 2).
Ventricular tachycardia
Differences were seen in patients with ventricular tachycardia; a tendency where older patients had more events (33 vs. 45 years. p = 0.057); more use of an ICD (33.3 vs. 87.5%, p = 0.01). Echocardiography demonstrated lower fractional RV shortening (39.9 vs. 22%, p = 0.033), and CMR showed that they had lower RVEF (39.6 vs. 32.2% p = 0.025) and higher LV mass (38.99 vs. 45.55, p = 0.045) and final diastolic LV volume (56.99 vs. 68.15 mL/m2, p = 0.045). No differences were seen in ECG findings, LVEF by echo or CMR, and no difference in RV volumes (Table 2).
Table 2 Clinical and demographic characteristics of arrhythmogenic cardiomyopathy cohort divided by the presence of ventricular tachycardia
| Variables | Ventricular tachycardia | ||
|---|---|---|---|
| No (n = 9) | Yes (n = 16) | p-value | |
| Men (%) | 6 | 12 | 0.673 |
| Age (years) | 33 (20-34) | 45 (28-55) | 0.057 |
| Weight (kg) | 80 (55-99) | 70 (60-81) | 0.637 |
| Height (cm) | 165 (162-172) | 168 (158.5-171.5) | 0.978 |
| BMI (kg/m2) | 28.3 (20.2-29.8) | 27.2 (21.4-27.6) | 0.522 |
| BSA (m2) | 1.9 (1.59-2.06) | 1.78 (1.6-1.91) | 0.718 |
| Incomplete RBBB (%) | 4 (44) | 9 (56.3) | 0.688 |
| T wave inversion | 4 (44) | 9 (56.3) | 0.688 |
| Corrected QT ms | 430 (424-470) | 438 (418-480) | 0.846 |
| Epsilon wave | 7 (77.8) | 10 (62.5) | 0.661 |
| Wide QRS (> 120 ms) | 5 (55.6) | 8 (50) | 1 |
| ICD (%) | 3 (33.3) | 14 (87.5) | 0.01 |
| Echocardiography | |||
| LVEF | 54.5 (49.2-55.5) | 55 (38-57) | 0.776 |
| RV shortening | 39.9 (27.5-41.5) | 22 (13-35) | 0.033 |
| TAPSE (mm) | 18.5 (14-26.5) | 18 (16.20) | 0.75 |
| Tricuspid gradient (mmHg) | 18 (13-30) | 18 (16-20) | 0.395 |
| sPAP (mmHg) | 30 (24-40.5) | 31 (25-41) | 1 |
| VAC (TAPSE/sPAP) | 0.65 (0.38-1.04) | 0.63 (0.58-0.67) | 0.916 |
| Cardiac magnetic resonance | |||
| LVEF | 47 (46-53.5) | 46.2 (39.4-57) | 0.718 |
| RVEF | 39.6 (36.2-46) | 32.2 (26.7-36.1) | 0.025 |
| Mass (g/m2) | 38.99 (29.02-45.41) | 45.55 (44.87-57.43) | 0.045 |
| LV final diastolic volume (mL/m2) | 56.99 (55.5-60.83) | 68.15 (64.88-74.54) | 0.045 |
| LV final systolic volume (mL/m2) | 29.11 (26.85-33.59) | 39.27 (35.71-43.62) | 0.14 |
| LV beat volume (mL/m2) | 27.17 (25.69.34.43) | 30.22 (26.7-39.52) | 0.301 |
| RV final diastolic volume (mL/m2) | 90.29 (74.07-113.76) | 85.78 (49.38-103.53) | 0.656 |
| RV final systolic volume (mL/m2) | 48.24 (31.29-68.81) | 55.35 (37.47-68.60) | 0.717 |
| RV stroke volume (mL/m2) | 29.61 (28.17-44.95) | 25.63 (11.92-34.07) | 0.126 |
| Gadolinium enhancement | 6 (66.7) | 13 (81.3) | 0.63 |
Values in interquartile ranges unless stated otherwise. IRBBB: incomplete right bundle branch block; LV: left ventricle; RV: right ventricle; EF: ejection fraction; sPAP: pulmonary arterial systolic pressure; VAC: ventriculo-arterial coupling.
Discussion
Our study demonstrated that in ACM, VAC correlates with lower RVEF and LVEF and the RV stroke volume, suggesting increased fibrofatty replacement; furthermore, end-diastolic LV volumes, RVEF, and shortening correlate with electrical outcomes measured by CMR and echo, respectively (Fig. 3).
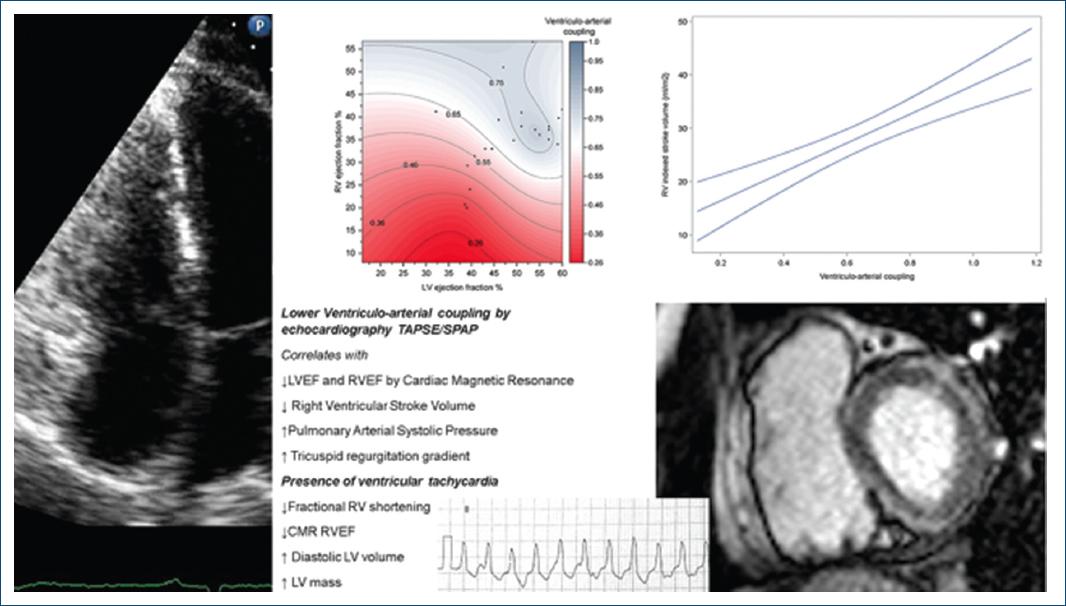
Figure 3 Illustrates the relationship between venoarterial coupling (TAPSE/sPAP) and the presence of ventricular tachycardia, with echocardiogram and cardiac magnetic resonance parameters.
Over the past decade, there has been increasing appreciation for the importance of RV coupling, which provides a valuable method for assessing performance by describing the energy transfer between ventricular contractility and arterial afterload (vascular stiffness)7. We found that VAC, measured by echocardiogram, correlates with CMR-measured LVEF, RVEF, and RV stroke volume, confirming the relationship between mechanical RV performance, possibly suggesting an increased RV involvement and poor RV performance in these patients8.
The RV EF is an essential parameter to define morphofunctional abnormalities in ACM patients, as well as for outpatient patient monitoring. Although CMR imaging remains the gold standard for defining this variable, limited availability in non-cardiovascular centers, the time required for proper image acquisition (not suitable for hemodynamically unstable patients), and the cost associated with magnetic resonance imaging hinder its widespread use in ACM patients. As a result, the existence of a non-invasive parameter that can be easily performed at the patient's bedside, with fast availability, such as VAC, becomes a useful tool.
In our study, the right VAC provides a suitable correlation with CMR-measured RVEF without replacing this study in the in-depth analysis of RV function.
Previous studies have addressed the presence of VT in ACM but did not explore LV volumes9. In our study, we found that increased diastolic LV volumes and low RVEF are associated with the presence or development of VT; interestingly, although RVAC correlated with RVEF, we did not find a statistically significant association with VT; the low number of patients could explain this but also that the presence of VT could be driven by other factors not explored. These findings are relevant since no studies have correlated the presence of an increased volume and electrical outcomes in ACM. A possible explanation is that the global disturbance of the cardiomyocytes and electrical uncoupling in ACM are more prominent in individuals with LV involvement, having a higher risk of VT. Overload RV induces increased Cx43 heterogeneity, thus translating in the clinical setting to a reduced RV fractional shortening and probably the development of electrical instability5,10.
Future perspective includes a more extensive study of VAC as it would allow to set up the prognosis in ACM, identifying patients at risk of electromechanical complications in a non-invasive way by the relationship with RV and LVEF also if the LV diastolic volume predicts the development of clinically important arrhythmias in ACM rather than RV volumes.
Limitations
Limitations in this study were the lower prevalence of the confirmed disease by two criteria although this gives the study a higher specificity. Despite the compelling data, this study proves that echo and CMR might be key in predicting electromechanical coupling in ACM. A more extensive population must be studied to confirm these findings.
Conclusions
This study remarks that in multi-imaging evaluation in recognition of electric-mechanical outcomes, we found that right VAC by echo correlates with RVEF and LVEF by CMR and RV stroke volume in patients with ACM. RVEF, LV mass, and end-diastolic volume correlate with clinically relevant arrhythmias highlighting that ACM has a biventricular involvement that is not limited to RV.











 nueva página del texto (beta)
nueva página del texto (beta)