Articles
Oxidative stress in a population with low blood lead concentrations, chronically exposed in a contaminated area of Zacatecas, Mexico
Estrés oxidativo en una población con bajas concentraciones de plomo en sangre, expuesta crónicamente en una zona contaminada de Zacatecas, México
-
Publication dates-
July 29, 2025
April , 2025
- Article in PDF
- Article in XML
- Automatic translation
- Send this article by e-mail
- Share this article +
ABSTRACT
Many studies have focused on the toxic health effects of low blood lead concentrations in populations chronically exposed to lead-polluted environments. However, few studies have examined oxidative stress in humans under such conditions. We studied a population chronically exposed to mining waste with low blood lead concentrations, high δ-ALAD activity, and high lipid peroxidation levels. A k-means analysis revealed low-high groups based on total antioxidant capacity (TAC), superoxide dismutase (SOD), and catalase (CAT) activities. Low TAC, SOD, and CAT were associated with higher blood lead concentrations, and low SOD was also associated with higher lipid peroxidation. The results suggest that oxidative stress may lead to oxidative damage in chronically lead-exposed populations, even at low blood lead concentrations. Thus, although pathophysiological changes may not be evident at small variations in blood lead concentrations, chronic exposure could cause oxidative/antioxidant changes at blood lead concentrations below 5 μg/dL.
Key words::
lipid peroxidation, total antioxidant capacity, catalase, superoxide dismutase, human lead intoxication
INTRODUCTION
Chronic lead exposure can produce intoxication even at low blood lead concentrations. Some studies found cardiovascular changes, oxidative alterations, and high cortisol concentrations in people with lead concentrations below 10 and 5 µg/dL (Lee et al. 2006, Gump et al. 2008, 2011). Even behavioral and cognitive functions in children were related to low blood lead concentrations (Calderón-Salinas et al. 1996, Mazumdar et al. 2011, Desrochers-Couture et al. 2018). Likewise, some studies reported changes in oxidative and antioxidant markers under similar conditions, though methodological difficulties in selecting populations and temporalities resulted in inconclusive findings (Roy and Kordas 2016, Vacchi-Suzzi et al. 2018).
-
Lee et al. 2006Graded associations of blood lead and urinary cadmium concentrations with oxidative-stress-related markers in the U.S. population: Results from the third national health and nutrition examination surveyEnvironmental Health Perspectives, 2006
-
Gump et al. 2008Low-level prenatal and postnatal blood lead exposure and adrenocortical responses to acute stress in childrenEnvironmental Health Perspectives, 2008
-
2011Low-level Pb and cardiovascular responses to acute stress in children: The role of cardiac autonomic regulationNeurotoxicology and Teratology, 2011
-
Calderón-Salinas et al. 1996Lead exposure in a population of Mexican childrenHuman and Experimental Toxicology, 1996
-
Mazumdar et al. 2011Low-level environmental lead exposure in childhood and adult intellectual function: A follow-up studyEnvironmental Health, 2011
-
Desrochers-Couture et al. 2018Prenatal, concurrent, and sex-specific association between blood lead concentrations and IQ in preschool Canadian childrenEnvironment International, 2018
-
Roy and Kordas 2016The relation between low-level lead exposure and oxidative stress: A review of the epidemiological evidence in children and non-occupationally exposed adultsCurrent Environmental Health Reports, 2016
-
Vacchi-Suzzi et al. 2018Low levels of lead and glutathione markers of redox status in human bloodEnvironmental Geochemistry and Health, 2018
Low blood lead concentrations are relevant for populations not occupationally exposed, but chronically exposed to this metal in the environment (Flores-Ramírez et al. 2012, Carrel et al. 2017, Castro et al. 2019). Changes smaller than 0.5 μg/dL of blood lead concentrations in children triggered medical and governmental actions in Flint, Michigan, USA (Hanna-Attisha et al. 2016, Ruckart et al. 2019). In Canada, the effect of lead-contaminated drinking water on children with blood lead concentrations lower than 5 μg/dL also translated into public actions (Bushnik et al. 2010, Ngueta et al. 2014). The CDC (2024) suggested changing the reference values for blood lead concentration from 5 to 3.5 μg/dL in 2021, in agreement with different multicentric studies done in the USA, Europe, and Asia that found damage at these conditions (Council on Environmental Health 2016, Nakayama et al. 2019).
-
Flores-Ramírez et al. 2012Exposición infantil al plomo en sitios contaminados [Children exposure to lead in contaminated sites]Salud Pública de México, 2012
-
Carrel et al. 2017High prevalence of elevated blood lead levels in both rural and urban Iowa newborns: Spatial patterns and area-level covariatesPLoS One, 2017
-
Castro et al. 2019Variability in the spatial density of vacant properties contributes to background lead (Pb) exposure in childrenEnvironmental Research, 2019
-
Hanna-Attisha et al. 2016Elevated blood lead levels in children associated with the Flint drinking water crisis: A special analysis of risk and public health responseAmerican Journal of Public Health, 2016
-
Ruckart et al. 2019The Flint water crisis: A coordinated public health emergency response and recovery initiativeJournal of Public Health Management and Practice, 2019
-
Bushnik et al. 2010Lead and bisphenol A concentrations in the Canadian populationHealth Reports, 2010
-
Ngueta et al. 2014Exposure of young children to household water lead in the Montreal area (Canada): The potential influence of winter-to-summer changes in water lead levels on children’s blood lead concentrationEnvironment International, 2014
-
CDC (2024)Childhood lead poisoning prevention. About the data: Blood lead surveillance, 2024
-
Council on Environmental Health 2016Prevention of childhood lead toxicityPediatrics, 2016
-
Nakayama et al. 2019Benefits of cooperation among large-scale cohort studies and human biomonitoring projects in environmental health research: An exercise in blood lead analysis of the environmental and child health international birth cohort groupInternational Journal of Hygiene and Environmental Health, 2019
Chronic exposure to lead causes alterations in different organs and systems due, among other causes, to oxidative damage processes such as high lipid peroxidation and impairing antioxidant responses like low activity of antioxidant enzymes such as SOD, CAT, and glutathione peroxidase (GPx), and changes in the antioxidant cofactor concentration (GSH) (Kim et al. 2015, Singh et al. 2015, Dobrakowski et al. 2017). Previous works on laborers chronically exposed to high lead levels described similar effects (Aguilar-Dorado et al. 2014, Rendón-Ramírez et al. 2014).
-
Kim et al. 2015Evaluation and management of lead exposureAnnals of Occupational and Environmental Medicine, 2015
-
Singh et al. 2015Toxic and essential metals in placenta and its relation with lipid peroxides/glutathione status in pre-term and full-term deliveriesAsian Journal of Medical Sciences, 2015
-
Dobrakowski et al. 2017Oxidative DNA damage and oxidative stress in lead exposed workersHuman and Experimental Toxicology, 2017
-
Aguilar-Dorado et al. 2014Eryptosis in lead-exposed workersToxicology and Applied Pharmacology, 2014
-
Rendón-Ramírez et al. 2014Effect of vitamin E and C supplementation on oxidative damage and total antioxidant capacity in lead-exposed workersEnvironmental Toxicology and Pharmacology, 2014
The present work aims to study a population that has long been exposed to lead from mining waste in soil and water in La Zacatecana, Zacatecas Mexico, to evaluate the adverse effects of chronic exposure to low concentrations of lead on the oxidant/antioxidant state, with emphasis on the reduction of antioxidant defenses, in the face of chronic oxidative aggression. This population has been exposed to lead by the dispersion of mining waste from soil and water contaminated by on-site mining activity and the arrival of water from other mines (Iskander et al. 1994, Santos-Santos et al. 2006). The lead values found in soil by Covarrubias et al. (2018) ranged from 29 to 3070 mg/kg, and in some areas, the lead concentration is seven times the reference value, which is 400 mg/kg according to the Official Mexican Standard NOM-147-SEMARNAT/SSA1-2004 (SEMARNAT-SSA 2007). García-Berumen (2015) also found lead concentrations in lagoon water from 0.2 to 0.4 mg/L, below the reference value of 0.5 mg/L indicated in NOM-001-SEMARNAT-2021 (SEMARNAT 2022).
-
Iskander et al. 1994Determination of mercury and other elements in La Zacatecana dam sediment in MexicoScience of The Total Environment, 1994
-
Santos-Santos et al. 2006Analysis of arsenic, lead and mercury in farming areas with mining contaminated soils at Zacatecas, MexicoJournal of the Mexican Chemical Society, 2006
-
Covarrubias et al. (2018Spatial variability of heavy metals in soil and sediments on “La Zacatecana” lagoon, MexicoApplied and Environmental Soil Science, 2018
-
SEMARNAT-SSA 2007Norma Oficial Mexicana NOM-147-SEMARNAT/SSA1-2004. Que establece criterios para determinar las concentraciones de remediación de suelos contaminados por arsénico, bario, berilio, cadmio, cromo hexavalente, mercurio, níquel, plata, plomo, selenio, talio y/o vanadio. Secretaría de Medio Ambiente y Recursos Naturales and Secretaría de Salubridad y AsistenciaDiario Oficial de la Federación. Mexico City, Mexico, March 2, 2007
-
García-Berumen (2015)Significancia de la actividad microbiana en las fluctuaciones de metales pesados (Hg, Pb y As) en la laguna “La Zacatecana”, Guadalupe, Zacatecas, 2015
-
SEMARNAT 2022Norma Oficial Mexicana NOM-001-SEMARNAT-2021. Que establece los límites permisibles de contaminantes en las descargas de aguas residuales en cuerpos receptores propiedad de la naciónSecretaría de Medio Ambiente y Recursos Naturales. Diario Oficial de la Federación, Mexico. March 11, 2022, 2022
We are unaware of other studies published in journals with international circulation that examine blood lead concentrations in this population, and local reports are inconsistent and unreliable. Interestingly, our data did not match the patterns found in workers with high blood lead concentrations (Aguilar-Dorado et al. 2014, Rendón-Ramírez et al. 2014, López-Vanegas et al. 2020). This discrepancy could lead to important insights into the effects of chronic lead exposure. A clustering produced by the k-means technique based on total antioxidant capacity (TAC), superoxide dismutase (SOD), and catalase (CAT) values showed that groups with low TAC and low activities of SOD and CAT had significantly higher blood lead concentrations, though below 10 μg/dL. The results show that oxidative damage did not directly correlate with blood lead concentration but was associated with alterations of antioxidant elements. Therefore, we propose that chronic lead exposure can generate oxidative damage by lowering the total antioxidant capacity and the activity of antioxidant enzymes.
-
Aguilar-Dorado et al. 2014Eryptosis in lead-exposed workersToxicology and Applied Pharmacology, 2014
-
Rendón-Ramírez et al. 2014Effect of vitamin E and C supplementation on oxidative damage and total antioxidant capacity in lead-exposed workersEnvironmental Toxicology and Pharmacology, 2014
-
López-Vanegas et al. 2020Leukocytes apoptosis, TNF-α concentration and oxidative damage in lead-exposed workersToxicology and Applied Pharmacology, 2020
MATERIALS AND METHODS
Study population
The study was conducted in a total population of 3294 inhabitants. The sample size was estimated to detect an effect size distinguishing 1% of the population with lead poisoning, considered noncritical, from 5% as the threshold for acute exposure, with a significance level of 0.05 and 80% power. We used the pwr.p.test function of the pwr R package for a significance level of 0.05 and a power of 0.8; the effect size was calculated as h = 2arcsin (sqrt[0.05])-2arcsin(sqrt[0.01]).The calculation indicated 125 participants, but 150 initiated the study. This study is observational, transversal, and descriptive, providing a comprehensive overview of the population’s lead exposure and its effects on the oxidative/antioxidant status.
Participants were required to be born and reside at the study site. Children under nine and adults with complications or disabling chronic degenerative diseases were excluded. Participants were advised not to consume medications and vitamin supplements 15 days before the study, except for medications necessary for treating chronic diseases. Only 117 people concluded the study, 77 females and 40 males. The age ranged from 9 to 17 years for children (n = 47) and between 22 and 86 for adults (n = 70). All participants agreed to partake; the adults and guardians of the children signed their informed consent to comply with the ethical principles of the Helsinki Declaration; they responded to an announcement posted in a clinic that provides social security. The work was approved by the State Council of Bioethics of Zacatecas and the Ethics and Research Committee of the Hospital Regional de Alta Especialidad del Bajío, with records CNBMX-CEB-32-2011-100 and CONBIOETICA-11-CEI-004-20170731 CEI-31-15, respectively.
Blood sample
The blood samples were collected into Vacutainer tubes with heparin and transported to the laboratory at 4 ºC for the biochemical tests.
Clinical analysis
Blood glucose was measured on test strips and analyzed in the Roche Accu-Chek Glucose Meter; triacylglycerols (TG), total cholesterol (TC), HDL cholesterol (HDL-C), and LDL cholesterol (LDL-C) were quantified in the CZWNB Multifunction Meter using calibrator strips and new strips for each sample according to the manufacturer’s specifications. Results are expressed in mg/dL. Both devices have an accuracy greater than 95%, with a variation between 5 and 7%.
Spirometry and oximetry
A certified specialist physician performed and analyzed the spirometry using a Spirolab IPX1 MIR, interpreted the results, and diagnosed them as a percentage of forced vital capacity (FVC). A pulse oximeter measured the oxygen saturation, reported as a percentage.
Clinic history
A specialist physician took clinical histories with a focus on toxicologic history, emphasizing lead poisoning, in accordance with the working group’s experience with lead-exposed patients (Aguilar-Dorado et al. 2014, Hernández et al. 2019). Our anthropometric indicator was Body Mass Index (BMI) = weight/height² and its classifications.
-
Aguilar-Dorado et al. 2014Eryptosis in lead-exposed workersToxicology and Applied Pharmacology, 2014
-
Hernández et al. 2019Participation of phospholipase-A2 and sphingomyelinase in the molecular pathways to eryptosis induced by oxidative stress in lead-exposed workersToxicology and Applied Pharmacology, 2019
Blood lead concentration
The lead concentration in total blood was determined using the ESA 3010B voltametric instrument at the biochemistry department of CINVESTAV. The results, expressed in μg of lead per dL of total blood (μg/dL), were obtained after calibration with hi and low ESA certified standards with an accuracy of 87-104% and a detection limit of 0.5 μg/dL (Aguilar-Dorado et al. 2014).
-
Aguilar-Dorado et al. 2014Eryptosis in lead-exposed workersToxicology and Applied Pharmacology, 2014
δ-aminolevulinic dehidratase acid activity (δ-ALAD)
The enzymatic activity was measured according to Redig et al. (1991). First, 100 μL of total blood was mixed with ALA 20 mM per 1 h at 37 ºC; later, the reaction was stopped with trichloroacetic acid (TCA) 10%. Then, 750 μL of supernatant was obtained by centrifuging the mixture at 3000 rpm for 15 min. Finally, the supernatant containing porphobilinogen (PBG, a product of δ-ALAD activity) was mixed with 750 μL of Erlich’s reactive (p-dimethyl aminobenzaldehyde [p-DMAB] 1% in ethanol: HCl 1:1) to generate a colored compound measured in a spectrophotometer UV/VIS DU 650 Beckman at 555 nm. The δ-ALAD activity appears as nmol PBG/h/mL of erythrocytes.
-
Redig et al. (1991)Effects of chronic exposure to sublethal concentrations of lead acetate on heme synthesis and immune function in red-tailed hawksArchives of Environmental Contamination and Toxicology, 1991
Lipid peroxidation
The thiobarbituric acid-reactive substances (TBARS) were measured spectrophotometrically (Jain et al. 1989). Erythrocytes were incubated with BHT and TCA 10% for 2 h at 4 ºC. Then, the sample was centrifuged at 3000 rpm for 15 min to obtain a supernatant. A mixture of the supernatant, EDTA 0.1 M, and thiobarbituric acid (TBA) 1% was heated at 95 ºC for 15 min. Later, a spectrophotometer measured the reaction absorbance at 600 and 532 nm (Beckman DU 650 UV/VIS). We show equivalents of malondialdehyde (nmol MDA/mL of erythrocytes) as values of lipid peroxidation.
-
Jain et al. 1989Erythrocyte membrane lipid peroxidation and glycosylated hemoglobin in diabetesDiabetes, 1989
SOD activity
The xanthine-xanthine oxidase system was used to assay the SOD activity by inhibiting the reduction of nitroblue tetrazolium (NBT). An aliquot of hemolysate erythrocytes was mixed with xanthine 0.3 mM, EDTA 0.6 mM, NBT 150 μM, Na2CO3 400 mM, and BSA 1 g/L. Later, the mixture was incubated with xanthine oxidase for 20 min, and the reaction was then stopped with CuCl2 0.8 mM. The formed superoxide reacts with NBT, and the absorbance of the formazan produced was measured spectrophotometrically at 560 nm. The results are reported as units of SOD per g of hemoglobin (UI/g Hb) (Sun et al. 1988).
-
Sun et al. 1988A simple method for clinical assay of superoxide dismutaseClinical Chemistry, 1988
CAT activity
The assay measures the decomposition of H2O2 by catalase of a hemolysate of erythrocytes (Aebi 1984). The sample is mixed with H2O2 30 mM; the absorbance decreases proportionally to the CAT activity and is followed in a spectrophotometer at 240 nm for 30 s. The report uses units of CAT activity per g of hemoglobin (UI/g Hb).
-
Aebi 1984Catalasa in vitroMethods in Enzymology, 1984
TAC
The total antioxidant status in plasma was determined using the oxidation system of 2,2’-azino-di(3-ethyilbenzthiazoline-6-sulfonic acid) (ABTS) by myoglobin with hydrogen peroxide (H2O2) in a 96-well plate (Kambayashi et al. 2009). After 5 min of starting the reaction with H2O2, the formed colored reaction was measured at 600 nm in a microplate reader (ELISA Awareness Stat Fax 2100). The reported values for this assay are mM equivalents of Trolox.
-
Kambayashi et al. 2009Efficient assay for total antioxidant capacity in human plasma using a 96-well microplateJournal of Clinical Biochemistry and Nutrition, 2009
Statistical analysis
Contrary to experimental conditions where there are at least two groups with different levels of exposure to lead, we have only one population with long but mild exposure to the toxic, which causes low blood lead concentrations with minimal variation. We used simple machine-learning techniques to determine the effect of low blood lead levels. The antioxidant system is affected by lead directly because it inactivates some of its enzymes, and indirectly by producing oxidative effects that modulate the response of the antioxidant system. We included all the registered elements of the antioxidant system, TAC, SOD, and CAT, in a cluster analysis of the k-means type to see if it was possible to discriminate groups by similarity; that is, we wanted to determine if there was a numerical distinction of groups based solely on the values of the activity of CAT and SOD and the levels of TAC. Once the two groups were discriminated, we used principal components to estimate the involvement of these variables in distinguishing the groups. We then tested whether these groups had different blood lead concentrations. We also tested these groups for the corresponding levels of lipid peroxidation. By reducing the number of antioxidant elements in the analysis and eliminating the one with the highest weight at each step, we could test the difference in blood lead concentration and lipid peroxidation for the groups individually determined by TAC, SOD, and CAT. We used R software (R Core Team 2019) for k-means, principal components analysis, hypothesis testing, and graphics analysis. Standard t-test with α = 0.05 compared blood lead concentration and lipid peroxidation for the selected groups described above.
-
R Core Team 2019The R project for statistical computing, 2019
RESULTS
The studied population had 60% adults and 66% women. The children were 13 ± 2 years, whereas the adults were 50 ± 16 (Table I).
TABLE I
CHARACTERISTICS OF THE STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
CHARACTERISTICS OF THE STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
| Characteristics | Study population n = 117 | Women n = 77 (66%) | Men n = 40 (34%) | Children n = 47 (40%) | Adults n = 70 (60%) |
| Age (years) Range | 35 ± 22 (9-86) | 33 ± 19 (11-79) | 40 ± 28 (9-86) | 13 ± 2 (9-17) | 50 ± 16* (22-86) |
| Glucose in blood (mg/dL) Range | 83 ± 32 (50-315) | 81 ± 30 (52-315) | 87 ± 36 (50-213) | 73 ± 9 (50-89) | 90 ± 40* (51-315) |
| TG (mg/dL) Range | 138 ± 78 (50-500) | 144 ± 82 (50-500) | 127 ± 70 (50-417) | 111 ± 56 (50-359) | 155 ± 85* (60-500) |
| TC (mg/dL) Range | 161 ± 37 (100-268) | 168 ± 37 (100-268) | 149 ± 34* (103-265) | 138 ± 28 (100-215) | 176 ± 34* (116-268) |
| HDL-C (mg/dL) Range | 45 ± 10 (27-80) | 45 ± 10 (29-80) | 43 ± 10 (27-71) | 42 ± 8 (29-63) | 46 ± 11* (27-80) |
| LDL-C (mg/dL) Range | 90 ± 35 (41-200) | 93 ± 34 (45-200) | 85 ± 38 (41-200) | 74 ± 26 (45-180) | 101 ± 37* (41-200) |
| BMI Range | 26 ± 8 (14-70) | 27 ± 9 (14-70) | 24 ± 6* (16-40) | 20 ± 4 (14-34) | 30 ± 8* (19-70) |
| Classification by BMI (n [%]) Underweight Normal weight Overweight Obesity | 12 (10) 48 (41) 28 (24) 29 (25) | 8 (10) 26 (34) 19 (25) 24 (31) | 4 (10) 22 (55)** 9 (32) 5 (13)** | 12 (26) 27 (57) 7 (15) 1 (2) | 0 (0)** 21 (30)** 21 (30) 28 (40)** |
| Oximetry (%) | 95 ± 4 | 95 ± 3 | 94 ± 6 | 95 ± 5 | 94 ± 3 |
| FVC (%) Range | 82 ± 8 (52-98) | 82 ± 8 (52-97) | 81 ± 8 (62-98) | 83 ± 8 (65-98) | 81 ± 8 (52-94) |
| Diagnosis by spirometry (n [%]) Normal Obstruction Restriction | 91 (78) 22 (19) 4 (3) | 62 (81) 12 (16) 3 (4) | 29 (73) 10 (25) 1 (3) | 39 (83) 7 (15) 1 (2) | 52 (74) 15 (21) 3 (4) |
| Smoking (n [%]) | 6 (5) | 3 (4) | 3 (8) | 1 (2) | 5 (7) |
| Alcoholism (n [%]) | 14 (12) | 7 (9) | 7 (18) | 1 (2) | 13 (19)** |
The mean concentrations of glucose and lipid blood resided within the normal limits (Table I); however, some people scored outside the normal limits in the screening of glucose (8.5%), TG (21.4%), TC (11.1%), HDL-C (53.8%) and LDL-C (20.5%). Alterations of two or more lipid parameters affected 24% of the population. Blood glucose concentration was no different in men and women. Children consistently had lower values than adults (Table I), though some children were outside of upper limits in TG (5.1%), TC (1.7%), HDL-C (26.5%), and LDL-C (2.6%); there were no significant differences between girls and boys.
The BMI in the population was 26 ± 8; women had 12.5% higher BMI than men, and adults were 50% higher than children (p < 0.05). Overweight or obesity was present in 49% of the total population, predominating in adults, with 70%; 12 children were underweight (Table I).
All participants’ oxygen saturation was within normal limits (Table I), with the mean FVC (%) near the lower normal limit. Data did not show significant differences between women and men or children and adults. Of the total population, 22% were diagnosed with obstruction or restriction by spirometry, but men (28%) and women (20%) were not statistically different, nor were children (17%) and the adult population (25%) (Table I).
Smoking and alcoholism were present in 5 and 12% of the total population, respectively, higher in adults (7% and 19%) than in children (2% and 2%) (p < 0.05). There were no differences between men and women for these addictions (Table I).
The population showed a high incidence of chronic clinical diseases (86%), with no statistical difference between genders, but adults had a higher incidence than children (p < 0.05). There were more total incidences in adults (94%) than in children (74%), p < 0.05, but no difference was observed between women (87%) and men (85%) (Table II). The more frequent chronic clinical diseases or damage in the population were dermatitis (65%) and motor incoordination (46%), followed by respiratory, gastrointestinal, ophthalmic, and urinary disorders, along with diabetes and dyslipidemia. However, percentages for men and women were only different for diabetes (23 and 9%, respectively). Chronic clinical diseases were more frequent in adults than in children (p < 0.05), except for ophthalmic diseases (Table II). Diseases or damage with a low incidence (< 5%) in the total population do not appear in Table II.
TABLE II
CHRONIC CLINICAL DISEASES OF STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
CHRONIC CLINICAL DISEASES OF STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
| Chronic clinical diseases | Study population n = 117 (%) | Women (66%) n = 77 (%) | Men (34%) n = 40 (%) | Children (40%) n = 47 | Adults (60%) n = 70 |
| Total patients without incidence | 16 (14) | 10 (13) | 6 (15) | 12 (26) | 4 (6) |
| Total patients with incidence | 101 (86) | 67 (87) | 34 (85) | 35 (74) | 66 (94)* |
| Total of incidence | 306 | 191 (62) | 115 (38) | 60 (20) | 246 (80) |
| Dermatitis | 76 (65) | 47 (61) | 29 (73) | 23 (49) | 53 (76)* |
| Motor incoordination | 54 (46) | 34 (44) | 20 (50) | 10 (21) | 44 (63)* |
| Respiratory infections | 46 (39) | 28 (36) | 18 (45) | 12 (26) | 34 (49)* |
| Gastrointestinal | 31 (26) | 21 (27) | 10 (25) | 6 (13) | 25 (36)* |
| Ophthalmic infections | 28 (24) | 19 (25) | 9 (23) | 7 (15) | 21 (30) |
| Hypertension | 26 (22) | 16 (21) | 10 (25) | 0 (0) | 26 (37)* |
| Urinary infections | 18 (15) | 14 (18) | 4 (10) | 2 (4) | 16 (23)* |
| Diabetes | 16 (14) | 7 (9) | 9 (23)* | 0 (0) | 16 (23)* |
| Dyslipidemia | 11 (9) | 5 (6) | 6 (15) | 0 (0) | 11 (16)* |
The means for blood lead concentration, lipid peroxidation, TAC, and the activities of SOD, CAT, and δ-ALAD in the total population seem to be within the range for non-lead exposed populations (Table III). SOD activity in adults (39 ± 21) was 20% lower than in children (49 ± 24) with p < 0.0, table III.
TABLE III
PARAMETERS OF THE STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
PARAMETERS OF THE STUDY POPULATION: WOMEN, MEN, CHILDREN, AND ADULTS.
| Study population n = 117 | Women n = 77 | Men n = 40 | Children n = 47 | Adults n = 70 | |
| Blood lead concentration (μg/dL) Range | 2.1 ± 2.0 (0.5-10.8) | 2.1 ± 2.1 (0.5-10.8) | 2.2 ± 1.8 (0.5-7.7) | 1.8 ± 1.7 (0.5-10.8) | 2.4 ± 2.1 (0.5-10.7) |
| δ-ALAD activity (nmol PBG/h/mL) Range | 707 ± 218 (187-1336) | 722 ± 223 (187-1336) | 679 ± 208 (297-1300) | 725 ± 208 (360-1300) | 695 ± 225 (187-1336) |
| Lipid peroxidation (nmol MDA/mL) Range | 1.06 ± 0.22 (0.61-1.83) | 1.07 ± 0.23 (0.61-1.83) | 1.06 ± 0.19 (0.67-1.62) | 1.05 ± 0.20 (0.61-1.62) | 1.07 ± 0.23 (0.62-1.83) |
| TAC (mM equivalents of Trolox) Range | 346 ± 246 (30-813) | 356 ± 251 (30-813) | 327 ± 237 (52-795) | 353 ± 243 (52-813) | 341 ± 250 (30-810) |
| SOD activity (UI/g Hb) Range | 43 ± 22 (5-115) | 45 ± 23 (11-115) | 40 ± 20 (5-92) | 49 ± 24 (12-115) | 39 ± 21* (5-107) |
| CAT activity (UI/g Hb) Range | 0.45 ± 0.28 (0.15-1.42) | 0.43 ± 0.25 (0.15-1.42) | 0.48 ± 0.34 (0.18-1.35) | 0.48 ± 0.30 (0.16-1.42) | 0.43 ± 0.27 (0.15-1.35) |
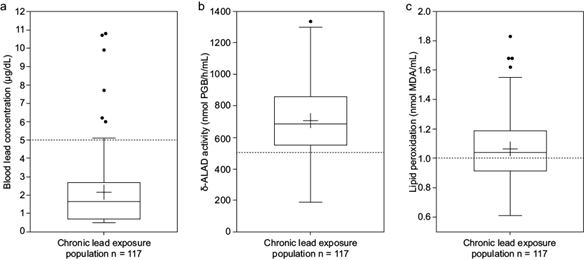
Blood lead concentrations in 94% of the total population were below 5 µg/dL, with a median of 1.5 µg/dL (Fig. 1a). Only seven people exhibited values higher than 5 and lower than 11 µg/dL, which appeared as outliers in the graph. The δ-ALAD activity had a median of 700 nmol PBG/h/mL. Only 12% of the population had values lower than 500 nmol PBG/h/mL, which, in this work, were considered the normal limit of δ-ALAD activity (Fig. 1b). The median of lipoperoxidation was 1.04 nmol MDA/mL for the total population, indicating that more than 50% of the population (57%) has lipid oxidation above 1, which we take as the reference value for unexposed populations with basal oxidation (Fig. 1c).
Thumbnail
Fig. 1
Boxplot of (a) blood lead concentration, (b) δ-ALAD activity, and (c) lipid peroxidation of the chronic lead exposure population. The figures show quartiles. The cross shows the mean. Dotted lines indicate the reference values or operational divisions.
Boxplot of (a) blood lead concentration, (b) δ-ALAD activity, and (c) lipid peroxidation of the chronic lead exposure population. The figures show quartiles. The cross shows the mean. Dotted lines indicate the reference values or operational divisions.
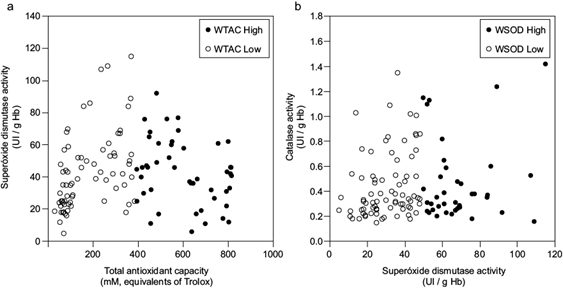
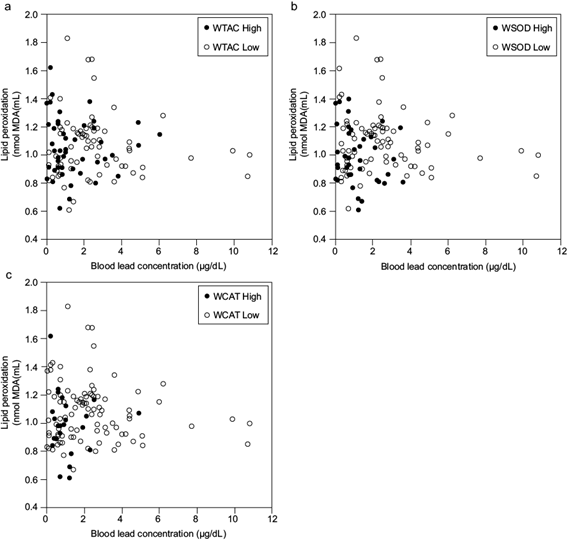
The values of antioxidant enzymes SOD, CAT, and TAC were distributed into two groups by the k-means statistical method. By projecting the groups in a principal component analysis (PCA) plane, we estimated each component’s weight in the splitting. We labeled the groups according to low and high levels for our determinants as weighted factors (WTAC, WSOD, and WCAT). The element with the highest weight was TAC, followed by SOD. Figure 2a shows that TAC separates the groups the most (86%). After eliminating TAC, the analysis for SOD and CAT resulted in SOD contributing 99% of the splitting (Figure 2b). Ultimately, only CAT participated in the analysis, yielding two groups. The similarity of antioxidant indicators alone seems to contribute little to accounting for the relation between blood lead concentration and lipid peroxidation: the scatter plot of blood lead concentration against lipid peroxidation shows no correlation. Labeled values in figure 3 correspond to grouping determined by TAC, SOD, and CAT.
Thumbnail
Fig. 2
Scatter plots of total antioxidant capacity (TAC) vs. superoxide dismutase activity (SOD) classified according to k-means clustering. (a) Low-values (open circles) and high-values groups (closed circles) of WTAC, and (b) superoxide dismutase activity vs. catalase activity (CAT) with points marked as open circles for the low-values group and closed circles for the high-values group of WSOD. WTAC: weight of factor TAC, WSOD: weight of factor SOD activity.
Scatter plots of total antioxidant capacity (TAC) vs. superoxide dismutase activity (SOD) classified according to k-means clustering. (a) Low-values (open circles) and high-values groups (closed circles) of WTAC, and (b) superoxide dismutase activity vs. catalase activity (CAT) with points marked as open circles for the low-values group and closed circles for the high-values group of WSOD. WTAC: weight of factor TAC, WSOD: weight of factor SOD activity.
Thumbnail
Fig. 3
Scatter plots of blood lead concentration vs. lipid peroxidation labeled by k-means clustering for (a) WTAC, (b), WSOD, and (c) WCAT. Open circles represent low-value groups and closed circles high-value groups TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
Scatter plots of blood lead concentration vs. lipid peroxidation labeled by k-means clustering for (a) WTAC, (b), WSOD, and (c) WCAT. Open circles represent low-value groups and closed circles high-value groups TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
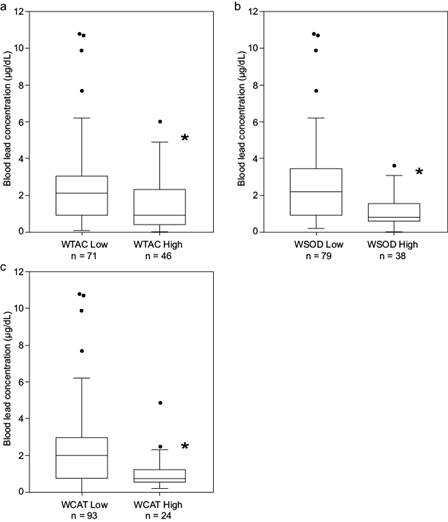
However, data aggregated according to the division into groups WTAC, WSOD, and WCAT, clearly differentiated patients with higher blood lead concentration. The comparison of blood lead concentration values corresponding to groups determined by TAC, SOD, and CAT resulted in a statistically significant difference with higher blood lead concentrations corresponding to lower enzyme activity or TAC values (Fig. 4).
Thumbnail
Fig. 4
Box plots of blood lead concentration by k-means clustering given by (a) WTAC, (b) WSOD, and (c) WCAT. The figures show quartiles. *p < 0.05 according to the Wilcoxon test (p = 0.00156, p = 0.00014, and p = 0.00214, respectively). TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
Box plots of blood lead concentration by k-means clustering given by (a) WTAC, (b) WSOD, and (c) WCAT. The figures show quartiles. *p < 0.05 according to the Wilcoxon test (p = 0.00156, p = 0.00014, and p = 0.00214, respectively). TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
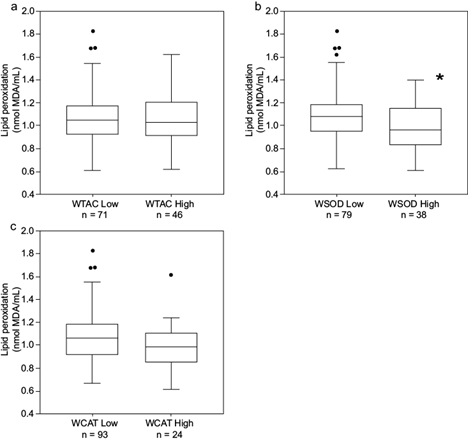
Lipid peroxidation did not follow the same pattern. Only the WSOD group yielded a difference in oxidative effect, though WCAT was close to producing similar results (Fig. 5).
Thumbnail
Fig. 5
Boxplot of lipid peroxidation by k-means clustering based on (a) WTAC, (b) WSOD, and (c) WCAT. The figures show quartiles. *p < 0.05 according to the Wilcoxon test (p = 0.9666, p = 0.0156, and p = 0.0888, respectively). TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
Boxplot of lipid peroxidation by k-means clustering based on (a) WTAC, (b) WSOD, and (c) WCAT. The figures show quartiles. *p < 0.05 according to the Wilcoxon test (p = 0.9666, p = 0.0156, and p = 0.0888, respectively). TAC: total antioxidant capacity, SOD: superoxide dismutase, CAT: catalase, WTAC: weight of factor TAC, WSOD: weight of factor SOD activity, WCAT: weight of factor CAT activity.
DISCUSSION
Clinical conditions
We studied children and adults of both genders to better represent the population. Adults showed more metabolic alterations like overweight, obesity, hyperglycemia, and dyslipidemia than children (Engin 2017, WHO 2021). People have malnutrition and deficient physical activity, including some children with low weight. Also, the population showed a high incidence of pathological alterations, with a higher frequency in adults. Respiratory alterations (obstruction or restriction) are consistent with suburban populations near agricultural activity with high dust dispersion and poor hygienic-dietary conditions due to limited access to public services (Anticona and San-Sebastián 2014, Shahar et al. 2019). The frequent dermatological alterations may be due to unprotected outdoor activities (Schuch et al. 2017).
-
Engin 2017The definition and prevalence of obesity and metabolic syndromeAdvances in Experimental Medicine and Biology, 2017
-
WHO 2021Draft recommendations for the prevention and management of obesity over the life course, including potential targets, 2021
-
Anticona and San-Sebastián 2014Anemia and malnutrition in indigenous children and adolescents of the Peruvian Amazon in a context of lead exposure: A cross-sectional studyGlobal Health Action, 2014
-
Shahar et al. 2019Factors associated with poor socioeconomic status among Malaysian older adults: An analysis according to urban and rural settingsBMC Public Health, 2019
-
Schuch et al. 2017Sunlight damage to cellular DNA: Focus on oxidatively generated lesionsFree Radical Biology and Medicine, 2017
Molecular and clinical alterations in low blood lead concentrations
Motor incoordination and gastrointestinal diseases could be associated with chronic lead intoxication, as indicated in other studies on the effects of low or high blood lead concentrations, which also found alterations in behavior, memory, hormonal, and cognitive functions (Calderón-Salinas et al. 1996, Mazumdar et al. 2011, Hernández et al. 2019). At the molecular level, some studies report low GSH concentrations associated with lead exposure, even at low blood lead concentrations (Ahamed et al. 2007, Singh et al. 2015, Vacchi-Suzzi et al. 2018).
-
Calderón-Salinas et al. 1996Lead exposure in a population of Mexican childrenHuman and Experimental Toxicology, 1996
-
Mazumdar et al. 2011Low-level environmental lead exposure in childhood and adult intellectual function: A follow-up studyEnvironmental Health, 2011
-
Hernández et al. 2019Participation of phospholipase-A2 and sphingomyelinase in the molecular pathways to eryptosis induced by oxidative stress in lead-exposed workersToxicology and Applied Pharmacology, 2019
-
Ahamed et al. 2007Low level lead exposure and oxidative stress: Current opinionsClinica Chimica Acta, 2007
-
Singh et al. 2015Toxic and essential metals in placenta and its relation with lipid peroxides/glutathione status in pre-term and full-term deliveriesAsian Journal of Medical Sciences, 2015
-
Vacchi-Suzzi et al. 2018Low levels of lead and glutathione markers of redox status in human bloodEnvironmental Geochemistry and Health, 2018
Lead exposure of the population by environmental contamination
The soil and water in the area where the population lives contain high concentrations of lead, ranging from 29 to 3070 mg/kg for soil and 0.2 to 0. 4 mg/L for water. Despite some bioremediation actions, lead is still present in the soil in different physical and chemical forms in the vicinity of the population, and people are exposed differently depending on their occupation, outdoor and indoor activities, and hygienic dietary habits, among other factors (Gulson et al. 1994, Santos-Santos et al. 2006, González-Dávila et al. 2012, Covarrubias et al. 2018). Such conditions made this population suitable for studying the effects of chronic environmental lead exposure on the oxidative status and the antioxidant response. The population had low blood lead concentrations, partly because people do not drink or use contaminated water, and partly because soil lead is not bioavailable due to its physical and chemical properties, as suggested by other studies. Environmental exposure is present for everyone, but only those involved in agricultural or construction activities have significant exposure. However, low bioavailability of lead in soil could affect the general population (Gulson et al. 1994, Santos-Santos et al. 2006, Covarrubias et al. 2018, Levin et al. 2020).
-
Gulson et al. 1994Lead bioavailability in the environment of children: Blood lead levels in children can be elevated in a mining communityArchives of Environmental Health, 1994
-
Santos-Santos et al. 2006Analysis of arsenic, lead and mercury in farming areas with mining contaminated soils at Zacatecas, MexicoJournal of the Mexican Chemical Society, 2006
-
González-Dávila et al. 2012Plants and soil contamination with heavy metals in agricultural areas of Guadalupe, Zacatecas, MexicoEnvironmental contamination, 2012
-
Covarrubias et al. 2018Spatial variability of heavy metals in soil and sediments on “La Zacatecana” lagoon, MexicoApplied and Environmental Soil Science, 2018
-
Gulson et al. 1994Lead bioavailability in the environment of children: Blood lead levels in children can be elevated in a mining communityArchives of Environmental Health, 1994
-
Santos-Santos et al. 2006Analysis of arsenic, lead and mercury in farming areas with mining contaminated soils at Zacatecas, MexicoJournal of the Mexican Chemical Society, 2006
-
Covarrubias et al. 2018Spatial variability of heavy metals in soil and sediments on “La Zacatecana” lagoon, MexicoApplied and Environmental Soil Science, 2018
-
Levin et al. 2020Lead seasonality in humans, animals, and the natural environmentEnvironmental Research, 2020
Although people are chronically exposed to lead, only seven participants had blood lead concentration above the reference value for non-occupationally exposed populations (5 μg/dL), and 94% of the people were under this reference value (CDC 2024). The low blood lead concentration might also indicate a kinetic distribution that accumulates lead in bone, as some epidemiological studies on pregnant women and animal models report (Maldonado-Vega et al. 1996, 2002, Olchowik et al. 2014, Flores et al. 2018, Rygiel et al. 2020). In general, despite inhabiting a polluted area with a high lead concentration in soil and water for a long time, populations dwelling in areas with mining waste have low blood lead concentrations (Kim et al. 2008, Schoof et al. 2016, Soto-Ríos et al. 2017).
-
CDC 2024Childhood lead poisoning prevention. About the data: Blood lead surveillance, 2024
-
Maldonado-Vega et al. 1996Lead: Intestinal absorption and bone mobilization during lactationHuman and Experimental Toxicology, 1996
-
2002The effects of dietary calcium lactation on lead in bone mobilization: Implications for toxicologyHuman and Experimental Toxicology, 2002
-
Olchowik et al. 2014The influence of lead on the biochemical properties of bone tissue in ratsAnnals of Agricultural and Environmental Medicine, 2014
-
Rygiel et al. 2020Trimester-specific association of prenatal lead exposure with infant cord blood DNA methylation at birthEpigenetics Insights, 2020
-
Kim et al. 2008Investigation on health effects of an abandoned metal mineJournal of Korean Medical Science, 2008
-
Schoof et al. 2016Assessment of blood lead level declines in an area of historical mining with a holistic remediation and abatement programEnvironmental Research, 2016
-
Soto-Ríos et al. 2017Elevated blood lead levels in children associated with living near mining waste sites in Guerrero/MexicoEnvironments, 2017
Only 12% of the population had low δ-ALAD activity, consistent with low blood lead concentration and a low inhibitory effect on this erythrocyte enzyme. With these low blood lead concentrations and their slight effect on enzymatic activity, it is impossible to see a correlation such as that found in populations with high blood lead concentrations (Ahamed et al. 2006, Rendón-Ramírez et al. 2014).
-
Ahamed et al. 2006Delta-aminolevulinic acid dehydratase inhibition and oxidative stress in relation to blood lead among urban adolescentsHuman and Experimental Toxicology, 2006
-
Rendón-Ramírez et al. 2014Effect of vitamin E and C supplementation on oxidative damage and total antioxidant capacity in lead-exposed workersEnvironmental Toxicology and Pharmacology, 2014
Oxidative damage and antioxidant response associated with environmental lead exposure
The high lipid peroxidation responds to chronic exposure and low blood lead concentrations; however, it resembles that found in lead-exposed workers with high blood lead concentrations (Sugawara et al. 1991, Aguilar-Dorado et al. 2014, Hernández et al. 2019). This high oxidation could be due to other comorbidity factors, but the data analysis did not show any factor that explains high oxidation (Schuch et al. 2017).
-
Sugawara et al. 1991Lipid peroxidation and concentration of glutathione in erythrocytes from workers exposed to leadBritish Journal of Industrial Medicine, 1991
-
Aguilar-Dorado et al. 2014Eryptosis in lead-exposed workersToxicology and Applied Pharmacology, 2014
-
Hernández et al. 2019Participation of phospholipase-A2 and sphingomyelinase in the molecular pathways to eryptosis induced by oxidative stress in lead-exposed workersToxicology and Applied Pharmacology, 2019
-
Schuch et al. 2017Sunlight damage to cellular DNA: Focus on oxidatively generated lesionsFree Radical Biology and Medicine, 2017
Alterations in the antioxidant mechanisms are direct and indirect effects of lead intoxication in populations chronically exposed to environmental and occupational lead, as seems to be the case in this population studied with environmental exposure (Rendón-Ramírez et al. 2014, Jangid et al. 2016, Lopes et al. 2016, Shefa and Héroux 2017, Covarrubias et al. 2018). This effect is discernible in our case, where the groups with low WTAC, WSOD, and WCAT have the highest blood lead concentrations.
-
Rendón-Ramírez et al. 2014Effect of vitamin E and C supplementation on oxidative damage and total antioxidant capacity in lead-exposed workersEnvironmental Toxicology and Pharmacology, 2014
-
Jangid et al. 2016Effect of lead on human blood antioxidant enzymes and glutathioneInternational Journal of Biochemistry Research and Review, 2016
-
Lopes et al. 2016Lead exposure and oxidative stress: A systematic reviewReviews of Environmental Contamination and Toxicology, 2016
-
Shefa and Héroux 2017Both physiology and epidemiology support zero tolerable blood lead levelsToxicology Letters, 2017
-
Covarrubias et al. 2018Spatial variability of heavy metals in soil and sediments on “La Zacatecana” lagoon, MexicoApplied and Environmental Soil Science, 2018
The analysis shows that low WSOD activity is associated with high values of lipid peroxidation. Therefore, in chronic conditions, even at low blood lead concentrations, there is an effect on the antioxidant system elements that could yield oxidative damage, like that of workers with high lead exposure. Although there is no apparent damage in the studied population, there seems to be a chronic effect due to exposure to this environmental risk (Sugawara et al. 1991, Lopes et al. 2016). New research paths could explore subclinical lead intoxication levels to delve deeper into the physiopathology of intoxication processes in populations chronically exposed to lead, such as particular enzyme sensitivities or adaptive responses to chronic damage.
-
Sugawara et al. 1991Lipid peroxidation and concentration of glutathione in erythrocytes from workers exposed to leadBritish Journal of Industrial Medicine, 1991
-
Lopes et al. 2016Lead exposure and oxidative stress: A systematic reviewReviews of Environmental Contamination and Toxicology, 2016
High lipid peroxidation indicates that TAC was insufficient to prevent oxidative damage, and SOD could be sensitive to this lipid peroxidation. The activity shortage of some of their enzymatic components, such as CAT and SOD, could account for the diminished ability of the antioxidant response, despite TAC sustaining normal levels, probably compensated by increments of other antioxidant elements such as uric acid, bilirubin, NADP, or GSH (Kirkman et al. 1999, Flora 2009). These antioxidant enzymes could be sensitive to chronic exposure even at low blood lead concentrations.
-
Kirkman et al. 1999Mechanisms of protection of catalase by NADPH. Kinetics and stoichiometryThe Journal of Biological Chemistry, 1999
-
Flora 2009Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposureOxidative Medicine and Cellular Longevity, 2009
Damage from low lead exposure and low blood lead concentrations
Chronic exposure to lead may trigger conditions such as the kinetics distribution of lead, contributing to its accumulation in organs like bone, or its urinary elimination, which can yield low blood lead concentrations that may still cause systemic damage discernible by clinical and biochemical tests (Khalil et al. 2009). In this way, lead concentrations in blood would not reflect the concentrations of this metal in tissues after long-term exposure. Lead distribution and accumulation in bones have already been documented (Nilsson et al. 1991, Maldonado-Vega et al. 2002, Flores et al. 2018). Lead accumulation in tissues can generate pathophysiological effects on antioxidant enzymes and trigger chronic oxidative stress and damage, which could explain chronic pathologies in the population and is consistent with low TAC, SOD, and CAT. Such phenomena could occur in populations living in environments within accepted lead concentrations or with a high lead concentration and low bioavailability (Gulson et al. 1994, Levin et al. 2020).
-
Khalil et al. 2009Association of cumulative lead and neurocognitive function in an occupational cohortNeuropsychology, 2009
-
Nilsson et al. 1991Kinetics of lead in bone and blood after end of occupational exposurePharmacology and Toxicology, 1991
-
Maldonado-Vega et al. 2002The effects of dietary calcium lactation on lead in bone mobilization: Implications for toxicologyHuman and Experimental Toxicology, 2002
-
Flores et al. 2018Effect of plant grown Pb and Zn geoacumulation in 300-year-old mine tailings of Zacatecas, MexicoEnvironmental Earth Sciences, 2018
-
Gulson et al. 1994Lead bioavailability in the environment of children: Blood lead levels in children can be elevated in a mining communityArchives of Environmental Health, 1994
-
Levin et al. 2020Lead seasonality in humans, animals, and the natural environmentEnvironmental Research, 2020
CONCLUSIONS
The statistical analysis enabled us to detect changes in the antioxidant system associated with oxidative damage at low blood lead concentrations in this population, with precarious health status and long lead environmental exposure. Our study suggests chronic oxidative stress and impaired health status could derive from chronic lead exposure despite the low bioavailability of this metal. The effects of low blood lead concentration in the oxidative system suggest lowering the reference values of blood lead.
This population has oxidative stress. Additional studies could estimate the effects of other health problems, such as sun exposure, poor hygiene, and diet. The necessary population interventions, prevention programs, and public policies for environmental remediation are only possible based on solid data and careful review.
ACKNOWLEDGMENTS
The authors thank Lic. Angélica González Ceja and Héctor Camacho Pérez for their administrative and technical assistance; MD Maria Concepción Flores Barajas for her work in the application and interpretation of spirometry and oximetry in each participant, and Dr. Silvana Marina Flores Barajas for her participation in the design and review of algorithms regarding data capture and refinement.
REFERENCES
- Aebi H. (1984). Catalasa in vitro. Methods in Enzymology 105, 121-126. https://doi.org/10.1016/S0076-6879(84)05016-3 Links
- Aguilar-Dorado I.C., Hernández G., Quintanar-Escorza M.A., Maldonado-Vega M., Rosas-Flores M. and Calderón-Salinas J.V. (2014). Eryptosis in lead-exposed workers. Toxicology and Applied Pharmacology 281 (2), 195-202. https://doi.org/10.1016/j.taap.2014.10.003 Links
- Ahamed M., Verma S., Kumar A. and Siddiqui M.K. (2006). Delta-aminolevulinic acid dehydratase inhibition and oxidative stress in relation to blood lead among urban adolescents. Human and Experimental Toxicology 25 (9), 547-553. https://doi.org/10.1191/0960327106het657oa Links
- Ahamed M. and Siddiqui M.K. (2007). Low level lead exposure and oxidative stress: Current opinions. Clinica Chimica Acta 383 (1-2), 57-64. https://doi.org/10.1016/j.cca.2007.04.024 Links
- Anticona C. and San-Sebastián M. (2014). Anemia and malnutrition in indigenous children and adolescents of the Peruvian Amazon in a context of lead exposure: A cross-sectional study. Global Health Action 7(1), 1-8. https://doi.org/10.3402/gha.v7.22888 Links
- Bushnik T., Haines D., Levallois P., Levesque J., van Oostdam J. and Viau C. (2010). Lead and bisphenol A concentrations in the Canadian population. Health Reports 21 (3), 1-12. Links
- Calderón-Salinas J.V., Valdez-Anaya B. and Mazúñiga-Charles M.A. and Albores-Medina A. (1996). Lead exposure in a population of Mexican children. Human and Experimental Toxicology 15, 305-311. https://doi.org/10.1177/096032719601500406 Links
- Carrel M., Zahrieh D., Young S.G., Oleson J., Ryckman K.K., Wels B., Simmons D.L. and Saftlas A. (2017). High prevalence of elevated blood lead levels in both rural and urban Iowa newborns: Spatial patterns and area-level covariates. PLoS One 12 (5), 1-17. https://doi.org/10.1371/journal.pone.0177930 Links
- Castro I.E., Larsen D.A., Hruska B., Parsons P.J., Palmer C.D. and Gump B.B. (2019). Variability in the spatial density of vacant properties contributes to background lead (Pb) exposure in children. Environmental Research 170, 463-471. https://doi.org/10.1016/j.envres.2018.12.069 Links
- CDC (2024). Childhood lead poisoning prevention. About the data: Blood lead surveillance. Centers for Disease Control and Prevention [online]. https://www.cdc.gov/lead-prevention/php/data/blood-lead-surveillance.html 14/06/2024 Links
- Council on Environmental Health (Perrin L.B., Lowry J.A., Ahdoot S., Baum C.R., Bernstein A.S., Bole A., Brumberg H.L., Campbell C.C., Pacheco S.E., Spanier A.J. and Trasande L.) (2016). Prevention of childhood lead toxicity. Pediatrics 138 (1), 1-15. https://doi.org/10.1542/peds.2016-1493 Links
- Covarrubias S.A., Flores T.J.A., Maldonado V.M., Avelar G.F.J. and Peña C.J.J. (2018). Spatial variability of heavy metals in soil and sediments on “La Zacatecana” lagoon, Mexico. Applied and Environmental Soil Science 2018 (1), 1-8. https://doi.org/10.1155/2018/9612412 Links
- Desrochers-Couture M., Oulhote Y., Arbuckle T.E., Fraser W.D., Séguin J.R., Ouellet E., Forget-Dubois N., Ayotte P., Boivin M., Lanphear B.P. and Muckle G. (2018). Prenatal, concurrent, and sex-specific association between blood lead concentrations and IQ in preschool Canadian children. Environment International 121, 1235-1242. https://doi.org/10.1016/j.envint.2018.10.043 Links
- Dobrakowski M., Pawlas N., Kasperczyk A., Kozłowska A., Olewińska E., Machoń-Grecka A. and Kasperczyk S. (2017). Oxidative DNA damage and oxidative stress in lead exposed workers. Human and Experimental Toxicology 36 (7), 744-754. https://doi.org/10.1177/0960327116665674 Links
- Engin A. (2017). The definition and prevalence of obesity and metabolic syndrome. Advances in Experimental Medicine and Biology 960, 1-17. https://doi.org/10.1007/978-3-319-48382-5_1 Links
- Flora S.J. (2009). Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposure. Oxidative Medicine and Cellular Longevity 2 (4), 191-206. https://doi.org/10.4161/oxim.2.4.9112 Links
- Flores de la Torre J.A., Mitchell K., Ramos-Gómez M.S., Guerrero Barrera A.L., Yamamoto F.L. and Avelar G.F.J. (2018). Effect of plant grown Pb and Zn geoacumulation in 300-year-old mine tailings of Zacatecas, Mexico. Environmental Earth Sciences 77 (386), 1-9. https://doi.org/10.1007/s12665-018-7563-7 Links
- Flores-Ramírez R., Rico-Escobar E., Núñez-Monreal J.E., García-Nieto E., Carrizales L., Ilizaliturri-Hernández C. and Díaz-Barriga F. (2012). Exposición infantil al plomo en sitios contaminados [Children exposure to lead in contaminated sites]. Salud Pública de México 54 (4), 383-392. https://doi.org/10.1590/s0036-36342012000400008 Links
- García-Berumen J.A. (2015). Significancia de la actividad microbiana en las fluctuaciones de metales pesados (Hg, Pb y As) en la laguna “La Zacatecana”, Guadalupe, Zacatecas. M.Sc. Thesis. Centro de Investigación y de Estudios Avanzados, Instituto Politécnico Nacional. Irapuato, Guanajuato, Mexico, 107 pp. Links
- González-Dávila O., Gómez-Bernal J.M. and Ruiz-Huerta E.A. (2012). Plants and soil contamination with heavy metals in agricultural areas of Guadalupe, Zacatecas, Mexico. In: Environmental contamination (Srivastava J., Ed.). InTech, London, United Kingdom, pp. 37-50. Links
- Gulson B.L., Davis J.J., Mizon K.J., Korsch M.J., Law A.J. and Howarth D. (1994). Lead bioavailability in the environment of children: Blood lead levels in children can be elevated in a mining community. Archives of Environmental Health 49 (5), 326-31. https://doi.org/10.1080/00039896.1994.9954982 Links
- Gump B.B., Stewart P., Reihman J., Lonki E., Darvill T., Parson P.J. and Granger D.A. (2008). Low-level prenatal and postnatal blood lead exposure and adrenocortical responses to acute stress in children. Environmental Health Perspectives 116 (2), 249-255. https://doi.org/10.1289/ehp.10391 Links
- Gump B.B., MacKenzie J.A., Bendinskas K., Morgan R., Dumas A.K., Palmer C.D. and Parson P.J. (2011). Low-level Pb and cardiovascular responses to acute stress in children: The role of cardiac autonomic regulation. Neurotoxicology and Teratology 33 (2), 212-219. https://doi.org/10.1016/j.ntt.2010.10.001 Links
- Hanna-Attisha M., LaChance J., Sadler R.C. and Champney S.A. (2016). Elevated blood lead levels in children associated with the Flint drinking water crisis: A special analysis of risk and public health response. American Journal of Public Health 106 (2), 293-290. https://doi.org/10.2105/AJPH.2015.303003 Links
- Hernández G., Villanueva-Ibarra C.A., Maldonado-Vega M., López-Vanegas N.C., Ruiz-Cascante C.E. and Calderón-Salinas J.V. (2019). Participation of phospholipase-A2 and sphingomyelinase in the molecular pathways to eryptosis induced by oxidative stress in lead-exposed workers. Toxicology and Applied Pharmacology 371, 12-19. https://doi.org/10.1016/j.taap.2019.03.025 Links
- Iskander F.Y., Vega-Carrillo H.R. and Manzanares A.E. (1994). Determination of mercury and other elements in La Zacatecana dam sediment in Mexico. Science of The Total Environment 148 (1), 45-48. https://doi.org/10.1016/0048-9697(94)90372-7 Links
- Jain S.K., McVie R., Duett J. and Herbst J.J. (1989). Erythrocyte membrane lipid peroxidation and glycosylated hemoglobin in diabetes. Diabetes 38 (12), 1539-1543. https://doi.org/10.2337/diab.38.12.1539 Links
- Jangid A.P., Shekhawat V.P.S., Pareek H., Yadav D., Sharma P. and John P.J. (2016). Effect of lead on human blood antioxidant enzymes and glutathione. International Journal of Biochemistry Research and Review 13 (1), 1-9. https://doi.org/10.9734/IJBCRR/2016/26992 Links
- Kambayashi Y., Binh N.T., Asakura H., Hibino Y., Hitomi Y., Nakamura H. and Ogino K. (2009). Efficient assay for total antioxidant capacity in human plasma using a 96-well microplate. Journal of Clinical Biochemistry and Nutrition 44 (1), 46-51. https://doi.org/10.3164/jcbn.08-162 Links
- Khalil N., Morrow L.A., Needleman H., Talbott E.O., Wilson J.W. and Cauley J.A. (2009). Association of cumulative lead and neurocognitive function in an occupational cohort. Neuropsychology 23 (1), 10-19. https://psycnet.apa.org/doi/10.1037/a0013757 Links
- Kim H.C., Jang T.W., Chae H.J., Choi W.J., Ha M.N., Ye B.J., Kim B.G., Jeon M.J., Kim S.Y. and Hong Y.S. (2015). Evaluation and management of lead exposure. Annals of Occupational and Environmental Medicine 27 (30), 1-9. https://doi.org/10.1186/s40557-015-0085-9 Links
- Kim S., Kwon H.J., Cheong H.K., Choi K., Jang J.Y., Jeong W.C., Kim D.S., Yu S., Kim Y.W., Lee K.Y., Yang S.O., Jhung I.J., Yang W.H. and Hong Y.C. (2008). Investigation on health effects of an abandoned metal mine. Journal of Korean Medical Science 23 (3), 452-458. https://doi.org/10.3346/jkms.2008.23.3.452 Links
- Kirkman H.N., Rolfo M., Ferraris A.M. and Gaetani G.F. (1999). Mechanisms of protection of catalase by NADPH. Kinetics and stoichiometry. The Journal of Biological Chemistry 274 (20), 13908-13014. https://doi.org/10.1074/jbc.274.20.13908 Links
- Lee D.H., Lim J.S., Song K., Boo Y. and Jacobs D.R. (2006). Graded associations of blood lead and urinary cadmium concentrations with oxidative-stress-related markers in the U.S. population: Results from the third national health and nutrition examination survey. Environmental Health Perspectives 114 (3), 350-354. https://doi.org/10.1289/ehp.8518 Links
- Levin R., Zilli Vieira C.L., Mordarski D.C. and Rosenbaum M.H. (2020). Lead seasonality in humans, animals, and the natural environment. Environmental Research 180 (108797), 1-11. https://doi.org/10.1016/j.envres.2019.108797 Links
- Lopes A.C.B.A., Peixe T.S., Mesas A.E. and Paoliello M.M. (2016). Lead exposure and oxidative stress: A systematic review. Reviews of Environmental Contamination and Toxicology 236, 194-236. https://doi.org/10.1007/978-3-319-20013-2_3 Links
- López-Vanegas N.C., Hernández G., Maldonado-Vega M. and Calderón-Salinas J.V. (2020). Leukocytes apoptosis, TNF-α concentration and oxidative damage in lead-exposed workers. Toxicology and Applied Pharmacology 392 (114901), 1-8. https://doi.org/10.1016/j.taap.2020.114901 Links
- Maldonado-Vega M., Cerbón-Solorzano J., Albores-Medina A., Hernández-Luna C. and Calderón-Salinas J.V. (1996). Lead: Intestinal absorption and bone mobilization during lactation. Human and Experimental Toxicology 15 (11), 872-877. https://doi.org/10.1177/096032719601501102 Links
- Maldonado-Vega M., Cerbón-Solorzano J. and Calderón-Salinas J.V. (2002). The effects of dietary calcium lactation on lead in bone mobilization: Implications for toxicology. Human and Experimental Toxicology 21 (8), 204-214. https://doi.org/10.1191/0960327102ht281oa Links
- Mazumdar M., Bellinger D.C., Gregas M., Abanilla K., Bacic J. and Needleman H.L. (2011). Low-level environmental lead exposure in childhood and adult intellectual function: A follow-up study. Environmental Health 10 (24), 1-7. https://doi.org/10.1186/1476-069X-10-24 Links
- Nakayama S.F., Espina C., Kamijima M., Magnus P., Charles M.A., Zhang J., Wolz B., Conrad A., Murawski A., Iwai-Shimada M., Zaros C., Caspersen I.H., Kolossa-Gehring M., Meltzer H.M., Olsen S.F., Etzel R.A. and Schüz J. (2019). Benefits of cooperation among large-scale cohort studies and human biomonitoring projects in environmental health research: An exercise in blood lead analysis of the environmental and child health international birth cohort group. International Journal of Hygiene and Environmental Health 222 (8), 1059-1067. https://doi.org/10.1016/j.ijheh.2019.07.005 Links
- Ngueta G., Prévost M., Deshommes E., Abdous B., Gauvin D. and Levallois P. (2014). Exposure of young children to household water lead in the Montreal area (Canada): The potential influence of winter-to-summer changes in water lead levels on children’s blood lead concentration. Environment International 73, 57-65. https://doi.org/10.1016/j.envint.2014.07.005 Links
- Nilsson U., Attewell R., Christoffersson J.O., Schütz A., Ahlgren L., Skerfving S. and Mattsson S. (1991). Kinetics of lead in bone and blood after end of occupational exposure. Pharmacology and Toxicology 68 (6), 477-84. https://doi.org/10.1111/j.1600-0773.1991.tb01273.x Links
- Olchowik G., Widomska J., Tomaszewski M., Gospodarek M., Tomaszewska M. and Jagiełło-Wójtowicz E. (2014). The influence of lead on the biochemical properties of bone tissue in rats. Annals of Agricultural and Environmental Medicine 21 (2), 278-281. https://doi.org/10.5604/1232-1966.1108591 Links
- R Core Team (2019). The R project for statistical computing. R Core Team Foundation for Statistical Computing [online]. https://www.r-project.org/ 01/02/2024 Links
- Redig P.T., Lawler E.M., Schwartz S., Dunnette J.L., Stephenson B. and Duke G.E. (1991). Effects of chronic exposure to sublethal concentrations of lead acetate on heme synthesis and immune function in red-tailed hawks. Archives of Environmental Contamination and Toxicology 21 (1), 72-77. https://doi.org/10.1007/BF01055559 Links
- Rendón-Ramírez A.L., Maldonado-Vega M., Quintanar-Escorza M.A., Hernández G., Arévalo-Rivas B.I., Zentella-Dehesa A. and Calderon-Salinas J.V. (2014). Effect of vitamin E and C supplementation on oxidative damage and total antioxidant capacity in lead-exposed workers. Environmental Toxicology and Pharmacology 37 (1), 45-54. https://doi.org/10.1016/j.etap.2013.10.016 Links
- Roy A. and Kordas K. (2016). The relation between low-level lead exposure and oxidative stress: A review of the epidemiological evidence in children and non-occupationally exposed adults. Current Environmental Health Reports 3 (4), 478-492. https://doi.org/10.1007/s40572-016-0115-y Links
- Ruckart P.Z., Ettinger A.S., Hanna-Attisha M., Jones N., Davis S.I. and Breysse P.N. (2019). The Flint water crisis: A coordinated public health emergency response and recovery initiative. Journal of Public Health Management and Practice 25 (Suppl. 1), S84-S90. https://doi.org/10.1097/PHH.0000000000000871 Links
- Rygiel C.A., Dolinoy D.C., Perng W., Jones T.R., Solano M., Hu H., Téllez-Rojo M.M., Peterson K.E. and Goodrich J.M. (2020). Trimester-specific association of prenatal lead exposure with infant cord blood DNA methylation at birth. Epigenetics Insights 13, 1-11. https://doi.org/10.1177/2516865720938669 Links
- Santos-Santos E., Yarto-Ramírez M., Gavilán-García I., Castro-Díaz J., Gavilán-García A., Rosiles R., Suárez S. and López-Villegas T. (2006). Analysis of arsenic, lead and mercury in farming areas with mining contaminated soils at Zacatecas, Mexico. Journal of the Mexican Chemical Society 50 (2), 57-63. Links
- Schoof R.A., Johnson D.L., Handziuk E.R., Landingham C.V., Feldpausch A.M., Gallagher A.E., Dell L.D. and Kephart A. (2016). Assessment of blood lead level declines in an area of historical mining with a holistic remediation and abatement program. Environmental Research 150, 582-591. https://doi.org/10.1016/j.envres.2015.12.028 Links
- Schuch A.P., Moreno N.C., Schuch N.J., Menck C.F.M. and García C.C.M. (2017). Sunlight damage to cellular DNA: Focus on oxidatively generated lesions. Free Radical Biology and Medicine 107, 110-124. https://doi.org/10.1016/j.freeradbiomed.2017.01.029 Links
- SEMARNAT-SSA (2007) Norma Oficial Mexicana NOM-147-SEMARNAT/SSA1-2004. Que establece criterios para determinar las concentraciones de remediación de suelos contaminados por arsénico, bario, berilio, cadmio, cromo hexavalente, mercurio, níquel, plata, plomo, selenio, talio y/o vanadio. Secretaría de Medio Ambiente y Recursos Naturales and Secretaría de Salubridad y Asistencia. Diario Oficial de la Federación. Mexico City, Mexico, March 2. Links
- SEMARNAT (2022). Norma Oficial Mexicana NOM-001-SEMARNAT-2021. Que establece los límites permisibles de contaminantes en las descargas de aguas residuales en cuerpos receptores propiedad de la nación. Secretaría de Medio Ambiente y Recursos Naturales. Diario Oficial de la Federación, Mexico. March 11, 2022. Links
- Shahar S., Vanoh D., Mat Ludin A.F., Singh D.K.A. and Hamid T.A. (2019). Factors associated with poor socioeconomic status among Malaysian older adults: An analysis according to urban and rural settings. BMC Public Health 19 Suppl. 4 (549), 1-12. https://doi.org/10.1186/s12889-019-6866-2 Links
- Shefa S.T. and Héroux P. (2017). Both physiology and epidemiology support zero tolerable blood lead levels. Toxicology Letters 280, 232-237. https://doi.org/10.1016/j.toxlet.2017.08.015 Links
- Singh L., Agarwal P., Anand M. and Taneja A. (2015). Toxic and essential metals in placenta and its relation with lipid peroxides/glutathione status in pre-term and full-term deliveries. Asian Journal of Medical Sciences 7 (1), 34-39. https://doi.org/10.3126/ajms.v7i1.12808 Links
- Soto-Ríos M.L., Juárez-Pérez C.A., Rendón-Gandarilla F.J., Talavera-Mendoza O. and Aguilar-Madrid G. (2017). Elevated blood lead levels in children associated with living near mining waste sites in Guerrero/Mexico. Environments 4 (41), 1-9. https://doi.org/10.3390/environments4020041 Links
- Sugawara E., Nakamura K., Miyake T., Fukumura A. and Seki Y. (1991). Lipid peroxidation and concentration of glutathione in erythrocytes from workers exposed to lead. British Journal of Industrial Medicine 48 (4), 239-242. https://doi.org/10.1136/oem.48.4.239 Links
- Sun Y., Oberley L.W. and Li Y. (1988). A simple method for clinical assay of superoxide dismutase. Clinical Chemistry 34 (3), 497-500. https://doi.org/10.1093/clinchem/34.3.497 Links
- Vacchi-Suzzi C., Viens L., Harrington J.M., Levine K., Karimi R. and Meliker J.R. (2018). Low levels of lead and glutathione markers of redox status in human blood. Environmental Geochemistry and Health 40 (4), 1175-1185. https://doi.org/10.1007/s10653-017-0034-3 Links
- WHO (2021). Draft recommendations for the prevention and management of obesity over the life course, including potential targets. World Health Organization [online]. https://www.who.int/publications/m/item/who-discussion-paper-draft-recommendations-for-the-prevention-and-management-of-obesity-over-the-life-course-including-potential-targets 10/03/2023 Links