INTRODUCTION
The pollutants found in groundwater can be classified as organic and inorganic, and within the latter, the inorganic pollutants include cations, like heavy metals, and anions, like semimetal and non-metal elements, most of them naturally present in soils and rocks (Postigo et al. 2018). One of the most abundant anions present in drinking water is fluoride, and depending on its concentration and amounts ingested it may be beneficial or detrimental to human health (Cai et al. 2015). Groundwater pollution by fluoride is one of the most critical and severe environmental concerns for the developing world due to high toxicity and serious health problems at concentrations above 1.0 mg/L (Rasool et al. 2018). In groundwater, fluoride concentrations can naturally occur from trace amounts to concentrations above 25 mg/L (Bibi et al. 2017). The World Health Organization has specified acceptable fluoride concentrations in drinking water of 1.5 mg/L (WHO 2022).
Various methods are accessible for removing fluoride from water, the most representative are precipitation-coagulation, membrane-based processes, ion exchanges, and adsorption processes (Waghmare and Arfin 2015). Precipitation methods add chemical substances like coagulants and aids in the subsequent precipitation of a poorly soluble fluoride salt as insoluble fluorapatite. The elimination of fluoride is possible thanks to separating solids from the liquid. Calcite can remove fluoride from water by adsorption and precipitation, and magnesium oxide also acts as a precipitating agent. Aluminum salts are frequently used in defluorination (Chai et al. 2013, Gai et al. 2015, Suneetha et al. 2015). Even when the ion exchange, electrocoagulation, and membrane exchange processes are effective and capable of removing fluoride to an acceptable level, they are considered expensive treatment methods and require frequent regeneration and cleaning of the scaling and fouling (Velazquez-Jimenez et al. 2015). Therefore, a non-toxic, environmentally friendly, and economically viable treatment is necessary, as is the spherical agglomeration technique (SAT), as demonstrated by Bailón-Salas et al. (2018).
The SAT has proven to be an effective heavy metal removal method (Alcázar-Medina et al. 2024). It consists of four stages: 1) heavy metal precipitation performed by a controlled dosage of a precipitation agent in a specific pH range; 2) hydrophobization step, aimed to change the hydrophilic nature of the surface of the metal hydroxide using a surfactant; 3) wetting with n-heptane to unify the hydrophobic chains of the surfactant; and 4) the agglomeration step, where the initiator (CaCl2) which acts as a bridge between the particles, allows their union and nuclei formation (González-Valdez et al. 2013, Bailon-Salas et al. 2018), which is essential for agglomeration process (Kawashima and Capes 1974, Keshwani et al. 2015). Performing SAT under controlled physical-chemical parameters such as temperature, pH magnitude, and stirring speed (Alcázar-Medina et al. 2024).
The agglomeration of particles is a complex phenomenon that combines repulsive and attractive forces (Yu et al. 2008). It takes place when the materials have a binding force superior to the force of separation exerted on the particles (Jono et al. 2000), generated due to the energy barrier created by the electrostatic forces of the colloid, for which it is necessary to neutralize the burden of the colloid, this by modifying the pH or the addition of surfactant agents (Louise 1993). Surfactants in aqueous medium have a hydrophilic and a hydrophobic group. The hydrophobic group tends to interfere with the hydrogen bonds of the water molecules since these are formed between the hydrophilic head groups, redistributing the water in the vicinity of the hydrophobic group (Guo et al. 2018, Xu et al. 2020). In this way, adsorbing the surfactant on the surface with its hydrophilic head group positively oriented towards the negatively charged surface (due to electrostatic attraction) and its hydrophobic group oriented away from the surface, making the surface repellent to water, which implies a change in hydrophilic nature of the colloidal surface to hydrophobic (Yin et al. 2009, Rosen and Kunjappu 2012).
The SAT has been used for the removal of metallic and semimetal elements (Zn, Ni, Cu, Cd, Mn, and As), reaching removal efficiencies of up to 99.92% (Proal-Nájera et al. 1997), using sodium oleate as hydrophobizing agent. The disadvantage of using chemical surfactants is the high concentration of residual sodium (Alcázar-Medina et al. 2024). Mohapatra et al. (2011) previously removed fluoride by adsorption in mixed-phase iron monoxides prepared through the mediation-precipitation technique using cetyltrimethylammonium bromide. However, this type of surfactant of industrial origin used in the second stage of the SAT can have a negative effect on human health and the aquatic medium due to its high toxicity and almost null biodegradability (Li et al. 2013). In counterpart, natural surfactants (biotensioactive), such as saponins, have advantages over their industrial counterparts in biodegradability, low toxicity, ecological acceptability, and efficiency under extreme conditions (Liu et al. 2017).
Saponins are an essential group of secondary metabolites in vegetables widely distributed throughout the vegetable kingdom. These biomolecules can be divided into two main classes: Triterpenoid glycosides and steroids, whose structure varies according to the number of sugar units united in different positions (Nguyen et al. 2020).
The family of the Agavaceae is recognized as an important source of steroidal saponins (Nava-Cruz et al. 2015). Puente-Garza et al. (2017) identified the saponins as glycosides of chlorogenic, hecogenin, and tigogenin in the leaves of Agave salmiana sp. (A. salmiana), with a total content of 6.3% of dry material, with the plants acclimated in open environments.
The contamination of groundwater by toxic elements, mainly fluoride, in some wells of communities in Mexico has been reported by Martínez-Cruz et al. (2020), where the concentrations of this element significantly exceed the limits established by Mexican normativity (SSA 2021). This work aimed to investigate the optimal fluoride removal conditions using the spherical agglomeration technique, focusing on the absorbent material (aluminum-activated or freshly prepared aluminum hydroxide) and its dosage. This study also aimed to examine the effectiveness of the foliar material of A. salmiana as a biosurfactant in removing fluoride from aqueous solutions. The spherical agglomeration technique was chosen over traditional adsorption methods to enhance the manipulation and stability of the sludge formed, prevent the re-dissolution of fluoride into the medium, and ensure the stability of the formed agglomerates. This approach aims to optimize the hydrophobization of colloids.
METHODOLOGY
Reactants and equipment
To carry out the removal experiments, a solution with a fluoride concentration of 5 mg/L was used, prepared from a fluoride standard of 1000 mg/L (Hanna instruments, HI 4010-03) and deionized water. For the adsorption experiments and the precipitation stage of SAT, two adsorbents were tested: Activated alumina (AA) (Karal, CAS No. 1344-28-1), and freshly prepared aluminum hydroxide (FPAH), formulated through the reaction between AlCl3 (Fermont, CAS No. 10025-77-1 MEX) and Ca(OH)2 (Jalmek, CAS No. 1305-62-0 MEX). For the hydrophobization stage, A. salmiana extract was prepared according to the methodology used by Hernández et al. (2005) and González-Valdez et al. (2013), using ethyl alcohol absolute (Fermont CAS No. CAS: 64-17-5 MEX). N-heptane (SIGMA CHEMICAL) and CaCl2 (Fermont, CAS No. 10043-52-4 MEX), were used for humectation and agglomeration stages, respectively.
For the AA and FPAH characterization, an X-ray fluorescence (XRF) elemental analysis was performed using an X-Supreme 8000 (Oxfords Instruments, United Kingdom). The fluoride concentration of the samples was determined by an Orion Versa Star Pro (Thermo Fisher Scientific Inc, USA) multiparametric meter with a specific ion electrode (Orion9609NWP) with the use of total ionic strength adjustment buffer (Tisab with CDTA, Orion 940909). Fluoride samples and standard fluoride solutions were diluted 1:1 with a total ionic strength adjustment buffer.
Adsorption batch studies
To select the best adsorbent for fluoride removal, AA and FPAH were tested in doses of 0.1, 0.15, and 0.2 g/L in batch adsorption experiments performed with aqueous models with an initial fluoride concentration of 5.0 mg/L, similar to those found in some wells with high fluoride content (López-Guzmán et al. 2019).
The experiments were carried out in 1 L acrylic containers with baffles, a stirring of 300 rpm provided by a 2.5 cm x 5.0 cm propeller; also, these were carried out at room temperature and initial pH 7.0 (added calcium hydroxide when necessary) with a duration of 20 minutes, the time required to carry out the first stage (precipitation) of the SAT process. A sample volume of 250 mL was used for each experiment, where the required adsorbent (AA or FPAH) and dose (0.1, 0.15 or 0.2 g/L) was added as soon as stirring began. Each adsorption experiment was performed in triplicate.
The preparation method of FPAH is as follows: First, the necessary amount of AlCl3 was diluted in the aqueous model to generate the established adsorbent dose. Once the pH was stabilized, Ca(OH)2 was added to the solution to form FPAH, and the pH was carefully adjusted until the established operating value was reached (pH 7.0).
Precipitates characterization
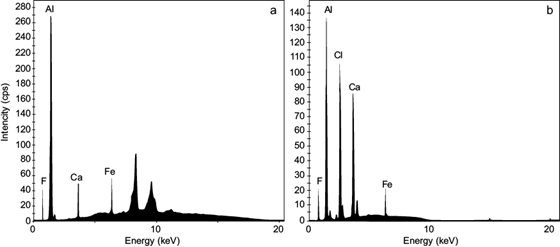
After each adsorption experiment, the water samples were filtered through 0.45 μm membrane filters. The obtained filtered solid was dried, finely pulverized, and analyzed by X-ray fluorescence.
Agave salmiana extract preparation
The plant material was collected in the town of Amado Nervo, Nombre de Dios, Durango, Mexico. The leaves were deposited in plastic bags and stored at 4 ºC until processing. Plant material was identified as Agave salmiana in the herbarium of the CIIDIR-Durango, Instituto Politécnico Nacional, Mexico. Then, they were washed, and spines and cuticle were removed. The material was dried at 40 ºC, cut and minced. Ethanolic extraction was performed according to Hernández et al. (2005) and González-Valdez et al. (2013), and the liquid obtained was evaporated at room temperature to get the solid extract.
Spherical agglomeration technique application (SAT)
Once the best adsorbent was selected based on results from adsorption experiments, SAT was carried out to lower the adsorbent dose.
Usually, SAT consists of four stages: Precipitation, with a length of 20 minutes, where the pH of the sample is adjusted as required for metal precipitation; the 30-minute hydrophobization stage, achieved by the addition of hydrophobizing agent; the wetting stage, which lasts for 15 min, where a conditioning substance is added to cover the hydrophobic solid phase formed in the second stage of SAT (Wu et al. 2015). Finally, in the agglomeration stage (fourth stage, 90 min), CaCl2 is added to ensure the growth of the formed nuclei.
For this study, the initial pH was adjusted to 7 using calcium hydroxide and hydrochloric acid solutions as needed. Also, the first stage was modified to achieve precipitation/adsorption, the adsorbent used was FPAH, selected according to the results from experiments described in the methodology. FPAH was added in doses of 0.06, 0.08, 0.1, 0.15, and 0.2 g/L for the first stage of SAT. As hidrophobizing agent in the second SAT stage, different doses (0.3, 0.6, and 0.9 gExt/gCont in the sample) of A. salmiana leaves extract were added. In the third stage, n-heptane (C7H16) was added in a 2:1 molar ratio (two moles of n-heptane/mol of aluminum). Finally, in the agglomeration stage, CaCl2 was added at a ratio of 1:1 (parts of Ca2+ by part of pollutant) concerning the stoichiometric proportion of the contaminant. At the end of each experiment, the formed agglomerates were separated from the aqueous solution using the Whatman #40 filter paper (Proal-Nájera et al. 1997, Bailón-Salas et al. 2018).
Fluoride quantification
The final fluoride concentration in water was measured with a specific ion electrode (Orion9609NWP) using a total ionic strength adjustment buffer (Tisab with CDTA, Orion 940909). Fluoride samples and standard fluoride solutions were diluted 1:1 with the buffer. The fluoride concentration was determined by an Orion Versa Star Pro (Thermo Fisher Scientific Inc. USA) multiparametric meter.
Statistical analysis
SAT experiment results were analyzed through an analysis of variance (Anova), verifying the assumptions of normality, independence, and homogeneity of variance. The Pearson coefficient of determination (R2) was estimated to verify the model’s goodness of fit through Statistica 7. Subsequently, mean comparisons were made with Fisher’s minimum significant difference test (LSD) to determine the effects of the dose of adsorbent, the dose of extract, and the effect of the interaction. Finally, a quadratic response surface model was obtained for SAT experiments.
RESULTS AND DISCUSSION
Adsorption batch studies for fluoride removal
Table I shows the results of residual fluoride from adsorption experiments described in the methodology. The residual concentration of all the experiments carried out with FPAH was within the limits established by the WHO and the Mexican regulations for drinking water (1.5 mg/L) in a range of final fluoride concentration of 0.59 to 0.74 mg/L (Fig. 1), obtaining removal yields in the range of 85.20 to 88.28% of fluoride adsorption, within 20 minutes of experiment length. On the other hand, in the case of the use of AA as an adsorbent using the exact dosages, lower levels of fluorine adsorption were presented, obtaining concentrations above the NOM-127-SSA-1994 (SSA 2021), with removal yields of this element in the range of 39.34 to 48.66%. The results obtained with the treatment with FPAH in situ showed, in all cases, better results than those where activated alumina was used. Previous studies suggest that FPAH has a particle size of 100 to 200 nm (Gogoi et al. 2020), the more significant adsorption of F- is due to smaller particles, this generates a higher specific surface, especially to an external surface, and greater access to the pores and the internal surface area (Cheng et al. 2014), while the particle size of the activated alumina used was 75 µm (200 mesh).
TABLE I F- Total REMOVAL BY ADSORPTION USING TWO DIFFERENT ADSORBENTS (INITIAL FLUORIDE CONCENTRATION: 5.0 mg/L).
| Adsorbent | AAx | FPAHy | ||
| Dose (g/L) | [F-]final (mg/L) | F- Removal (%) | [F-]final (mg/L) | F- Removal (%) |
| 0.2 | 2.57 ± 0.1 | 39.3 ± 2 | 0.59 ± 0.1 | 88.28 ± 1 |
| 0.15 | 2.70 ± 0.2 | 46.0 ± 3 | 0.63 ± 0.1 | 87.46 ± 2 |
| 0.10 | 3.03 ±0.1 | 48.6 ± 2 | 0.74 ± 0.1 | 85.20 ± 1 |
x Activated alumina; y Freshly prepared aluminum hydroxide.
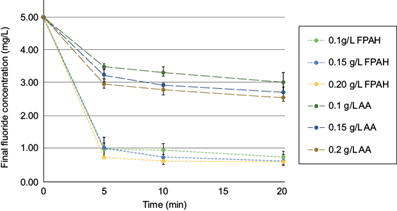
Fig. 1 Comparison between adsorbent type and its dosage for the final fluoride concentration in aqueous models (FPAH = freshly prepared aluminum hydroxide; AA = activated alumina).
In this study, it was found that the removal range achieved with the FPAH was between 85.20 to 88.28% (Table I), these results coincide with those obtained by Dhawane et al. (2018), where one of the most significant effects in fluoride removal efficiency is the initial concentration of this element. Likewise, our results suggest that the smaller particle size of FPAH allows more significant adsorption of fluoride ions due to a larger external surface and greater access to the pores of the internal surface. Besides, the results obtained with FPAH coincide with those obtained by Zhang and Jia (2016), who found the highest levels of fluoride adsorption within the first 20 minutes (> 85% of removal for all cases), being an ideal time for the precipitation stage of SAT. Dhawane et al. (2018) reported the removal of fluoride in aqueous models using NaOH as a pH control agent, obtaining a maximum removal of 86.1% at pH 7.0 from a 10.0 mg/L solution with a dose of adsorbent based on the initial fluoride concentration. On the other hand, Liu et al. (2011) reported the removal of this element using aluminum hydroxide prepared in situ, obtaining removal percentages similar to those reported in this work but with initial fluoride concentration values of 4 and 10 mg/L, which suggests that the fluoride removal achieved in water is based on the proportion of the applied adsorbent dose. However, these works were carried out using precipitation and reaction agents such as NaOH. This study used calcium hydroxide as a precipitation and pH control agent. Since the World Health Organization (WHO 2022) states that calcium can provide essential health benefits, it is recommended that this element be added to drinking water.
Likewise, the results demonstrate the convenience of using freshly prepared aluminum hydroxide. Under the same experimental conditions, up to five times the adsorption capacity can be observed, thus allowing a smaller amount of reagent to comply with the established regulations for drinking water (SSA 2021).
The results obtained here are in good agreement with the studies conducted by Liu et al. (2011) and Gai et al. (2015), where for activated alumina adsorbent, the maximum adsorption capacity for fluoride removal is about 14.5 mg/g. In contrast, for freshly prepared aluminum hydroxide, the maximum fluoride adsorption capacity is more than 110 mg/g in a pH range of 5.0 to 7.2. This indicates that freshly prepared aluminum hydroxide has significantly higher adsorption capacity compared to activated alumina for fluoride removal.
X-ray fluorescence (XRF) analysis of precipitates
XRF analysis of solid samples from the fluoride removal experiments indicates the presence of aluminum and fluoride (in small quantities) in those precipitates, both in the activated alumina and aluminum hydroxide precipitates. This coincides with El Diwani et al. (2022)), who used XRF to characterize the precipitates, revealing the presence of fluoride in the precipitated particles (Fig. 2).
Spherical agglomeration technique for fluoride removal
The adsorbent doses used in the experiments described in the previous section were in a range of 0.1 to 0.2 g/L, obtaining final concentrations within the established regulations (SSA 2021). However, adsorption processes can present limitations in the stability and separation of contaminants due to the irregular shape of the particles. These properties affect the handling and disposal of the generated waste, complicating its management and increasing the challenges in water treatment. In addition, the irregular shape of the particles can lead to inconsistent sedimentation and hinder the effective separation of the adsorbent from the treated water (Yuskel and Dirim 2018). On the other hand, using SAT, a technique that produces spherical particles, and applying biosurfactant, a substance that enhances the separation of contaminants, presents a promising solution to these challenges. As previously demonstrated, SAT was applied to water samples, using FPAH as an adsorbent, given its better fluoride adsorption.
The practical implications of this research are evident in the results presented in table II. Applying SAT made it possible to meet the established regulations with an adsorbent dose as low as 0.1 g/L and a surfactant dose of 0.3 g/L (SSA 2021). However, this study on fluoride removal was conducted using different doses of A. salmiana extract (biosurfactant), with the highest achieved fluoride removal reaching 89.4%, which was obtained under the operating parameters established in the methodology section, with an adsorbent dose (FPAH) of 0.2 g/L and a surfactant dose of 0.3 gExt/gCont.
TABLE II FLUORIDE REMOVAL BY SAT AT PH 7.0, UNDER A FACTORIAL DESIGN 5X3 USING EXTRACTS OF Agave salmiana
| FPAHy dosage (Adsorbent) (g/L) | Extract dosage (gExt/gCont ) | pH Final | Final fluoride concentration (mg/L) | Fluoride removal (%) |
| 0.2 | 0.9 | 6.85 | 0.77 ± 0.1e | 84.6 ± 1 |
| 0.2 | 0.6 | 7.05 | 0.66 ± 0.1bc | 86.8 ± 1 |
| 0.2 | 0.3 | 6.93 | 0.53 ± 0.1ª | 89.4 ± 2 |
| 0.15 | 0.9 | 6.56 | 0.86 ± 0.1e | 82.8 ± 1 |
| 0.15 | 0.6 | 6.93 | 0.71 ± 0.1cd | 85.8 ± 1 |
| 0.15 | 0.3 | 6.69 | 0.63 ± 0.1b | 87.4 ± 1 |
| 0.1 | 0.9 | 6.77 | 1.02 ± 0.1f | 79.6 ± 1 |
| 0.1 | 0.6 | 6.87 | 1.05 ± 0.1f | 79 ± 1 |
| 0.1 | 0.3 | 7.02 | 0.91 ± 0.1e | 81.8 ± 1 |
| 0.08 | 0.9 | 6.80 | 1.24 ± 0.1g | 75.2 ± 1 |
| 0.08 | 0.6 | 6.94 | 1.24 ± 0.1g | 75.2 ± 1 |
| 0.08 | 0.3 | 7.01 | 1.08 ± 0.1f | 78.4 ± 1 |
| 0.06 | 0.9 | 6.94 | 1.62 ± 0.1i | 67.6 ± 1 |
| 0.06 | 0.6 | 7.09 | 1.59 ± 0.1i | 68.2 ± 1 |
| 0.06 | 0.3 | 6.98 | 1.50 ± 0.1h | 70.0 ± 1 |
| Drinking water normativity (SSA 2021): | 6.5 - 8.5 1 - 2 | 1.5 mg/L1 1.0 g/L2 | ||
Note: 1World Health Organization guidelines for drinking water quality (WHO 2022).
2Drinking Water quality standards in the Mexican normativity (SSA 2021).
a, b, c, d… i superscript: Represent differences from the Fisher LSD test.
yFreshly prepared aluminum hydroxide.
Similarly, it is possible to observe that with lower doses of adsorbent, such as 0.1 g/L, values lower than one ppm can be reached, also with a lower surfactant dose of 0.3 gExt/gCont (88.1% of fluoride removal). This trend is maintained throughout the experimental design, where higher percentages of fluoride removal are obtained when using lower dosages of surfactant (0.3 gExt/gCont) at all levels of adsorbent used. The use of lower doses of surfactant can lead to higher fluoride removal values due to a reduction in competition between surfactant molecules, optimizing the surface of the adsorbent and avoiding saturation of the active sites (Merkelbach et al. 2024). Due to this, it is necessary to optimize the application of the surfactant to guarantee the correct hydrophobization of the colloids and avoid the redissolution of the fluorides, maintaining the stability of the particles formed by the spherical agglomeration technique. Adequate concentrations also prevent micelles formation, improving removal efficiency (Schreiner et al. 2020).
The statistical analysis of the residual fluoride concentration of the random factorial linear model of blocks complied with the assumptions of normality, independence, and homogeneity of variance. The coefficient of determination (R2) for the experimental series was 0.972, indicating the reliability of the experimental design (p < 0.05). It also shows that the SAT applied for fluoride removal is adequate, given that high levels of removal of this element (89.5%) comply with the drinking water regulations (SSA 2021). Three-way ANOVA analysis performed for the residual concentration of fluoride, as the dependent variable, showed the effects of the adsorbent dose and that of the applied extract (Table III). Similarly, the Fisher LSD test showed significant differences between the different treatments. In the case of adsorbent concentration, significant differences were observed, up to 0.1 g/L of adsorbent. However, significant differences were observed between extract dosages when using adsorbent concentrations from 0.1 g/L of adsorbent, showing lower fluoride concentration when using a lower extract dosage at all adsorbent concentrations (0.3 gExt/gCont). These trends coincide with what was established by Yang et al. (2014), who mentions that the fluoride removal efficiency decreases as increase concentrations of coexisting anions, such as surfactants, increase due to competition for adsorption sites.
TABLE III ANOVA FOR THE RESIDUALS OF RESIDUAL FLUORIDE IN AQUEOUS MODELS, AFTER APPLICATION OF THE SPHERICAL AGGLOMERATION TECHNIQUE FOR THE 5X3 FACTORIAL DESIGN.
| Effect | Sum of squares | Degrees of freedom | Fcalculated | pvalue |
| Intercept | 47.5347 | 1 | 23120.00 | <0.001 |
| Adsorbent dosage | 4.97646 | 4 | 605.11 | <0.001 |
| Extract dosage | 0.23709 | 2 | 57.66 | <0.001 |
| Adsorbent dosage * Extract dosage | 0.04170 | 8 | 2.54 | 0.0308 |
The fluoride removal efficiencies obtained in this study, which were similar to those reported by Dubey et al. (2018) using aluminum and polyaluminum chloride in batch type reactors and continuous flow (final concentration of 0.6 mg/L of fluoride), demonstrate the practical applicability of our research. Moreover, the removal efficiency was greater than 87.3% using activated red mud as a permeable reactive barrier (Vinati et al. 2019), further highlighting the effectiveness of our approach in comparison to existing methods.
Findings from prior research in which the SAT was used demonstrate its effectiveness for removing non-metallic elements (González-Valdez et al. 2013). Research shows that biosurfactant use significantly affects the removal of contaminating inorganic elements (Alcázar-Medina et al. 2024).
Results obtained in fluoride removal experiments through SAT suggest that as in past studies (Bailón-Salas et al. 2018), foliar material extracts can adequately cover precipitated particles, helping to optimize the process of fluoride removal after spherical agglomeration to reach the fluoride concentrations recommended by the WHO. Previous works (Proal-Nájera et al. 1997, González-Valdez et al. 2013, Alcázar-Medina et al. 2024) have demonstrated a direct relationship between the presence of colloidal particles and the final concentration of the contaminating element, which reveals the effectiveness of the SAT in the agglomeration of particles. Therefore, a corresponding reduction in the number of particles dispersed in the solution is possible by a significant decrease in the final fluoride concentration after the particle agglomeration treatment.
It is important to point out that doses substantially lower than the LD50 of n-heptane (3000 mg/kg) are used, thus reducing any potential risk to human and environmental health. In addition, n-heptane remains attached to the agglomerated particles trapped in the filter, which further reduces direct exposure to this substance in the treated water.
Figure 3 shows the surface response graph constructed for the SAT experiments using FPAH, designed using a quadratic fit model, which demonstrates the behavior of the final fluoride concentration as a function of the effect of the initial concentration of the adsorbent (A) and the dose of A. salmiana leaf extract (S). The combined effect of both parameters was significant in the removal of fluoride. At the same time, the increase in the concentration of adsorbent can be the dominant factor since, in all cases, a greater removal of adsorbent allowed a greater removal of fluoride at an initial concentration of this element. These findings are consistent with those of Vinati et al. (2019), who achieved similar fluoride removal levels with activated red mud under similar batch operating conditions, demonstrating that adsorbent dosage is one of the most essential elements in fluoride removal in water. The results obtained allow to reduce the amount of reagents and still obtain a residual fluoride concentration close to the recommendations established by the WHO (1.5 mg/L).
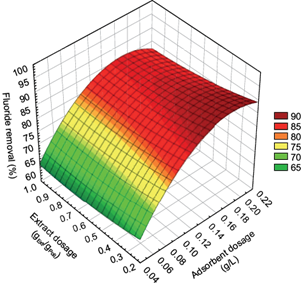
Fig. 3 Surface response for the fluoride removal present in water, using freshly prepared aluminum hydroxide as an adsorbent and Agave salmiana leaf extract as a surfactant, through the spherical agglomeration technique.
In addition, the response surface plot (Fig. 3) shows the removal efficiencies of fluoride in water as a function of previously established parameters, where it is possible to observe that there is a vast area that identifies the maximum efficiency between the different doses of extract and the different doses of adsorbent. Additionally, it is observed that a maximum percentage of fluoride removal is obtained in the generated response surface with low surfactant dosages (0.3 g of extract/g of pollutant). That demonstrates the ability of the surfactant to hydrophobicize the precipitated particles and thus avoid its redissolution in the liquid medium, which allows for predicting the result of fluoride removal efficiencies in water under a considerable range of concentrations of this element in the water to be treated.
Quadratic fit response surface graph for the residual concentration of fluoride, is represented by equation 1 (Ec. 1):
The application of the SAT was carried out successfully since it was possible to obtain large removal percentages, where it was possible to reach residual concentrations of fluoride that meet the water quality recommendations established by the WHO and therefore for the established regulations for drinking water.
CONCLUSIONS
Adsorption experiments from fluoride aqueous solution with a concentration of 5 mg/L, using activated alumina or aluminum hydroxide as precipitants, yielded higher removal efficiency when using aluminum hydroxide. The x-ray diffraction analysis from fluoride precipitates on activated alumina and fluoride on aluminum hydroxide showed the presence of fluoride in the filtrate of both adsorbent materials, which evidences the correct fluoride adsorption on both compounds.
The highest percentage of removal was achieved through the use of FPAH at dose of adsorbent of 0.2 g/L, however, it should be mentioned that it is possible to reduce the amount of reagents to be used and still comply with the fluoride values in drinking water recommended by the World Health Organization (1.5 mg/L).
Then, the removal of fluoride, a nonmetallic element, was demonstrated in aqueous models through the SAT using FPAH as adsorbent for the first SAT stage, using A. salmiana extract as hydrophobizing agent. The highest fluoride removals were obtained with lower doses of extract, which in turn allows using lower doses of adsorbent. The removal of fluoride through the SAT, reached removal levels of 89.5% and a residual concentration that complies with both the standards established by the WHO and with the standards established by the Mexican regulations for drinking water.











 nueva página del texto (beta)
nueva página del texto (beta)