Introduction
Seeds of leafy aerial parasitic plants (mistletoes) must be dispersed onto a suitable branch of the appropriate hosts for successful seedling establishment, with post-dispersal processes being crucial for recruitment (Sargent, 1995; Ladley and Kelly, 1996; López de Buen and Ornelas, 2002; Norton et al., 2002; Rawsthorne et al., 2012). Studies on seed dispersal of mistletoes have primarily considered the seed movement effect (Overton, 1994; Martínez del Rio et al., 1996), focusing on the consequences of directed bird dispersal from the perspective of the mistletoe-bird mutualistic interaction (e.g., Reid, 1991). Accordingly, these studies examined frugivore-generated seed shadows (the seed deposition pattern of a mistletoe population; sensu Schupp, 1993), infection patterns on host species associated with seed dispersal by frugivorous birds (Davidar, 1983; Reid, 1989; Aukema and Martínez del Rio, 2002), and seed and seedling survival on different hosts (Monteiro et al., 1992; Sargent, 1995; Yan and Reid, 1995; Norton and Ladley, 1998; López de Buen and Ornelas, 2002). Despite the well-studied consequences of directed bird dispersal in mistletoes, the effects of fruit size variation on seed germination processes have received limited attention, particularly those spanning from seed deposition by seed dispersers and adherence to the host branches with their viscin to haustorial penetration into host branches.
As far as we know, Psittacanthus Mart. (Loranthaceae) mistletoes depend on frugivorous birds for seed dispersal, and their foraging behavior strongly influences the spatial, often aggregated, distribution of these parasites within and between hosts (Monteiro et al., 1992; López de Buen and Ornelas, 1999; Arce-Acosta et al., 2016; Pérez-Crespo et al., 2016; Guerra et al., 2018). Fruit size of Psittacanthus species ranges from 5 mm long in P. sonorae (S. Watson) Kuijt to at least 24 mm in P. macrantherus Eichler (Ornelas et al., 2024a). Interspecific differences in fruit size could be explained by habitat differences, the identity, specificity and quality of host species, and/or by the identity and foraging behavior of seed dispersers (Aukema and Martínez del Rio, 2002; Watson, 2009; Ornelas et al., 2024b), although information is lacking for Psittacanthus species with very large fruits. Understanding intraspecific variation in fruit size is also important, as it could relate to selection and handling by fruit consumers to mediate seed dispersal and successful establishment, assuming that larger or heavier fruits and seeds have more resources for haustorium development, subsequent interaction with the host and for seedling survival (e.g., Gonzáles et al., 2007; Ramírez and Ornelas, 2009, 2012). Ramírez and Ornelas (2009) first showed that host-associated differences in fruit size traits affect its attractiveness and consumption by seed dispersal vectors. Larger fruits may be preferentially taken by frugivores with longer gut passage rates, achieving a greater likelihood of seed attachment, seed germination and seedling survival on most preferred host species and for long-distance seed dispersal (Ramírez and Ornelas, 2009; 2012). In Psittacanthus calyculatus G. Don and P. schiedeanus (Cham. & Schltdl.) G. Don, fruit size differs among mistletoes growing on different host tree species, with typically larger fruits in mistletoe growing on the most compatible host species (Lara et al., 2009; Ramírez and Ornelas, 2009; Rodríguez-Mendieta et al., 2018; Lara et al., 2021; Ornelas and Vásquez-Aguilar, 2023), but these differences vary across space in P. calyculatus (Díaz Infante et al., 2016; Rodríguez-Mendieta et al., 2018; Lara et al., 2021). Ultimately, host-associated differences in fruit size might induce reproductive isolation of mistletoe populations infecting different host species (Rodríguez-Mendieta et al., 2018; Lara et al., 2021; Ornelas and Vásquez-Aguilar, 2023). Host-associated differences across space of reproductive traits in other mistletoe species growing on different host species are associated with genetic structure in sympatry (Yule et al., 2016; Yule and Bronstein, 2018; de Vega et al., 2024), facilitating the formation of host races and eventually speciation via host race formation (Zuber and Widmer, 2009).
To better understand the germination process of the seed unit, Ornelas et al. (2024b) investigated the different phases of seed germination of manually attached seeds of nine Psittacanthus species inhabiting dry and humid environments placed under common experimental conditions, from seed attachment to early haustorial formation. The study by Ornelas and collaborators (2024b) revealed that seed germination in Psittacanthus mistletoes involves stages such as viscin drying, seed coat breaking, and cotyledon expansion. These seeds possess a seed coat that safeguards the embryo, and convey environmental cues, and produce copious liquid exudates during germination, which is likely aiding attachment and penetration. Also, the study by Ornelas et al. (2024b) highlighted variability in the shape, size and number of cotyledons across Psittacanthus species, indicating a strong phylogenetic signal in fruit length and fruit width, as well as in the average cotyledon number. Interestingly, a positive association with germination rate and germination speed was observed, after accounting for phylogeny (Ornelas et al., 2024b). However, the direct effects of fruit size on seed germination were not assessed in the studied Psittacanthus species. Thus, the aim of this study is to investigate the effects of fruit size (length, width, and weight), length of the cupular pedicel of ripe fruits, and the number of cotyledons on seed germination in Psittacanthus mayanus Standl. & Steyerm. (Loranthaceae). Understanding these relationships will provide insights into the adaptive significance of fruit size variation in mistletoes and its potential role in host-race formation.
Material and Methods
Study species
Psittacanthus mayanus commonly grows in seasonally dry deciduous and semideciduous forests to seasonally dry deciduous forests with columnar cacti from near sea level up to 500 m elevation (Kuijt, 2009). It is distributed in Mexico, Guatemala and Belize, mostly in the Yucatán Peninsula and Chiapas, representing two divergent lineages marked by historical demographic stability and isolation (Licona-Vera et al., 2018). The species frequently parasitizes leguminous trees (Fabaceae), mainly Lysiloma Benth., Lonchocarpus Kunth, Acacia Mill., Pithecellobium Mart., Inga Mill. and Haematoxylum Gronov., and less frequently Coccoloba L. (Polygonaceae), Conocarpus L. (Combretaceae), Crescentia L. (Bignoniaceae), and Bursera Jacq. ex L. (Burseraceae) trees (Kuijt, 2009). Psittacanthus mayanus (Fig. 1) can easily be distinguished from other Psittacanthus species encountered in the region (e.g., P. rhynchanthus (Benth.) Kuijt) by its more delicate habit, the presence of prophylls (two minute foliar organs, transversely placed in a leaf axil, as a bracteole), by having 4-5 cotyledons, non-foliaceous lowest triad bracts, and straight, thinner flowers (Kuijt, 2009; Licona-Vera et al., 2018; J. F. Ornelas, pers. obs.). In its southern range, however, P. mayanus is sometimes difficult to separate from other Psittacanthus species: P. schiedeanus, P. calyculatus, P. breedlovei Kuijt, and P. minor Kuijt (Kuijt, 2009). However, P. breedlovei has extremely narrow linear-lanceolate leaves, <1 cm wide (P. mayanus has leaves >2 cm wide, ovate, ovate-lanceolate), and P. calyculatus is a more robust plant with denser inflorescences and buds to 6 cm long (P. mayanus is a slender plant with few flowers and buds to 3 cm long), while P. minor is endemic to Nicaragua (Kuijt, 2009). The few-flowered inflorescences of P. mayanus produce red-orange flowers mainly visited by Pampa pampa (R. Lesson, 1832) (= Campylopterus curvipennis pampa (Deppe, 1830)) and Amazilia Lesson, 1843 hummingbirds (Trochilidae), and its ripe, purplish-black fruits are consumed by several bird species, mainly Patagioenas cayennensis Bonnaterre 1792, Myiozetetes similis Spix 1825, Pitangus sulphuratus Linnaeus 1766, Megarhynchus pitangua Linnaeus 1766, and Piranga roseogularis Cabot 1846 (Howell, 1972; Licona-Vera et al., 2018).

Figure 1: Morphology of Psittacanthus mayanus Standl. & Steyerm. A. Lysiloma latisiliquum (L.) Benth. (Fabaceae), a frequent leguminous tree species parasitized by P. mayanus; B. terminal inflorescence with buds and open flowers, each with cupular bract and long, orange floral petals, stamens and filament; C. developed unripe and ripe black fruits with prominent orange cupular pedicel. Photos by José Manuel García-Enríquez (A-B) and Juan Francisco Ornelas (C) at Calderitas on the road to Luis Echeverría, in the municipality of Othón P. Blanco, Chetumal, Quintana Roo, Mexico, in 2023.
Sampling
On November 21, 2023, we collected fully developed unripe and ripe fruits of Psittacanthus mayanus at three different locations near Calderitas on the road to Luis Echeverría, in the municipality of Othón P. Blanco, Chetumal (18°34'44.49''N, 88°15'35.54''W; 18°34'19.79''N, 88°15'38.32''W; 18°34'13.78''N, 88°15'38.14''W; at 4-5 meters above sea level), Quintana Roo, Mexico (Fig. 1). Seeds came from ripe fruits of three plants growing on three different individuals of Lysiloma divaricatum (Jacq.) J.F. Macbr. (Fabaceae), one of the most frequent host species in the Yucatán Peninsula. The fruits were taken to the laboratory (Instituto de Ecología, A.C., Xalapa, Veracruz, Mexico) to measure the length of the cupular pedicel, as well as the lengths and widths of ripe fruits using a digital caliper (Mitutoyo, Aurora, Illinois, USA), with a precision of 0.1 mm (following Ornelas and Vásquez-Aguilar, 2023). Fruits were also individually weighed with a digital scale (OHAUS PioneerTM, Newark, New Jersey, USA: error 0.01 g).
Seed germination and growth conditions
To perform the seed germination experiment (26 November 2023-5 March 2024), one-seeded ripe fruits (n = 37) had their exocarp manually removed by gently squeezing the tip of the fruit by hand. The seeds were then directly placed at 5 cm from each other and glued with their viscin onto the surface of wooden rectangle sticks of 30-50 cm long (1.5 cm thick ( 4.5 cm wide, approximately). During the experiment, the wooden rectangle sticks with naturally glued seeds were kept in a sheltered back porch/garden, exposed to natural environmental conditions during winter at a location 6 km W of the city of Xalapa, Veracruz, Mexico (19°30'25''N, 96°57'39''W; at 1348 m above sea level; see also Ornelas et al., 2024b). We maintained moisture levels by daily misting the sticks with distilled water using a spray bottle.
Throughout the experiment, we recorded and monitored the different stages of germination of each seed according to the results of a seed inoculation experiment for several species of Psittacanthus (Ornelas et al., 2024b), starting from seed inoculation or planting to cotyledon expansion. We documented the timing and the chronological development of each stage, tracking the drying of the viscin, the breakdown of the seed coat, and the onset and completion of cotyledon expansion. We also recorded the number of planted seeds that germinated and the number of cotyledons per germinated seed. In the context of our study, cotyledon expansion refers specifically to the opening of the cotyledons during the germination process, rather than subsequent growth phases (Ornelas et al., 2024b). This approach allowed us to monitor the different stages of germination on a surface under similar controlled experimental conditions (see also Ornelas et al., 2024b for further details). The experimental attached seeds were closely observed, photographed and followed for about 100 days, spanning stages from seed planting to presumed haustorium development (see further details in Ibarra-Laclette et al., 2022; Ornelas et al., 2024b).
Data analysis
We used the GerminaR package (Lozano-Isla et al., 2019) with the web application GerminaQuant for R environment (R Development Core Team, 2024) to analyze the probability of germination (expansion of cotyledons) of each experimentally inoculated seed, followed by approximately 100 days after inoculation. We calculated the following germination indices: (a) germination or germinability (GNP) as the percentage of seeds that expanded cotyledons from those inoculated during the time of the experiment; (b) mean germination time (MGT), which denotes the number of seeds germinated (cotyledons expanded) with respect to the number of seeds inoculated during the time of the experiment; (c) germination speed coefficient (GSP) as a measure of the rate and time-spread of germination; (d) mean germination rate (MGR), expressed as the reciprocal of MGT; (e) germination uncertainty (UNC), which evaluates the uncertainty associated with the relative distribution of the frequency of germination; (f) germination synchrony (SYN), where synchrony is equal to 1 when germination occurs at the same time, whereas synchrony near 0 denotes that at least two seeds complete the germination process at different times; (g) index of germination variance (VGT); (h) germination standard deviation (SDG); and (i) germination coefficient of variation (CVG). The germination speed coefficient (GSP) is synonymous with the coefficient of velocity of germination (CVG) (Jones and Sanders, 1987), as both indicate germination velocity. Germination variables were evaluated following Ranal and de Santana (2006), where n i is the number of seeds germinated (expanded cotyledons) in i th time; and k is the last day of the evaluation process for germination (for more information about these functions, see Lozano-Isla et al., 2019, and references therein).
To examine the relationships between fruit morphological traits and seed germination parameters in Psittacanthus mayanus, we implemented a multi-model analytical approach. Germination success (binary response: germinated/not germinated) was analyzed using a generalized linear model (GLM) with binomial error distribution and logit link function. The model included four fruit traits as predictor variables: cupular pedicel length (mm), fruit length (mm), fruit width (mm), and fruit weight (g). The significance of individual predictors was assessed using likelihood ratio tests with sequential addition of terms (Type I analysis of deviance), and model fit was evaluated using residual deviance and Akaike Information Criterion (AIC). We estimated the volume of the 37 mistletoe fruits assuming that they are ovoid or ellipsoidal in shape. Then, we correlated the length and width of the fruit with these volumes and the resulting correlations were positive and highly significant (length and ovoid volume: r = 0.893, P < 0.001; fruit length and ellipsoidal volume, r = 0.853, P < 0.001; fruit width and ovoid volume: r = 0.859, P < 0.001; fruit width and ellipsoidal volume: r = 0.920, P < 0.001), indicating that these correlations are not due to chance. Based on these results, we used the fruit length and fruit width values rather than adding fruit volume as an additional response variable.
For continuous response variables (days until germination, days until full cotyledon expansion, and cotyledon number), we fitted separate multiple linear regression models with the same set of predictor variables. Model assumptions were evaluated through diagnostic plots, including residual plots for homoscedasticity and normal Q-Q plots for residual normality. The significance of individual predictors was assessed using t-tests, with statistical significance set at α = 0.05. Model performance was evaluated using adjusted R² values and F-statistics. There is a significant risk primarily due to sampling error that our multivariate analysis might not have enough statistical power. This means that the ability to detect a true effect or relationship between variables is diminished. All statistical analyses were performed in R v. 4.0.2 (R Development Core Team, 2024).
Results
Seeds of Psittacanthus mayanus exhibited high germinability, with 89.19% (33 out of 37 seeds) completing germination during the 100-day experiment. The mean germination time (MGT) was 11.0 ± 1.4 days, with a germination speed (GSP) of 9.09% and a mean germination rate (MGR) of 0.091 time⁻¹ (the reciprocal of MGT, days). The synchronization index (SYN = 0.138) indicated relatively asynchronous germination among seeds, which was further supported by a high uncertainty index (UNC = 2.849). Germination showed considerable temporal variation, as evidenced by the germination variance (VGT = 67.188 time²), standard deviation (SDG = 8.197 time), and coefficient of variation (CVG = 74.516%).
The relationship between fruit morphological traits and germination parameters revealed that fruit width was the most influential predictor (Fig. 2). In the binomial model analyzing germination probability, fruit width was the only significant predictor (χ² = 4.125, df = 1, P = 0.042), with wider fruits associated with higher germination success (Table 1). Similarly, fruit width significantly influenced the time to germination (β = -17.534 ± 8.329, t = -2.105, P = 0.043), indicating that seeds from wider fruits germinated more rapidly (Fig. 3A). The number of cotyledons was also significantly related to fruit width (β =-0.422 ± 0.183, t =-2.309, P = 0.029), with wider fruits producing seeds with fewer cotyledons (Fig. 3B). Other fruit traits (pedicel length, fruit length, and fruit weight) showed no significant relationships with any of the measured germination parameters (all P > 0.05; Table 1).
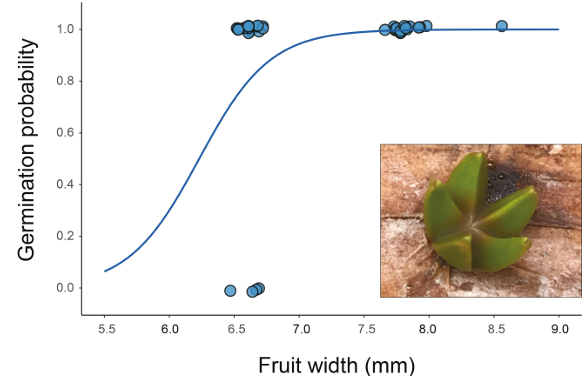
Figure 2: Relationship between fruit width and germination success in Psittacanthus mayanus Standl. & Steyerm. The logistic regression curve (blue line) shows the predicted probability of germination in relation to fruit width. Each point represents an individual seed (n = 37 seeds), where 1 indicates successful germination and 0 indicates no germination (logistic regression: χ² = 4.125, df = 1, P = 0.042).
Table 1: Effects of fruit morphological traits (pedicel length, fruit length, width and weight) on germination success, time to germination, and cotyledon number in Psittacanthus mayanus Standl. & Steyerm. seeds, based on (A) binomial generalized linear model (germination probability) and (B) linear regression models (days until germination and cotyledon number). *Significant at p < 0.05.
| Response variable | Predictor | Estimate ± SE | Test statistic | p -value |
| (A) Germination probability | Pedicel length | 0.618 ± 0.498 | X2 = 2.329 | 0.125 |
| Fruit length | -0.070 ± 0.848 | X2 = 0.037 | 0.848 | |
| Fruit width | 4.335 ± 4.782 | X2 = 4.125 | 0.042* | |
| Fruit weight | -9.173 ± 18.539 | X2 = 0.243 | 0.622 | |
| (B) Days until germination | Pedicel length | -4.887 ± 3.902 | t = -1.252 | 0.220 |
| Fruit length | -2.108 ± 6.515 | t = -0.324 | 0.748 | |
| Fruit width | -17.534 ± 8.329 | t = -2.105 | 0.043* | |
| Fruit weight | 96.255 ± 143.477 | t = 0.671 | 0.507 | |
| Cotyledon number | Pedicel length | 0.054 ± 0.093 | t = 0.587 | 0.562 |
| Fruit length | -0.147 ± 0.143 | t = 1.024 | 0.315 | |
| Fruit width | -0.422 ± 0.183 | t = 2.309 | 0.029* | |
| Fruit weight | 5.414 ± 3.421 | t = 1.583 | 0.125 |
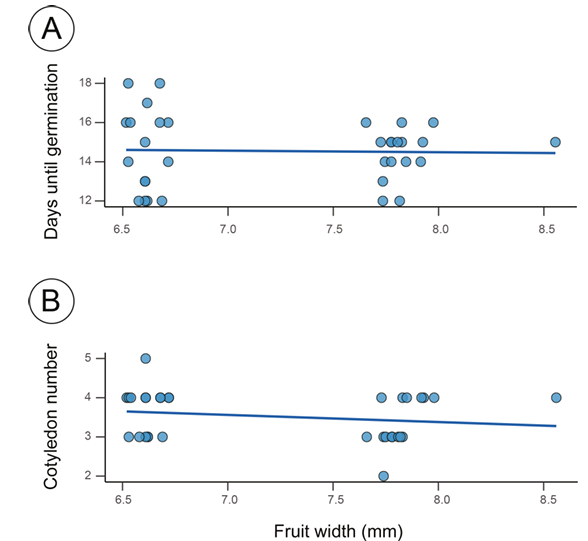
Figure 3: Relationship between fruit width and germination parameters in Psittacanthus mayanus Standl. & Steyerm. (A) Time to germination in relation to fruit width (linear regression: β = -17.534 ± 8.329, t = -2.105, P = 0.043). (B) Number of cotyledons in relation to fruit width (linear regression: β = -0.422 ± 0.183, t = -2.309, P = 0.029). Each point represents an individual seed that successfully germinated (33 out of 37 seeds). Blue lines represent fitted linear regressions.
Discussion
Germination is considered one of the most important stages influenced by environmental factors, with timing and abundance determining plant establishment and recruitment. For mistletoes, research has focused on the consequences of directed bird dispersal and effects of host provenance and host fate on seed germination to understand seedling establishment and seed deposition patterns. However, the effects of fruit size variation on seed germination processes, particularly the critical early stages from seed deposition by seed dispersers and host-branch adherence to the haustorial penetration into host branches, are poorly understood. Our study reveals significant patterns in the germination biology of Psittacanthus mayanus. Most notably, fruit morphology plays a crucial role in seed germination success, with fruit width emerging as the sole significant predictor among the morphological traits examined. This trait influences not only germination probability but also germination timing and cotyledon development. Seeds from wider fruits showed higher germination probability and faster germination rates, although they developed fewer cotyledons. These findings align with previous studies showing the importance of fruit morphology in mistletoe establishment (Sargent, 1995; Gonzáles et al., 2007).
Comparison of germination indices across Psittacanthus species reveals that P. mayanus exhibits remarkable germination characteristics. With a germination rate of 89.19% (fruit length = 8.9 mm, fruit width = 7.2 mm), it ranks among the highest within the genus, comparable to P. schiedeanus (87.5-92.3%) and P. macrantherus (88%) with much larger fruits (P. schiedeanus: fruit length = 14.4 mm, fruit width = 10.2 mm; P. macrantherus: fruit length = 24 mm, fruit width = 18 mm), and considerably higher than P. calyculatus (52.17%) and P. rhynchanthus (40-75%) with larger fruits as well (P. calyculatus: fruit length = 13.9 mm, fruit width = 9.9 mm; P. rhynchanthus: fruit length = 12.2 mm, fruit width = 9.2 mm; Ornelas et al., 2024b). The mean germination time of 11 days in P. mayanus represents one of the fastest germination responses in the genus, substantially quicker than lowest P. rhynchanthus (53-60 days) and second lowest P. calyculatus (18-25 days), though not as rapid as fastest P. schiedeanus (4-7.8 days; Ornelas et al., 2024b). The relatively high germination speed coefficient (GSP = 9.09) further supports this rapid germination pattern (Ornelas et al., 2024b). Notably, P. mayanus shows considerable asynchrony in germination timing (SYN = 0.138) and high variation in germination responses (CVG = 74.516%), suggesting a bet-hedging strategy that could enhance establishment success across variable environmental conditions or different host species, as observed in other mistletoe species (Norton and Ladley, 1998; López de Buen and Ornelas, 2002; Rodríguez-Mendieta et al., 2018). These germination characteristics might represent adaptations to the seasonally dry tropical deciduous forests where P. mayanus typically occurs (Kuijt, 2009), allowing rapid establishment during favorable conditions, while maintaining variability in germination timing as a risk-spreading strategy. The study by Ornelas et al. (2024b) revealed that seed germination in Psittacanthus mistletoes involves stages such as viscin drying, seed coat breaking, and cotyledon expansion. Seeds possess a seed coat that safeguards the embryo, conveys environmental cues, and produces copious liquid exudates during germination, likely aiding attachment and penetration. To ensure that the post-emergence environmental conditions are favorable for seedling growth and survival, some species have developed fast-response germination strategies, like P. schiedeanus and P. mayanus, and others remain seemly arrested, or their cotyledons remain unexpanded and encapsulated by dried viscin and seed coat (Ornelas et al., 2024b). Additionally, the variation in germination timing we observed could represent an adaptation to the seasonally dry tropical deciduous forest where P. mayanus occurs, similar to patterns observed in other mistletoes where temporal variation in seed dispersal and germination influences population structure (López de Buen and Ornelas, 1999; Watson, 2009). Moreover, this pattern might be influenced by the specific host preferences of P. mayanus in dry forest environments (Licona-Vera et al., 2018), where rapid establishment through efficient resource allocation could be particularly advantageous. However, potential explanations for the observed germination uncertainty (UNC), low germination synchrony (SYN), and high germination variance (VGT, SDG, CVG) in P. mayanus require increasing sample size in further research to explore the effects of timing, location, host species, and variation in mistletoe fruit traits.
The significant effect of fruit width, rather than other morphological traits, on germination success in P. mayanus provides new insights into the adaptive significance of fruit characteristics in mistletoes. The revealed importance of fruit width could be related to optimal resource allocation during seed development or to mechanical advantages during seed dispersal and attachment to host branches, as suggested for other Loranthaceae species (Watson, 2009; Ramírez and Ornelas, 2012). Our findings that wider fruits were associated with higher germination probability align with patterns observed across many plant groups and with previous studies on other Psittacanthus species that demonstrate the importance of fruit size in reproductive success. Studies across various plant taxa have shown that wider fruits often produce seeds with higher germination rates and faster germination times (see for example Murali, 1997; Kidson and Westoby, 2000; Gómez, 2004; van Mölken et al., 2005; Souza and Fagundes, 2014). This pattern is typically explained by the relationship between fruit width and seed resource allocation, in which wider fruits can accommodate seeds with more stored reserves to support the germination process. Moreover, the effect of fruit width on germination probability would be particularly relevant in mistletoes given their host dependence and specialized life history, where the timing of germination, host attachment and success in haustorial penetration need to coincide with suitable conditions on the host branch.
In Psittacanthus calyculatus, fruit size variation is associated with the identity and quality of the host species, suggesting differential host local adaptation (Rodríguez-Mendieta et al., 2018; Lara et al., 2021). Similarly, Ramírez and Ornelas (2009) found that larger fruits in P. schiedeanus were preferentially selected by avian dispersers and showed higher germination success after gut passage. The importance of fruit width specifically might be related to mechanical advantages during seed adhesion, as wider fruits could contain more viscin tissue, which is crucial for initial adhesion to host branches (Ibarra-Laclette et al., 2022). This aligns with previous findings in P. calyculatus where fruit size variation was shown to affect successful establishment through both direct effects on seed adhesion and indirect effects on bird handling behavior (Lara et al., 2009). Additionally, this pattern could reflect resource allocation trade-offs, where increased fruit width allows for optimal endosperm development, potentially providing more resources for early haustorium formation (Watson, 2009). The host specialization patterns observed in P. mayanus, primarily on leguminous trees in the Yucatán Peninsula (Kuijt, 2009; Licona-Vera et al., 2018), might also influence selection on fruit characteristics, as host bark characteristics and branch architecture can affect seed deposition patterns (López de Buen and Ornelas, 2002).
Germination response depends on phylogeny, life history traits and environmental signals (e.g., Xu and Du, 2023). Across the range of hemiparasitic mistletoe species, the host characteristics and the spatial and temporal environmental changes can lead to variation in selection pressures that could affect mistletoe growth and establishment. The first mistletoe-environment interaction occurs during the most fragile and critical early stages in the process of seed germination (Watson, 2009), and host dependence and environmental cues at these early life history stages become a potential bottleneck of population recruitment (Barga et al., 2017; Ornelas et al., 2024b). The negative relationship we found between fruit width and cotyledon number in P. mayanus represents an intriguing trade-off in early developmental strategies. This inverse relationship suggests a possible resource allocation pattern, where wider fruits invest more in endosperm or embryo development at the expense of cotyledon number. Such a trade-off aligns with recent findings by Ornelas et al. (2024b), who documented significant variation in cotyledon characteristics across Psittacanthus species, with a strong phylogenetic signal in both fruit dimensions and cotyledon number. After accounting for phylogeny, cotyledon number was negatively associated with mean germination time and germination uncertainty, while positively associated with mean germination rate and germination speed coefficient (Ornelas et al., 2024b). Interspecific variation in cotyledon number, seed coat characteristics, and in the timing and duration of seed germination stages highlights distinct processes, likely influenced by environmental differences. Then, the adaptive significance of fewer cotyledons in seeds from wider fruits might be related to the initial establishment phase, as demonstrated in other Loranthaceae species where cotyledon number influences early haustorial development (Watson, 2009). In P. schiedeanus, for example, Ibarra-Laclette et al. (2022) found that successful haustorium formation depends on coordinated gene expression during early development stages, suggesting that resource allocation during seed development could be crucial for establishment success. This trade-off might also reflect different strategies for host exploitation, as observed in P. calyculatus, where establishment success varies with host species and local conditions (Lara et al., 2021). The variation in cotyledon number could represent a bet-hedging strategy, similar to what has been observed in other mistletoe species where morphological plasticity enhances establishment success across different host species (Gonzáles et al., 2007).
Our findings on the relationship between fruit morphology and germination patterns in Psittacanthus mayanus have broader implications for understanding the evolution of the host-parasite interaction and population structure in mistletoes. The significant effect of fruit width on germination success could influence patterns of host specialization, particularly given that P. mayanus shows strong preferences for leguminous hosts in the Yucatán Peninsula (Kuijt, 2009; Licona-Vera et al., 2018). Host specificity, combined with variation in fruit characteristics, might contribute to the formation of host races, a phenomenon documented in other mistletoes (Zuber and Widmer, 2009; Yule et al., 2016; Yule and Bronstein, 2018; de Vega et al., 2024) and Psittacanthus species such as P. calyculatus (Pérez-Crespo et al., 2016) and P. schiedeanus (Ramírez and Ornelas, 2012). Indeed, experimental evidence has shown that successful seed germination and seedling establishment in Psittacanthus depend on the interaction between host provenance (the tree species from which fruits originate) and host fate (the tree species where seeds are deposited) (Lara et al., 2009; Ramírez and Ornelas, 2012). The role of avian dispersers in this system is particularly relevant, as frugivorous birds (i.e., various flycatcher species such as Myiozetetes similis, Tyrannus vociferans Swainson 1826 and Pitangus sulphuratus, and Ptilogonys cinereus (Swainson 1827) might selectively feed on fruits of different sizes, potentially affecting seed dispersal and gene flow patterns among host-specialized populations (López de Buen and Ornelas, 1999; Lara et al., 2009; Ramírez and Ornelas, 2009, 2012; Díaz Infante et al., 2016).
Conclusions
Our study reveals the critical role of fruit width in the germination success of Psittacanthus mayanus, contributing to our understanding of the reproductive biology of mistletoes. These findings build upon previous work demonstrating the importance of fruit characteristics in mistletoe establishment (Sargent, 1995; Watson, 2009), while highlighting new aspects of the relationship between fruit morphology and early developmental stages. Future research should investigate whether the patterns we observed are consistent across different host species and environmental conditions, particularly given the habitat and host range documented in Psittacanthus species from specialist to generalist species (e.g., López de Buen and Ornelas, 2002; Ornelas et al., 2018, 2019; Rodríguez-Mendieta et al., 2018). Additionally, experimental studies examining the mechanistic basis of the relationship between fruit width and cotyledon development could provide insights into the developmental processes underlying successful establishment, building on recent molecular studies of haustorium formation (Ibarra-Laclette et al., 2022). The potential role of climate change in affecting these relationships also merits investigation (Ornelas et al., 2018; Fontúrbel, 2020; Fadini et al., 2024), as environmental stress can influence both fruit development and seed germination, as well as establishment patterns in parasitic plants (Ramírez and Ornelas, 2012; Lara et al., 2021). Furthermore, investigating the genetic basis of fruit width variation and its relationship to host race formation could enhance our understanding of speciation processes in mistletoes (Pérez-Crespo et al., 2016; Licona-Vera et al., 2018). Lastly, from a conservation perspective, understanding these relationships is crucial as mistletoes are important components of forest ecosystems, serving as keystone species that support biodiversity through direct and indirect interactions (Watson and Herring, 2012). Such research directions would not only advance our understanding of mistletoe biology, but also contribute to broader questions in plant evolutionary ecology and host-parasite interactions.











 nueva página del texto (beta)
nueva página del texto (beta)



