Introduction
Mushroom cultivation is considered a relatively primitive agricultural activity for a highly technological industry. However, in each case, the continued production of successful crops requires practical experience and scientific knowledge. Pleurotus ostreatus (Jacq.) P. Kumm. is often seen as one of the easiest and most profitable mushrooms to grow at different scales, both commercially and experimentally. The cultivation of this species begins with (1) selection of a good quality fruiting body, (2) development of the mycelium, (3) preparation of substrate or compost, (4) care of the growth of the mycelium, (5) management of the development and fruiting of the mushrooms, and (6) careful harvesting of the fruiting body (Chang, 2009; Sánchez, 2010; Rodríguez-Estrada and Pecchia, 2017).
There are various cultivation methods to obtain fruiting bodies of fungi, among which are ready-to-grow kits, which include a mass of mushroom mycelium ready to enter the fruiting stage. Another culture method widely used in the industry is cultivation in a liquid medium, with which the efficient production of mycelium biomass and fungal metabolites is achieved. However, the cultivation in a solid medium is one of the methods applied on a large scale; it is a fermentation process that involves a solid matrix and is carried out with little free water, providing sufficient moisture to support the growth and metabolism of microorganisms, and which has been used to release phenolic compounds and improve the antioxidant capacity of plant materials (Singhania et al., 2009; Petre and Petre, 2016).
Species belonging to the genus Pleurotus (Fr.) P. Kumm. are saprophytic organisms that obtain nutrients from the substrate, such as grass, wood, and agricultural residues, through their mycelium, acquiring substances necessary for its development, such as carbon, nitrogen, vitamins, and minerals. For this reason, it has been reported that the substrates used to cultivate P. ostreatus affect the fungus's chemical, sensory characteristics, and functional properties (Oyetayo, 2013; Bellettini et al., 2019). Various agricultural and industrial wastes are used to cultivate Pleurotus spp., such as cereal straw, cotton waste, sawdust, cardboard, residues from the coffee industry, and wastepaper, among others (Nguyen and Ranamukhaarachchi, 2020). However, there is no evidence that spent coffee grounds (SCG) and potato peel residues (PPR) are used as Pleurotus cultivation substrates.
It has been demonstrated that SCG and PPR exert some techno-functional properties (water and oil retention capacity, capacity and stability of the emulsion, among others) and biological activity; thus, they are considered a potential additive for pharmaceutical and food industries (Ballesteros et al., 2014; Singh et al., 2022). Previous work evaluated the extraction of antioxidant compounds from SCG, finding a strong antioxidant activity attributed to the total phenolic and flavonoid content and the presence of chlorogenic and protocatechuic acids (Mussatto et al., 2011). Also, potato peel extracts exert potent antioxidant and antibacterial activity, which has also been attributed to phenolic components (Helal et al., 2020). Besides, the total phenolic, tannin, and flavonoid content was previously evaluated, as well as the antioxidant properties of P. ostreatus grown in various wastes and sawdust, obtaining the highest content of flavonoids, as well as inhibiting the DPPH (1,1-diphenyl-2-picrylhydrazyl) radical for fungi grown with these agro-industrial residues (Yılmaz et al., 2017). Based on the above, SCG and PPR could enhance the nutrient composition and phytochemicals profile.
Despite the use of agro-industrial waste as a substrate for mushroom cultivation, research has focused on the transformation of waste, underestimating the benefits that these transformations bring to the chemical and metabolite composition of the fruiting body and mycelium of the fungus, which could be an interesting source of compounds with potential use as food additives. Therefore, this study aimed to evaluate the effect of SCG and PPR as a substrate on the polyphenol composition and antioxidant and antimicrobial activity of P. ostreatus mycelium.
Materials and Methods
Chemicals and reagents
All the chemical products used were of analytical grade. Ethanol, urea, glacial acetic acid, hydrochloric acid (HCl), trichloroacetic acid (TCA), nitrate (NaNO2) Folin-Ciocalteu reagent, sodium carbonate (Na2CO3), aluminum chloride (AlCl3), 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azinobis-(-3 ethylbenzothia-zoline-6-sulfononic acid) diammonium salt (ABTS), 4,4,6-tripyridyl-S-triazine (TPTZ), sodium phosphate, potassium ferricyanide, iron chloride, were purchased from Sigma Chemicals®. Gallic acid, quercetin, chlorogenic acid, and ascorbic acid were acquired from the same supplier. In addition, potato dextrose agar (PDA) and brain heart infusion medium (BHI) were purchased from Difco®. Spent coffee grounds and potato peel residues were donated by commercial suppliers (CAFFENIO® and La Costeña®, respectively).
Obtaining mycelium
Filamentous P. ostreatus strain (IE-8), belonging to the fungal collection of the Plant-based Food Technology Department (Centro de Investigación en Alimentación y Desarrollo, Hermosillo, Mexico), was grown on Petri dishes containing PDA medium (IC403C, Yamato Scientific Co., Tokyo, Japan), at 25 °C for five days until mycelium entirely covered the surface, and stored at 4 °C. After that, 1/3 of the plate surface with mycelium was incorporated into the inoculum seed contained in plastic bags. Previously, wheat grains (Triticum aestivum L.) were hydrated (overnight), drained to remove water exceeds, and sterilized (SM300, Yamato Scientific Co., Tokyo, Japan) at 121 °C for 1 h to prepare the inoculum seed. The inoculated bags were stored in the dark at 28 °C until the mycelium completely covered the surface of the seed (white coloration). Subsequently, wheat straw was used as basal substrate and mixed at different ratios of supplementing residues, as follows: T1, wheat straw at 100%; T2, wheat straw at 80% + 10% of SCG + 10% of PPR; T3, wheat straw at 70% + 15% of SCG + 15% of PPR; T4, wheat straw at 60% + 20% of SCG + 20% of PPR. The wet and sterilized substrates were inoculated with the inoculum seed covered with mycelium (10% w/w) and stored in the dark at 28 °C until mycelium covered the surface. After that, samples were stored dark at 25 °C with a photoperiod of 12 h and a relative humidity between 80-90% for three weeks (Sánchez et al., 2002; Singh et al., 2022).
Obtaining extracts
Bioactive compounds from mycelium were extracted with a solvent mixture in a 1:10 proportion (water-ethanol, 1:1) by the ultrasound-assisted method (42 KHz, 25 °C, 30 min). The resultant mixture was filtered (with Whatman 1 filter paper) under a vacuum (FE-1500, Felisa, Mexico), concentrated under reduced pressure at 60 °C (RE301BW, Yamato Scientific Co., Tokyo, Japan), and lyophilized (DC401, Yamato Scientific Co., Tokyo, Japan). The dried hydroalcoholic extract from PO mycelium was stored at -20 °C under dark conditions until further analysis (Ainsworth and Gillespie, 2007).
Total phenolic content
Total phenolic content was determined by the Folin-Ciocalteu assay (Ainsworth and Gillespie, 2007). The extract (20 µl, 10 mg/ml water-ethanol, 1:1) was transferred into a well of a flat microplate (96-well) and mixed with 160 µl of distilled water, 60 µl of Na2CO3 (7% w/v), and 40 µl of Folin-Ciocalteu reagent (2 M). The resulting mixture was incubated in the dark at room temperature (25 °C) for 1 h. Subsequently, the absorbance was measured at 750 nm in a spectrophotometer (Multiskan GO, Thermo Scientific, USA). The results were calculated from a standard curve (62.5 to 1000 µg/ml) and expressed as mg equivalents of gallic acid per ml of extract (mg GAE/g).
Total flavonoid content
Total flavonoid content was determined by the NaNO2-Al(NO3)3-NaOH assay (Zhishen et al., 1999). The extract (500 µl, 10 mg/ml water-ethanol, 1:1) was mixed with 1 ml of NaNO2 (5% w/v), 1 ml of AlCl3 (10% w/v), and 10 ml of NaOH (1 M). The resulting mixture was adjusted to 25 ml with ethanol (70%) and incubated in the dark at 25 °C for 15 min. Subsequently, the absorbance was measured at 510 nm. The results were calculated from a standard curve (62.5 to 1000 µg/ml) and expressed as mg quercetin equivalents per ml (mg QE/g).
Total chlorogenic acid content
Total chlorogenic acid content was also determined (Griffiths et al., 1992). The extract (100 μl, 10 mg/ml water-ethanol, 1:1) was homogenized with 200 μl of urea (0.17 M), 200 μl of glacial acetic acid (0.1 M), and 500 μl of distilled water. The resulting mixture was mixed with 500 μl of NaNO2 (0.14 M) and 500 μl of NaOH (1 M), centrifuged at 2250 × g for 10 min at 4 °C (Sorvall ST18R, Thermo Fisher Scientific, Waltham, USA). The absorbance was measured at 510 nm. The results were calculated from a standard curve (62.5 to 1000 µg/ml) and expressed as mg equivalents of chlorogenic acid per ml (mg CAE/g).
Free-radical scavenging activity
The free-radical scavenging activity was determined using the DPPH assay (Molyneux, 2004). The extract (100 µl, 10 mg/ml water-ethanol, 1:1) was homogenized with 100 µl of DPPH solution (300 µM) and incubated in the dark at 25 °C for 30 min. The absorbance was measured at 517 nm. As a standard, ascorbic acid (70 µM) was used. The results were calculated as follows:
Where Abs A is the absorbance of the DPPH solution (t=0), and Abs B is the absorbance of the used antioxidant (t=30 min).
Radical cation scavenging activity
The radical cation scavenging activity was determined by the ABTS+ assay (Re et al., 1999). Before analysis, equal parts of the ethanolic solution of the ABTS radical (7 mM) and potassium persulfate (2.45 M) were homogenized in the dark at 10,000 rpm for 1 min. The resulting mixture was stored in the dark at 5 °C for 16 h. The radical formed (180 µl, adjusted at 0.8 abs in ethanol) was mixed with 20 µl of the extract (10 mg/ml water-ethanol, 1:1) and incubated in the dark at 25 °C for 10 min. The absorbance was measured at 734 nm. As a standard, ascorbic acid (70 µM) was used. The results were calculated as follows:
Where Abs A is the absorbance of the ABTS+ solution (t=0), and Abs B is the absorbance of the used antioxidant (t=10 min).
Reducing power ability
The reducing power ability was determined by the ferricyanide/Prussian blue assay (Işıl Berker et al., 2010). The extract (100 µl, 10 mg/ml water-ethanol, 1:1) was homogenized with 300 µl of phosphate buffer (0.2 M, pH 6.6) and 300 µl of potassium ferricyanide (1% w/v). The resulting mixture was incubated in the dark at 50 °C for 20 min in a water bath (Aquabath, Thermo Fisher Scientific, Ohio, USA). After that, the mixture was homogenized with 300 µl of TCA (10% w/v) and centrifuged at 4200 × g for 10 min at 4 °C. Then, 100 µl of the supernatant was mixed with 100 µl of distilled water and 250 µl of ferric chloride (0.1% w/v). As a standard, ascorbic acid (70 µM) was used. The absorbance was measured at 700 nm. The results were expressed as absorbance at 700 nm.
Ferric-reducing antioxidant power
The ferric-reducing antioxidant power was determined (Işıl Berker et al., 2010). The extract (20 µl, 10 mg/ml water-ethanol, 1:1) was homogenized with 150 µl of FRAP solution (10:1:1, 300 mM sodium acetate buffer in glacial acetic acid at pH 3.6 and 4.4.6-tripyridyl-S-triazine (TPTZ) in 40 nM of HCl and 20 mM of FeCl3). The resulting mixture was incubated in the dark at 25 °C for 8 min. The absorbance was measured at 595 nm. As a standard, ascorbic acid (70 µM) was used. The results were expressed as mg Fe2+ equivalents per ml (mg Fe2+/ml).
Antibacterial activity
The antibacterial activity was evaluated using the broth microdilution assay (Jorgensen and Turnidge, 2015) with slight modifications. Four standard bacteria strains were used, including two Gram-positive (Staphylococcus aureus Rosenbach 1884 ATCC® 29213 B and Listeria monocytogenes (Murray et al. 1926) Pirie 1940, ATCC® 33090) and two Gram-negative (Escherichia coli (Migula 1895) Castellani & Chalmers 1919, ATCC® 25922, and Salmonella typhimurium (Loeffler 1892) Castellani & Chalmers 1919, ATCC® 14028) strains. The strains were reactivated in liquid nutrient broth (BHI, brain heart infusion) at 37 °C for 24-48 h. After that, the strains' suspension was diluted with saline solution until the turbidity of 0.5 McFarland standard, BaSO4 (ca. 1.5 × 108 CFU/ml; 0.08 to 0.13 abs at 620 nm). Subsequently, the extract (20 µl, 10 mg/ml water-ethanol, 1:1) was homogenized with 80 µl of the diluted strains. As standard, gentamicin (0.025 mg/ml) was used, and BHI was used as a bacterial growth blank. The plates were incubated at 37 °C for 24 h, and the absorbance was read at 620 nm. The results were calculated as follows:
Where Abs A is the absorbance of the negative control at 24 h (bacteria without antibacterial), and Abs B is the absorbance of the used antibacterial at 24 h.
Statistical analysis
Experiments were conducted in triplicate with at least three independent trials (n=9). The results were presented as mean ± standard deviation (SD). The obtained data were subjected to a one-way analysis of variance (ANOVA). A Tukey-Kramer multiple comparison test was performed for mean separation (p≤0.05). In addition, a principal component analysis was performed to evaluate the relationships among the analyzed variables (SPSS, 2021).
Results
Polyphenol content
Table 1 shows the results of the total phenolic, flavonoid, and chlorogenic acid content of hydroalcoholic extract from PO mycelium. The phenolic content was greater (p≤0.05) than 40 mg GAE/g for T2-T4 treatments when compared to the T1-control sample (<40 mg GAE/g). Regarding flavonoid content, the results showed that T4 had the highest content, 45.04 mg QE/g (p≤0.05). The results showed no significant differences between treatments (p≥0.05) regarding chlorogenic acid content.
Table 1: Phenolic compound content of hydroalcoholic extract from Pleurotus ostreatus (Jacq.) P. Kumm. mycelium. T1=wheat straw at 100%; T2=wheat straw at 80% + 10% of SCG (spent coffee grounds) + 10% of PPR (potato peel residues); T3=wheat straw at 70% + 15% of SCG + 15% of PPR; T4=wheat straw at 60% + 20% of SCG + 20% of PPR. GAE=gallic acid equivalents; QE=quercetin equivalents; CAE=chlorogenic acid equivalents. Different letters indicate significant differences between treatments (p≤0.05).
| Treatments | Phenols | Flavonoids | Chlorogenic acid |
| (mg GAE/g) | (mg QE/g) | (mg CAE/g) | |
| T1 | 35.35 ± 2.19a | 39.67 ± 1.55a | 0.09 ± 0.01a |
| T2 | 42.38 ± 2.21b | 39.86 ± 1.47a | 0.09 ± 0.01a |
| T3 | 40.52 ± 2.43b | 42.30 ± 0.86a | 0.08 ± 0.01a |
| T4 | 44.48 ± 3.51b | 45.04 ± 1.22b | 0.08 ± 0.01a |
Antioxidant activity
Table 2 shows the results of the antioxidant activity of hydroalcoholic extract from PO mycelium. The results showed that positive control (ascorbic acid) exerts the highest (p≤0.05) antioxidant activity. However, when comparing the treatments, T3 and T4 showed higher DPPH inhibition (approximately 18%), and T4 showed higher ABTS+ inhibition (>30) (p≤0.05). In addition, no significant differences between treatments were found for RPA and FRAP values (p≥0.05).
Table 2: Antiradical activity and reducing power of hydroalcoholic extract from Pleurotus ostreatus (Jacq.) P. Kumm. mycelium. T1=wheat straw at 100%; T2=wheat straw at 80% + 10% of SCG (spent coffee grounds) + 10% of PPR (potato peel residues); T3=wheat straw at 70% + 15% of SCG + 15% of PPR; T4=wheat straw at 60% + 20% of SCG + 20% of PPR. DPPH=free-radical scavenging activity; ABTS+=radical cation scavenging activity; RPA=reducing power ability; FRAP=ferric reducing antioxidant power; ABS=Absorbance. Different letters indicate significant differences between treatments (p≤0.05).
| Treatments | DPPH (% Inhibition) | ABTS+ (% Inhibition) | RPA (Abs 700 nm) | FRAP (mg Fe2+/mL) |
| T1 | 4.76 ± 1.27a | 15.24 ± 1.77a | 0.31 ± 0.01a | 15.21 ± 0.72a |
| T2 | 8.09 ± 1.19b | 19.17 ± 3.25b | 0.31 ± 0.01a | 14.25 ± 0.72a |
| T3 | 18.41 ± 1.45c | 19.68 ± 3.36b | 0.31 ± 0.01a | 13.95 ± 0.68a |
| T4 | 18.93 ± 1.81c | 33.33 ± 1.78c | 0.32 ± 0.01a | 14.49 ± 1.50a |
| Vit C | 90.92 ± 1.05d | 94.48 ± 3.14d | 0.54 ± 0.03b | 25.71 ± 3.20b |
Antibacterial activity
Table 3 shows the results of the antibacterial activity of hydroalcoholic extract from PO mycelium. The results showed that positive control (gentamicin) exerts the highest (p≤0.05) antibacterial activity against S. aureus, L. monocytogenes, E. coli, and S. typhimurium. However, when comparing between strains, positive control and treatments (T1-T4) showed the highest effect (p≤0.05) against S. aureus. In addition, T2-T4 showed higher (p≤0.05) L. monocytogenes, E. coli, and S. typhimurium inhibition concerning T1.
Table 3: Antibacterial activity of hydroalcoholic extract from Pleurotus ostreatus (Jacq.) P. Kumm. mycelium (% Inhibition). T1=wheat straw at 100%; T2=wheat straw at 80% + 10% of SCG (spent coffee grounds) + 10% of PPR (potato peel residues); T3=wheat straw at 70% + 15% of SCG + 15% of PPR; T4=wheat straw at 60% + 20% of SCG + 20% of PPR. Different letters indicate significant differences between treatments (p≤0.05).
| Treatments | Gram-positives | Gram-negatives | ||
| Staphylococcus aureus Rosenbach 1884 | Listeria monocytogenes (Murray et al. 1926) Pirie 1940 | Escherichia coli (Migula 1895) Castellani & Chalmers 1919 | Salmonella typhimurium (Loeffler 1892) Castellani & Chalmers 1919 | |
| T1 | 68.57 ± 2.56a | 52.94 ± 2.63a | 36.11 ± 3.90a | 56.10 ± 2.18a |
| T2 | 68.10 ± 2.15a | 57.84 ± 2.40b | 43.33 ± 2.98ab | 60.98 ± 2.18b |
| T3 | 65.71 ± 2.56a | 58.82 ± 2.63b | 46.67 ± 2.98b | 61.38 ± 2.40b |
| T4 | 68.57 ± 3.13a | 58.33 ± 2.21b | 50.56 ± 2.51b | 65.45 ± 1.84b |
| Gentamicin | 72.38 ± 3.90b | 67.65 ± 2.63c | 68.33 ± 4.08c | 75.20 ± 3.91c |
Correlation between polyphenol content and bioactivity
Table 4 shows a Pearson correlation matrix of the evaluated parameters. The results showed a high positive association (>0.60) between phenolic content with DPPH, ABTS, and RPA values, and E. coli and S. typhimurium inhibition. Also, flavonoids content showed a positive association (>0.60) with DPPH and ABTS values, as well as with E. coli and S. typhimurium inhibition. Additionally, the association of S. aureus inhibition concerning phenolic and flavonoid content was low. Also, chlorogenic acid content showed the lowest association with the evaluated parameters.
Table 4: Pearson’s correlation matrix of the evaluated parameters. TPC=total phenolic content; TFC=total flavonoids content; CGA=chlorogenic acid; DPPH=free-radical scavenging activity; ABTS+=radical cation scavenging activity; RPA=reducing power; FRAP=ferric reducing antioxidant power; SA=Staphylococcus aureus Rosenbach 1884; LM=Listeria monocytogenes (Murray et al. 1926) Pirie 1940; EC=Escherichia coli (Migula 1895) Castellani & Chalmers 1919; ST=Salmonella typhimurium (Loeffler 1982) Castellani & Chalmers 1919. *Significant at p≤0.05. **Significant at p≤0.01.
| TPC | TFC | CGA | DPPH | ABTS+ | RPA | FRAP | SA | LM | EC | ST | |
| TPC | 1.000 | 0.528** | -0.644** | 0.734** | 0.674** | 0.716** | -0.312* | -0.012 | 0.573** | 0.777** | 0.727** |
| TFC | 1.000 | -0.348* | 0.819** | 0.768** | 0.487** | -0.029 | 0.051 | 0.377* | 0.625** | 0.624** | |
| CGA | 1.000 | -0.608** | -0.489** | -0.598** | 0.255 | 0.003 | -0.500** | -0.654** | -0.577** | ||
| DPPH | 1.000 | 0.765** | 0.560** | -0.139 | 0.074 | 0.556** | 0.805** | 0.714** | |||
| ABTS+ | 1.000 | 0.533** | -0.069 | 0.133 | 0.448** | 0.745** | 0.743** | ||||
| RPA | 1.000 | -0.280* | -0.003 | 0.497** | 0.653** | 0.588** | |||||
| FRAP | 1.000 | 0.211 | -0.138 | -0.080 | -0.049 | ||||||
| SA | 1.000 | 0.416** | 0.284* | 0.241 | |||||||
| LM | 1.000 | 0.853** | 0.837** | ||||||||
| EC | 1.000 | 0.962** | |||||||||
| ST | 1.000 |
Multivariate analysis
Figure 1 shows a principal component analysis, which was carried out to evaluate the differences between treatments and evaluated parameters. The first two components show a 56.86% and 13.44% variance, respectively, indicating that 70.3% of the total variation can be explained in the first two components. This graph shows a separation of the treatments regarding the antioxidant and antimicrobial activity; for example, the T4>T3 treatment presented higher total phenolic and flavonoid content, antiradical activity, and reducing power than other treatments. Besides, T1 and T2 showed weak inhibition of pathogenic microorganisms.
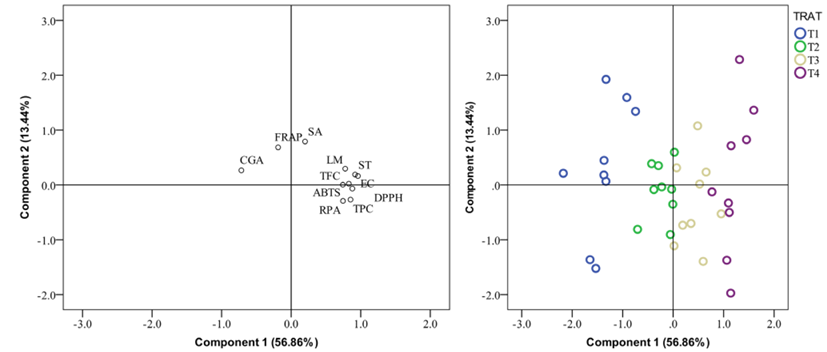
Figure 1: Principal component analysis of the evaluated parameters. TPC=total phenolic content; TFC=total flavonoids content; CGA=chlorogenic acid; DPPH=free-radical scavenging activity; ABTS+=radical-cation scavenging activity; RPA=reducing power; FRAP=ferric reducing antioxidant power; SA=Staphylococcus aureus Rosenbach; LM=Listeria monocytogenes (Murray et al. 1926) Pirie 1940; EC: Escherichia coli (Migula 1895) Castellani & Chalmers 1919; ST: Salmonella typhimurium (Loeffler 1892) Castellani & Chalmers 1919. T1=wheat straw at 100%; T2=wheat straw at 80% + 10% of SCG (spent coffee grounds) + 10% of PPR (potato peel residues); T3=wheat straw at 70% + 15% of SCG + 15% of PPR; T4=wheat straw at 60% + 20% of SCG + 20% of PPR.
Discussion
Edible mushrooms are known to be rich sources of secondary metabolites, particularly polyphenol compounds, which exhibit antioxidant activity (Valverde et al., 2015). It is well known that antioxidant activity is the ability of a substance to inhibit oxidative degradation (Shahidi and Zhong, 2015). The antioxidant activity of edible mushroom mycelium has been demonstrated (Kalyoncu et al., 2010a). Regarding antibacterial activity, it has been reported that the fruiting body and mycelium of edible mushrooms contain compounds with this property (Akyüz and Kirbag, 2009).
In this context, a previous investigation by González-Palma et al. (2016) demonstrated the antioxidant activity of P. ostreatus mycelium. The study evaluated extracts obtained from various growth stages of P. ostreatus, both fresh and dried samples. The results indicated that the aqueous extract of P. ostreatus mycelium exhibited significantly higher total polyphenol content and antioxidant activity than extracts from other growth stages. This suggests that the observed antioxidant activity could be attributed to the extract’s polyphenols and other bioactive compounds. In 2011, Vamanu et al. evaluated the impact of different nitrogen sources on the antioxidant activity of P. ostreatus M2191 mycelium cultivated under submerged cultivation. The research emphasized these mycelium ethanol extracts' inherent antioxidants and free radical scavenging properties, suggesting their potential role in health promotion and prevention of diseases associated with oxidative stress. Furthermore, the study revealed higher antibacterial effectiveness of these extracts against Gram-positive than Gram-negative bacteria (S. aureus > E. coli). Interestingly, the choice of nitrogen source was a critical factor in determining the suitability of these extracts as nutraceuticals and bioactive agents.
The study by Krupodorova et al. (2024) investigated the diverse biological activities exhibited by various strains of P. ostreatus. The authors discovered that the ethyl acetate extract obtained from P. ostreatus 2462 displayed notable antibacterial activity against S. aureus. In contrast, the ethyl acetate extract from P. ostreatus 2460 exhibited pronounced efficacy against E. coli. Additionally, the study demonstrated a range of DPPH• inhibition percentages, spanning from 30.9 to 61%, dependent on the specific strain of P. ostreatus and the solvents utilized for mycelium extract preparation, which was higher than in our work. The ethanol extracts from P. ostreatus 551, 2460, 2461, and 2462 showed higher DPPH• inhibition percentages than ethyl acetate extracts, except for P. ostreatus 1685. Moreover, the total phenolic content across all fungal extracts analyzed fell within the range of 2.76 to 7.52 mg of GAE per gram, which was lower than this work.
Furthermore, previous research has explored the quantification of polyphenols and the evaluation of antioxidant and antibacterial activity in ethanol extracts derived from P. ostreatus mycelium PQMZ91109 (Vamanu, 2012). The results showed the presence of total phenolic (approx. 0.66 mg GAE/g) and flavonoid content (approx. 3.9 mg QE/g), as well as high antiradical activity against radical 1,1-diphenyl-2-picrylhydrazyl (DPPH•, approx. 30% of inhibition at 10 mg/ml) and reducing power (approx. 0.5 abs). Also, the results showed a higher antibacterial effect against Gram-positive than Gram-negative bacteria (S. aureus > L. innocua > E. coli), as observed in our work. Another study demonstrated that ethanol extracts from P. ostreatus and P. eryngii (DC.) Quél. exert antioxidant (DPPH•, 6 and 11% of inhibition, respectively) and antibacterial activity against S. aureus, E. coli, and S. typhimurium (Kalyoncu et al., 2010b). A previous study indicates that hot-water extract from P. ostreatus mycelium showed antioxidant activity against DPPH• and ABTS•+ radicals (approx. 96 and 55% of inhibition, respectively) and RPA (1.1 Abs), as well as antibacterial activity against S. aureus, Bacillus subptilis (Ehrenb.) Cohn, Bacillus cereus Frankland & Frankland, and Pseudomonas aeruginosa (Schroeter) Migula, associated with the presence of phenols, tannins, and flavonoids; hence, it was recommended to cosmetic and pharmaceutical industries (Morris et al., 2017).
Moreover, Cardoso et al. (2017) analyzed the mycelia of Agaricus bisporus (J.E. Lange) Imbach and P. ostreatus, emphasizing the potential of in vitro cultivation for enhancing bioactive compound production. They found that P. ostreatus mycelium, grown on solid cultivation, exhibited superior antioxidant activity and higher concentrations of ergosterol and phenolic compounds than its fruiting body. This mycelium showed DPPH• scavenging activity and β-carotene bleaching inhibition, suggesting its significant antioxidant activity.
Similarly, a previous investigation demonstrated the antioxidant activity of P. citrinopileatus Singer extracts. The results revealed that ethanol extract (approx. 60% of inhibition) showed higher DPPH and RPA than hot-water and cold-water extracts (approx. 30% of inhibition by both) (Lee et al., 2007). It has been reported that there is a higher antiradical DPPH• value in aqueous than chloroform extracts from P. djamor (Rumph. ex Fr.) Boedijn mycelium (approx. 9 and 4% of inhibition, as well as higher ABTS values in aqueous than chloroform extracts (approx. 82 and 22% of inhibition (Kalyoncu et al., 2010a).
The antioxidant and antibacterial activity of edible mushrooms mycelium has been demonstrated; an investigation evaluated the antioxidant activity of aqueous and ethanol extracts from Ganoderma mycelium (G. australe (Fr.) Pat., G. neojaponicum Imazeki, and G. lucidum (Curtis) P. Karst., obtained after fermentation in wheat grains. The results showed that ethanol extract from G. neojaponicum showed the highest antioxidant activity (DPPH• and FRAP), which was related to the high total phenolic content values (Subramaniam et al., 2014). Furthermore, a previous investigation by Hsu et al. (2022) examined the polyphenol composition of Tuber magnatum Picco (white truffle) mycelium cultivated on substrates with varying ratios of soybean and red adlay (ratio 4:0, 3:1, 2:2, 1:3 and 0:4). This study found that both aqueous and ethanol extracts showed enhanced antioxidant capacities as the proportion of red adlay increased. This was evidence by elevated values in DPPH radical scavenging, FRAP, and RPA assays. These results align with our findings.
In the same way, the metabolites recovery and antioxidant activity was also investigated for the extract from Agaricus brasiliensis Fr. mycelium, obtained after fermentation, using different residues as substrates (grape pomace, apple pomace, pineapple pomace, and pineapple peel). After fermentation, the results showed higher total phenolic and flavonoid content in mycelium methanol extracts obtained from pineapple peel, pineapple pomace, and grape pomace substrates than wheat substrates. With respect to antioxidant activity (DPPH•), mycelium methanol extracts from pineapple peel and apple pomace showed higher antiradical activity than other treatments. Based on the above, the potential of mycelium mushrooms to develop new food additives was demonstrated (Mokochinski et al., 2015). Moreover, the polyphenol content and antioxidant activity of natural extracts have been extensively correlated (Reis et al., 2012; Muflihah et al., 2021).
Conclusions
This study shows that coffee bagasse and potato peel significantly affect the antioxidant and antimicrobial properties of Pleurotus ostreatus mycelium. The extracts from T2-T4 showed the highest content of phenols and the highest values of inhibition of the DPPH radical. In addition, T4 presented the highest content of flavonoids and the highest ABTS+ antiradical activity. There were no differences between the chlorogenic acid content treatments, the reducing power ability, and the ferric-reducing antioxidant power. In addition, T2-T4 exerts antimicrobial activity against Gram-positive and Gram-negative bacteria. SCG and PPR residues used in the formulation of substrates for mushroom cultivation increase the polyphenol content and improve the bioactivity of the hydroalcoholic extract from P. ostreatus.











 nueva página del texto (beta)
nueva página del texto (beta)



