Introduction
Burmanniaceae is a family of ca. 100 species of frequently achlorophyllous, mycoheterotrophic herbs (often associated with arbuscular mycorrhizal Glomeromycetes, Merckx et al., 2010; Zhao et al., 2021), distributed in tropical, subtropical, and some mild-temperate wet regions of southeast Asia, Australasia, Africa, and the Americas (Jonker, 1938; Dahlgren et al., 1985; Maas et al., 1986; Maas-van de Kamer, 1998). Its evolutionary relationships within the monocotyledons were unclear before the advent of DNA sequence-based studies. Based on morphological characters, different authors considered Burmanniaceae to be closely related to either Amaryllidaceae, Campynemataceae, Haemodoraceae, Hydrocharitaceae, Iridaceae, Melanthiaceae, Orchidaceae, Philydraceae, Taccaceae, Triuridaceae, Corsiaceae, and/or Thismiaceae; together with the last two families, Burmanniaceae used to be classified in its own order, Burmanniales (Jonker, 1938; Dahlgren et al., 1985; Maas et al., 1986; Maas-van de Kamer, 1998). Thismiaceae has been treated as part of Burmanniaceae in some classifications (e.g., by Jonker, 1938, and Maas et al., 1986).
Multiple phylogenetic studies based on molecular data indicate that Burmanniaceae is nested in the order Dioscoreales, which also includes the families Afrothismiaceae, Dioscoreaceae, Nartheciaceae, Taccaceae, Thismiaceae and Trichopodaceae (Stevens, 2017, continuously updated, and references therein). These studies suggest that Thismiaceae and Afrothismiaceae are both more closely related to Taccaceae than to Burmanniaceae. However, the evolutionary relationships within the Dioscoreales (and the recognition of some families) are still unsettled (Stevens, 2017, continuously updated, and references therein).
Burmannia L. is the largest genus of Burmanniaceae, with ca. 63 currently recognized species, with the same overall distribution of the family. Many species of Burmannia are autotrophic (photosynthetic), while others are holo-mycoheterotrophic (completely mycoheterotrophic) or mixotrophic (partly mycoheterotrophic). Most of the holo-mycoheterotrophic (“saprophytic” in the older literature, e.g., Jonker, 1938; Maas et al., 1986) species of the genus are from Asia and Africa. Of the ca. 20 New World species of the genus, Burmannia tenella Benth. is the only holo-mycoheterotrophic representative (Maas et al., 1986).
Burmannia tenella is widespread in tropical South America. It has been collected in Colombia, Venezuela, Guyana, Ecuador, Brazil, Peru, and Bolivia (Maas et al., 1986). In the present contribution, we report this species for the first time in Central America based on recent collections from Costa Rica, present an overview of its geographic distribution, and provide an updated key to the species of Burmannia in Mesoamerica.
Material and Methods
Field work and identification
We collected plants of Burmannia in Costa Rica, province Puntarenas, Cantón Coto Brus, San Vito, Finca Los Jilgueros (8°47'19.3''N, 82°59'14.3''W, 1450 m), in 2023 and 2024, which were preserved either in liquid (80% ethanol, 20% water) or pressed and dried. These plants (Fig. 1) did not match any of the previously known species of Burmannia for Costa Rica or other parts of Mesoamerica (Maas and Maas-van de Kamer, 1994; Maas-van de Kamer and Maas, 2003), and were identified as B. tenella using the monograph of Neotropical Burmanniaceae of Maas et al. (1986) and their revision of Burmanniaceae of Colombia (Maas and Maas-van de Kamer, 1988).
Examined collections and geographic data
Specimens of the genus Burmannia from Costa Rican (CR and USJ) and Peruvian herbaria (HOXA and USM) were physically examined, among which we discovered an additional, previous collection of B. tenella from Costa Rica, identified only to family level (Rodríguez et al. 11756, CR). Other herbarium materials (including type specimens of B. tenella and its synonym B. amazonica Schltr.) were studied through high-resolution digital images found on GBIF.org (GBIF, 2024, continuously updated), JSTOR Global Plants (JSTOR, 2024), or websites of individual herbaria: K (Kew, 2024) and COL (Universidad Nacional de Colombia, 2024). These images represent specimens from the following herbaria (acronyms of all herbaria follow Thiers, 2024, continuously updated): AAU, B, BM, BR, C, COL, E, FI, G, GH, HBG, K, LE, MBM, MG, MO, NY, P, RB, S, SP, TCD, U, and US. These data were complemented with unequivocal photographic evidence from the citizen science website iNaturalist.org (iNaturalist, 2024a).
A distribution map was created with QGIS v. 3.24.0 (QGIS, 2024), using the geographic coordinates of herbarium specimens and iNaturalist observations (Fig. 2). For herbarium specimens lacking geographic coordinates and approximate elevation but with reasonably precise locality information, coordinates were inferred using Google Earth Pro v. 7.3.6.9345 (Google, 2022). For such specimens from Brazil, coordinates (when available) were obtained from Reflora (2024, continuously updated), but corrected with Google Earth Pro when obviously erroneous. These inferred coordinates and elevations are indicated between parentheses in the specimens listed below.
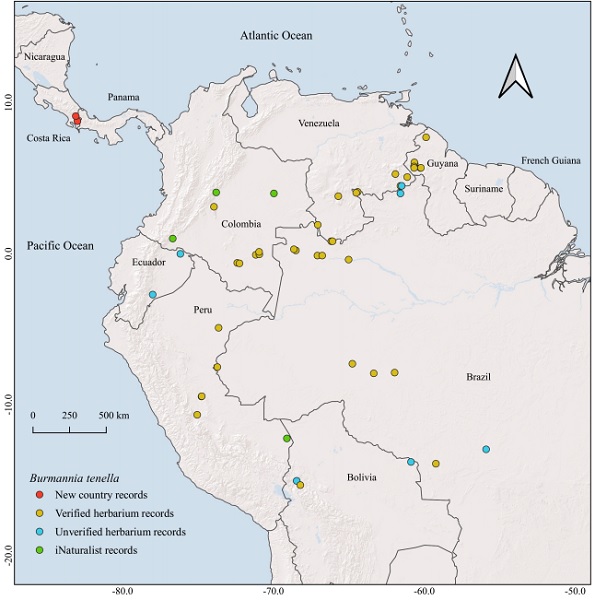
Figure 2: Distribution of Burmannia tenella Benth., based on herbarium records with geographic coordinates. “Verified herbarium records” correspond to herbarium collections seen by the authors. “Unverified herbarium records” correspond to collections not seen by the authors, but identified as B. tenella by P. J. M. Maas and/or H. Maas-van de Kamer (see Results).
Results
Taxonomy
Burmannia tenella Benth., Hooker’s J. Bot. Kew Gard. Misc. 7: 12. 1855.
TYPE: BRAZIL. Amazonas, in sylvis arenosis fl. Vaupés (“from the sandy woods along the Rio Uaupés”, from protologue), I.1853, R. Spruce 2835 (holotype: K!, putative isotypes (“Prope Panurè ad Rio Uaupès, X.1852-I.1853”; possibly different gatherings that were given the same collection number by Spruce; Panurè=Ipanoré, 00°18'08''N, 68°38'33''W, 85 m): B!, BM!, BR!, C!, CA, E!, G!, GH!, K! (two sheets), LE!, MG!, NY!, OXF, P!, TCD!, W).
= Burmannia amazonica Schltr., Verh. Bot. Vereins Prov. Brandenburg 47: 102. 1905. TYPE: BRAZIL. Amazonas, Rio Marmelos, near falls, Rio Madeira (“in der Campina bei den Wasserfällen des Marmellos”, from protologue; 07°52'S, 61°58'W, 53 m), III.1902, E. H. G. Ule 6124 (holotype: B!, isotype: HBG!).
Distribution: Burmannia tenella has been collected and/or photographically documented in Costa Rica (newly recorded here; province of Puntarenas), Colombia (departments of Amazonas, Caquetá, Meta, Putumayo and Vaupés), Venezuela (states of Amazonas and Bolívar), Guyana (administrative regions of Barima-Waini and Cuyuni-Mazaruni), Ecuador (provinces of Morona Santiago and Napo), Brazil (states of Acre, Amazonas, Mato Grosso, Rondônia and Roraima), Peru (departments of Huánuco, Madre de Dios, and Pasco), and Bolivia (departments of La Paz and Santa Cruz), at elevations from 120 to ca. 1580 m (see examined material below and Fig. 2; Jonker (1938), Maas et al. (1986), and Maas and Maas-van de Kamer (1988), cite additional collections and/or duplicates of this species from South America). The two Costa Rican locations of B. tenella are ca. 38 km apart; both are in Puntarenas province, within the Térraba river basin (the largest river in the country). Burmannia tenella very likely occurs in additional political subdivisions within the cited South American countries, but it has escaped detection by collecting botanists.
Habitat and phenology: Burmannia tenella occurs in rain forest habitats (Maas et al., 1986; Maas and Maas-van de Kamer, 1988), including in Costa Rica. Throughout its distribution area, it has been recorded flowering all year (Maas et al., 1986). In Costa Rica, it has been collected with flowers and fruits in January and February.
Material examined (new country record): COSTA RICA. Province Puntarenas, Cantón de Coto Brus, San Vito, Cañas Gordas, Finca Los Jilgueros, cerca de la laguna, 1450 m, 8°47'19.3''N, 82°59'14.3''W, 31.I.2023, L. Álvarez 994 (USJ (spirit)); Cantón de Coto Brus, San Vito, Finca Los Jilgueros, cerca de la laguna, 1450 m, 8°47'19.3''N, 82°59'14.3''W, 23.II.2024, B. Gamboa 2417 (CR, MO (to be distributed), USJ); Buenos Aires, P.N. La Amistad, Cuenca Térraba-Sierpe, Potrero Grande, La Lucha, alrededores del Cerro Amuo, 1500 m, 9°07'18.96''N, 83°05'57.33''W, 20.II.2008, A. Rodríguez et al. 11756 (CR).
Additional examined material: BOLIVIA. La Paz (Larecaja), Mapiri, (15°19'S, 68°13'W), VII-VIII.1892, M. Bang 1563 (E (pro parte), K); Mapiri, 2500 ft. (762 m), V.1886, H. H. Rusby 858 (E, K, US (two sheets)); Mapiri region, San Carlos, 850 m, 12.II.1927, O. Buchtien 459 (U, US (pro parte)). BRAZIL. Acre, Serra da Moa, Rio Moa, 6 km above school, (7°30'S, 73°43'W), 25.IV.1971, G. T. Prance et al. 12443 (NY, U, US). Amazonas, Prope San Gabriel da Cachoeiras ad Rio Negro, (00°07'S, 67°05'W), III.1852, R. Spruce 2299 (K (two sheets)); Rio Negro, an feuchten Stellen, Manáos (Manaus), VII.1900, E. H. G. Ule 5212a (B); Brazilian Amazonia, Basin of Rio Negro, Tapuruquara, forest on terra firme, (0°24'S, 65°01'W), 27.X.1971, G. T. Prance et al. 15607 (NY); Rio Curicuriari, 10 km above mouth, Caatinga forest, (0°14'S, 66°51'W), 3.XI.1971, G. T. Prance et al. 16061 (NY, U); Rio Purus, Rio Ituxi, Fortaleza Savanna, Rio Puciari tributary of Rio Ituxi, 20 km above mouth, (7°16'50.9''S, 64°46'04.1''W) 30.VI.1971, G. T. Prance et al. 13832 (NY, U). Mato Grosso, Formiga, L. Telegr., (610 m, 13°54'S, 59°14'W), III.1918, J. G. Kuhlmann s.n. (RB-55097). Rondônia, highway between Humaita and Porto Velho, 60 km S of Humaita, at crossing of Sao Joao stream 1-5 km N highway, W of stream, (07°55'S, 63°21'W), 4.V.1982, A. J. Fife et al. 4027 (NY); Roraima, Auaris Mission, Rio Auaris, 2600 ft. (792 m), (4°02'28.0''N, 64°30'47.9''W), G. T. Prance et al. 20068 (NY, U); vicinity Auaris, Territorio do Roraima, 800 m, 04°06'N, 64°25'W, 28.VII.1974, G. T. Prance et al. 21459 (NY, U). COLOMBIA. Amazonas, corregimiento de Araracuara, carretera a Puerto Arturo, ca. al aeropuerto, sitio “La Nevera”, 350 m, (0°36'01''S, 72°23'51''W), 21.IX.1987, G. Galeano and J. Huitoto 1299 (COL). Amazonas - Vaupés, Rio Apaporis, entre el Rio Pacoa y el rio Kananarí, 250 m, (00°04'S, 71°10'W), 4.VII.1951, R. E. Schultes and I. Cabrera 12889 (US); Rio Apaporis, Cachivera de Jirijirimo y alrededores, 250 m, (00°02'S, 70°56'W), 7.VII.1951, R. E. Schultes and I. Cabrera 12959 (US); Rio Apaporis, Cachivera de Jirijirimo y alrededores, 250 m, 12.VIII.1951, R. E. Schultes and I. Cabrera 13521 (US). Vaupés, Rio Kananarí, Cerro Isibukuri, 250 m, (00°07'40''N, 70°56'54''W), 28.X.1951, R. E. Schultes and I. Cabrera 14470 (US). Caquetá, Río Caqueta, Araracuara, rastrojo inundable 20 años, (0°37'59''S, 72°14'59''W), 1.XI.1990, A. van Dulmen and N. Matapi 32 (U). Meta, Sierra de La Macarena, montañas centrales, flanco norte, 1400 m, (3°6'55''N, 73°56'26''W), 25.XII.1949, W. R. Philipson and J. M. Idrobo 1931 (COL). GUYANA. No date, Appun 686B (K). Barima-Waini, Kaituma, N.W. District, (07°43'N, 59°53'W), VI.1897, G. S. Jenman s.n. 7270 (K); Kaituma, 15.VI.1897, E. F. im Thurn 180 (K (two sheets, including drawings)); Kaituma River, 12 miles up, 15.III.1945, D. B. Fanshawe F2409 (5145) (K, US). Cuyuni - Mazaruni, approximately 2 km N of Maipuri Falls, Karowrieng River, 570-600 m, 05°41’N, 60°15’W, 24.XII.1989, L. J. Gillespie and D. R. Smart 2886 (COL, K, U, US); Pakaraima Mountains, Mt. Latipú, Mazaruni R., 400-800 m, 5°57'N, 60°38'W, I.1980, P. J. M. Maas and L. Y. T. Westra 4188 (U); Pakaraima Mts., Imbaimadai, approx. 2 miles NE of Base Camp 1, 675 m, 5°41'35.7''N, 60°13'22.4''W, 23.I.2004, K. M. Redden et al. 2361 (US); Pakaraima Mts., E bank of Mazaruni River, 1.14 miles SW of Base Camp 6 towards ABC Falls, 492 m, 6°03'32''N, 60°39'’34.4''W, K. M. Redden 2847 (US). Upper Mazaruni Distr., on the trail along Wabaru Creek to Waramadon Mt., 0.75-3 mi. S of Waramadon Amerindian Village ca. 20 mi. above Kamarang, 1650 ft. (503 m), 5°47'N, 60°41'W, 17.X.1985, H. E. Robinson 85-0044 (US (two sheets)); 3¼ miles upstream on Saydak Creek from its junction with Kako river, 470 m, 5°41'30''N, 60°40'30''W, 19.IV.1979, P. J. Edwards 1350 (K). PERU. Huánuco, Province Pachitea, region of Pucallpa, western part of the “Sira Mountains” and adjacent lowland, c. 24 km SE to c. 26 km ESE of Puerto Inca, from the beginning of the mountain rainforest next to the “Campamento Pato Rojo”, 1080 m, 9°27'S, 74°46'W, 5.I.1998, B. Wallnöfer 15-050188 (U); SW slope of the Rio LlullaPichis watershed, on the ascent of Cerros del Sira, Camp 3 (Laguna), 1290 m, 9°26'S, 74°45'W, 12.VII.1969, F. Wolfe 12301 (U). Loreto, Prov. Requena, Centro agro-forestal “Jenaro Herrera”, road to Varillal, km 5, 120 m, (04°54'S, 73°38'W), 18.XI.1985, P. J. M. Maas et al. 6333 (U, USM). Pasco, Oxapampa, Distrito Villa Rica, Sector Ubiriqui camino a Apizuzu. bosque primario, 1574 m, 10°39'44''S, 75°03'02''W, 17.VI.2009, R. Vásquez et al. 35910 (HOXA, MO, USM). VENEZUELA. Amazonas, VI.1854, R. Spruce 3733 (K) (the label does not indicate the country, but it is not from the Brazilian state of Amazonas; see Discussion); Dpto. Atapabo, trail from Río Cunucunuma to Huachamacari, 200-400 m, 03°49'N, 65°42'W, 7.XI.1988, R. Liesner 25965 (MO, U); Río Negro, 1 km south of Cerro de La Neblina Base Camp, which is on Rio Mawarinuma, in low area with stream, 140 m, 00°50'N, 66°10'W, 27.II.1984, R. Liesner and W. R. Buck 16277 (MO, U); Río Negro, Neblina Massif, mouth of the canon Grande along the Río Mawarinuma, just E of Puerto Chimo, 00°50'N, 66°05'W, 15.VII.1984, G. Davidse and J. S. Miller 27116 (MO, U (pro parte)); San Carlos (de Rio Negro), (80 m, 01°55'13''N, 67°03'37''W), no date, R. Spruce 2836 (K (pro parte), P). Bolivar, 17 km E of El Pauji by road and 64 km W of Santa Elena by road, 4 km N of highway, Río Las Ahallas, 850 m, 04°30'N, 61°30'W, 30.X.1985, R. Liesner 19218 (MO, U); Distr. Piar, bosques medianos entre el caserío de Kukenán y el Rio Churí, al E del Churi-tepui, en el Valle del Rio Karuay inferior, 900 m, 5°17'N, 61°55'W, 17.XI.1984, O. Huber 9776 (K, U); Dist. Roscio; 3 km S of El Pauji, 900 m, 04°90'N (04°30'N), 61°35'W, 19.X.1985, B. K. Holst and R. L. Liesner 2331 (MO, U); Quebrada El Jaspe (Rio Caco), afloramientos de Jaspe colorado, 47 Km al N de Santa Elena, al S del Rio Yuruani, 5°5'N, 61°8'W, 25.VIII.1976, J. A. Steyermark et al. 112348 (U).
Additional collections not seen by us, but identified as Burmannia tenella by P. J. M. Maas and/or H. Maas-van de Kamer (specialists on the taxonomy of Burmanniaceae); these specimens correspond to the “unverified herbarium records” in Fig. 2: BOLIVIA. La Paz, Bautista Saavedra, Área Natural de Manejo Integrado Apolobamba, Pauje Yuyo, 940 m, 15°02'12.0''S, 68°27'26.0''W, 7.IX.2004, L. Cayola et al. 1104 (MO). Santa Cruz, Velasco, La Meseta, the trail between Lago Caimán and the cascades “Arco Iris”, 520 m, (13°47'S, 60°53'W), 11.IV.1996, N. Ritter 3087 (MO). BRAZIL. Mato Grosso Serra da Chapada, pr. São Jeronymo, (12°56'50.6''S, 55°54'13.3''W), 11.VI.1903, G. O. A. Malme 3493 (S). ECUADOR. Morona Santiago, Cordillera Cutucú, ridge between the Itzintza and Chupiasa, 1143 m, (2°42'S, 78°00'W), 1940, W. H. Camp E-1251 (S). Napo, Lagunas de Cuyabeno, NE part of the first lake, swamp forest near high water level of the lake, 300 m, (0°00'S, 76°10'W), 24.VIII.1981, J. S. Brandbyge et al. 36069 (AAU). GUYANA. No date, C. F. Appun 686B (K). PERU. Loreto, Maynas, distrito Fernando Lores, Tamshiyaco, Quebrada Tamishiyaco, Trocha de Ambores, ca 4 km above mouth of creek, (900 m, ca. 04°00'N, 61°35'W), 24.III.1973, S. T. McDaniel and M. Rimachi Y. 17249 (IBE, MO). VENEZUELA. Amazonas, primary forest on lateritic soil, mammal study site at Hauchica, 11 km northeast of San Carlos de Rio Negro, 13.XI.1977, R. L. Liesner and J. Hall 3455 (MO). Bolívar, 17 km E of El Pauji by road and 64 km W of Santa Elena by road, 4 km N of highway, Río Las Ahallas, 850 m, ca. 4°30'N, 61°30'W, 28.X.1985, R. L. Liesner 19046 (MO); loc. cit., 29.X.1985, R. L. Liesner 19136 (MO).
Records from iNaturalist: COLOMBIA. Amazonas, Leticia, Macedonia, -3.9994368339, -69.9671605602 (03º59’58”N, 69º58’01”W, ca. 140 m), 25.VII.2023, observation by ashdevos (iNaturalist, 2024b). Meta, Acacías, 4.0663972222, -73.7925916667 (04°03'59''N, 73°47'33''W, ca. 885 m), 29.IV.2024, observation by sruffner (iNaturalist, 2024c). Putumayo, Villagarzón, 1.0095785006, -76.6715317592 (01°00'34''N, 76°40'17''W, ca. 507 m), 30.XII.2022, observation by natujuano (iNaturalist, 2024d). PERU. Madre de Dios, Tambopata, ASA Finca Las Piedras, -12.2268033333, -69.1079778333 (12°13'36''S, 69°06'28''W, ca. 266 m), 6.VIII.2022, observation by geoffgalice (iNaturalist, 2024e).
The specimen M. E. Engels 3777 (MBM), from the Brazilian state of Mato Grosso, is erroneously identified as Burmannia tenella in both GBIF (2024, continuously updated) and Reflora (2024, continuously updated) at the time of writing of this article. It has green stems and small but evident leaves and appears to correspond to B. flava Mart. (see Discussion).
Discussion
The occurrence of Burmannia tenella in Central America represents a significant disjunction from its previously known geographical distribution. All other recorded occurrences of this species are located east of the Andes in South America, specifically within the southern and western Amazon basin and the Essequibo and southern Orinoco basins in the Guiana Shield. These are part of two phytogeographic regions recognized by Gentry (1982): Amazonia (including the Guiana subregion) and Guyana Highlands, respectively. Both are part of the so-called Amazon Biome or Amazon Subregion (the latter a biogeographic region defined by Morrone, 2000). In Colombia, where the Northern Andes split into three major branches divided by large river basins, B. tenella has not been recorded west of the eastern range (Cordillera Oriental), nor has it been recorded north of the department of Meta (in the center of the country) (Maas, 2016). The Costa Rican collections of B. tenella are more than 1170 km apart from the closest known locations in South America (from the Colombian department of Meta) and represent the northernmost known localities of this species. The northernmost reaches of the Andes (i.e., the Serranía de Perijá and the Cordillera de Mérida in Colombia and Venezuela) extend somewhat farther north than the Costa Rican localities of B. tenella.
Merckx et al. (2013) pointed out that, among mycoheterotrophic plants, disjunct distributions are shown mainly by tropical species. They mention that several of these disjunct taxa are known from very few collections, and thus the apparent disjunctions may represent sampling artifacts. It is possible that B. tenella occurs (and has escaped detection) in western Colombia and even Panama, but this species is known from more than 50 herbarium collections from South America.
Burmannia tenella can be recognized from other New World congeners by being completely achlorophyllous and having white, very thin stems. Other species of Burmannia from the Americas are either fully photosynthetic with well-developed leaves (e.g., B. kalbreyeri Oliv.), have a rosette of small, green or greenish leaves at the base of the stem (e.g., B. flava), or at least have green or greenish stems (e.g., B. capitata (Walter ex J. F. Gmel.) Mart.) (Maas et al., 1986). Burmannia tenella is the only American species whose fruits are dehiscent by three reflexed, septicidal valves, and form a lantern-like structure when open (Fig. 1). The valves separate from the rest of the fruit wall from apex to base; the three remaining ribs form the “frame” of the lantern-like structure (see figures 2(3C) and 18e in Maas et al., 1986). The fruits of other Burmannia species open by several narrow, transverse slits on the external walls of each of the three locules or are irregularly dehiscent by withering of the membranous fruit wall (Maas et al., 1986). Burmannia tenella is also unique among American congeners in that it lacks rhizomes and has relatively thick (0.7-2 mm) and short roots. Most Burmannia species (and other Burmanniaceae) have rhizomes and thinner, longer, filiform roots (Imhof, 1999; Imhof et al., 2013).
As with other holo-mycoheterotrophic species of the family, Burmannia tenella grows only on the leaf litter in the dark, wet forest understory (Maas et al., 1986). Most other Neotropical species of Burmannia, which are photosynthetic, grow in more open habitats, including savannas and other grasslands; B. kalbreyeri is unique in the genus in that it grows as a pendulous epiphyte (Jonker, 1938; Maas et al., 1986).
Very little is known about other aspects of the natural history of Burmannia tenella. Pollinators are unknown, but flowers of the closely related B. lutescens Becc. (see below) produce nectar and were seen to be visited by two genera of mosquitoes (Culicidae, Diptera) at dawn and the early evening in Malaysia (Kato, 1996; Momose et al., 1998). Seeds of Burmanniaceae are characterized by their minute size (“dust diaspores”, apparently dispersed by wind, similar to those of most Orchidaceae). However, the seeds of B. tenella are quite long and narrow compared to those of other species of Burmannia and might be better dispersed by water (Dahlgren et al., 1985; Maas et al., 1986; Leake, 1994; Maas-van de Kamer, 1998).
Interestingly, both Jonker (1938) and Maas et al. (1986) indicated that Burmannia tenella is most closely related to the Southeast Asian and Australasian B. lutescens (TYPE: MALAYSIA. Ragiato di Sarawak. Mattang, IV.1866, O. Beccari 1505, FI!. Distributed in the Bismarck Archipelago, Borneo, Java, Peninsular Malaysia, New Guinea, Sulawesi, Sumatra, Vanuatu and Vietnam; Merckx et al., 2013; Nuraliev et al., 2018; POWO, 2024). Maas et al. (1986) even suggested that both taxa might represent the same species; this hypothesis has not yet been tested. Kato (1996) and Nuraliev et al. (2018) published photographs of living B. lutescens from Malaysia and Vietnam, respectively.
The famed English botanist and explorer Richard Spruce (1817-1893) was the first to collect herbarium specimens of Burmannia tenella. In fact, he collected this species at least four different times: in São Gabriel da Cachoeira along the Rio Negro (Brazilian state of Amazonas), in January 1852 (Spruce 2299); in Panuré (nowdays Ipanoré) along the Rio Uaupés (Brazilian state of Amazonas), in January 1853 (Spruce 2835, type of B. tenella); in an unspecified place in the Venezuelan Amazonas in April 1854 (Spruce 3733 (this specimen has been erroneously attributed to the Brazilian state of Amazonas by Maas et al. (1986), GBIF (2024) and Reflora (2024). The label does not indicate the country, but it had to be collected in Venezuela (or even possibly in Colombia just across the border from San Carlos de Rio Negro, see below) according to the summary of Spruce´s itinerary in Amazonia published by Prance, 1971; see also Steyermark, 1981)); and in San Carlos de Rio Negro (Venezuelan state of Amazonas), without date (Spruce 2836). The latter collection number is puzzling, because it is immediately above that of the type of the species, but from a relatively distant locality (ca. 480 km by river); from Ipanoré he first went downstream to São Gabriel da Cachoeira and then upstream to San Carlos de Rio Negro. Spruce first arrived in San Carlos on 11 April 1853. He made it his headquarters for 20 months, making several long and short excursions from there, and left San Carlos for the last time on 23 November 1854 (Spruce, 1908; Prance, 1971; Porter, 1996).
Maas et al. (1986) remarked that it could not be ascertained whether the many specimens with the same collection number as the holotype of B. tenella (Spruce 2835) should be considered as isotypes, because their labels contain some data that differ from those on the label of the holotype. A very similar situation occurs with specimens bearing the type collection number of Gymnosiphon cymosus (Benth.) Benth. & Hook. f. (Spruce 2829, also Burmanniaceae), collected along the Uaupés river in January 1853, with others also indicating the Brazilian locality of Panuré (Ipanoré), and others even indicating as having been collected in the Venezuelan locality of San Carlos de Rio Negro in March-May 1854 (Maas et al., 1986). It may be that Spruce decided to use the same collection numbers of plants that he collected along the Uaupés river, for some of the specimens of the same species that he collected a few months later in San Carlos de Rio Negro, as this latter part of his voyage was particularly difficult (Spruce, 1908).
Burmannia tenella is one of only six or seven American species of Burmannia (of the 20 species in the New World) that occur outside South America. The others are B. bicolor Mart., B. biflora L., B. capitata, B. flava, B. kalbreyeri, and “Burmannia sp. A” (a still undetermined entity, see below). All these also occur in South America except for B. biflora, which is restricted to the southeastern United States of America (states of Alabama, Georgia, Florida, Louisiana, Mississippi, North Carolina, South Carolina, Texas and Virginia) and Cuba (province of Pinar del Rio), and “Burmannia sp. A” (apparently endemic to Costa Rica, if different from B. bicolor). Burmannia bicolor is widespread in South America but also occurs in Cuba.
A dichotomous key for the identification of the five known species of Burmannia in Mesoamerica (adapted from Maas and Maas-van de Kamer, 1994, and Maas-van de Kamer and Maas, 2003) is presented below. Mesoamerica is defined here in the same way as in Flora Mesoamericana, i.e., as the region encompassing the seven Central American countries, plus the Mexican states of Campeche, Chiapas, Quintana Roo, Tabasco, and Yucatán. “Burmannia sp. A”, a still undetermined entity based on Weston 10216 (CR), is closest to the South American B. bicolor and B. stuebelii Hieron & Schltr. (Maas-van de Kamer and Maas, 2003); more material is needed to ascertain its identity. Costa Rica is the only country where all five species are known to occur (again, if “Burmannia sp. A” is different from B. bicolor).
Key to the species of Burmannia in Mesoamerica
1a. Plants epiphytic, leaves 2-25 cm long; Costa Rica and Panama (also Colombia, Ecuador, Peru, and Venezuela) ...... Burmannia kalbreyeri Oliv.
1b. Plants terrestrial; leaves 0.1-2 cm long ...... 2
2a. Inflorescences capitate, with >6 flowers separated by <1 mm; flowers not or barely winged, white or yellowish white (rarely blue with yellow); Mexico to Panama (also eastern and southern United States of America, Greater Antilles, and widespread in South America southwards to Argentina) ...... Burmannia capitata (Walter ex J. F. Gmel.) Mart.
2b. Inflorescences lax, with 1-6 flowers, separated by >1 mm; flowers conspicuously winged, yellow, greenish yellow or blue with yellow ...... 3
3a. Stems and flowers completely white; fruits longitudinally dehiscent; Costa Rica (also in Colombia, Venezuela, Guyana, Ecuador, Brazil, Peru, and Bolivia) ...... Burmannia tenella Benth.
3b. Stems at least partially green, flowers greenish, yellow, or blue with yellow; fruits dehiscent by transverse slits ...... 4
4a. Flowers unicolored, yellow or greenish yellow; inflorescence with 1-2 flowers; Mexico to Panama (also southern Florida in the United States of America and widespread in South America southwards to Argentina) ...... Burmannia flava Mart.
4b. Flowers bicolored, blue with yellow; inflorescences with ca. 6 flowers; Costa Rica ...... “Burmannia sp. A”
Author contributions
LAA conceived the study. LAA and BGR carried out the field studies. LAA and MAB examined herbarium specimens and compiled the distribution information about the species and the bibliography. LAA prepared the figures. LAA and MAB wrote the manuscript. All authors contributed to the discussion, revision and final approval of the manuscript.
Funding
This study was supported by Osa Conservation, the fieldwork was funded by Fondation Franklinia (2022-03/Conserving the Rare and Endemic Trees of the AmistOsa region, Southern-Pacific Costa Rica). Additional funding was provided by the Vicerrectoría de Investigación, Universidad de Costa Rica, for the research project B6-771 “Colección del Herbario USJ” registered in the Centro de Investigación en Biodiversidad y Ecología Tropical.











 nueva página del texto (beta)
nueva página del texto (beta)




