INTRODUCTION
Exaiptasia diaphana is a symbiotic anemone and a model organism to study various processes in cnidarians such as symbiosis, nutrition, and immunological mechanisms (Lehnert et al. 2012, Matthews et al. 2017, Rädecker et al. 2018, Mansfield and Gilmore 2019, Dungan et al. 2020). This anemone maintains a facultative symbiosis with microalgae of the Symbiodiniaceae family (Weis et al. 2008). Cnidarians exhibit nocturnal behavior (Lewis and Price 1975, Sebens and De Riemer 1977) and establish symbiosis with microalgae, which represents a challenge for the study of basal parameters, since both are governed by different biological rhythms (Sorek et al. 2014). Biological rhythms allow organisms to anticipate and adapt to natural environmental changes (Roenneberg and Merrow 2005, Bednářová et al. 2013). Symbiotic cnidarians exhibit heterotrophic feeding habits at night (Lewis and Price 1975) and use the energy provided by their endosymbiont algae during the day; this complicates the study of their rhythmic behavior. Furthermore, they show differential behavior during the day and night; for example, non-symbiotic anemones lose their circadian rhythm when kept under constant light or darkness (Aguillon et al. 2024). In symbiotic organisms, the tentacles of anemones and the corals that allow them to feed expand at night, whereas those with a higher density of symbionts expand during the day (Sebens and De Riemer 1977).
Lunar cycles are important for the reproduction of marine animals (Raible et al. 2017). In corals, lunar cycles regulate spawning (Harrison et al. 1984), although factors such as temperature can modify this process (Lin and Nozawa 2023). In addition, they have a correlation with the deposition of calcareous structures called dissepiments and with the growth of the calcareous skeleton of coral colonies (Winter and Sammarco).
Biological rhythms are key in controlling physiology and, therefore, the immune system (Fagiani et al. 2022). In mammals, studies have found that the circadian cycle is related to the migration of cells of the innate immune system and changes in the expression of immune molecules (Reviewed in Scheiermann et al. 2013). Furthermore, when organisms face an immunological challenge, they respond best in their active phase (Reviewed in Westwood et al. 2019). In cnidarians, the relationship between biological rhythms and the immune system has not yet been studied.
As in other invertebrates, cnidarians have an innate immune system whose function is to maintain the health of the animal (Palmer 2018). An immunological pathway commonly used by invertebrates is the proPO system, or the phenol oxidase (PO) pathway (Cerenius et al. 2008). The proPO system is activated by proteins capable of binding to peptidoglycans and lipopolysaccharides in various invertebrates, such as insects (Zhao et al. 2018), crustaceans (Liu et al. 2011), and cnidarians (Lehnert et al. 2012). Once activated, it generates cytotoxic free radicals, binding agents, and opsonin, which protect the animal without synthesizing melanin (Zhao et al. 2007, Cerenius and Söderhäll 2021). The synthesis of melanin as a final product, known as melanization, encapsulates potentially harmful microorganisms (Zhao et al. 2007, Cerenius et al. 2008). For example, Palmer et al. (2010) showed that after damage, melanin deposits appear in some corals almost instantaneously.
In some cnidarians, PO has been studied as a response to various diseases and bleaching (Mydlarz et al. 2009, Palmer et al. 2010, Rivera-Ortega and Thomé 2018). However, the study of the basal immune response and its relationship with biological rhythms has not been considered. Studying the immune response under basal conditions is important since biological processes in any living organism alternate between periods of high, low, or no activity (Cardinali 2005). Therefore, our aim was to characterize the fluctuation of PO in healthy E. diaphana individuals, both symbiotic and aposymbiotic, throughout the day and during the phases of full moon and new moon. In addition, to understand whether symbionts affect the immune response, PO fluctuations were compared in both symbiotic and aposymbiotic E. diaphana.
MATERIALS AND METHODS
Experimental design
To explore whether biological cycles influence the immune response, the experiments were done at different times throughout the day and during 2 lunar phases (full moon and new moon) using 6 symbiotic anemones and 6 aposymbiotic anemones. In each lunar phase, mucus was collected every 6 h in 2 cycles per 24 h; during the first cycle, samples were taken at 8:00 p.m., 2:00 a.m., 8:00 a.m., and 2:00 p.m. The next day, measurements of the second cycle were resumed at 2:00 p.m., 8:00 p.m., 2:00 a.m., and 8:00 a.m. Mucus extracted from the 6 symbiotic anemones and 6 aposymbiotic anemones (n = 48) was grouped in duplicate per lunar phase and per symbiotic state. In total, 348 anemones were used. The experiment was carried out from September to November 2023.
To determine symbiont density, 6 symbiotic anemones were used per hour of sampling. This experiment was done in a cycle for each lunar phase. In total, 48 anemones were used.
Organisms
The symbiotic anemones were collected from the aquariums of the Unidad Académica de Sistemas Arrecifales (Instituto de Ciencias de Mar y Limnología, Universidad Nacional Autónoma de México), where they grew under natural conditions. The aposymbiotic anemones came from a laboratory culture of several years. After their collection, the anemones were acclimatized for 14 d to avoid the aquarium effect. Subsequently, anemones were maintained individually in 6-well plates with filtered seawater (0.22 µm) under a light/dark regime (full moon = 67.9-985 lux; new moon = 67.4-838 lux) and at natural temperature (full moon = 28.87-29.60 °C; new moon = 28.47-29.48 °C) in a humid windowless laboratory. Anemones were fed once a week with nauplii of Artemia spp.
Mucus extraction
Mucus was extracted from anemones exhibiting extended tentacles and column. Before extraction, the water was completely removed from each well with a disinfected transfer pipette, wiping off the remaining water with a sterilized cotton swab. The mucus expelled by the anemone after 2 min was collected with a micropipette and stored in individual microtubes. Finally, 10 mL of filtered seawater was added to each well.
Evaluation of the immune response
To determine the basal immune response in E. diaphana, mucus PO activity was measured. We used 96-well plates in 90-µL reactions in triplicate, as described below. Each well contained 30 µL of mucus (n = 6 anemones) and 30 µL of pyrogen-free water. Subsequently, the plate was covered with aluminum and incubated for 5 min at 35 °C with shaking (60 rpm). The reaction was started by adding 30 µL of L-Dopa substrate [2.5 mM]; measurements were immediately taken in a plate reader. The absorbance reading was performed at 450 nm every minute for 10 min. The activity of PO is presented as the change in absorbance per microliter of mucus per minute.
Symbiont density
To count symbionts, the anemones were weighed and placed in microtubes with 500 µL of filtered seawater. They were homogenized manually with a plastic homogenizer until no tissue was visible. Symbionts were purified by 3 centrifugation cycles (14,000 rpm) for 1 min. They were resuspended in 500 µL of filtered seawater and fixed by adding lugol (30%). Algae were counted in a hemocytometer and calculated per milliliter. The total cell number count per anemone was normalized to grams of wet weight.
Statistical analysis
Data normality and homoscedasticity were evaluated with the Shapiro-Wilks and Levene tests, respectively. Because the data did not meet the assumptions, nonparametric statistical tests were used. To compare PO activity with the interaction between the moon and symbiotic state, the Kruskal-Wallis test and post hoc Mann-Whitney tests were used. A Pearson correlation was applied between PO activity and symbiont density. Statistical analyses were performed in Rstudio v. 4.1.0 (Posit Team 2024).
RESULTS
Immune response
Circadian cycle
The activity of PO in both symbiotic and aposymbiotic anemones showed natural variability throughout the day, with 2 acrophases. The highest peak of activity was at 2:00 p.m., with symbiotic anemones at full moon showing lower PO activity compared to aposymbiotic anemones. A second smaller, but equally represented, peak was detected at 2:00 h (Fig. 1). Despite showing some biological variability, data showed no statistically significant differences (H [7] = 7.09, P = 0.41) throughout the day.
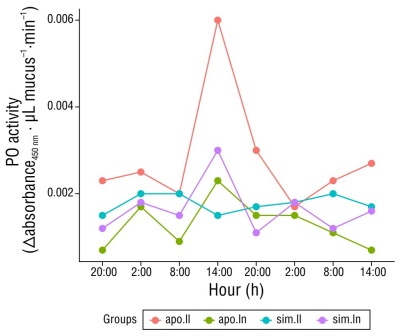
Figure 1 Variation of phenol oxidase (PO) activity during the day in Exaiptasia diaphana. The activity of PO was calculated in samples taken every 6 h for 48 h. Each mucus sample corresponds to a group of 6 anemones. The results of one estimation are shown; repetitions had very similar results. Abbreviations: aposymbiotic anemones at full moon (apo.ll), aposymbiotic anemones at new moon (apo.ln), symbiotic anemones at full moon (sim.ll), and symbiotic anemones at new moon (sim.ln).
Lunar cycle
The analysis of PO activity, measured as the interaction between the lunar phase and the symbiotic state, showed statistically significant differences (H [15] = 17.47, P = 0.0005). According to the post hoc analysis, PO activity in aposymbiotic anemones was found to be significantly higher at full moon than at new moon and higher than in symbiotic anemones for both moons (P < 0.05). Furthermore, PO activity in symbiotic anemones at full moon was significantly higher than that observed in aposymbiotic anemones at new moon (P = 0.032) (Fig. 2).
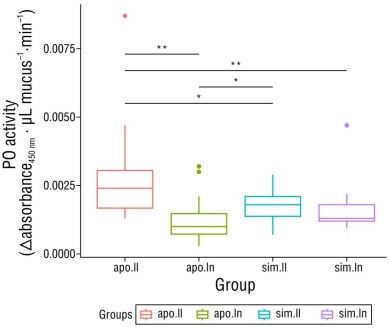
Figure 2 Comparison of phenol oxidase (PO) activity with respect to the symbiotic state and the lunar phase using the Kruskal-Wallis test. Boxplots represent the mean, first and third quartiles, confidence interval (95%), and outlier points (red dot, green dots, and purple dot). Each sample corresponds to the activity values of 6 anemones. The horizontal black lines show the comparisons between the different treatments; statistical significance of comparisons is represented by asterisks (*P < 0.05; **P < 0.01). Abbreviations: aposymbiotic anemones at full moon (apo.ll), aposymbiotic anemones at new moon (apo.ln), symbiotic anemones at full moon (sim.ll), and symbiotic anemones at new moon (sim.ln).
Symbiont density
We found a negative relationship between PO activity and symbiont density in symbiotic anemones (R 2 = 0.51, P = 0.04) (Fig. 3). Furthermore, symbiont density varied significantly depending on the time of day (H [3] = 8.55, P = 0.035). According to the post hoc analysis, symbiont density was significantly higher at 14:00 h (P = 0.006, Fig. 4a). Taking into account the lunar phase, we found that symbiont density was similar for both the full moon (H [3] = 4.84, P = 0.18) and the new moon (H [3] = 4.32, P = 0.22) (Fig .4b).
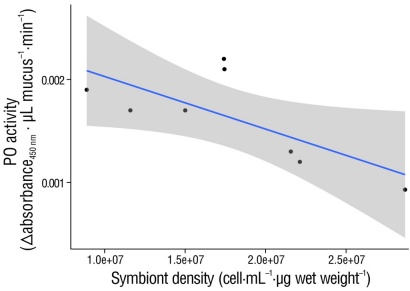
Figure 3 Relationship between phenol oxidase (PO) activity and symbiont density using a Spearman correlation test. The activity of PO decreases as symbiont density increases (cell·mL-1·µg wet weight). The density of symbionts was determined in 48 anemones, grouped into 6 anemones for each sampling. The 95% confidence level is shaded.
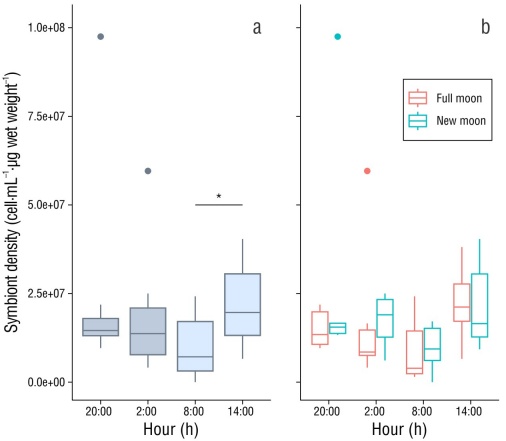
Figure 4 Comparison of symbiont density with respect to time of day and lunar phase using the Kruskal-Wallis test. (a) Symbiont density considering only the time of day, without taking into account the lunar phase. (b) Symbiont density considering lunar phase and time of day. Boxplots represent the mean, first and third quartiles, confidence interval (95%), and outliers. The asterisk (*) shows significant differences (P = 0.035) in the density of symbionts at 14:00 h compared to the other samples.
DISCUSSION
In this study, PO activity was measured in the mucus of E. diaphana under basal conditions. We determined that both symbiotic and aposymbiotic anemones show PO activity throughout the day. Therefore, E. diaphana shows constitutive immunity, and the proPO system is part of its basal immunity. The above agrees with what was proposed by Palmer (2018), who suggests that, though immunity is commonly approached as a response activated by damage signals, the immune system operates and remains active in a constitutive manner, even if it is not being challenged by a threat.
Immunity fluctuation in the circadian cycle
The PO activity in the mucus of E. diaphana fluctuated throughout the day, both in symbiotic and aposymbiotic anemones, with the highest PO activity at 2:00 p.m. No previous studies track PO activity throughout the day in cnidarians; our results provide a background for how the circadian cycle of E. diaphana affects its basal immunity.
The variability of basal immunity throughout the day could be consistent with what was observed at the transcriptomic level in symbiotic cnidarians. Gong et al. (2023) found that the corals Acropora pruinosa and Pocillopora damicornis differentially transcribe certain genes at night compared to the morning. At night, transcribed genes are mostly related to the immune system, metabolism, and signal transduction, whereas during the day, transcribed genes relate to transport and catabolism. However, Levy et al. (2011) reported that Acropora millepora genes associated with antioxidant activity have expression peaks at night, whereas those associated with metabolism are more expressed during the day. Furthermore, some studies report that crustaceans have a bimodal variation of PO in a diurnal cycle (She et al. 2019), whereas others have reported greater activity during the day (Rund et al. 2016) or even constant PO activity (Pfenning-Butterworth et al. 2022). These studies illustrate the importance of considering the circadian cycle in studies of the immune system because it could help explain some variations.
Immunity fluctuations in response to the lunar cycle
The PO activity in E. diaphana mucus was slightly higher with the full moon than with the new moon in anemones of both symbiotic states. In corals, reproduction and growth are synchronized with lunar cycles (Boch et al. 2011). However, despite the extensive information on the influence of the moon on coral reproduction, information on the immune system is scarce and practically non-existent in E. diaphana. Given that the values of PO activity in the mucus of E. diaphana showed different behavior in the full moon than in the new moon, we can suggest that the basal constitutive immunity in this anemone also obeys the lunar cycles, regardless of its symbiotic state. This finding constitutes a novel contribution of the present study.
Symbiont density
We found a negative relationship between symbionts and the immune response that coincides with studies on the coral Orbicella faveolata, which reported a reduction in the expression of genes related to immunity (Fuess et al. 2020). Other studies have positively correlated immune activity with antioxidant activity and melanin synthesis (Changsut et al. 2022). Finally, it has been hypothesized that, in corals, the loss of symbionts after bleaching confers some protection, since a lower incidence of diseases has been observed (Merselis et al. 2018). It is possible that symbiosis in cnidarians depends on reduced immune activity that facilitates this relationship.
CONCLUSIONS
The study of constitutive immunity and its relationship with biological cycles in E. diaphana is a scarcely explored field. The present study expands the understanding of PO activity as an immune response under basal conditions in aposymbiotic and symbiotic anemones, which showed basal constitutive immunity under laboratory conditions. The response varied according to biological rhythms, showing greater activity during the day and low activity at night. Furthermore, we observed that lunar cycles influenced PO activity, which was greater in the full moon phase. We also found that symbiotic status had an effect on the immune response, probably because symbiont density was negatively correlated with PO activity, with higher symbiont density observed at 14:00 h. These results not only provide evidence for constitutive immunity, but also illustrate the complexity of holobiont relationships. Future studies focusing on the relationship between immunity and biological cycles are important to achieve a more adequate interpretation of biological responses to stressors.











 texto en
texto en 



