INTRODUCTION
Coral reefs are characterized by complex three-dimensional structures of calcium carbonate (CaCO3) developed mainly through the interaction of 2 geo-ecological processes: (1) CaCO3 gross production, driven primarily by scleractinian corals, and (2) bioerosion, derived from the activity of numerous organisms that remove carbonate directly from the reef matrix (Chave et al. 1972, Perry 2011), as well as processes of cementation, lithification, and physical exportation of materials (Cornwall et al. 2021). As mentioned in Sheppard et al. (2018), production and erosion processes are present in a reef in ecological balance. However, a positive balance (i.e., production rate greater than erosion rate) is necessary for the reef to grow. Despite the importance of erosion processes in reef ecosystems, many studies have focused mainly on the production processes of coral CaCO3 (Perry and Lange 2019); in the case of bioerosion, the number of species-specific studies is much smaller and limited to a few sites and environmental gradients (Brown et al. 2020, Perry et al. 2023). Understanding the role of bioeroders is particularly relevant in the current context, where increasing human pressures (e.g., rising temperatures, coastal development, and overfishing) are affecting not only calcifiers but also bioerosion processes, which can vary at different spatial and temporal scales (Perry et al. 2014, Schönberg et al. 2017, Molina-Hernández et al. 2022). Currently, the scenarios observed reflect those reported by Molina-Hernández et al. (2020), in which the low levels of bioerosion are determining the direction (i.e., accretion or net erosion) and speed with which carbonate balances shift towards neutral scenarios (~1-0 kg CaCO3·m-2·y-1) in various highly degraded Caribbean reefs.
Sea urchins are important bioeroders in coral reefs (Bak 1994, Glynn and Manzello 2015). Historically, the main eroding urchin in the tropical Atlantic Ocean has been Diadema antillarum (Bak 1994, Hutchings 2011). However, given the mass mortality of this species as a result of a disease outbreak in the 1980s, its populations drastically decreased throughout the region (Lessios et al. 1984). At present, most Caribbean reefs are characterized by low urchin abundance (Hughes et al. 2010). Diadema antillarum has shown localized recovery in certain sites (Jorgensen et al. 2008, Keller 2011, Johnston et al. 2021), but recent disease outbreaks have caused new mortality events (Levitan et al. 2023), which could be reversing recovery trends in its populations (Tuohy et al. 2020). In the Gulf of Mexico, current urchin populations are larger than in the Caribbean; however, they are dominated by urchins of the genus Echinometra (González-Azcárraga 2009, Morales-Quijano et al. 2017, Johnston et al. 2021). Mass mortality events have not only reduced D. antillarum populations, but also their contribution to coral reef bioerosion, going from values of up to 5.3 kg CaCO3·m-2·y-1 (Scoffin et al. 1980) to only 0.1 kg CaCO3·m-2·y-1 (Perry et al. 2014). Currently, parrotfishes are considered the dominant bioerosion agents in the Caribbean (Perry et al. 2014, Molina-Hernández and Álvarez-Filip 2024).
Rates of urchin bioerosion have decreased by up to 90% due to low population densities in the Caribbean (Perry et al. 2014). However, the population of urchins could be underestimated as most surveys are performed during the day, when urchins retreat into their shelters (Mills et al. 2000, Tuya et al. 2004, Young and Bellwood 2011). Most urchin species are nocturnal and have a peak in their movement patterns (movement on the substrate) during the night when they feed. Their activity decreases as dawn breaks, and urchins are cryptic during the day (Smith et al. 2024). Likewise, under experimental conditions, 90% of sea urchins show negative phototaxis and seek refuge when exposed to ultraviolet light (Adams 2001). However, it remains to be confirmed whether this behavior in natural conditions is related to other variables such as depth and solar radiation (Adams 2001). In addition, reef structural heterogeneity can influence the population density of sea urchins; as depth increases, colonies tend to have flatter morphologies (due to decreased light), and the amount of sand increases, so there is less space available for shelter (Griffin et al. 2003). This is important because erosive species (D. antillarum, Echinometra spp., and Eucidaris tribuloides) prefer rocky and coral substrates (Celaya-Hernández et al. 2008, González-Azcárraga 2009), with bigger population densities at depths of 3-5 m for D. antillarum (Sellers et al. 2010), 1-6 m for Echinometra viridis (Griffin et al. 2003), and 1-3 m for E. tribuloides (McPherson 2018).
In the Campeche Bank, and in most of the Gulf of Mexico, sea urchin research has focused mainly on the taxonomy (Durán-González et al. 2005, Solís-Marín et al. 2013) and the distribution and species composition of all echinoderm groups (Vázquez-Bader et al. 2008). Few studies have focused on the taxonomic diversity, the description of urchin habitat, and variations in D. antillarum population density between day and night, although the latter has been addressed in a very lax manner (Celaya-Hernández et al. 2008, González-Azcárraga 2009). Almost all the information available for the southeastern Gulf of Mexico is restricted to the Veracruz Reef System (VRS) (Durán-González et al. 2005, Celaya-Hernández et al. 2008, González-Azcárraga 2009).
For this region, some authors report Echinometra lucunter as one of the most abundant species at shallow depths, both in the crest area and in the reef lagoon, where it is associated with rocky-coral substrate (Lara et al. 1992, Celaya-Hernández et al. 2008). González-Azcárraga (2009) found that population density and species diversity are greater in the Gulf of Mexico than in the Mexican Caribbean, and observed higher values for the 2 variables at shallow depths (1-10 m) than at greater depths (10-15 m) in both regions. However, in recent years, D. antillarum has shown low population densities (~0.5 ind·m-2) in both regions (Celaya-Hernández et al. 2008, González-Azcárraga 2009).
The above indicates that the study on the ecological functionality of sea urchins with erosive potential (i.e., D. antillarum, E. viridis, E. lucunter, and E. tribuloides) still presents gaps, both ecological and geographical, in the Gulf of Mexico and, especially, in the Campeche Bank. Therefore, in this study, we evaluated and compared the abundance of sea urchins and their bioerosion potential between diurnal and nocturnal visual surveys conducted at 3 sites with different characteristics of reef heterogeneity. Due to the nocturnal habits of sea urchins, we expected greater variation in urchin abundance between day and night in locations with greater reef heterogeneity and, consequently, higher bioerosion rates.
MATERIALS AND METHODS
Study area
Cayo Arenas (Fig. 1) belongs to a set of reefs on the Campeche Bank in the Gulf of Mexico (Logan et al. 1969, Sanvicente-Añorve et al. 2014). The Campeche Bank does not have reef barriers, which leaves the platform devoid of physical protection from wave and wind energy, and open to the influence of the Loop Current in the western part of the Gulf of Mexico (Logan et al. 1969). Cayo Arenas is considered a platform reef with complex reef structures, called “walls”, with depths up to 30 m (Tunnell and Chávez 2013). In addition, it is a remote reef with limited access located ~200 km from the coastal area. Various authors have classified these reef structures based on their location: the northeastern structure, which is the largest formation with 2 emergent zones, and the southeastern structure, the smallest of the structures with one small emergent zone, which receives the greatest energy from the wind and waves along with the Northeast wall. The eastern structure has a lagoon on the windward side and a cay with vegetation on sand blocks and coral skeletons; due to its location, the structure is protected from wave and wind energy (Logan et al. 1969, Chávez et al. 2007, Tunnell and Chávez 2013).
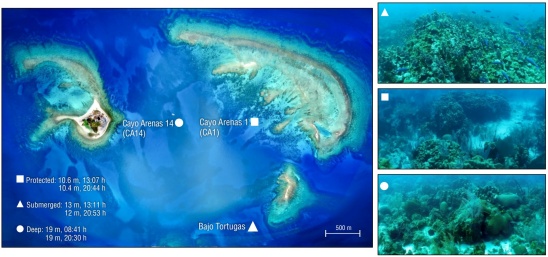
Figure 1 Location of the 3 sampling sites in Cayo Arenas, Campeche Bank. The photos show the representative characteristics of the coral community and the depth and sampling schedule for each Site. The photo of Cayo Arenas is an orthomosaic composed of photos taken by drone; the flights and construction of the orthomosaic were done by Lorenzo Alvarez-Filip. Lorenzo Alvarez-Filip and Esmeralda Pérez-Cervantes took the photos of the reef.
The study area covered 3 sites (Bajo Tortugas, Cayo Arenas 1 [CA1], and Cayo Arenas 14 [CA14]), located in the protected zone of the reef, with different depths and benthic components, such as coral, sand, and hard substrate (i.e., calcareous matrix that is not covered by coral tissue). Bajo Tortugas is a submerged reef that was sampled between 12 and 13 m depth. This site had a well-developed coral community, with a coral cover of 23%, absence of sand cover, and a hard substrate cover of 77%, which made it the site with the greatest heterogeneity and, therefore, available habitat. In Bajo Tortugas, the reef structure was mainly formed by corals with massive and large morphologies such as Orbicella spp. and Colpophyllia natans. In contrast, CA1 was located leeward in the eastern zone and had discontinuous coral patches separated by large sand areas. In addition, this site presented 20% coral cover, 11.6% sand cover, and 68.4% hard substrate cover. Therefore, CA1 showed medium reef heterogeneity. Cayo Arenas 14 was located in the leeward reef lagoon and was sampled down to 19 m. The site had a mixed composition of corals and algae, with 12% coral cover, 8.5% sand cover, and 79.5% hard-substrate cover. Compared to CA1, CA14 had more continuous coral patches, but low reef heterogeneity.
Field work
Diurnal and nocturnal visual surveys were conducted following the ReefBudget methodology (Perry and Lange 2019). For day and night surveys, we deployed 10 × 1 m band transects. However, for night surveys, transects were modified to 20 × 0.5 m to focus on a smaller area and make data collection with the diving lights more efficient during sampling. The transects were 10 m2 for all cases. In each transect, the number of urchins per species and the size of the testa were recorded following size classes of 20 mm (i.e., 0-20, 21-40, 41-60, 61-80, and 81-100 mm). The records were made considering only eroding species, including D. antillarum, E. viridis, E. lucunter, and E. tribuloides. All surveys were conducted with scuba diving equipment in July 2023. Daytime surveys were conducted between 08:00 h and 14:00 h, whereas night durveys were conducted one hour after sunset at all sites (i.e., around 20:00 h). Figure 1 shows the exact monitoring schedules for each site. Current intensity and reef depth complicated monitoring efforts; therefore, the number of transects varied between sites and between diurnal and nocturnal surveys. In CA1, 5 transects were done during the day at 8 m depth and 7 transects at night at 9.8-10 m depth. In Bajo Tortugas, 6 transects were done during the day at 7.5-11.2 m depth and 5 transects at night at 8.5-11 m depth. Finally, in CA14, 6 transects were done for both times of the day, at 18.5 m depth during the day and 17 m depth at night. For all sites, monitoring was only conducted once a day and once at night. The sampling depths were random because transects were deployed on coral substrate.
Estimation of abundance and rate of urchin bioerosion
Urchin abundance per site (number of individuals) was estimated from the surveys. Urchin bioerosion rate was estimated according to species identity, the number and size of urchins (based on testa diameter), and constants from previously published species-specific regression equations compiled in Perry and Lange (2019). These equations describe the relationship between the size of urchin and the bioerosion values (g urchin·d-1) based on the analysis of stomach content, reported for different urchin sizes (see Perry and Lange 2019). The annual rate was calculated from the daily rate multiplied by 365 (days of the year), and was standardized to 1 m2; values are reported in kg CaCO3·m-2·y-1 (Perry and Lange 2019). This approach assumes constant bioerosion rates throughout the year.
Benthic description of sites
Coral, sand, and hard substrate cover data were obtained through orthomosaic analysis. The information collected helped to determine the reef heterogeneity of the sites based on the amount of coral cover, hard substrates, and sand. Underwater images were obtained with scuba diving using 2 cameras equipped with waterproof housing. For each site, orthomosaics were constructed using underwater photogrammetry following the methodology proposed by Hernández-Landa et al. (2020). This consisted of delimiting plots of ~400 m2 with transects and polyvinyl marks (6 m × 0.6 m). Each plot was divided in 2 by the central mark and each half was photographed by 2 divers swimming at a speed of ~5 m·min-1. Photographs were taken 2 m above the average depth of the reef bottom. Depth was recorded at the beginning and end of each transect with a standard dive computer. For each site, ~4,000 images were obtained with a high level of overlap (>70%). The images were processed in AgiSoft Metashape v. 2.0.3 (Agisoft 2024) with Structure from Motion algorithms. To build orthomosaics and digital elevation models, 3 steps were followed: (1) initial processing (internal and external orientation of the camera and creation of sparse clouds); (2) generation of point clouds and meshes; and (3) digital surface modeling (DSM) and orthomosaic construction. Because the images did not have geolocation, the polyvinyl marks were used as a contrast measure to transform the model into absolute measurements (Hernández-Landa et al. 2020).
The orthomosaics were analyzed in ArcMap v. 10.6 (ESRI 2017). Sections of each plot were joined along the center mark using spatial snap extension. One orthomosaic was constructed per site, and the scleractinian coral colonies were identified to the species level (Human and Deloach 2002, Lang and Marks 2018). For each colony, coral cover of living tissue was manually digitized following its contour, considering colonies ≥5 cm2. Data matrices were created to estimate the cover (cm2) at each site. For each site, vector layers of coral cover and sand cover were generated in an area of 380 m2. The cover of other hard substrates (i.e., calcareous matrix that is not covered by coral tissue) was estimated by subtracting the digitized area from the total area analyzed.
Data analysis
To explore potential differences between sites and sampling times (day and night monitoring), we used 2 generalized linear models (GLM). We opted for GLM instead of linear models due to their ability to fit distributions of response variables that do not follow a normal distribution. The first model analyzed urchin abundance (total number of individuals observed [1]), whereas the second evaluated bioerosion rates (kg CaCO3·m-2·y-1 [2]). To evaluate differences between individuals between day and night we used abundance instead of density (ind·m-2) because the model requires discrete data (Crawley 2005). However, we present the results using density units to facilitate comparison with previous studies. We fit the models using the ‘glm()’ function. The dependent variables were urchin abundance (Y1) and bioerosion rate (Y2), whereas predictor variables included the site (Bajo Tortugas, CA1, and CA14), the survey time (diurnal or nocturnal), and the interaction between both variables. Data were not transformed for analysis; however, different distributions were used to address the non-normal distribution for the response variables: (1) a Poisson distribution with log link (log) for abundance and (2) a Gaussian distribution with identity link function for the rate of urchin bioerosion. Model validation was carried out using residual plots generated with the ‘simulateResiduals()’ function of the ‘DHARMa’ package. Subsequently, for each model, we performed a 2-way analysis of variance (ANOVA) (Type II Wald chi-square tests using the ‘Anova()’ function from the ‘car’ package. We performed post hoc tests using adjusted marginal means analyses with the Bonferroni correction with the ‘emmeans()’ function from the package of the same name. All analyses were performed in R v. 2023.12.0 (R Core Team 2023).
The models used were mathematically represented as follows:
where Y1 and Y2 represent the dependent variables (urchin abundance and bioerosion rate, respectively); β0 is the intercept; β1 and β2 are the coefficients associated with the survey time (X1) and the site (X2), respectively; β3 corresponds to the interaction coefficient between survey time and site (X1·X2); and ϵ is the residual error term, which follows a normal distribution.
RESULTS
Urchin abundance
Cayo Arenas had greater urchin abundance during the night (14.5 ± 3.8 ind) than during the day (2.6 ± 0.8 ind). This pattern was observed at the sites Bajo Tortugas and CA1 (Fig. 3). For the 3 sites, the most abundant species was D. antillarum, both in the diurnal (1.4 ± 0.4 ind) and nocturnal (10.2 ± 3.3 ind) surveys, followed by E. viridis (0.18 ± 0.1 ind and 3.9 ± 1.2 ind, respectively), and E. lucunter (1 ± 0.4 ind and 0.16 ± 0.2 ind, respectively); E. tribuloides was only observed during the night (0.2 ± 0.2 ind). Site evaluation showed that Bajo Tortugas had lower urchin abundance in diurnal surveys (2.7 ± 0.95 ind) than in nocturnal surveys (28.4 ± 09.5 ind); the same occurred for CA1 (diurnal: 5.6 ± 1.9 ind; nocturnal: 16.8 ± 3.2 ind). In CA14, there was only one record of the species D. antillarum at night (Table 1).
Table 1 Abundance and estimates of diurnal and nocturnal bioerosion rates of the species of eroding urchins in Cayo Arenas.
| Site | Species | Abundance (ind) | Bioerosion rate (kg CaCO3·m-2·y-1) | ||
| Diurnal | Nocturnal | Diurnal | Nocturnal | ||
| Bajo Tortugas | Diadema antillarum | 2.2 ± 0.7 n = 13 | 24.6 ± 9.0 n = 123 | 0.28 ± 0.1 | 3.85 ± 1.5 |
| Echinometra viridis | 0.17 ± 0.2 n = 1 | 03.6 ± 1.0 n = 18 | 0.003 ± 0.003 | 0.05 ± 0.01 | |
| Echinometra lucunter | 0.33 ± 0.2 n = 2 | - | 0.005 ± 0.003 | - | |
| Eucidaris tribuloides | - | 0.2 ± 0.2 n = 1 | - | 0.008 ± 0.008 | |
| Site mean | 2.7 ± 0.95 n = 16 | 28.4 ± 9.5 n = 142 | 0.29 ± 0.1 | 3.9 ± 1.49 | |
| Cayo Arenas 1 | Diadema antillarum | 2.2 ± 1.0 n = 11 | 8.4 ± 1.0 n = 59 | 0.29 ± 0.12 | 1.1 ± 0.13 |
| Echinometra viridis | 0.4 ± 0.2 n = 2 | 7.6 ± 2.4 n = 53 | 0.003 ± 0.003 | 0.09 ± 0.03 | |
| Echinometra lucunter | 3.0 ± 1.0 n = 15 | 0.43 ± 0.4 n = 3 | 0.04 ± 0.013 | 0.007 ± 0.007 | |
| Eucidaris tribuloides | - | 0.43 ± 0.4 n = 3 | - | 0.003 ± 0.003 | |
| Site mean | 5.6 ± 1.9 n = 28 | 16.8 ± 3.2 n = 118 | 0.33 ± 0.13 | 1.18 ± 0.12 | |
| Cayo Arenas 14 | Diadema antillarum | - | 0.17 ± 0.17 n = 1 | - | 0.02 ± 0.02 |
| Site mean | n = 0 | 0.17 ± 0.17 n = 1 | - | 0.02 ± 0.02 | |
| Species | Diadema antillarum | 1.4 ± 0.4 n = 24 | 10.2 ± 3.3 n = 183 | 0.2 ± 0.06 | 1.5 ± 0.5 |
| Species mean | 5.9 ± 1.8 n = 207 | - | 0.86 ± 0.3 | - | |
| Echinometra viridis | 0.18 ± 0.1 n = 3 | 3.9 ± 1.2 n = 71 | 0.002 ± 0.001 | 0.05 ± 0.01 | |
| Species mean | 2.1 ± 0.7 n = 74 | - | 0.03 ± 0.008 | - | |
| Echinometra lucunter | 1.0 ± 0.4 n = 17 | 0.16 ± 0.2 n = 3 | 0.013 ± 0.006 | 0.003 ± 0.003 | |
| Species mean | 0.6 ± 0.2 n = 20 | - | 0.008 ± 0.003 | - | |
| Eucidaris tribuloides | - | 0.2 ± 0.2 n = 4 | 0 ± 0 | 0.003 ± 0.002 | |
| Species mean | 0.11 ± 0.1 n = 4 | - | 0.002 ± 0.001 | - | |
| Cayo Arenas | Mean | 2.6 ± 0.8 n = 44 | 14.5 ± 3.8 n = 261 | 0.2 ± 0.06 | 1.55 ± 0.53 |
Data are shown as the mean ± standard error and number of individuals (n). Diurnal surveys were conducted between 08:00-13:00 h and nocturnal surveys at around 20:00 h (see Fig. 2). No individuals found for the species monitored transect (-).

Figure 2 Graphic representation of the number of Diadema antillarum urchins during nocturnal surveys at the 3 sampling sites. A gradient is observed in the abundance of urchins that decreases from left to right: Bajo Tortugas (5 urchins), Cayo Arenas 1 (3 urchins), and Cayo Arenas 14 (no urchins). Lorenzo Alvarez-Filip and Esmeralda Pérez-Cervantes took the photos of the urchins.
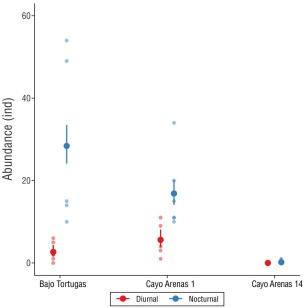
Figure 3 Urchin abundance (number of individuals) in the Cayo Arenas reef, Campeche Bank. The points indicate the predicted means for each site and survey type (diurnal and nocturnal), and the error bars represent the 95% confidence intervals (CI). Both calculations come from the adjusted marginal means based on the generalized linear model. Non-overlapping 95% CI are significantly different.
The 2-way ANOVA showed differences in urchin abundance between sites (χ2(2) = 33, P < 0.05), between diurnal and nocturnal surveys (χ2(1) = 93.9, P < 0.05), and between the interaction of these variables (χ2(2) = 14, P < 0.05). The post hoc test showed differences between diurnal and nocturnal abundance in Bajo Tortugas (P < 0.05) and in CA1 (P < 0.05) (Fig. 3).
Bioerosion potential
The bioerosion potential for Cayo Arenas was significantly higher in nocturnal surveys (1.55 ± 0.53 kg CaCO3·m-2·y-1) than in diurnal surveys (0.2 ± 0.06 kg CaCO3·m-2·y-1). In Cayo Arenas, the species with the greatest contribution to the bioerosion potential, both during the day and at night, was D. antillarum (0.19 ± 0.06 kg CaCO3·m-2·y-1 and 1.5 ± 0.53 kg CaCO3·m-2·y-1, respectively), followed by E. viridis (0.002 ± 0.001 kg CaCO3·m-2·y-1 and 0.05 ± 0.014 kg CaCO3·m-2·y-1, respectively), E. lucunter (0.013 ± 0.006 kg CaCO3·m-2·y-1 and 0.003 ± 0.003 kg CaCO3·m-2·y-1, respectively), and E. tribuloides (0.003 ± 0.002 kg CaCO3·m-2·y-1 in nocturnal surveys). In Bajo Tortugas, the diurnal bioerosion rate was 0.29 ± 0.1 kg CaCO3·m-2·y-1 and the nocturnal bioerosion rate was 3.9 ± 1.49 kg CaCO3·m-2·y-1. In CA1, diurnal and nocturnal bioerosion rates were 0.33 ± 0.13 kg CaCO3·m-2·y-1 and 1.18 ± 0.12 kg CaCO3·m-2·y-1, respectively; in CA14, the nocturnal bioerosion rate was 0.02 ± 0.02 kg CaCO3·m-2·y-1 (Table 1).
The statistical analyses used to compare bioerosion rates between sites and between diurnal and nocturnal surveys showed differences in the bioerosion rate between sites (χ 2 (2) = 14.8, P < 0.05) and between the 2 types of survey (χ 2 (1) = 11.4, P < 0.05). Furthermore, urchin bioerosion rates in Cayo Arenas showed a significant interaction between both variables (χ2(2) = 12.7, P < 0.05). The post hoc test showed differences between sites and between the 2 types of survey (Fig. 4); however, only Bajo Tortugas showed differences between nocturnal and diurnal bioerosion rates (P < 0.05) (Fig. 4).
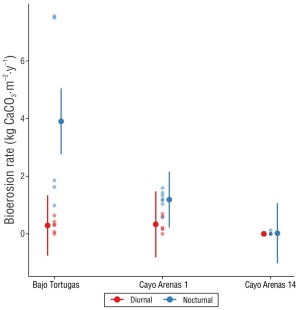
Figure 4 Contribution of sea urchins to the bioerosion potential in the Cayo Arenas reef, Campeche Bank. The points indicate the predicted means for each site and the type of survey (diurnal and nocturnal), and the error bars represent the 95% confidence intervals (CI). Both calculations come from the adjusted marginal means based on the generalized linear model. Non-overlapping 95% CI are significantly different
DISCUSSION
In this study, we found significant changes in population densities and bioerosion potential between diurnal and nocturnal surveys in Cayo Arenas. In addition, the most abundant species with the greatest changes in diurnal and nocturnal abundance was D. antillarum. Due to its high nocturnal abundance, D. antillarum boosted the total bioerosion potential, which is, the summed effect of the 4 species of eroding urchins found in the study sites. Furthermore, we observed that the site with the greatest changes between day and night for both variables was the site that had the reef with the greatest reef heterogeneity, that is, with continuous coral patches and without sand patches (Bajo Tortugas). At the site with the greatest depth and largest sand patches, only one urchin (CA14) was recorded. In general, the results indicated that bioerosion rates are proportional to the abundance of urchins (Figs. 3, 4), therefore, in sites where they are still abundant, such as in Cayo Arenas, urchins have an important role in the carbonate dynamics in the reef.
Our findings evidence that there are still reef sites dominated by D. antillarum, despite historical and current reports of the mortality of this species (Lessios 2016, Levitan et al. 2023). Currently, the density of urchins in the Caribbean continues to be zero for some sites (Kuffner et al. 2019) and very low for others, such as for the back reefs in the Florida Keys, where the average density of D. antillarum urchins ranges between 0.017 to 0.026 ind·m-2 (Chiappone et al. 2013), or the front reefs of the northern Mexican Caribbean (Molina-Hernández et al. 2020). Studies that have reported the abundance of urchins in the Gulf of Mexico have recorded values similar to those of Cayo Arenas. For example, in VRS, average urchin densities have been estimated between 0.646 (González-Azcárraga 2009) and 1.4 ind·m-2 (Horta-Puga et al. 2017), whereas, for the reef banks of Flower Gardens and Stetson Bank in the northern Gulf of Mexico, reports show average densities between 0.28 to 1 ind·m-2 (Nuttal et al. 2020, Johnston et al. 2021). In these studies, the data were obtained during diurnal surveys, which suggests that there is a possibility that urchin densities in VRS and Flower Gardens are higher; therefore, it is recommended to confirm these findings in these and other systems in the Gulf of Mexico through nocturnal surveys, particularly in reefs with high structural heterogeneity.
Regarding the species reported for Cayo Arenas, the one with the highest density was D. antillarum (diurnal and nocturnal surveys), which highlights the importance of the populations of this species in a regional context, since D. antillarum has experienced severe population losses throughout its range (Chiappone et al. 2002, González-Azcárraga 2009, Levitan et al. 2023). The 2 species of the genus Echinometra recorded in this study had much lower densities compared to what was reported in the reef systems of Veracruz, Mexico (González-Azcárraga 2009, Morales-Quijano et al. 2017). For example, in the Lobos-Tuxpan Reef System, Veracruz, average densities for E. viridis and E. lucunter were 6.8 ind·m-2 and 3.6 ind·m-2, respectively (Morales-Quijano et al. 2017), whereas in the VRS, the values recorded for these species were 2.5 and 0.34 ind·m-2, respectively (González-Azcárraga 2009). These values are higher than those reported for D. antillarum in the same studies (0.59 ± 0.18 ind·m-2, Fig. 3) and those recorded for all urchin species in this study in diurnal and nocturnal surveys. This suggests that, in reefs with low abundance of D. antillarum, E. viridis and E. lucunter have become the main reef eroders due to their greater abundance (Dunn et al. 2017, Morales-Quijano et al. 2017), which could be reflected in a reduced bioerosion potential, as is currently occurring in Caribbean reefs (Perry et al. 2014). Unfortunately, there are few records of urchin populations, such as D. antillarum, prior to the mass mortality event of the 1980s. The only source of evidence we found is for the Flower Gardens reefs, where D. antillarum densities were reported to range between 0.5 to 1.40 ind·m-2 before the mass mortality event (Johnston et al. 2021), which is slightly higher than what was found in the present study.
Our results show a clear pattern between surveys carried out during the day and those conducted at night. All urchin species had greater densities during the night, when they are most active. This could be related to urchins coming out at night to search for food because this protects them against predators (Shulman 2020, Smith et al. 2024). This is particularly important in reefs with high structural heterogeneity given that greater heterogeneity (González-Azcáraga 2009, Tuohy et al. 2020) provides more refuge spaces for urchins, which reduces their energy expenditure to avoid predators (Bodmer et al. 2021) and even modulates their functionality in the reef (Lee and Hessen 2006). This suggest that shelter quality can minimize predator attacks (Carpenter 1984). The more evident changes in D. antillarum between day and night could be due to the greater distances traveled by this species during the night (3.7 m, Tuya et al. 2004) compared to those traveled by E. viridis and E. lucunter (10 and 5 cm, respectively; Shulman 2020). This was most evident in Bajo Tortugas, the site with high reef heterogeneity, which not only had the highest urchin densities but the greatest difference between day and night urchin densities, probably because the calcareous structures provide them with greater refuge from natural predators during the day. Conversely, CA14 was predominantly covered by large patches of sand and had the lowest population density. Site CA1 represented an intermediate case; this site was covered by small coral patches interspersed with sand patches and had medium urchin density, without significant changes between day and night. This shows that the abundance of urchins in Cayo Arenas is still subject to reef heterogeneity.
Bioerosion is key in coral reefs because it modulates other processes such as reef accretion and sediment production (Perry and Alvarez-Filip 2019). Our observations show that there are healthy populations of urchins, particularly of D. antillarum, in Cayo Arenas, which have the potential to play an important role in carbonate dynamics. The bioerosion rates reported in this study are the first for Campeche Bank reefs. However, we were able to compare our rates with those of VRS, where studies report a diurnal urchin bioerosion rate, mainly related to E. viridis, of 0.43 kg CaCO3·m-2·y-1 (Cabrera-Rivera 2022). This value triples when compared to the nocturnal erosion of Cayo Arenas. An important finding of our study is that the potential for nocturnal urchin bioerosion in our study area (1.55 ± 0.53 kg CaCO3·m-2·y-1) can exceed the CaCO3 production by corals, even in reefs with high gross production such as Bonaire reefs (>5 kg CaCO3·m-2·y-1, Perry et al. 2018). For example, Bajo Tortugas would have to have exceptionally high gross production (~9 kg CaCO3·m-2·y-1) to remain in a state of net accumulation. The results reported here suggest that the surveys conducted during the day could be underestimating the density and bioerosion values; however, more studies are needed in other reefs to corroborate this.
Although not analyzed in this study, it is possible that differences in abundances observed between diurnal and nocturnal samplings are also reflected in the estimation of other key functions performed by urchins, such as herbivory, since this function also depends on species, size, and abundance (Ogden and Lobel 1978, Lange and Perry 2019, Williams 2022). Future research will be necessary to characterize possible variations, not only in urchin erosion rates, but also herbivory along different environmental gradients. Considering the relevant functional spectrum of urchins in reef systems, this information will contribute, in a comprehensive manner, to the implementation of conservation and management strategies (Molina-Hernández and Álvarez-Filip 2024), especially at sites with reported increase in urchin abundance (Myhre and Acevedo-Gutiérrez 2007, Vermeij et al. 2010, Keller 2011, Johnston et al. 2021).
Although it has been observed that reefs in good condition can withstand high erosion rates (Hoey and Bellwood 2008), any impact that affects calcification processes will have serious implications for the delicate balance between erosion and CaCO3 production, potentially causing an abrupt transition to net erosive states (Cornwall et al. 2021). This is particularly relevant in reef systems such as Cayo Arenas, where despite no documented mass bleaching events or coral diseases, any event that results in significant coral mortalities will increase the impact of urchin bioerosion, which could lead to immediate imbalances in net production rates with negative consequences at the reef scale. An example of this was observed in the reefs of the tropical eastern Pacific, where an increase of up to 42% in the rate of urchin bioerosion was estimated after a mass coral mortality event, causing widespread degradation of reefs in the region (Alvarado et al. 2016).
In general terms, our study showed that the size of the urchin populations is larger than previously recognized and that they have a high erosive potential in the reef system of Cayo Arenas, Campeche Bank. It is still necessary to explore in more detail the ecological factors that could influence this circumstance, such as reef zones, the heterogeneity of the reef substrate, depth gradients, macroalgae cover, and the presence of natural predators. Nevertheless, it is likely that the remoteness from the coast has provided Cayo Arenas with a certain level of protection from direct anthropogenic threats, which contributes to the conservation of coral reefs and indirectly benefits the habitat of D. antillarum, maintaining it as the most abundant eroding urchin compared to other reefs in the Gulf of Mexico and the Caribbean. Furthermore, given that fishing pressure is relatively low due to the distance from the coast (Ocaña et al. 2019), it is possible that ecological interactions of predation could even control urchin population booms and, therefore, erosion pressure on these reefs. However, there is still little information about this reef and the rest of Campeche Bank and it is extremely important to continue and encourage long-term research in this system. With this, important information can be generated to allow us to understand ecological processes, responses to anthropogenic threats, possible recovery mechanisms of urchin populations, and important reef functions of urchin in other reefs of the Atlantic Ocean, where their populations continue to be decimated.
CONCLUSIONS
Cayo Arenas is a reef with high densities of eroding urchins, which was more evident during the night when the urchins emerge from their shelters and are easier to observe. This pattern was observed in the site with the greatest reef heterogeneity, and especially with the species D. antillarum, which had the highest population density. In this reef system, urchins are still subject to natural conditions such as substrate heterogeneity, depth, and environmental changes (i.e., between day and night). Likewise, these organisms continue to perform geo-ecological functions, since they show a high bioerosion potential dominated by D. antillarum. Continued monitoring is important in this area to understand the dynamics of urchin populations and their possible interaction with natural predators and geo-ecological variables in the face of the effects of climate change on these remote reefs.











 texto en
texto en 



