Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista mexicana de fitopatología
versión On-line ISSN 2007-8080versión impresa ISSN 0185-3309
Rev. mex. fitopatol vol.43 no.spe Texcoco 2025 Epub 01-Dic-2025
https://doi.org/10.18781/r.mex.fit.2024-33
Scientific Article
Microbial control of Meloidogyne incognita on Capsicum chinense under an organic production system
1 Tecnológico Nacional de México/Campus Conkal, División de Estudios de Posgrado e Investigación, Avenida Tecnológico, Conkal, Yucatán, CP. 97345, México.
Background/objective.
Habanero pepper (Capsicum chinense) cultivation is limited by root-knot nematodes such as Meloidogyne incognita, and its control is achieved through the repeated application of synthetic nematicides. An alternative is the application of microbial agents. The objective of this study was to evaluate the control of M. incognita with native Trichoderma spp. in C. chinense, in an organic production system.
Materials and Methods.
Seedlings of the 45-day-old Izamal cultivar were used and transplanted with a substrate based on soil, bocashi and volcanic rocks (6:3:1), three inoculations of native strains of Trichoderma asperellum (Ta13-17), T. erinaceum (Te10- 15) and their combination were applied at the time of transplantation, and eight and 15 days later, as control treatments: a nematicide (Vydate®) and water. Four destructive samplings were carried out during the crop cycle, and the variables were the severity with the galling index, number of eggs and females per g of root.
Results.
In relation to the control, the combination of T. asperellum (Ta13-17) and T. erinaceum (Te10-15) caused significantly lower severity of the nematode estimated with the AUDPC. The apparent infection rate using the Weibull model (1/b) and final galling index (4.13%), also showed the lowest average reproduction of the nematode (number of eggs and females per g of root). With the combination of T. asperellum (Ta13-17) and T. erinaceum (Te10-15) the crop yield was significantly improved by 47.26 and 34.25 % in relation to the control and Vydate®, respectively.
Conclusion
. In an organic production system of habanero pepper for the control of M. incognita; the individual application of T. asperellum (Ta13-17) significantly decreased the gall index; however, when combined with T. erinaceum (Te10-15) it improved nematode control (86.17%).
Keywords: Habanero pepper; Trichoderma asperellum; T. erinaceum; Effectiveness; Phytonematode
Antecedentes/Objetivo
. El cultivo de chile habanero (Capsicum chinense) es limitado por nematodos agalladores como Meloidogyne incognita, su control se realiza con la aplicación recurrente de nematicidas sintéticos. Una alternativa es con la aplicación de especies de agentes microbianos. El objetivo del estudio fue evaluar el control de M. incognita con especies nativas de Trichoderma spp. en C. chinense, en un sistema de producción orgánica.
Materiales y Métodos.
Se utilizaron plántulas del cultivar Izamal de 45 días, y trasplantadas con sustrato a base de suelo, bocashi y rocas volcánicas (6:3:1), se aplicaron tres inoculaciones de cepas nativas de Trichoderma asperellum (Ta13-17), T. erinaceum (Te10-15) y su combinación; al momento del trasplante, ocho y 15 días posteriores, como tratamientos testigo: un nematicida (Vydate®) y agua. Se realizaron cuatro muestreos destructivos durante el ciclo del cultivo, y las variables fueron la severidad con el índice de agallamiento, número de huevos y hembras por g de raíz.
Resultados.
En relación al testigo, la combinación de T. asperellum (Ta13-17) y T. erinaceum (Te10-15) causó significativamente menor severidad del nematodo estimado con el ABCPE. La tasa de infección aparente mediante el modelo Weibull (1/b) e índice de agallamiento final (4.13 %), también mostró, los menores promedios de reproducción del nematodo (número de huevos y hembras por g de raíz). Con la combinación de T. asperellum (Ta13-17) y T. erinaceum (Te10-15) se mejoró significativamente el rendimiento del cultivo en un 47.26 y un 34.25 % en relación al testigo y al Vydate®, respectivamente.
Conclusión.
En un sistema de producción orgánico de chile habanero para el control de M. incognita; la aplicación individual de T. asperellum (Ta13-17) disminuyó de manera significativa el índice de agallamiento; sin embargo, cuando se combinó con T. erinaceum (Te 10-15) mejoró el control del nematodo (86.17 %.).
Palabras clave: Chile habanero; Trichoderma asperellum; T. erinaceum; Efectividad; Fitonematodo
Introduction
Vegetable crops in tropical and subtropical areas largely depend on proper management of root-knot nematodes (Sikora and Fernández, 2005; Ulloa et al., 2016), especially Meloidogyne spp., which are difficult to control due to their wide host range, high reproduction rates, and parasitic lifestyle (Manzanilla et al., 2004; Guerrero-Abad et al., 2021).
In general, M. incognita induces the formation of galls (hypertrophy and hyperplasia) in roots, which block the flow of water and nutrients. As a result of vascular system malfunction, plant growth is reduced, causing chlorosis, wilting, flower and fruit drop, and yield losses (Hernández et al., 2011). Management of these nematodes has relied on the use of organophosphate and carbamate nematicides (Marbán and Manzanilla, 2012). However, due to their toxicity and environmental persistence, there is an increasing focus on evaluating alternative control methods compatible with agroecosystem health (Xie et al., 2015), such as Trichoderma spp., which act as antagonists of root pathogens (Szabó et al., 2013).
The efficiency of Trichoderma spp. in regulating populations of root parasites such as nematodes depends mainly on the strain origin, its interaction with the host, its ability to adapt to the environment where it is introduced, and the type of nematode parasitism (Zhang et al., 2014). Research on the use of antagonistic microorganisms is steadily increasing (Hallman et al., 2009; Corazon-Guivin et al., 2024).
Among the microorganisms that parasitize nematodes, fungi are especially promising because they have shown strong potential (Martínez-Canto et al., 2023). Trichoderma spp. are active mycoparasites that have been widely studied for the biocontrol of fungi causing foliar and root diseases (Martínez-Canto et al., 2021; Natsiopoulos et al., 2024). Based on this, the objective of this study was to evaluate, under protected conditions, the individual and combined application of two Trichoderma species on Capsicum chinense for the control of Meloidogyne incognita.
Materials and Methods
This study was conducted under protected conditions (greenhouse) and in the Phytopathology Laboratory of the Tecnológico Nacional de México/Campus Conkal, located on Avenida Tecnológico S/N, between 21°02’ and 21°08’ N latitude and 89°29’ and 89°35’ W longitude.
Experiment setup. Forty-five-day-old seedlings of habanero pepper cv. Izamal (red at full maturity) were transplanted into 5-kg bags filled with a substrate composed of soil, bocashi, and volcanic rock in a 6:3:1 ratio. The bags were arranged in a randomized complete block design inside a tunnel-type greenhouse with a plastic roof cover and walls lined with anti- aphid mesh. Five treatments were established: 1) T. asperellum (Ta13-17), 2) T. erinaceum (Te10-15), 3) a combination of T. asperellum (Ta13-17) and T. erinaceum (Te10-15), and two controls: 4) Vydate® (a.i. oxamyl 24% of chemical synthesis) applied at 2 mL L⁻¹ of water at the time of transplanting, and 5) an untreated control, consisting of plants without nematode management.
Isolation of Trichoderma spp. strains and inoculation in pepper plants. The Trichoderma strains evaluated were provided by the Fungal Culture Collection of the Phytopathology Laboratory at Campus Conkal, identified as T. asperellum (Ta13-17) (Celis-Perera et al., 2021) and T. erinaceum (Te10-15) (Martínez et al., 2021). For use, the strains were reactivated on potato dextrose agar (PDA) medium and incubated at 28 °C for 15 days. The fungal inoculum was then prepared (1×10³ spores mL⁻¹). Before transplanting, this inoculum was applied separately and in combination to the soil, followed by two additional inoculations at eight and 15 days after transplanting.
Preparation of Meloidogyne incognita inoculum and plant inoculation. From established populations of M. incognita maintained under protected conditions in Solanum lycopersicum crops, galled roots were collected and dissected to obtain nematode egg masses. These were then surface-disinfested to recover nematode eggs. Disinfestation was carried out with 2% sodium hypochlorite for two minutes, followed immediately by successive rinses with tap water using sieves with mesh numbers 45, 100, 200, 325, and 400 (Ayoub, 1980). Nematode egg inoculation was applied to the substrate contained in 5- kg bags, with each bag inoculated with 500 larvae eggs (Martínez et al., 2023).
Variables for estimating M. incognita control. To assess treatment effectiveness, the variables of galling severity and reproduction were evaluated at 56, 90, 122, and 137 days after transplanting (dat). For severity, a six-class gall index scale was used (Taylor and Sasser, 1978). The midpoint of each class was used to perform the following analyses with epidemiological parameters: Area Under the Disease Progress Curve (AUDPC), apparent infection rate using the inverse of parameter b (1/b) with the Weibull model, and final severity using the Yfinal parameter (Pennypacker et al., 1980; Campbell and Madden, 1990).
To evaluate the effect of treatments on M. incognita reproduction, eggs and females were counted per gram of root. Habanero pepper roots were fragmented, homogenized, and two grams were sampled. One gram was blended for 10 s with 20 mL of chlorine and 30 mL of tap water, then filtered through sieves with mesh sizes 45, 100, 200, 325, and 400. The collected eggs were washed with running water and counted using a nematode counting chamber under a compound microscope at 4× magnification. The other gram of root was stained with pre-prepared acid fuchsin by placing 1 g of root in a flask, adding 2 mL of acid fuchsin with 50 mL of tap water, and heating to boiling. After cooling, roots were rinsed with tap water to remove excess stain, and glycerin was added for preservation and later evaluation. Females were separated and counted from the stained roots under a stereomicroscope (Moo et al., 2018; Martínez et al., 2023).
Agronomic variables. In each of the four samplings (56, 122, and 137 dat), variables associated with crop productivity were measured. Plant height was recorded after removing plants from the bags and separating shoots from roots. Using a measuring tape, the distance from the plant apex to the stem base was measured. For leaf and stem dry weight, samples were placed in kraft paper bags and dried in an oven at 50 °C for 10 days until constant weight was reached. Root dry weight was obtained after washing the roots and evaluating disease severity, followed by drying, using the same procedure as for stems and leaves. Root volume was determined by water displacement using a 1000 mL graduated cylinder. Yield was estimated from eight harvests during the crop cycle, recording fruit weight, polar diameter, and equatorial diameter.
Results analysis. Epidemiological and nematode reproduction analyses, as well as agronomic variables, were processed using SAS software version 9.4 for Windows. For nematode reproduction variables, analyses of variance (ANOVA) were performed at the specified sampling dates. Mean comparisons were carried out using Tukey’s test (p≤0.05).
Results and Discussion
Microbial control effect of M. incognita in habanero pepper. During the first sampling at 56 dat, the combined treatment of T. asperellum (Ta13-17) and T. erinaceum (Te10-15) showed the lowest gall index, a trend that continued through the final sampling at 137 dat, with an 86.17% reduction. This was followed by T. asperellum (Ta13-17) alone with a 65.26% reduction, while the untreated control without fungal inoculants showed the highest nematode damage, averaging 29.88% (Figure 1). Similar results were reported by Moo- Koh et al. (2018), where the interaction between two strains, T. citrinoviride (Th33-58) and T. harzianum (Th33-59), reduced root damage severity by 83%. Likewise, Affokpon et al. (2011) reported a lower gall index with T. asperellum (T-12) at nine weeks compared with the uninoculated control.
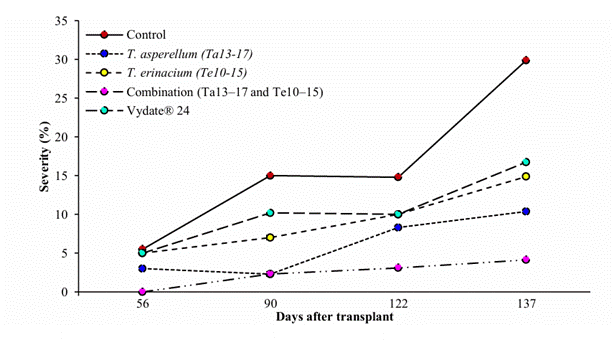
Figure 1 Curves of the progress of the gall index in the organic cultivation of C. chinense, during the period of 56, 90, 122 y 137 days after transplant.
Microbial control effect on epidemiological parameters. The AUDPC values showed that the combination of T. asperellum (Ta13-17) and T. erinaceum (Te10-15) resulted in the lowest disease progress, with 138.19 unit % day⁻¹, followed by T. asperellum alone with 376.94 unit % day⁻¹. This indicated that disease control was improved in these two treatments compared with the control. In an integrated management study of root-knot nematodes (Nacobbus aberrans and M. incognita) using biosolarization and T. viride, 457 unit % day⁻¹ was recorded (Magallanes, 2021), also showing that the accumulation of organic matter in association with antagonistic and saprophytic organisms can reduce M. incognita populations (Pérez et al., 2019).
Disease progression was fitted to the Weibull model, which allowed estimation of the apparent infection rate through the inverse of parameter b (1/b) (Pennypacker et al., 1980). The treatments that resulted in the lowest apparent infection rates (disease progression speed) were the combination T. asperellum (Ta13-17) / T. erinaceum (Te10-15), Vydate®, and T. erinaceum, demonstrating that the application intervals were suitable for disease control. In contrast, the untreated control showed the highest disease intensity (Table 1). At the end of the experiment, the Yfinal parameter showed that the combination of Ta13- 17 and Te10-15 provided the best disease control, with a final severity of 4.13%, which was significantly lower (p≤0.01) than the other treatments, all of which showed at least 30% final disease severity (Table 1). The antagonistic effect of Trichoderma spp. in nematode control is associated with their mechanisms of action, including the production of secondary metabolites such as viridin, gliotoxin, and gliovirin, which inhibit not only fungal growth but also egg hatching and the mobility of nematode juvenile stages (Zin and Badaluddin et al., 2020) (Table 1).
Table 1 Effect of treatments on the control of M. incognita estimated with epidemiological parameters in the organic cultivation of C. chinense.
| Treatment | AUDPC (unit % day -1) | Apparent infectionrate 1/b (unit % day -1) | r2 (adjustmentof the modeloWeibull) | Yfinal(%) |
|---|---|---|---|---|
| *T. asperellum* (Ta13-17) | 376.94 c $^z$ | 0.0076 ab | 0.97 | 10.38 ab |
| *T. erinaceum* (Te10-15) | 778.25 b | 0.0072 b | 0.91 | 14.88 ab |
| Combinación | 138.19 c | 0.0044 c | 0.93 | 4.13 b |
| Control | 1202.63 a | 0.0091 a | 0.90 | 29.88 a |
| Vydate® | 815.75 b | 0.0074 b | 0.90 | 16.75 ab |
| DMS* | 293.86 | 0.0016 | - | 11.56 |
ZMeans with the same letters are not statistically different (Tukey, ≤0.05). *Minimal Significant Difference (P≤0.05).
Number of eggs per gram of blended root. At 56 days after transplanting, the combination (Ta13-17 and Te10-15) showed no eggs, and maintained control of this variable with reductions of 81% at 122 days and 77% at 137 days after transplanting compared with the control (Table 2). Moo-Koh et al. (2018) evaluated the interaction of T. simmonsii (Th09-06) / T. harzianum (Th33-59) and achieved a 59.3% reduction in egg numbers. The treatment with T. asperellum alone caused a 37.65% reduction in egg numbers at 137 days after transplanting. Similarly, when T. asperellum strain Ta.90 was applied to tomato plants, egg numbers decreased by 50% (Hernández et al., 2015). The reduction in egg numbers is a consequence of parasitism detected in egg masses and eggs, confirming the effect observed under in vitro conditions, where T. asperellum FbMi6 inhibited egg hatching and caused mortality of M. incognita juveniles (Saharan et al., 2023).
Table 2 Effect of treatments on the number of M. incognita eggs in organic cultivation of C. chinense.
| Treatment | Number of eggs per gram of liquefied root | |||
|---|---|---|---|---|
| 56 dat y | 90 dat | 122 dat | 137 dat | |
| *T. asperellum* (Ta13-17) | 70.25 bc z | 86.50 b | 1214.25 a | 1361.75 ab |
| *T. erinaceum* (Te10-15) | 105.75 bc | 491.75 ab | 1372.25 a | 1487.25 ab |
| Combination | 0.00 c | 55.50 b | 335.25 a | 495.25 b |
| Control | 394.25 a | 944.25 a | 1787.00 a | 2184.25 a |
| Vydate® | 302.25 ab | 843.75 ab | 1540.25 a | 1554.50 ab |
| DMS* | 233.81 | 822.92 | 1978.01 | 1530.49 |
ydat: Days after transplant. Z Means with the same letters are not statistically different (Tukey, ≤0.05). *Minimal Significant Difference (P≤0.05).
Number of females per gram of stained root. At 90 days after transplanting, the treatment with T. asperellum (Ta13-17) reduced the number of females per gram of stained root by 75% compared with the control. A similar effect was observed with the combination T. asperellum (Ta13-17) / T. erinaceum (Te10-15), which maintained nematode reproduction control through 122 days after transplanting. By the end of the crop cycle (137 days after transplanting), the combination (Ta13-17 and Te10-15) reduced the number of females per gram of stained root by 77.31% and 73.65% compared with the control and Vydate®, respectively (Table 3). In another study with tomato, combinations of T. simmonsii (Th09- 06) / T. harzianum (Th33-59), T. virens (Th27-08) / T. harzianum (Th33-59), and T. virens (Th43-13) / T. ghanense (Th26-52) reduced reproduction by 90.1%, 88.1%, and 31.5%, respectively (Moo-Koh et al., 2018). At the end of the crop cycle, the individual treatment with T. asperellum (Ta13-17) reduced reproduction by 43.81% compared with the control.
Table 3 Effect of treatments on the number of M. incognita females in organic cultivation of C. chinense.
| Treatmet | Number of females per gram of dyed root | |||
|---|---|---|---|---|
| 56 dat y | 90 dat | 122 dat | 137 dat | |
| *T. asperellum* (Ta13-17) | 4.50 ab z | 4.50 a | 25.75 a | 27.25 ab |
| *T. erinaceum* (Te10-15) | 11.25 a | 13.25 a | 29 a | 35.25 ab |
| Combination | 0 b | 4.50 a | 6.25 a | 11 b |
| Control | 8.50 ab | 18 a | 41.77 a | 48.5 a |
| Vydate® | 7.50 ab | 37 a | 32.50 a | 41.75 ab |
| DMS* | 10.98 | 33.79 | 35.65 | 34.43 |
ydat: Days after transplant. Z Means with the same letters are not statistically different (Tukey, ≤0.05). *Minimal Significant Difference (P≤0.05).
It has also been suggested that the properties of Trichoderma lie in its ability to parasitize different life stages of M. incognita (Sharon et al., 2011). In particular, T. asperellum improves the tolerance of nematode-infected plants by enhancing biochemical and physiological traits, especially the production of secondary metabolites such as phenolic compounds, which hinder nematode reproduction (Saharan et al., 2023).
Effect of microbial control on crop performance. For the agronomic variables, although no statistically significant differences were observed among treatments, greater effects on plant height, foliar biomass, and root volume were recorded with the combination of Trichoderma strains (Ta13-17 and Te10-15), with averages of 30.85 cm, 95.49 g, and 55.06 cm³, respectively, compared with the individual application of Trichoderma spp. strains. The lowest plant height was observed with the Vydate® treatment, averaging 23.64 cm. Notably, the combination of Trichoderma strains (Ta13-17 and Te10-15) also had a positive effect on stem diameter (Table 4). Application of Trichoderma spp. strains, studied as antagonists of M. incognita, suppressed nematode populations and reduced galling, which tended to improve crop growth, as reflected in the agronomic variables of habanero pepper. Growth promotion occurs through a specific interaction with Trichoderma and the production of indole-3- acetic acid, resulting in increased plant biomass (Contreras et al., 2009; Martínez et al., 2011). However, this response does not occur when the host interacts with a pathogen. In such cases, biocontrol microorganisms like Trichoderma redirect resources to activate defense mechanisms known as induced systemic resistance, through jasmonic acid, salicylic acid, or ethylene pathways, which explains the outcomes observed in this study (Hermosa et al., 2013; Nawrocka et al., 2013).
Table 4 Effect of treatments in the control of M. incognita on the agronomic variables of the organic cultivation of C. chinense.
| Treatment | Plant height(cm) | Stem diameter(mm) | Foliar Biomass(g) | Root Volume(cm3) |
|---|---|---|---|---|
| *T. asperellum* (Ta13-17) | 28.84±2.68 a | 7.84±0.82 ab | 81.9±13.54 a | 48.94±6.3 a |
| *T. erinaceum* (Te10-15) | 26.76±1.36 a | 7.16±0.75 ab | 88.43±15.96 a | 49.31±8.35 a |
| Combination | 30.85±3.14 a | 7.96±0.71 a | 95.49±11.83 a | 55.06±7.05 a |
| Control | 30.41±1.26 a | 7.47±0.67 ab | 76.41±11.46 a | 40.88±6.24 a |
| Vydate® | 23.64±2.94 a | 6.72±0.82 b | 79.88±12.83 a | 44.38±7.76 a |
| DMS* | 9.32 | 1.23 | 52.26 | 16.00 |
Z Means with the same letters are not statistically different (Tukey, ≤0.05). *Minimal Significant Difference (P≤0.05).
Effect of treatments on fruit yield through the control of M. incognita in the organic cultivation of C. chinense. Yield and fruit size were evaluated across eight harvests, and analysis of variance showed significant differences among treatments (p≤0.01). The highest yields were obtained with treatments inoculated with Trichoderma spp. The treatments with T. asperellum (Ta13-17) and the combination of strains (Ta13-17 and Te10-15) not only outperformed the untreated control but also exceeded the nematicide treatment with Vydate®. In the first case, yield reached 244.78 g plant⁻¹, and in the second, 231.08 g plant⁻¹. The largest fruit size was recorded in the treatment with T. erinaceum (Te10-15) (Table 5). Some studies suggest that microbial antagonists may also reduce crop yield due to fungal competition (Meyer et al., 2001), interactions with crop-associated fungi (Harman, 2006), or root suppression, factors that should be considered in future biocontrol studies. However, such an effect was evident in this study (Table 5).
Table 5 Effect of treatments on fruit yield in C. chinense cultivation.
| Treatment | Yield (g plant -1) | Number of fruits | EquatorialDiameter(cm) | Polar Diameter(cm) |
|---|---|---|---|---|
| *T. asperellum* (Ta13-17) | 244.78±20.95 a | 23.59±1.85 a | 2.89±1.02 b | 4.63±1.64 b |
| *T. erinaceum* (Te10-15) | 210.4±17.65 ab | 20.97±1.69 a | 4.51±1.16 a | 11.23±3.97 a |
| Combination | 231.08±18.41 a | 18.94±1.27 ab | 2.89±1.02 b | 3.94±1.39 b |
| Control | 121.85±11.88 c | 12.91±1.29 b | 2.94±1.04 b | 3.94±1.39 b |
| Vydate® | 151.93±15.78 bc | 17.59±1.81 ab | 3.05±1.08 b | 4.79±1.69 b |
| DMS* | 66.47 | 6.18 | 0.45 | 1.13 |
Z Means with the same letters are not statistically different (Tukey, ≤0.05). *Minimal Significant Difference (P≤0.05).
Conclusions
Treatments that included inoculations with T. asperellum (Ta13-17) improved the control of M. incognita, reducing severity by 65.26%, while the combination of Trichoderma strains (Ta13-17 and Te10-15) achieved an 86.17% reduction. Nematode reproduction variables decreased by 75-85% in the number of eggs per gram of blended root and females per gram of stained root at 122 days after transplanting. Similarly, these treatments increased fruit production per plant by 40-50% compared with the control. The AUDPC and the apparent infection rate estimated with the Weibull model showed a significant reduction in the gall index with the individual application of T. asperellum (Ta13-17); however, when combined with T. erinaceum (Te10-15), gall formation was reduced even further.
Limitations
The experiments in this study were conducted under protected conditions, and the results must be validated in the field, since both biotic and abiotic factors influence the effectiveness of antagonists and the plant’s response. It is recommended to detect genes involved in systemic resistance during the interaction between antagonistic fungi and the plant for nematode control.
Conflict of interest
The authors declare no conflict of interest.
Funding
TecNM Project: Microbial Control of Phytopathogens in Tropical Vegetables (21013.24-P)
Author contributions
Citlally Guadalupe Puc-Flores; execution of experiments, sampling, and measurement of variables. Felicia Amalia Moo-Koh; activation of fungal isolates, preparation of treatments, and manuscript drafting. José María Tun Suárez; critical review of the experiment and manuscript drafting process. Eduardo Villanueva-Couoh; design and guidance of the experiments and variable measurements, manuscript review, and final editing. Jairo Cristóbal-Alejo; study supervision, research analysis and design. Leader of the research project. Participated in data analysis, critical review of the manuscript, and its final approval.
REFERENCES
Affokpon, A, Coyne, D, Htay, C, Dossou, R, Lawouin, L, et al. (2011). Biocontrol potential of native Trichoderma isolates againstroot- knot nematodes in West African vegetable production systems. Soil Biology and Biochemistry 43: 600-608. https://doi.org/10.1016/j.soilbio.2010.11.029 [ Links ]
Ayoub, SM. (1980). Plant Nematology an Agricultural TrainingAid. Department of Food and Agriculture Divisionof Plant Industry Laboratory Services Nematology. Editorial Nema aid publications, Sacramento, CA, USA. 157 p. [ Links ]
Campbell, C, Madden, V. (1990). Introduction to plant disease epidemiology. John Wiley & Sons Inc. New York, USA. 532. https://www.cabidigitallibrary.org/doi/full/10.5555/19912305030 [ Links ]
Celis-Perera, SE, Moo-Koh, FA, Reyes-Ramírez, A, Tun-Suárez, JM and Cristóbal-Alejo, J. (2021). In vitro antagonism of Trichoderma asperellum Samuels, Lieckf. & Nirenberg (Ta13-17) against phytopathogenic fungi of Solanum lycopersicum L. Revista Protección Vegetal 36:1-7. https://censa.edicionescervantes.com/index.php/RPV/article/view/1154 [ Links ]
Contreras-Cornejo, HA, Macías-Rodríguez, L, Cortés-Penagos, C and López-Bucio, J. (2009). Trichoderma virens, a plant beneficial fungus, enhances biomassproduction and promoteslateral root growththrough an auxin-dependent mechanism in Arabidopsis. Plant Physiology 149: 1579-1592. https://doi.org/10.1104/pp.108.130369 [ Links ]
Corazon-Guivin, MA, Rengifo del Aguila, S, Corrêa, RX, Cordova-Sinarahua, D, Costa Maia, L, et al. (2024). Native arbuscular mycorrhizal fungi promote Plukenetia volubilis growth and decrease the infection levels of Meloidogyne incognita. Journalof Fungi 10:451. https://doi.org/10.3390/jof10070451 [ Links ]
Guerrero-Abad, JC, Padilla-Domínguez, A, Torres-Flores, E, López Rodríguez, C, Guerrero-Abad, RA, et al. (2021). A pathogen complex between the root knot nematode Meloidogyne incognita and Fusarium verticillioides results in extreme mortality of the inka nut (Plukenetia volubilis). Journalof Applied Botanyand Food Quality. https://doi.org/10.5073/JABFQ.2021.094.019 [ Links ]
Hallman, J, Davies, KG and Sikora, R. (2009). Biological control using microbial pathogens, endophytes and antagonists. In: Perry, R.N., Moens, M., Starr, J.L. (Eds.), Root-knot Nematodes. CAB International, Wallingford, UK. 380-411. https://doi.org/10.1079/9781845934927.0380 [ Links ]
Harman, GE. (2006). Overview of mechanism and uses of Trichoderma spp. Phytopathology 96: 190-194. https://doi.org/10.1094/PHYTO-96-0190 [ Links ]
Hermosa, R, Rubio, M, Cardoza, R, Nicolás, C, Monte, E, et al. (2013). The contribution of Trichoderma to balancing the costs of plant growth and defense. International Journal of Microbiology 16:69-80. https://doi.org/10.2436/20.1501.01.181 [ Links ]
Hernández, M, Sánchez, M, García, J, Mayek, N, González, J, et al. (2011). Caracterización molecular y agronómica de aislados de Trichoderma spp. nativos del noroeste de México. Revista Colombiana de Biotecnología 13:176-185. https://repositorio.unal.edu.co/bitstream/handle/unal/48718/28009-99239-1-PB.pdf?sequence=2&isAllowed=y [ Links ]
Hernández-Ochandía, D, Rodríguez, MG, Peteira, B, Miranda, I, Arias, Y, et al. (2015). Efecto de cepas de Trichoderma asperellum Samuels, Lieckfeldt y Nirenberg sobre el desarrollo del tomate y Meloidogyne incognita (Kofoid y White) Chitwood. Revista de Protección Vegetal 30: 139-147. http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1010-27522015000200008 [ Links ]
Magallanes, M. (2021). Paquetetecnológico para el manejo de los nematodos Naccobus aberrans y Meloidogyne incognita en tomate de invernadero. Colegiode Postgraduados. Institución de enseñanza e investigación en ciencias agrícolas. Campus Montecillo. Fitosanidad. Fitopatología. 81-86. http://colposdigital.colpos.mx:8080/jspui/bitstream/handle/10521/4621/Magallanes_Tapia_MA_DC_Fitopatologia_2021.pdf?sequence=1&isAllowed=y (Consulta, enero 2024). [ Links ]
Manzanilla, H, Kenneth, E and Bridge, J. (2004). Plant diseases causedby nematodes. Pp. 637-716. In: Chen, ZX., Chen, SY, Dickson, DW (eds). Nematology Advancesand Perspectives. NematodeManagement and Utilization. CAB International, Wallingford, UK. https://doi.org/10.1079/9780851996462.0637 [ Links ]
Marbán, M and Manzanilla, R. (2012). Chemical and non-chemical tactics to control plant-parasitic nematodes. Pp:729-759. In: Manzanilla-López, RH and Marbán-Mendoza, N (eds.). Practical plant nematology. Mundi-Prensa. Madrid España.Colegio de Postgraduados. Montecillo, Edo. de Méx., México. https://repository.rothamsted.ac.uk/item/8qwx6/chemical-and-non-chemical-tactics-to-control-plant-parasitic-nematodes [ Links ]
Martínez-Canto, OJ, Cristóbal-Alejo, J, Tun-Suárez, JM y Reyes-Ramírez, A. (2021). Detección de genes Epl1 y Sm1 en Trichoderma spp. antagonistas contra hongos fitopatógenos. Ecosistemas y Recursos Agropecuarios 8:1-8. https://doi.org/10.19136/era.a8n2.2791 [ Links ]
Martínez-Canto, OJ, Cristóbal-Alejo, J, Tun-Suárez, JM and Reyes-Ramírez, A. (2023). Trichoderma erinaceum and Trichoderma virens in the control of Meloidogyne incognita in Solanum lycopersicum. Agrociencia 58:1-13. https://doi.org/10.47163/agrociencia.v57i8.2784 [ Links ]
Martínez, MA, Roldán, A and Pascual, AJ. (2011). Interaction between arbuscular mycorrhizal fungi and Trichoderma harzianum under conventional and low fertilization field condition in melon crops: Growth response and Fusarium wilt biocontrol. Applied Soil Ecology 47: 98 -105. https://doi.org/10.1016/j.apsoil.2010.11.010 [ Links ]
Meyer, SLF, Roberts, DP, Chitwood, DJ, Carta, LK, Lumsden, RD, et al. (2001). Application of Burkholderia cepacia and Trichoderma virens, alone and in combinations, against Meloidogyne incognita on bell pepper. Nematropica 31: 75-86. https://journals.flvc.org/nematropica/article/view/69615 [ Links ]
Moo-Koh, FA, Cristóbal-Alejo, J, Reyes-Ramírez, A, Tun-Suárez, JM, Gamboa-Angulo, M, et al. (2018). Incompatibilidad interespecífica de especiesde Trichoderma contra Meloidogyne incognita en Solanum lycopersicum. Scientia Fungorum 47:37-45. https://scientiafungorum.org.mx/index.php/micologia/article/view/1191/1380 [ Links ]
Natsiopoulos, D, Topalidou, E, Mantzoukas, S and Eliopoulos, PA. (2024). Endophytic Trichoderma: Potential and prospects for plant health management. Pathogens 13: 548. https://doi.org/10.3390/pathogens13070548 [ Links ]
Nawrocka, J, and Malolepsza, U. (2013). Diversity in plant systemicresistance induced by Trichoderma. Biological Control 67:149-156. https://doi.org/10.1016/j.biocontrol.2013.07.005 [ Links ]
Pennypacker, S, Knoble, H, Antle, C and Madden, L. (1980). A flexible model for studyingplant disease progression. Phytopathology 70: 232-235. https://doi.org/10.1094/Phyto-70-232. [ Links ]
Pérez, EA, Cid del Prado, VI, Alatorre, RR, Suárez, EJ, Rodríguez, GM, et al. (2019). Efecto de la biofumigación y Pochonia chlamydosporia en el manejo de nematodos nodulares en tomate. Nematropica 49:172-180. https://journals.flvc.org/nematropica/article/view/119471 [ Links ]
Saharan, R, Patil, J, Yadav, S, Kumar, A and Goyal, V. (2023). The nematicidal potentialof novel fungus, Trichoderma asperellum FbMi6 against Meloidogyne incognita. Scientific Reports 13:1-7. https://doi.org/10.1038/s41598-023-33669-z [ Links ]
Sharon, E, Chet, I and Spiegel, Y. (2011). Trichoderma as a Biological ControlAgent. Pp:183-201. In: Davies, K and Spiegel, Y. (eds) Biological Control of Plant-Parasitic Nematodes: Progress in Biological Control Vol. 11. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-9648-8_8 [ Links ]
Sikora, R and Fernández, E. (2005). Nematode parasites of vegetables. Pp: 319-392. In: Luc, M, Sikora, RA, Bridge, J (eds.), Plant Parasitic Nematodes in Subtropical and Tropical Agriculture. CAB International, Wallingford, UK. https://doi.org/10.1079/9780851997278.0319 [ Links ]
Szabó, M, Urbán, P, Virányi, F, Kredics, L and Fekete, C. (2013). Comparative gene expression profiles of Trichoderma harzianum proteases during in vitro nematodes egg-parasitism. Biological Control 67:337-343. https://doi.org/10.1016/j.biocontrol.2013.09.002 [ Links ]
Taylor, AL and Sasser, JN. (1978). Biology, identification and control of root-knot nematodes (Meloidogyne species). NorthCarolina State University, Graphics, 111p. [ Links ]
Ulloa, M, Wang, C, Saha, S, Hutmacher, RB, Stelly, DM, et al. (2016). Analysis of root-knot nematode and Fusarium wilt disease resistance in cotton (Gossypium spp.) using chromosome substitution lines from two alien species. Genetica 144:167-179. https://doi.org/10.1007/s10709-016-9887-0 [ Links ]
Xie, H, Yan, D, Mao, L, Wang, Q, Li, Y, et al. (2015). Evaluation of methyl bromidealternatives efficacy againstsoil-borne pathogens, nematodes and soil microbial community. Plos One 10:1-12. https://doi.org/10.1371/journal.pone.0117980 [ Links ]
Zhang, S, Gan, Y and Xu, B. (2014). Efficacy of Trichoderma longibrachiatum in the control of Heterodera avenae. BioControl 59:319-331. https://doi.org/10.1007/s10526-014-9566-y [ Links ]
Zin, N and Badaluddin, N. (2020). Biological functions of Trichoderma spp. for agricultura applications. Annals of Agricultural Sciences 65: 168-178. https://doi.org/10.1016/j.aoas.2020.09.003 [ Links ]
Received: November 23, 2024; Accepted: September 19, 2025











 texto en
texto en 


