Introduction
For crocodilians, successful nesting has an important implication on population dynamics, and it is an excellent indicator of how these species adapt to environmental conditions (Ugarte et al., 2013). The embryo development is one of the most critical periods of crocodilians life cycle, associated with increased predation risk due to exposure in less secure areas (Magnusson, 1982; Brazaitis et al., 1996); therefore, oviposition site choice can have an influence on developing embryos and offspring phenotypes (Doody et al., 2009; López-Luna et al., 2020). The nesting and hatching success of crocodilians can be affected by six major factors: fertility, predation, extreme temperatures, moisture conditions, erosion of nest sites, nest abandonment by females and human disturbance to nest sites (Mazzotti, 1989); however, all these parameters depend on when, where, and how females lay eggs into nests and the environmental conditions present during egg incubation (Mazzotti, 1989).
In Mexico, the history of crocodilian nesting observations began in the 1970s (Álvarez del Toro, 1974), but detailed information on the nesting behavior of the American crocodile (Crocodylus acutus, Cuvier, 1807) did not begin until two decades later by Casas-Andreu (2003). Subsequent studies were conducted by Valtierra-Azotla (2007) on the coast of the state of Jalisco. Charruau et al. (2010, 2011, 2012, 2022) conducted studies on an isolated population in an atoll of the Mexican Caribbean, while Cedillo-Leal et al. (2013) and Barragán-Lara et al. (2021) showed information on the nesting ecology and incubation conditions in Oaxaca, and González-Desales et al. (2016) reported information on the reproductive biology of a population from La Encrucijada Biosphere Reserve, Chiapas.
The studies mentioned above do not mention the presence of communal nesting areas. Communal egg-laying in reptiles is defined as the laying of eggs by at least two conspecific gravid females sharing the same nest area or even nest cavity, throughout the nesting season (Espinoza & Lobo, 1996; Doody et al., 2009). There is also the possibility of communal nesting between different species within the same family (Chou, 1979; Vitt, 1986; Krysko et al., 2003) or between different families (Enge et al., 2000; Alfonso et al., 2012; Elsey et al., 2013).
The American crocodile is the most widely distributed crocodile species in the Americas, with an extensive neotropical distribution range and inhabits the Atlantic and Pacific coasts, including the Caribbean and insular areas (Thorbjarnarson et al., 2006). This species is classified as Vulnerable across its range by the International Union for Conservation of Nature (IUCN) Red List, and recently some populations have been transferred to Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) (Rainwater et al., 2022).
The nesting season of C. acutus shows a latitudinal pattern, determined by temperature and precipitation, indicating that nesting begins earlier in lower latitudes (Thorbjarnarson, 1989; Casas-Andreu, 2003). In Oaxaca, nesting occurs along the coastal areas (Álvarez del Toro, 1974), with courtship occurring likely in late January and February (Casas-Andreu, 2003), extending the nesting season from March to May, and egg laying between March and April (Cedillo-Leal et al., 2013). The mean incubation period is 77 days (Barragán-Lara et al., 2021). The hatching usually occurs in the late May and June (Cedillo-Leal et al., 2013). This species builds two types of nests, mound and hole (Mazzotti, 1989); the hole nest is the most frequently found in the Mexican Pacific slope, which is associated with sandy beach sites (Cedillo-Leal et al., 2013; González-Desales et al., 2016).
In respect to communal egg-laying by C. acutus females, Alonso-Tabet et al. (2014) described conspecific egg laying in relatively small areas and called as gregarious nesting in Cuba. Regarding to interspecific communal nesting, some anecdotal observations of communal nesting between C. acutus and different reptiles have been reported in Mexico (Escobedo-Galván et al., 2019; Charruau et al., 2021), and in other countries such as USA (Kushlan, 1982), Haiti (Thorbjarnarson, 1984, 1988), and Cuba (Alonso-Tabet et al., 2014). To our knowledge, only the available information of Alonso-Tabet et al. (2000) showed conspecifics communal nesting between C. acutus gravid females (Alonso-Tabet et al., 2000; Rodríguez-Soberón et al., 2002; Alonso-Tabet et al., 2014).
Therefore, the nesting success for communal egg-laying (CEL) in crocodilian nests has not been studied in depth. Specifically, until now, spatial analyses or descriptions of CEL areas for C. acutus in Mexico have not been carried out. Our aim in this study was to describe the CEL of the species in a tropical lagoon on the central coast of Oaxaca, Mexico.
Materials and methods
Study area. The study was carried out in Palmasola lagoon, located near Puerto Escondido in the Municipality of Santa María Colotepec, on the central coast of Oaxaca (Fig. 1). This lagoon extends parallel to the shoreline, feeds off the fluff from the Colotepec River and maintains temporary connection with the sea between June and July (Cedillo-Leal et al., 2013). The vegetation around the Palmasola lagoon are mangroves (Rhizophora mangle L. 1753, Laguncularia racemose (L.) Gaertn, Avicennia germinans (L.) L., and Conocarpus erectus L.). This lagoon is considered an important site for the conservation of the American crocodile based on the large number of organisms reported in population studies (García-Grajales & Buenrostro-Silva, 2014).
Sampling and data collection. An intensive search around the lagoon by daily walks was carried out during the 2018 nesting season, starting in mid-February, to detect and identify potential sites for egg laying. Once a nest track was detected, a thin rigid rod was used to detect the exact location of the nest, with the possibility of breaking one or more eggs during this search process. This procedure is frequently used in the search for American crocodile nests in Mexico; however, no official document related to this topic has ever been described.
Each nest detected was georeferenced (Garmin 64s, <3m precision) and carefully excavated to determine the clutch size. Prior to their removal from the nests, the eggs were marked with pencil on the uppermost surface in order to maintain their original orientation (Charruau, 2012). During the counting and extraction of eggs, egg viability was determined by observing the banding (Ferguson, 1985). All eggs were counted, including the infertile and broken eggs. Latex gloves were used during the handling of nests to avoid leaving traces and smells on the eggs, as well as on the sites where the nests were located (Cedillo-Leal et al., 2013).
Each egg was weighed (We) with a digital bascule of 1000 g (Techmel, ± 1 g precision). The maximum length (MaLE), and minimum length (MiLE) were measured with a digital caliper (AIRAJ model ARZ-150, ± 0.1 mm precision). Then, all eggs except those that were broken were returned to their initial site within the nest.
The distance from the center of the nest to the nearest trees, and the distance from the center of the nest to the nearest body of water were measured using a tape 50 m in length (Cedillo-Leal et al., 2013). Canopy coverage was estimated using photographs taken with a fisheye lens (Aukey) for a Smartphone, with the help of a tripod placed above the nest based on the methods described by Charruau (2012). The photographs were then analyzed by the software Gap Light Analyzer v.2.0 (Cary Institute of Ecosystem Studies, Millbrook).
Data analysis. To determine the spatial distribution of the C. acutus nests in the Palmasola lagoon, the georeferenced points of nests were analyzed through the QGis 3.16 software to determine the type of spatial distribution (Random, clustered, or regular) based on a square grid of 50 m2. In the first case (random arrangement), there is no spatial correlation between the locations of the points (nests), but in the second case (clustered arrangement), there are subgroups of points that tend to be significantly closer to each other, relative to other subgroups of points; and finally, in the third case (regular arrangement), points tend to stay equidistant from each other (Valdiviezo, 2019). Then, using this information crocodile nests were categorized as isolated nests (IN) when they were found far from other nests (> 2.5 m away) or as communal nests (CN) when they were found with multiple clutches in very close proximity (<2.5 m between nests) (Rodríguez-Soberón et al., 2002; Murray et al., 2019).
For each CEL site found, the measurements of egg sizes were compared between the nests through an analysis of variance (ANOVA). In addition, an ANOVA test was used to detect differences among MaLE, MiLE, and We of CEL nests. Lastly, the importance for each CEL site was determined based on the abundance of nests found in each of them (Gómez-González et al., 2017). Descriptive statistics of clutch characteristics are presented with mean, standard deviations (SD), and range values. All statistics analyses were carried out with Past 4.08 statistical software (Hammer et al., 2001), and the significance level for all tests was considered at P<0.05.
Results
Of the 27 nests registered, five were found towards the end of the incubation period. The laying period lasted 25 days (from 16 February to 13 March 2018), and the hatching period lasted 14 days (14-28 May 2018). The mean incubation period was 77.6 ± 5.89 days (n= 22), and all nests were classified as hole nests. Ten nests were considered failed because they suffered flooding due the effects of high-tides, and three nests were predated.
In respect to spatial distribution of nests, 88.9% (n= 24) were in five clustered arrangements (CEL) and the rest (n= 3) were classified as isolated nests. Three CEL sites were in the south of the lagoon and the rest (two CEL and three isolated nests) were found at the north of the lagoon (Fig. 2).
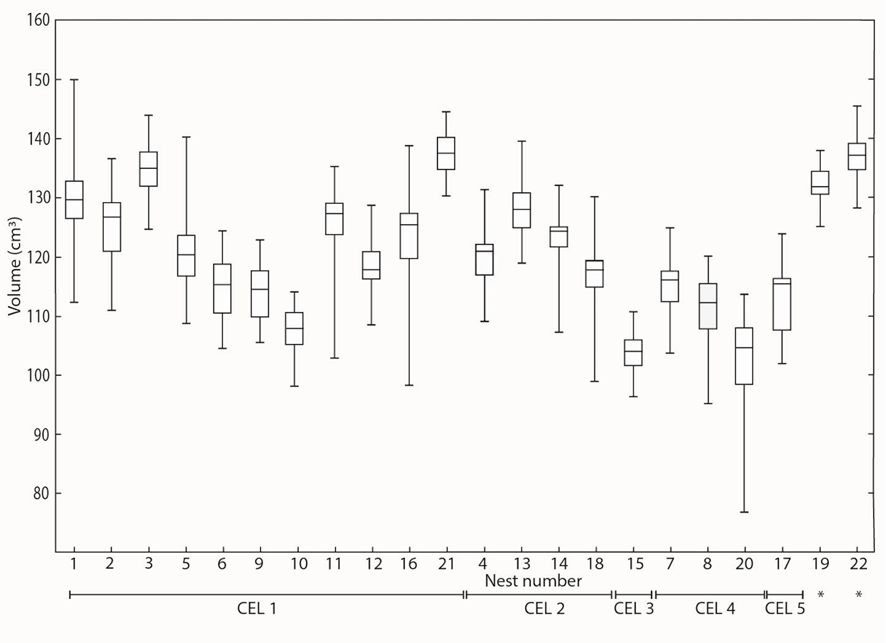
Clutch characteristics of the CEL sites and isolated nests can be found in Table 1. Briefly, 737 eggs were counted in 22 nests, with a mean clutch size of 30.7 ± 7.8 eggs per nest. The most important was CEL 1 which presented a grouping of 13 nests, with a mean distance between nests of 0.70 cm, and a mean clutch size of 34.5 ± 7.1 eggs per nest. In CEL 1, differences among nests were found for MaLE (F13, 448 = 27.451; p<0.001), MiLE (F13, 448 = 92.455; p<0.001), and We (F13, 448 = 97.549; p<0.001). In addition, statistical differences were found in the volume of eggs among nests (F13, 448 = 102.4, p<0.0001; Fig. 3). Egg fertility in CEL 1 varied between 70.6% and 97.1% (average: 89.2%), with an average of infertile eggs of 11.2%, and embryonic death of 2.8% (Table 2). In the case of isolated nests, statistical comparisons could not be made as for only one nest information was obtained.
Table 1 Clutch characteristics of Crocodylus acutus nests in a communal egg-laying area of Oaxaca. CEL (Communal Egg-laying Sites), † (Isolated nests). Maximum length (MaLE), Minimum length (MiLE), Weight (We), Standard Deviation (SD).
| CEL | Nest number | Clutch size | MaLE ± SD (mm) | MiLE ± SD (mm) | Average We ± SD (g) |
|---|---|---|---|---|---|
| CEL 1 | 1 | 40 | 76.45 ± 2.58 | 46.42 ± 0.84 | 93.88 ± 4.96 |
| 2 | 19 | 76.09 ± 3.29 | 45.77 ± 0.61 | 94.07 ± 3.72 | |
| 3 | 35 | 75.57 ± 2.16 | 47.64 ± 0.8 | 99.73 ± 2.86 | |
| 5 | 46 | 73.02 ± 2.58 | 45.83 ± 0.97 | 89 ± 3.83 | |
| 6 | 31 | 72.18 ± 1.97 | 44.94 ± 0.57 | 84.87 ± 3.08 | |
| 9 | 30 | 72.62 ± 2.22 | 44.67 ± 0.86 | 83.93 ± 3.60 | |
| 10 | 34 | 71.08 ± 1.98 | 43.88 ± 0.75 | 79.1 ± 3.50 | |
| 11 | 34 | 72.94 ± 1.92 | 46.89 ± 0.89 | 90.68 ± 4.54 | |
| 12 | 33 | 77.17 ± 2.12 | 44.12 ± 0.69 | 88.62 ± 4.94 | |
| 16 | 43 | 75.86 ± 2.85 | 45.54 ± 1.16 | 92.34 ± 4.42 | |
| 21 | 34 | 74.23 ± 2.29 | 48.48 ± 1.03 | 98.02 ± 3.23 | |
| 24 | - | - | - | - | |
| 27 | - | - | - | - | |
| Mean | 34.5 ± 7.1 | 74.3 ± 2 | 45.8 ± 1.4 | 90.4 ± 6.2 | |
| CEL 2 | |||||
| 4 | 30 | 74.29 ± 2.29 | 45.31 ± 0.61 | 88.86 ± 4.88 | |
| 13 | 38 | 75.89 ± 2.25 | 46.34 ± 0.83 | 94.12 ± 3.19 | |
| 14 | 21 | 74.44 ± 2.31 | 45.82 ± 0.82 | 90.85 ± 3.39 | |
| 18 | 33 | 73.35 ± 2.32 | 45.07 ± 0.67 | 86.03 ± 3.76 | |
| Mean | 30.5 ± 7.1 | 74.5 ± 1 | 45.6 ± 0.6 | 90 ± 3.4 | |
| CEL 3 | 15 | 29 | 70.77 ± 1.67 | 43.28 ± 0.68 | 76.1 ± 3.20 |
| 25 | 31 | - | - | - | |
| Mean | 29 | 70.77 ± 1.67 | 43.28 ± 0.68 | 76.1 ± 3.20 | |
| CEL 4 | 7 | 39 | 72.84 ± 1.97 | 44.89 ± 0.55 | 85.91 ± 3.14 |
| 8 | 20 | 73.64 ± 2.36 | 43.65 ± 1.10 | 81.69 ± 5.32 | |
| 20 | 26 | 69.42 ± 3.10 | 43.42 ± 1.27 | 73.65 ± 5.85 | |
| Mean | 28.3 ± 9.7 | 72.7 ± 2.2 | 44 ± 0.8 | 80.4± 6.2 | |
| CEL 5 | 17 | 17 | 73.47 ± 1.79 | 44.42 ± 1.08 | 83.57 ± 3.56 |
| 26 | - | - | - | - | |
| Mean | 17 | 73.47 ± 1.79 | 44.42 ± 1.08 | 83.57 ± 3.56 | |
| † | 19 | 28 | 75.42 ± 1.63 | 47.19 ± 0.62 | 92.26 ± 3.68 |
| † | 22 | 29 | 76.43 ± 1.89 | 47.80 ± 0.76 | 101.98 ± 2.81 |
| † | 23 | 17 | - | - | - |
| Mean | 30.7±7.83 | 75.9 ± 0.7 | 47.5 ± 0.4 | 97.1 ± 6.9 |
Table 2 Viability, mortality and natality of the American crocodile eggs in a communal egg-laying area of Oaxaca. CEL (Communal Egg-laying Sites), * Predated nests, ** Flooded nests, ● Assisted nests, () number of eggs at the end of the embryonic period after being predated, † (Isolated nests).
| CEL | Nest number | Number of eggs (n=24) | Viability % (n=22) | Embrio Mortality % (n=22) | Infertile eggs % (n=22) | Natality % (n=17) |
|---|---|---|---|---|---|---|
| CEL 1 | **1 | 40 | 87.50 | 2.50 | 10 | - |
| *2● | 19 (3) | 84.21 | 5.26 | 10.53 | 33.33 | |
| 3● | 35 | 94.29 | 0 | 5.71 | 84.84 | |
| 5● | 46 | 89.13 | 8.70 | 2.17 | 26 | |
| **6 | 31 | 70.97 | 0 | 29.03 | - | |
| **9 | 30 | 80 | 3.33 | 16.67 | - | |
| **10 | 34 | 97.06 | 0 | 2.94 | 28 | |
| *11 | 34(0) | 91.18 | 2.94 | 5.88 | - | |
| **12 | 33 | - | 0 | 0 | - | |
| 16● | 43 | 97.67 | 0 | 2.33 | 32.43 | |
| 21● | 34 | 70.59 | 2.94 | 26.47 | 45.45 | |
| 24 | - | - | - | - | 100 | |
| 27 | - | - | - | - | 100 | |
| CEL 2 | *4● | 30 (20) | 90 | 0 | 10 | 45 |
| **13 | 38 | 94.74 | 5.26 | 0 | - | |
| **14 | 21 | 95.24 | 4.76 | 0 | - | |
| **18 | 33 | 90.91 | 0 | 9.09 | - | |
| CEL 3 | 15 | 29 | 86.21 | 3.45 | 10.34 | 100 |
| 25● | 31 | - | - | - | 93.54 | |
| CEL 4 | 7 | 39 | 94.87 | 5.13 | 0 | 94.20 |
| 8 | 20 | 95 | 0 | 5 | 100 | |
| **20 | 26 | 80.77 | 3.85 | 15.38 | - | |
| CEL 5 | 17 | 17 | 82.35 | 0 | 17.65 | 100 |
| 26 | - | - | - | - | 100 | |
| † | **19 | 28 | 100 | 0 | 0 | - |
| † | 22 | 29 | 86.21 | 13.79 | 0 | 55.55 |
| † | 23● | 17 | - | - | - | 41.17 |
In general, the mean distance from the nest to the water edge was 13.6 ± 4.8 m (range: 3.7-24.1 m), and the mean distance between the nest and the closest tree was 3.3 ± 2.6 m (range: 0.35-10 m). The mean canopy coverage was 24.8 ± 26.9% (range: 0-95%). Fifteen nests showed a certain percentage of tree coverage, while the rest of the nests were exposed to direct solar radiation.
Discussion
Our field observation is the first documented case of communal egg-laying of the American crocodile in the Pacific slope. Although there are several studies (Kushlan, 1982; Throbajarnarson, 1984, 1988; Rodríguez-Soberón et al., 2002; Alonso Tabet et al., 2000, 2014) that reveal egg oviposition by at least two gravid females sharing the same area are more common, the nesting success for communal egg-laying in crocodilian nests has not been studied (Escobedo-Galván et al., 2019). Maybe, the first analysis approach should be the comparison of nesting success between communal and isolated nests over the same reproductive period to understand the importance of gregarious nesting areas.
Our observations at Palmasola lagoon have demonstrated the existence of communal egg-laying sites with differences in the number of eggs that they contain, and can be an indicator of active selection of nesting areas by females of different sizes. Further studies are needed to assess the reproductive effort through long-term systematic monitoring of the study area, mainly to provide new insights into the ecological and evolutionary consequences of communal egg-laying on the Mexican Pacific coast.
Palmasola lagoon has become an important nesting area for American crocodile on the coast of Oaxaca, mostly because it is a lagoon of small dimensions (0.67 km). On the other hand, to the north of Palmasola is Los Naranjos lagoon, a site where no evidence of nesting of this species has been found (García-Grajales & Buenrostro-Silva, 2014). In contrast, the south of Palmasola lagoon is in contact with a beach that is narrow (less than 50 m in width), situated along the coastline, and frequently exposed to the action of the waves and the tides.
In the whole region, some factors represent a serious threat to nesting success (Thorbjarnarson, 1996; Platt et al., 1995; Somaweera et al., 2013; González Desales et al., 2021). For example, the flooding of nests is considered to be one of the main causes of embryo mortality in some crocodile species (Pooley, 1969; Webb et al., 1977; Magnusson, 1982; Mazzotti et al., 1988). Coastal upwellings (mar de fondo) are an upward movement of water parcels (big waves) in the sea heading to the continent, that is maintained over a reasonably long period (several days to weeks; Kämpf & Chapman, 2016). Due to the distance between crocodile nesting areas and the sea, coastal upwellings produce excess moisture in the substrate of the nests, which reduces the ability to diffuse oxygen on the surface of the shell, causing malformations, suffocation and subsequently the death of the developing embryo (Mazzotti et al., 1988; Thorbjarnarson, 1989; Ji et al., 2002). Since 2011, this phenomenon has been reported in the coastal region of Oaxaca (Cedillo-Leal et al., 2013), threatening the nesting success of this species. With this in mind, there is evidence that increasing global temperatures associated with global warming could lead to doubling effects of El Niño events (Cai et al., 2014), causing coastal upwellings with greater intensity, which could lead to variability and failure of nesting seasons.
The Monte Cabaniguan-Ojo de Agua wildlife refuge in Cuba experiences a frequent loss of nests by flooding of incubation chambers due to waves and tides (Alonso-Tabet et al., 2014). Therefore, the geomorphological characteristics of nesting areas are a key element in the females’ choice and maybe an indicator of suitability of the chosen nesting site (Alonso-Tabet et al., 2014).
Predation is another factor that affects nesting success (Somaweera et al., 2013; González Desales et al., 2023). However, predation in our study was not a significant factor that affected nesting success, similarly to what Cedillo-Leal et al. (2013) reported. Maybe, the overall condition of females and abundance of nests (Lang, 1987; Campos et al., 2016; González-Desales et al., 2023) in the communal egg-laying area are the causes of why predation was low. Even, Casas-Andreu (2003) mentioned that there was no predation during his study on the coast of Jalisco. Until now, several studies in Mexico have been focused on nesting ecology of C. acutus on the Mexican Pacific slope (Casas-Andreu, 2003; Valtierra-Azotla, 2007; González Desales et al., 2016), and only in some studies (Casas-Andreu, 2003; Escobedo-Galván et al., 2019) the presence of communal nesting has been mentioned in anecdotal form. Unfortunately, the nesting ecology of this crocodile species in communal egg-laying areas has not been described at depth; therefore, the knowledge about the benefits of communal oviposition in crocodilians remain unknown (Goodwin & Marion, 1977; Kushlan & Kushlan, 1980; Dugan et al., 1981; Hunt & Ogden, 1991).
Communal egg-laying could be beneficial when suitable oviposition sites are constrained by high population densities, as is the case of Palmasola lagoon (García-Grajales & Buenrostro-Silva, 2014), or unclear environmental factors exist (Doody et al., 2009) such as coastal upwellings. Maybe, C. acutus females carefully select nesting sites in zones with limited nesting areas, using those most likely to ensure a successful outcome (Thorbjarnarson, 1989; Charruau et al., 2022), exhibiting minimal territoriality and greater tolerance with other females (Charruau et al., 2010). Therefore, territoriality and nest site competition among females reduced to the maximum is a strategy that benefits the species, reducing predation risk (Doody et al., 2009) as well as creating a save environment where one female does not have to protect the nesting area alone (Danforth, 1991).
Until now in Mexico, crocodile clutch size has been evaluated in isolated nests. Under this scenario, clutch size (30.7 ± 7.83 eggs) is similar to what reported in other areas (27.9 ± 13.2 eggs for Cuitzmala, Jalisco - Casas-Andreu, 2003; 34.28 ± 6.74 eggs for La Encrucijada, Chiapas - González-Desales et al., 2016), and it did not suffer a great variation in relation to what Cedillo-Leal et al. (2013) reported (35.3 ± 9.9 eggs). Even, the characteristics of the eggs found in Palmasola lagoon are similar to those reported at La Encrucijada (González-Desales et al., 2016), which indicates that the females are middle-aged (Ferguson, 1985). In addition, Thorbjarnarson (1996) hypothesized that there is a relationship between the weight and number of crocodilian eggs with female size, but in contrast, González-Desales et al. (2016) argues that egg characteristics have a relationship with the net primary productivity; therefore, they hypothesized that at sites with more productivity, females have more eggs and these have a greater weight with respect to females encountered in less productivity sites. Faced with this situation, we have no evidence to support any of the arguments described above; therefore, further studies are needed to assess reproductive efforts to better understand the relationships associated with clutch size, characteristics of the eggs, and female size.
The mean distance between the nests to the water’s edge, as well as the nests to the closest trees, are similar to those reported for other American crocodile populations (Cupul-Magaña et al., 2002; Casas-Andreu, 2003; Charruau et al., 2010; Cedillo-Leal et al., 2013; González-Desales et al., 2016). We speculate that the low incidence of predation is due to the short distance between nests and water’s edge, due to parental care of the mother as a strategy to avoid predation (Lang, 1987; Charruau & Hénaut, 2012; Campos et al., 2016; Barão-Nobrega et al., 2018). Moreover, communal egg-laying in Palmasola lagoon could represent a general benefit of protection of nests, coupled with a short distance from water, maximizing offspring fitness (Refsnider & Janzen, 2010; Somaweera & Shine, 2013; Escobedo-Galván et al., 2019).











 nueva página del texto (beta)
nueva página del texto (beta)