1. Introduction
The development of functional materials often involves synthesizing novel materials to enable new applications. Nanomaterials have garnered significant interest due to their small size and potential across various industries, including biomedical and electronic fields. By reducing materials to the nanoscale, new functions and quantum effects can be achieved, particularly with semiconductor and metal nanoparticles. These advancements are notable in fields like biology and medicine, where gold nanoshells, quantum dots, and super-paramagnetic nanoparticles are used for imaging and drug delivery among others [1,2].
Silver (Ag) is valued for its exceptional properties, including electrical conductivity, antimicrobial effects, and optical capabilities, making it superior to other metals. Its low toxicity allows for use in disinfectants, drug delivery, and textiles. Silver oxide exists in multiple phases: AgO, Ag2O, Ag2O3, and Ag3O4, each with unique optical and electrical properties. These oxides are utilized in photocatalysis, photovoltaic applications, energy storage, and flexible electronics. Methods such as thermal oxidation, electron beam evaporation, and chemical vapor deposition are commonly used to grow silver oxide thin films [3-6].
Recent literature highlights the successful preparation of silver nanoparticles within bacterial cellulose membranes using triethanolamine (TEA) as a stabilizer and reducing agent. This approach yields well-dispersed spherical particles, with TEA’s low content resulting in a mix of silver and silver oxides [7].
This study, “Synthesis and Characterization of Silver/Silver Oxide Thin Film via Chemical Bath Deposition (CBD)” presents a unique and economical technique for employing CBD to create homogeneous Silver/Silver Oxide thin films. It draws attention to the novel pairing of silver and silver oxide in a single film, which exhibits encouraging qualities for use in photovoltaics, sensors, and catalysis, among other fields. In addition, the study offers a thorough examination of the thin films’ optical and structural qualities, connecting synthesis parameters to material attributes. In comparison with earlier studies, this work provides an examination of the structural and optical properties, demonstrates how synthesis parameters directly affect material performance, and offers substantial improvements in the uniformity and control of the film properties.
Chemical bath deposited Silver/silver oxide thin films are expected to demonstrate superior optical and structural properties relative to those deposited by alternative methods. The synthesis parameters of CBD, including temperature, pH, and reactant concentrations, will have a significant impact on the morphology, crystallinity, and optical absorption of these thin films. In addition, silver and oxides are expected to be mixed with a single film at the same time to improve the performance in device applications.
2. Experimental procedure
Soda lime glass substrates were immersed into dilute hydrochloride acid (HCl) for 24 hours after which the substrates were washed with distilled water and then dried in air to ensure good adhesion and uniformity of films onto the substrate. A 100 ml beaker was employed as bath container with a synthetic foam cover which serve not only to protect the reaction baths from dust and other environmental impurities, but also to suspend the substrates in such a way as not to touch the bottom or wall of the bath container. The reaction baths were thoroughly stirred using magnetic stirrer; in that it gives uniform and homogeneous stir. The thermometer aided in maintaining the temperature of the baths and was regulated by turning off or on the heater when the temperature was about to exceed or fall below the range of 40-55◦C. Triethanolamine (TEA) was added slowly to 0.001 M of AgNO3 in 100 ml beaker using a syringe until 2 ml of it was dropped in each beater while the baths were under constant stirring on a magnetic stirrer with distilled water added to make a total volume of 80ml in each bath. Four pre-treated glass substrates carefully suspended from synthetic foam covers were immersed vertically in each of the bath solutions. The baths were kept at 40-55◦C on an electric heater. The first two substrates were removed one from each bath at about 60 minutes, washed with distilled water and dried in air. Subsequent ones were removes at an interval of 25 minutes; each washed and dried in air after which the samples were taken for characterization. The experimental set up is shown in Fig. 1 while Table I shows the time of synthesis of Ag/AgO thin film, the volume of TEA used, the molarities of AgNO3 and the total volume of the bath solutions.
TABLE I Deposition parameters of Ag/AgO thin film.
| Time of Deposition (hour) |
Volume of TEA (ml) |
Molarities of AgNO3 (M) |
Vol. of bath solution (ml) |
| 1.00 | 2.0 | 0.001 | 80.0 |
| 1.25 | 2.0 | 0.001 | 80.0 |
| 1.50 | 2.0 | 0.001 | 80.0 |
| 1.75 | 2.0 | 0.001 | 80.0 |
It is known that silver ions can be complexed by ammonium hydroxide according to the following reactions:
In excess of NH4OH,
The complexing reaction of silver ions with TEA follows a similar pathway [8].
This suggests that silver nitride undergoes decomposition and depending on the experimental conditions and amount of reagent, either Ag and AgO or possible mixture of AgO, Ag2O and higher oxides are deposition onto the substrate. However, the final product depends on the amount of TEA in solution. If enough TEA is added to completely dissolve the previously formed precipitate of AgOH, i.e. Ag2O, and then an Ag film is deposited onto the substrate. Just couples of drops less of TEA produced a film of AgO onto the substrate.
3. Results and discussion
3.1. Physical appearance
A colour change of AgNO3 was observed, from a colourless solution to a brownish solution, which became clear under constant stirring upon the addition of TEA, attributable to the dissolution of the initially formed precipitate. The deposited films were opaque, and the opacity increased as the deposition time increased. Although, in each case, there was uniformity and homogeneity in the dispersion of the film on the substrate, indicating that the thickness of the thin film deposits increased with time, culminating in a more reflective surface. The opacity of the film and the reflective surface were at maximum when excess TEA was added solution.
3.2. X-ray diffractometer
The X-ray diffraction spectra of the as-deposited Ag/AgO thin films are presented in Fig. 2 with 2θ values varying from 20◦C to 80◦C and Cu-kα radiations of wavelength λ ≈ 1.5406 Å.
The appearance of Bragg peaks: 38.05◦C, 44.15◦C, 64.31◦C, and 77.41◦C were correlated with standard JCPDSICDD diffraction patterns from PDF-2 sets 1-43 database and there exists conformity which corresponds to the preferred orientation planes of (111), (200), (220) and (311) respectively confirming the presence of Ag/AgO films. Following the inverse law relationship of full width at half maximum β (FWHM); Eq. (5) was used to deduce the average crystallite size which was valued as 0.21 nm after calculation.
where λ is Cu-kα radiation wavelength (1.5406 Å), β is full width at half maximum (FWHM) in radian, θ is the Bragg angle. Using the Bragg’s equation of Eq. (6), the interplanar spacing d hkl was established to be averagely 1.7698A for the grown Ag/AgO thin film.
where n corresponds to the order of the fringes and equals 1 for a special case. Other crystallographic parameters determined were macrostrain, є and dislocation density, ρ given by Eqs. (7) and (8) respectively. The interplanar spacing and the macrostrain decreases as the 2θ value increases.
Equation (8) was established by Williamson and Smallman; where a is the lattice constant and D is the particle size. Table II shows the values calculated for as-deposited Ag/AgO thin film at the temperature range of 40◦ to 55◦.
TABLE II Summary of structural parameters of as-deposited Ag/AgO thin film.
| Bath temp. (◦C) |
Particle size (nm) |
Dislocation3
density, ρ(Å −1) |
Microstrain, є |
Interplanar spacing, dhkl (Å) |
| 40-55 | 0.2 | 0.65 | 0.37 | 1.77 |
These parameters in Table II, contribute to the film’s enhanced optical and electronic properties. The small particle size suggests high surface area and quantum effects, beneficial for applications in sensors and catalysis. The moderate dislocation density implies the presence of crystalline defects, potentially increasing catalytic activity, while the macrostrain indicates structural strain that could modify the film’s band structure. The interplanar spacing (1.77 Å) reflects the crystallographic orientation, essential for tailoring film properties in optoelectronics [9].
3.3. Optical analysis
Figure 3 show the optical absorption spectra of Ag/AgO thin films without taking reflection and scattering into account which was measured in the wavelength range of 200-1100 nm for the as-deposited films. The Ag/AgO thin films showed a high absorbance value.
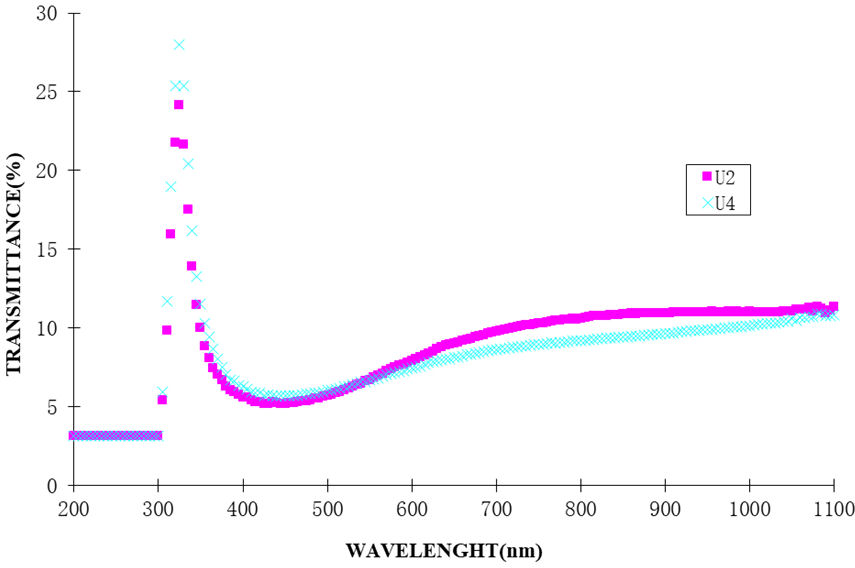
The transmittance spectra for the films is shown in Fig. 4. Despite the slight increase of transmittance through the visible spectrum, there was stability in its measurement at wavelength λ ≈ 1100 nm. Thicker Ag/AgO thin-film films exhibit lower transparency, especially in the visible spectrum, according to their transmittance spectra. This is corroborated by recent studies, which show that increased thickness of silver oxide thin films reduces transmittance mainly because of increased light absorption and scattering. In the visible spectrum, for instance, films that are thicker than 100 nm have transmittance below 20%, according to Patel et al. [10]. According to Wang et al. [11], developments in chemical bath deposition processes have made it possible to better control layer thickness, which has improved both transparency and electrical conductivity.
Figures 5 and 6 depicts the variation of absorption coefficient and refractive index with photon energy. The values of absorption coefficient were calculated using Eq. (9):
where α is the absorption coefficient; t is film thickness, I 0 and I t are the intensities of initial and transmitted light respectively.
The absorption coefficient (α) is related to the incident photon energy by:
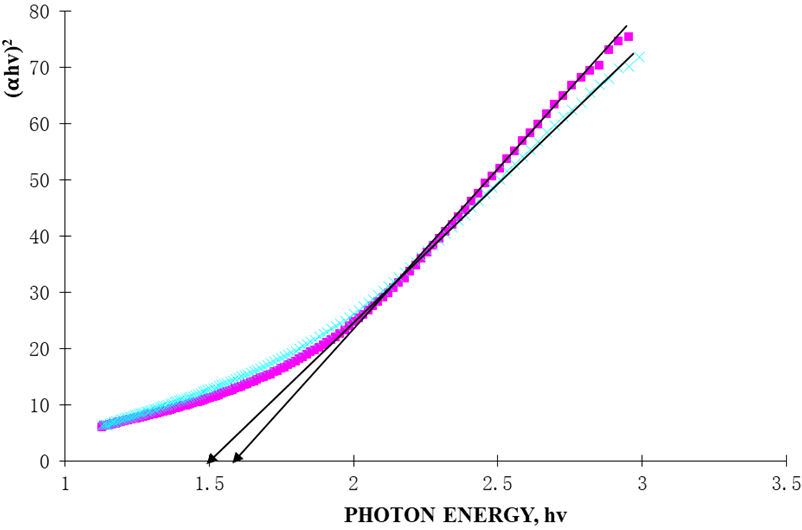
where K is a constant, E g is the energy band gap and n is a constant equal to 2 for direct band gap. Figure 7, shows the variation of (αhν)2 versus hν for Ag/AgO films. The graph gave the direct band gap values, obtained by extrapolating the linear line on hv axis as 1.5 and 1.6 eV for the films.
3.4.Morphological analysis
The scanning electron micrographs of the Ag/AgO thin film coated on glass substrates shed light on the surface morphology of the film as shown in Fig. 8. The micrographs show a black, granular surface with different-sized and shaped grains. Notably, there are no holes or cracks evident on the surface, demonstrating the consistency and strength of the deposition. Nonetheless, the grains were uniformly dispersed, with certain regions exhibiting agglomeration. Approximately 20 µm is the average grain size, as determined by ImageJ software [12, 13]. This grain size is in line with similar research [14, 15] that highlights the impact of deposition conditions on thin film microstructure. The uneven grain distribution seen in the SEM pictures is characteristic of films deposited via chemical bath deposition (CBD), where the nucleation and development of grains are greatly influenced by bath temperature, pH, and reactant concentration, among other variables [16].
The significance of optimizing these parameters to attain the intended grain size and morphology has been brought to light by recent developments in the synthesis of Ag/AgO thin films using CBD. For example, Wang et al. [11] work from 2023 showed that changing the bath solution’s pH may greatly lower agglomeration and enhance the uniformity of grain distribution. In a similar vein, Liu et al. [17] discovered that regulating the deposition temperature might improve the optical and electrical properties of Ag/AgO thin films by increasing their crystallinity and decreasing their structural defects.
3.5. Energy dispersive X-Ray analysis
The EDX spectrum in Fig. 9, recorded along the cross-section of the sample reveals the presence of Ag crystallite. The diagram shows the presence of a peak associated to the Ag (≈ 22:20%) atoms. Almost disappearing peak of oxygen suggest that it is a contribution from the glass substrates giving an elemental composition of Ag/O-Si-Ca-Al-Na-Mg-S. Traces of these elements shows that the starting materials (TEA and AgNO3) could contain some impurities. It is not unexpected that traces of Si, Ca, Al, Na, Mg, and S were observed, as these elements are common components of the glass substrate, which contains them as active ingredients. Daniel et al. [18] has reported similar phenomenon for soda lime glass substrate.
4. Conclusions
Using Ag+ - triethanolamine (TEA) as the precursor, an aqueous chemical bath deposition technique was used to synthesize Silver/Silver oxide thin films at temperatures between 40 and 55 oC. This confirmed the hypothesis that CBD can produce films with better optical and structural properties than other methods. The resultant films had a poor transmittance of about 12% and a high absorbance of ¸80%, exhibiting uniformity and reflection. The films appear to be suited for applications requiring spectrally selective films, such as electronics, optoelectronics, photothermal, and related applications, based on their direct energy band gap (E g < 2 eV). The study demonstrated how the synthesis factors affected the Ag/AgO films’ morphology and optical properties. As a potent reducing agent and a silver ion chelating agent, TEA had a dual purpose in regulating nucleation, particle development, and eventually particle size. The deposition method may be fine-tuned to make films with the necessary silver oxide content by tuning TEA concentration. This would increase the films’ potential for use in solar cells, thermal control window coatings, anti-reflective coatings, and other cutting-edge technical fields. These results support the hypothesis that CBD can synthesize high-performance Ag/AgO thin films for device applications by highlighting the significance of meticulously regulating the synthesis conditions to obtain the required film properties.











 nueva página del texto (beta)
nueva página del texto (beta)