INTRODUCTION
The coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), is a significant global health challenge with over 83 million confirmed cases as of December 2020. The World Health Organization (WHO) has declared the COVID-19 pandemic on March 11, 20201. COVID-19 is associated with a wide range of clinical syndromes, including respiratory, cardiac, thrombotic, and inflammatory complications2-4, and it is essential to identify patients who are at high risk of these morbidities and poor outcomes.
Monoclonal gammopathy of undetermined significance (MGUS) is a premalignant plasma cell disorder occurring in approximately 5% of patients over the age of 705. A small proportion of MGUS patients progresses to multiple myeloma, lymphoma, or AL amyloidosis (risk of progression of 1%/year5). While MGUS is otherwise considered a generally "benign" condition, MGUS patients have several clinical features that are relevant to consider when assessing their risk during the COVID-19 outbreak.
In addition to their older median age (72 years5) and medical comorbidities6, MGUS patients have previously been shown to have increased susceptibility to infections partly related to immunoparesis7,8, greater incidences of thrombotic events compared to the general population9,10, and higher mortality compared to expected age-matched controls5. Given these factors, it is important to study the vulnerability of MGUS patients to COVID-19 infection and its complications; therefore, we assessed our local experience of MGUS patient outcomes during this pandemic.
METHODS
Retrospective chart reviews of all consecutive patients with MGUS seen at the University of Calgary Medical Clinics (an academic medical center) between 2014 and 2020 were completed. Patients were managed according to local institution guidelines. This project has been approved by the Health Research Ethics Board.
Statistical analysis
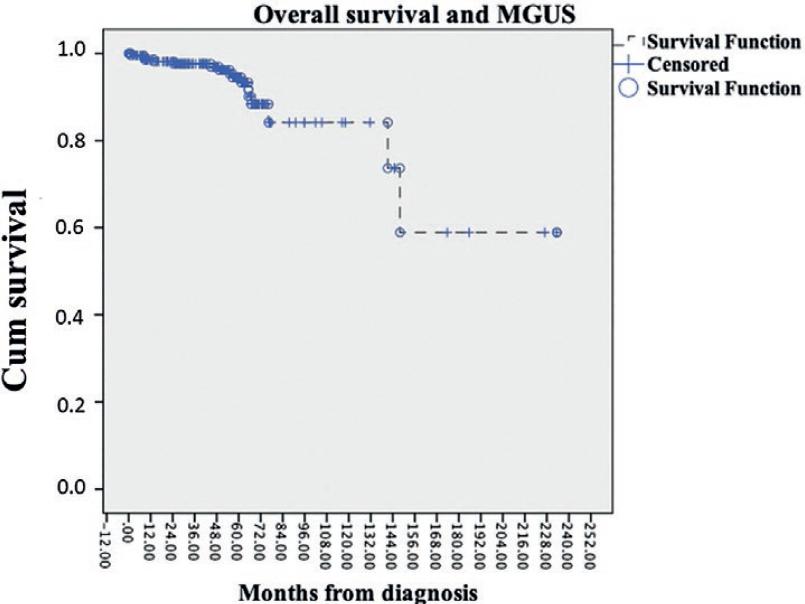
All statistical analyses were performed using the SPSS 24.0 software. Independent samples t-test was used to compare mean values (Table 1). Kaplan-Meier survival curve analysis was performed for the assessment of median survival and for the generation of figure 1.
Table 1 MGUS clinic patient characteristics
| Entire MGUS cohort (n = 228) Median |
Patients who died before the pandemic (n = 7) | Patients who died during the pandemic (n = 10)
Mean |
Independent sample t-test p-value | |
|---|---|---|---|---|
| Age | 73 | 83.0 | 75.2 | 0.4 |
| Male | 128 (56%) | 6 | 7 | 0.1 |
| Overall survival (months) | Not reached | 40.6 | 53.2 | 0.3 |
| Hemoglobin | 136 | 143 | 138.5 | 0.9 |
| Lymphocytes | 1.6 | 1.7 | 1.0 | 0.9 |
| Creatinine | 90 | 138.5 | 124 | 0.4 |
| Serum M protein (g/L) | 3.5 | 5.84 | 5 | 0.2 |
| Bone marrow plasma cells (n = 153) | 3% | 5 (n = 4) | 4.5 (n =5) | 0.1 |
| NT-proBNP | 124 | 4751 | 696 | 0.01 |
| Frequency | ||||
| IgG MGUS | 115 (50.5%) | 2 (28.6%) | 6 (60.0%) | |
| IgA MGUS | 20 (8.7%) | 1 (14.3%) | 2 (20%) | |
| IgM MGUS | 35 (15.3%) | 1 (14.3%) | 1 (14.3%) | |
| Biclonal MGUS | 8 (3.5%) | 0 | 0 | |
| Light chain MGUS | 44 (19.3%) | 3 (43.0%) | 0 | |
| Heavy chain and light chain MGUS | 6 (2.6%) | 0 | 1 (10%) | |
| MGUS risk | ||||
| Low | 56 (24.6%) | 0 | 4(40%) | |
| Low-intermediate | 83 (36.4%) | 1 (14.3%) | 4(40%) | |
| High-intermediate | 30 (13.2%) | 2 (28.6%) | 1 (10%) | |
| High | 2 (0.9%) | 0 | 0 | |
| Light chain MGUS | 44 (19.3%) | 3 (43.0%) | 0 | |
| Biclonal MGUS | 8 (3.5%) | 0 | 0 | |
| Missing data | 5 (2.2%) | 1 (14.3%) | 1 (10%) | |
| Immunoparesis | ||||
| None | 146 (64%) | 3 (42.9%) | 6 (60%) | |
| One immunoglobulin decreased | 45 (19.7%) | 1 (14.3%) | 3 (30%) | |
| Two immunoglobulins decreased | 25 (11.0%) | 3 (42.9%) | 1 (10%) | |
| Three immunoglobulins decreased | 1 (0.4%) | 0 | 0 | |
| Missing data | 11 (4.8%) | 0 | 0 | |
| Previous medical comorbidities | ||||
| Previous venous thromboembolism (DVT/PE) |
9 | 0 | 2 | |
| Atrial fibrillation | 33 | 4 | 3 | |
| Hypertension | 97 | 2 | 5 | |
| Diabetes | 52 | 0 | 3 | |
| Autoimmune conditions | 49 | 0 | 0 | |
| (includes psoriasis, rheumatoid arthritis, rheumatic polymyalgia, chronic inflammatory demyelinating polyneuropathy, hypothyroidism, Crohn's disease, multiple sclerosis, microscopic colitis, systemic lupus erythematosus, Sjogren's syndrome, GAD65 positive Stiff syndrome, pernicious anemia, anti-MAG antibody, Behcet's disease, and urticarial vasculitis) | ||||
MGUS: monoclonal gammopathy of undetermined significance.
RESULTS
A total of 228 patients with MGUS followed at the University of Calgary Medical Clinic were assessed. Clinical characteristics are reported in table 1. Median age of the group was 73 years. Since the beginning of the pandemic, all MGUS cases have been followed up by virtual appointments; 190 patients have been assessed through telemedicine.
At the time of analysis, 211 patients are alive, 7 patients died before the start of the pandemic, and 10 patients died since the pandemic declaration by the WHO on March 11, 2020. Three patients died in 2019 compared to 10 since the COVID-19 pandemic declaration, suggesting an excess of deaths in the year 2020. The median overall survival (OS) for the 228 patients was not reached (Fig. 1). Median follow-up was 55 months. There were no significant differences between the mean age and the OS of the patients who died before versus during the pandemic (age 83.0 vs. 75.2 years, p = 0.4, OS 40.6 vs. 53.2 months, p = 0.3) (Table 1).
Of the 10 patients who died during the pandemic, one patient died of COVID-19 a week after the diagnosis. Among the remaining nine MGUS patients, four patients had COVID-19 testing which was negative. One patient died of complications with viscous perforation with gastrointestinal (GI) involvement of AL amyloidosis, two patients died of an exacerbation of chronic cardiac comorbidities, and six patients died of undocumented or unknown causes.
At the onset of the pandemic in the beginning of year 2020, patients were eligible to have COVID-19 testing done if they had fulfilled the criteria for testing in our province which included the presence of fevers, new or worsening cough, dyspnea, rhinorrhea, chills, sore throat, painful swallowing, headache, muscle or joint ache, malaise, fatigue, severe exhaustion, nausea, vomiting, diarrhea, unexplained loss of appetite, loss of sense of smell or taste, or conjunctivitis. From the end of May 2020 to November 2020, testing criteria were expanded such that anyone was eligible for COVID-19 testing regardless of symptoms. Testing was done by nucleic acid amplification using primers targeting the E (envelope protein) gene of SARS-CoV-2. In the general population of 4.4 million in the province of Alberta, 1,650,655 people had been tested, 100,428 cases were positive, and 1046 patients died as of December 30, 202011. In the 228 MGUS clinic population, 85 patients were tested (37.3%). Three patients so far have tested positive for SARS-CoV-2; one patient died of COVID-19 and the other two are alive at the time of submission. The incidence of positive cases among our MGUS patient cohort is too small to understand the impact of the expanded local COVID-19 testing eligibility which included testing asymptomatic patients.
The one patient with MGUS who died of COVID-19 a week after testing positive was a male in his late 80s whose comorbidities included advanced dementia and hypertension. He did not have immunoparesis. The other two patients with MGUS and COVID-19 were a female in her 70s with hypertension, hyperparathyroidism, without immunoparesis, and a male in his 30s with end-stage renal disease, obesity, hypertension, and hypothyroidism, who had immunoparesis involving IgG.
In the MGUS cohort, five patients have progressed; two patients developed lymphoma, one patient developed smoldering myeloma, one patient developed AL amyloidosis and symptomatic multiple myeloma, and one patient progressed to AL amyloidosis with GI involvement (duodenal and gastric). All progression events occurred before the pandemic onset, except for the one patient who developed AL amyloidosis with GI involvement. This patient had a complication of viscous perforation and died. Median time to progression in the five patients was 44.6 months. Six patients deemed to have monoclonal gammopathy of clinical significance (MGCS) were transferred to the cancer center to expedite required investigations.
Nine patients had venous thromboembolisms (VTE), and all VTE events occurred before the pandemic onset (Table 1). One patient had a pulmonary embolism (PE), seven patients had deep vein thrombosis (DVT), and one patient had both a PE and a DVT.
Immunoparesis was found in 71 (31.1%) out of 228 patients. Forty-five patients (19.7%) had one affected immunoglobulin, 25 patients (11%) had two affected immunoglobulins, and 1 patient (0.4%) had decreased levels of three immunoglobulins. Data on immunoglobulin levels at diagnosis were missing in 11 patients.
Discussion
The province of Alberta has declared a public health emergency as it undergoes a recurrent rapid surge in COVID-19 infection rates with the latest reported active cases of 14,555 as of January 2021 since the nadir of 339 active cases back in June 2020. The number of active cases continues to rise with over 1000 new cases diagnosed daily. In the small MGUS clinic population presented here, COVID-19 testing has been done in 37.3% of the patients (85 out of 228 patients) compared with the testing rate of 37.5% in the general population within the province (1,650,655 people out of approximately 4.4 million). The incidence rate of COVID-19 in the MGUS clinic is 1.3% (3 out of 228 patients), compared with the rate in the general population within the province which stands at around 2.28% (100,428 out of 4.4 million). Unfortunately, the incidence rate in the general Alberta population continues to increase at the time of this writing (January 2021).
To assess the true susceptibility of MGUS patients to COVID-19, consistent testing is essential. It is possible that many MGUS patients are asymptomatic and well. Potentially, a number of MGUS patients may be reluctant to seek medical attention12 due to fear of COVID-19 despite developing new symptoms. With their medical complexity, older age, and frailty, patients may be offered comfort care without COVID-19 screening when severe illnesses occur.
With small patient numbers, our findings are inconclusive as to whether the older age, presence of comorbidities, inherent susceptibility to infections, and thrombotic tendencies of MGUS patients7,10 truly confer additional risk for poor outcomes during this pandemic. There were no significant differences found in the mean age, OS, or in the selected biochemical parameters (Table 1) of MGUS patients who died before or after the pandemic onset. Patient numbers were small with low incidences of COVID-19 to assess other endpoints such as intensive care unit (ICU) admission or ventilator support requirements.
Neither the MGUS risk score (based on one point for each of non-IgG monoclonal protein, abnormal free light chain ratio, and serum monoclonal protein ≥ro g/L13) nor immunoparesis appeared to be associated with death in our patient cohort. While MGUS patients carry a higher risk of thrombosis than the general population, an increase in VTE events was not seen since the pandemic onset.
Of seven MGUS patients with COVID-19 showed that despite having high-risk comorbidities, none of the patients required ICU care, and six of the seven patients recovered from the infection. A recent case series14 of 7 MGUS patients with COVID-19 showed that despite having high risk comorbidities, none of the patients required ICU care, and 6 of the 7 patients recovered from the infection. Similarly, in a report of 127 patients with plasma cell disorders who had COVID-19, including 20 MGUS patients15, their older age (>65) or hypogammaglobulinemia was not associated with death or ICU admissions.
Overall, our study with a small patient number did not identify increased COVID-19-related risk in MGUS patients. In future studies, it would be important to assess whether COVID-19 infection affects the rate of progression from MGUS to symptomatic disease secondary to asymptomatic inflammatory states contributing to disease progression. Larger population-based studies with longer follow-up periods are required to understand the full impact of COVID-19 on the MGUS patients' outcomes. Furthermore, as per the latest International Myeloma Working Group recommendations, all patients with MGUS are encouraged to receive vaccinations against COVID-19. Ongoing data collection will be essential to assess MGUS patients' immunological response to vaccination.











 nueva página del texto (beta)
nueva página del texto (beta)